Abstract
In this study, optimized conditions were established for diatomite grinding, which is a natural inorganic mineral with inherently high oil absorption capacity. Diatomite surface was modified with a fluorocarbon chemical and stearic acid via facile methods for enhancing compatibility between polypropylene and diatomite. Polypropylene/diatomite composites were generated in a twin screw extruder with/without using compatibilizer, and nonwoven structures were produced via meltblown technique. Pore size and void content analyses showed that addition of diatomite led to thicker fibers (1–17 µm (the neat polypropylene) vs. 1–32 µm (2 wt.% diatomite containing polypropylene)). Diatomite incorporation into polypropylene resulted in a rigid and brittle structure and a worsened oil absorption property (rust inhibitor oil absorption capacity: 1184% ± 105% (the neat polypropylene) vs. 718% ± 78% and 1089% ± 136% (2 wt.% diatomite containing polypropylene)). Increasing oil viscosity resulted in increased discrepancy among the oil absorption capacities of the neat polypropylene and diatomite containing polypropylene. Analysis of variance tests showed no changes or statistically insignificant differences in oil absorbency.
Introduction
Industrial oil sorbent textiles possessing a total turnover of $106.8 billion have an important position in technical textiles market. The most important reasons of this situation are the ever-increasing usage of oil and oil derivatives in industry and everyday life and the increased environmental conscious. Consequently, necessity of textile materials with improved oil absorbency property has been increasing day by day.
Sorbent materials should be both oleophilic and hydrophobic, i.e. they should have a high capacity to sorb oil and repel water. They are classified into three categories: natural organic vegetable-based, natural inorganic, and synthetic organic materials [1–3]. Natural organic sorbent materials contain a lot of agricultural products such as sugarcane bagasse, saw dust, straw, milkweed, kenaf, cotton, wool, and kapok [4,5]. All of these products will become water wet and sink, carrying the oil with them [6]. It is known that these materials are subjected to a surface modification for improving their oil absorptive properties [7–12]. Inorganic mineral sorbents include stearate-treated calcium carbonate, expanded perlite, expanded vermiculite, zeolite, exfoliated graphite, activated carbon, organoclay, basalt fibers, and diatomite (DE), which are also known as sinking sorbents [4,13–15]. Mineral sorbents are generally disliked as they have numerous shortcomings, such as contamination of sea beds and harmful effects to aquatic habitats. They also tend to release some of the sorbed oil while sinking because of the low retention capacity of some of the solids [6,16–18]. Synthetic organic materials are the most widely used sorbents, which are made from high-molecular-weight polymers, such as polyurethane and polypropylene (PP). They have good hydrophobic and oleophilic properties and high oil sorption capacity.
Absorbent minerals have been used since the 1950s for cleaning purposes of industrial spillages. DE, which is one of the absorbent minerals, represents a very rare occurrence—a silica mineral that has an elaborate structure worked by nature into a labyrinth of tiny holes. No other silica source that is mined or chemically prepared has such a structure [19]. The combination of the natural silica composition, the overall structure of the diatom particles, and the network of holes in the structure are responsible for its porous structure and thus high water and oil absorption capacity (150%–300%) [20,21].
DE has its origin from a siliceous, sedimentary rock consisting principally of the fossilized skeletal remains of diatom, a unicellular aquatic plant related to the algae, during the tertiary and quaternary periods [22,23]. DE consists of a wide variety of shape and sized diatoms, typically 10–200 mm, in a structure containing up to 80%–90% voids [24]. DE’s highly porous structure, low density, and high surface area resulted in a number of industrial applications as filtration media for various beverages and inorganic and organic chemicals as well as an absorbent for pet litter and oil spills. Although DE has a unique combination of physical and chemical properties, its use as an absorbent aid material in synthetic nonwoven structures has not been investigated so far.
Considering cost, oil absorption, and environmental issues, we tried to combine PP and DE in one single structure. To that end, DE was treated with a fluorosilane chemical, a fluorocarbon (FC) chemical, and a saturated fatty acid, and their surface properties related to water and oil wettability and absorbency were evaluated in our previous study [25]. In this study, the surface untreated/treated DEs were incorporated into PP matrix for generation of meltblown nonwoven structures and their oil absorption property was investigated for the first time.
Experimental
Materials
Materials, chemicals, and their functions used in the study.

Methods
Before the methods are explained, a flowchart is given in Figure 1 for a better understanding of the processing steps used in this study.
Flowchart of the processing steps.
Grinding
Considering the literature that there is an inverse proportionality between particle size and surface area [28,29], DE was ground for the purpose of enhancing specific surface area and thus to increase the absorption capacity of DE. Besides, reduction in particle size would enable easier processing during meltblown nonwoven production. Deniz [20] showed that DE could be ground easily although it contains high amount of silicon dioxide (SiO2) meaning that there would be no problem in terms of grinding process.
DE samples were dry and wet ground in a Fritsch Pulverisette 5 planetary ball mill at 400 r/min using balls with 0.5 and 5 mm diameter. The material of the mortars and balls was zirconium oxide. The milling process was conducted in time intervals of 2 min milling, followed by a 4-min break to avoid excessive heating of the mortar and sample.
Fluorocarbon (FC) surface treatment
Here, 3.3 ml (10 wt.%) FC compound was added to 90 ml of distilled water containing 10 g of DE and stirred for 15 min using a magnetic stirrer. The slurry was repeatedly washed with distilled water and then centrifuged at 4000 r/min for 9 min. Then the slurry was dried and cured at 110℃ for 5 min [26].
Stearic acid (SA) surface treatment
DE was surface treated with stearic acid (SA) (10 wt.%) in dry state in a Cyclomix 5 (Hosokawa Micron, The Netherlands) high shear mixer. It can be used in the drying, coating, mixing, agglomeration, and densification of particulate materials and has a nominal volume capacity of 5 l. The mixer has four pairs of flat-bladed impellers placed from the bottom to the top (Figure 2). Related details can be found elsewhere [30]. The DE surface coating was carried out at 80℃ for 15 min with a rotation speed of 1500 r/min. After completing the coating process, a quick cooling to room temperature and discharging were carried out.
Schematic diagram of a Cyclomix high shear mixer [30].
Generation of compounds and nonwoven structures
Notation and amounts for PP/diatomite (90/10 w/w) compounds.
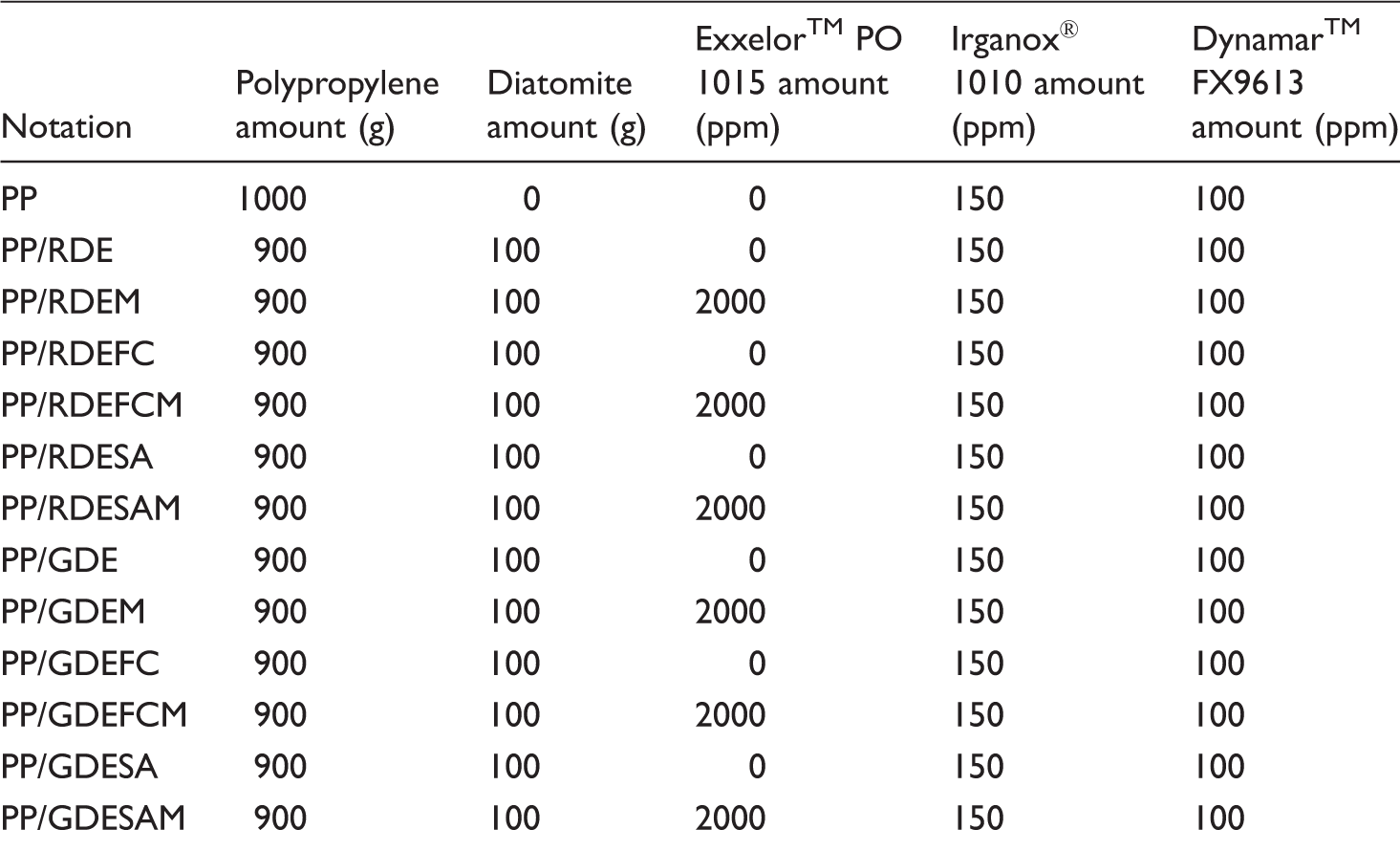
PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.
Meltblown production parameters.

Characterization
Brunauer, Emmett and Teller analysis
The specific surface area of the DE samples was determined using multi-point measurements on a Micromeritics Gemini VII 2390 instrument by the Brunauer, Emmett and Teller (BET) gas adsorption method. The samples were dried at 150℃ for 24 h and degassed at 200℃ for 5 h under vacuum prior to measurement.
Particle size analysis
Particle size and particle size distribution analyses were performed on Mastersizer 2000 laser diffraction particle size analyzer (Malvern Instruments Ltd, UK) from wet dispersions. A micro-volume (200 mg) sample feeder collected triplicate measurements in 2-s intervals. The obscuration limits were set to 5%–15%.
Contact angle and surface energy analyses
In order to determine hydrophilic/hydrophobic and oleophilic/oleophobic character of the untreated/treated DE and DE incorporated meltblown structures, contact angle measurements were carried out on the Theta Lite Optical Goniometer (KSV Instruments Ltd, Finland). DE samples were prepared in a hydraulic pellet press and dried at 80℃ overnight in an air circulating oven before measurement. Contact angles were measured using the sessile drop method with 3 µl distilled water and linseed oil droplets for the judgment of hydrophobicity and oleophobicity, respectively. Measurements were taken after the water/linseed oil droplet came into contact with the DE for 30 s. In total, 10 values were recorded and their average was calculated.
Contact angles of ethylene glycol, formamide, and diiodomethane of the raw and surface-treated DEs were also measured for surface free energy calculations by using Van Oss-Chaudhury methods.
Melt flow index analysis
The melt flow index (MFI) values of the neat PP and PP/DE compounds were detected according to ASTM 1238 using an MFI analysis instrument (Devotrans EA3, Istanbul, Turkey) [31]. The flow measurement was conducted by pushing the samples through a 2-mm diameter die at 230℃ with 2.16 kg mass. Results are expressed in grams per 10 minutes (g/10 min).
Differential scanning calorimetry analysis
Melting peak temperature (Tm), melting enthalpy (ΔHm), crystallization temperature (Tc), and % degree of crystallinity (χc) of the PP/DE compounds and nonwovens generated were determined with the help of a Perkin Elmer PYRIS Diamond™ differential scanning calorimeter. Heating and cooling scans were performed under nitrogen atmosphere at 10℃/min over the temperature range from 20℃ to 220℃. The first heating scans were considered for evaluation. Crystallinity of the samples was determined by adapting equation (1) as follows

Scanning electron microscope analysis
For morphological analysis, the samples were coated with gold (Au) using Emitech K950X sputter coater to avoid charge build-up and analyzed by LEO 440 scanning electron microscope equipped with a Quartz Xone energy dispersive X-ray (EDX) analysis system.
Pore size analysis
The pore sizes of the nonwoven structures were determined using a CFP-1005AQC capillary flow porometer (PMI Porous Materials, Inc., Ithaca, NY, USA) according to the ASTM F316-03 [33]. A Silwick by PMI with a surface tension of 20.1 dynes/cm was used as the immersion liquid. The sample immersed in the immersion liquid was pretreated by lowering it to 80 kPa below atmospheric pressure and deairing so that no bubbles remained in the sample. The measuring diameter was 20 mm. Dry air was passed through the sample and the gas pressure was increased in stages, measuring the gas flow rate at each point. The pressure P50 (PSI) at which the flow rate with the sample immersed in the liquid (wet flow rate) was 50% of the flow rate when not immersed (dry flow rate) was determined, and the mean flow pore size was calculated by equation (2).

In the formula, d50 is the mean flow pore size (µm), r is the surface tension of the immersion liquid, which was 20.1 (dynes/cm), and the constant C is 0.451 (µm·cm·PSI/dynes). Measurement was performed three times and the average value was calculated. Minimum (d10) and maximum (d97) flow pore sizes were also calculated according to the equation (2) [34].
Void content analysis
Void content measurement of the meltblown nonwoven structures was carried out from the images acquired from a Leica DFC450 optical microscope (Leica, Germany, Magnification: ×40) using Image Proplus 6.0 program. This involved shining a strong light through a sample and observing any shadows formed by voids. Three replications were applied to obtain average values.
Oil absorption measurements
The oil absorption capacity was measured by the gravimetric method. Meltblown structures were conditioned at 80℃ for 24 h in an air circulating oven prior to the measurements. ASTM D1483 standard was used as the test method which involves the titration of linseed oil onto a known amount of DE (1 g) until free oil appears [35]. Besides, three different oils (rust inhibitor oil, hydraulic oil, and diesel oil) were used for oil absorption measurements. The average of three measurements was used for analysis.
Statistical analysis
Oil absorption test results were statistically evaluated in Design Expert® 6.06 software by considering the grinding, compatibilizer, surface treatment, and oil type as independent variables and oil absorption property as the dependent variable. In order to judge the statistical importance of the variations, analysis of variance (ANOVA) tests were applied. The “p” values were examined as to whether the parameters were significant or not. If the “p” value of a parameter is greater than 0.05 (p > 0.05), the parameter will not be important and should be ignored.
Results and discussion
Particle size and morphology of the DE
Particle size distribution of the diatomite samples.

RDE: raw diatomite; GDE: ground diatomite; D: dry grinding; W: wet grinding; Ø: bead diameter.
The mean particle size (d50) of the raw DE was determined to be 20.26 µm. Its d10 and d90 values were 8.53 and 37.54 µm, respectively. Particle size of the DEs decreased to a large extent after grinding. Processing conditions were optimized according to the dry grinding. Two different beads (Ø: 0.5 and 5 mm) and four different grinding times (48 min (active grinding: 16 min), 78 min (active grinding: 26 min), 120 min (active grinding: 40 min), and 240 min (active grinding: 80 min) were employed. In addition, combined grindings were also performed by using different beads. Especially, the mean particle size (d50) was taken into account for judging the grinding performance. According to the results, the smallest particle sizes were obtained with a grinding time of 240 min (d10: 0.72 µm, d50: 4.12 µm, d90: 10.26 µm). A grinding time of 48 min delivered similar results (d10: 1.55 µm, d50: 4.69 µm, and d90: 11.57 µm). Wet grinding, which was also performed with a grinding time of 48 min, exhibited nearly the same results (d10: 0.97 µm, d50: 5.30 µm, d90: 18.39 µm for GDEW48-Ø5). Considering the particle sizes obtained and the easiness of the process, dry grinding with a total grinding time of 48 min was decided to use for compound and nonwoven generation. It was observed that usage of smaller beads together or separately with 5 mm beads did not work in terms of particle size reduction.
The morphological analysis showed that the diatom frustules in the raw DE were mostly well preserved, having cylindrical or disk shape (Figure 3, RDE). In spite of a short processing time of 48 min, dry grinding obviously deteriorated the porous structure of the DE (Figure 3, GDED), while wet grinding damaged it to less extent (Figure 3, GDEW).
SEM images of the neat (RDE), dry ground (GDED), and wet ground (GDEW) diatomite.
Surface area of the DE
We have shown in our previous study that surface area of the DE increased after grinding only to a small extent (RDE: 0.23 ± 0.06 m2/g, GDE: 2.25 ± 0.03 m2/g) [25]. Pore size of the DE increased after grinding (RDE: 163.7 Å, GDE: 201.8 Å). Grinding did not change the pore volumes considerably. As indicated in the literature [36–38], calcination carried out at high temperatures up to 1600℃ causes the generation of siloxane bridges on the DE backbone surface which leads to closing of the pores. Closed pores on the DE surface were not able to be detected by the BET N2-adsorption method and thus very low surface area was detected.
Surface modification of the DE
In order to increase physical interactions between hydrophilic DE and hydrophobic PP and thus contributing to enhanced compatibility in PP/DE compounds, hydrophobic surface treatments were performed on the DE. According to the results obtained from our previous study [25], it can be clearly said that the both surface treatment chemicals were successfully attached to the DE samples’ surfaces either physically (SA) or chemically (FC).
Wettability and oil absorptivity of the DE
Our previous study gives a detailed report on wettability and oil absorption properties of the raw, ground, and surface-treated DEs [25]. It was shown that the inherently hydrophilic and oleophilic structure of the DE could be changed to either a hydrophobic/oleophobic or hydrophobic/oleophilic one by FC or SA treatment, respectively. It was determined that the grinding process reduced the oil absorption property of the DE.
Surface energy
Surface energy values of the materials used.

PP: polypropylene; RDE: raw diatomite; FC: fluorocarbon; SA: stearic acid.
MFI analysis of the PP/DE compounds
It is known in the literature that maleic anhydride was used for PP and silica composites as a compatibilizer [39–41]. Therefore, we used a maleic anhydride-grafted PP in addition to the DE surface treatment for achieving an enhanced compatibility between PP and DE.
MFI values of the PP/DE (90/10 w/w) compounds.

PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.
Thermal behavior of the PP/DE compounds and nonwovens
Figures 4 and 5 present the differential scanning calorimetry (DSC) thermograms of the raw DE containing PP compounds and nonwovens, representatively. Thermal behavior results of all compounds and nonwovens are given in Table 7. The neat PP has a melting temperature of 164.4℃, a crystallization temperature of 122.0℃, and a crystallinity value of 30.6%. Independent of the grinding process, surface treatment, and compatibilizer usage, addition of DE led to only a slight change in thermal behavior of the PP/DE (90/10 w/w) compounds. 2 wt.% DE containing nonwoven samples delivered similar results. But, the values were different from those of the compounds. Nonwoven samples exhibited slightly lower Tm and Tc values than those of the compounds. But, their degrees of crystallinity values were determined to be too low when compounds were considered, although DE incorporation changed them only slightly. Generally, it can be said that incorporation of DE into PP did not affect the thermal behavior of the compounds and nonwovens.
DSC thermograms of the PP/raw diatomite (90/10 w/w) compounds. PP: polypropylene; RDE: raw diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid. DSC thermograms of the PP/raw diatomite (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid. DSC data of the PP/DE (90/10 w/w) compounds. PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.


Morphology of the PP/DE compounds and nonwovens
Before going into details related to the analysis results of the DE containing PP meltblown composite structures, it should be pointed out that high torque values were recorded and rigid and brittle structures were obtained during meltblowing process, when the DE amount was adjusted to equal or higher than 2 wt.%. In addition, increasing the DE amount led to a distinctive separation of the PP and DE as if the DE was sprayed separately as a powder mass through the spinnerets of the meltblown equipment. It can be said that observation of high torque values is related with the DE loading amount rather than its particle size, since even the raw DE has the biggest particle size of 37 µm, while the die diameter of the meltblown equipment is 380 µm (Table 3). In order to verify that rigid and brittle structures were obtained, we performed an EDX analysis on the cross section of a 2 wt.% DE incorporated PP nonwoven, which is presented in Figure 6. It can be said that the elements Si and O belonging to DE accumulated more on the fiber periphery. Therefore, DE-embedded PP meltblown nonwovens presented a rigid and brittle structure since DE has a typical Mohs hardness of 5–6. To our knowledge, this phenomenon is the first observation in the literature and could be attributed to the incompatibility between PP and DE and to hot air flow with very high attenuation speeds, which is used for melt blowing of primary filaments into very fine fibers. Because of these problems mentioned above, we only generated the PP/DE nonwovens with the highest possible amount of 2 wt.% DE.
EDX analysis of the 2 wt.% raw diatomite containing PP nonwoven.
Scanning electron microscope (SEM) images of the raw DE containing PP compounds and nonwovens in Figures 7 and 8, respectively. The compounds and nonwovens with the ground DEs were not shown here. PP/DE compounds exhibited DE domains varying between 2 and 8 µm independent of DE surface treatment and compatibilizer (Figure 7). A distinctive observation of the DE particles was only achieved on the PP/RDEFC and PP/RDESAM nonwovens due to the low amount of DE. Generally, it can be said that the PP/DE nonwovens showed smaller DE particles with several micrometers (Figure 8). Considering the initial mean particle sizes of the raw and ground DEs (d50: 20.26 µm (RDE), 4.69 µm (GDE), and d90: 37.54 µm (RDE), 11.57 µm (GDE)), it is obvious that the DE particle size reduced to a large extent when processed with PP in melt stage. Besides, the neat PP nonwoven showed a fiber diameter ranging from 1 to 17 µm, while fiber diameter of the DE containing PP composite nonwovens changed from 1 to 32 µm indicating a fiber thickening, which will be discussed in pore size and void content analyses.
SEM images of the PP/raw diatomite (90/10 w/w) compounds. PP: polypropylene; RDE: raw diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid. SEM images of the PP/raw diatomite (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.

Pore size and void content of the PP/DE nonwovens
Minimum (d10), maximum (d97), mean (d50) flow pore diameter of the neat and 2 wt.% diatomite containing polypropylene meltblown composite structures.

PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.
Void content results of the nonwoven structures are also given in Table 8. Selected optical microscopy images (on the left) and corresponding software images (on the right) of the raw DE containing PP used for the void content calculations are displayed in Figures 9 and 10. The neat PP showed a void content of 1.2% ± 0.3%, while DE containing PP nonwoven structures exhibited higher void contents varying between 9.6% ± 1.2% and 52.0% ± 1.6%. Accordingly, addition of the DE increased interfiber voids. Void content calculations are based on background gray level of the images. As might be expected, thicker fibers lead to an increased background gray level due to having a lower surface coverage, which result in higher void content.
Optical microscopy (left) and software (right) images of the PP/raw diatomite (98/2 w/w) nonwovens without compatibilizer. PP: polypropylene; RDE: raw diatomite; FC: fluorocarbon; SA: stearic acid. Optical microscopy (left) and software (right) images of the PP/raw diatomite (98/2 w/w) nonwovens with compatibilizer. PP: polypropylene; RDE: raw diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.

Considering both the pore size and the void content analyses, it can be said that incorporating DE into PP resulted in thicker fibers.
Wettability and oil absorptivity of the PP/DE nonwovens
Contact angle measurements of the PP/DE nonwovens with oil were not able to be performed since oil droplet spread out quickly once it contacted the nonwoven surface due to oleophilic character of the PP.
Oil absorption property of the PP/DE composite nonwovens was tested with four oils. As shown in Figure 11, linseed oil absorption of the neat PP is very high, being 3068% ± 90%. But, addition of DE worsened the linseed oil absorption capacity to a large extent independent of the grinding, surface treatment, and compatibilizer usage. Individual samples such as PP/RDEFC, PP/GDE, PP/GDEFC, and PP/GDESAM presented relatively higher linseed oil absorption values between 1750% ± 108% and 1809% ± 8% within the DE containing samples. But, considered as a whole, they changed between 1299% ± 110% and 1570% ± 54%.
Linseed oil absorbency of the PP/DE (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.
The purpose of using linseed oil was to meet the standard’s requirement [35], although it is not an oil type, which is encountered in industrial oil spills. In order to observe the oil absorption behavior of the nonwovens under real conditions, we used three synthetic-based oils additionally, which have different kinematic viscosities. Rust inhibitor oil delivered the following results (Figure 12): The neat PP had an oil absorption value of 1184% ± 105%. Independent of the grinding, surface treatment, and compatibilizer usage, DE containing nonwovens showed reduced oil absorption capacities changing between 718% ± 78% and 1089% ± 136%. PP/RDEM and PP/RDEFC nonwovens presented the highest oil absorption values within the DE containing samples. Usage of hydraulic oil delivered similar results as follows (Figure 13): Oil absorption capacity of the neat PP nonwoven was determined to be 2566% ± 167%, and incorporation of DE reduced the oil absorption values to a large extent. The general trend did not change when diesel oil was used (Figure 14); The neat PP exhibited an oil absorption value of 2921% ± 86% and DE added nonwovens showed reduced oil absorption capacities between 1276% ± 24% and 1523% ± 42%. Paying closer attention to all oil absorption results, it is noticeable that the discrepancies among the values of the neat PP and PP/DE nonwovens increase. This observation could be explained with the viscosity values of the oils used. Increasing kinematic viscosity resulted in increased discrepancy among the oil absorption values of the neat PP and DE containing PP. Accordingly, addition of DE reduced the oil absorbency more adversely when more viscous oil was used.
Rust inhibitor oil absorbency of the PP/DE (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid. Hydraulic oil absorbency of the PP/DE (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid. Diesel oil absorbency of the PP/DE (98/2 w/w) nonwovens. PP: polypropylene; RDE: raw diatomite; GDE: ground diatomite; M: maleic anhydride; FC: fluorocarbon; SA: stearic acid.
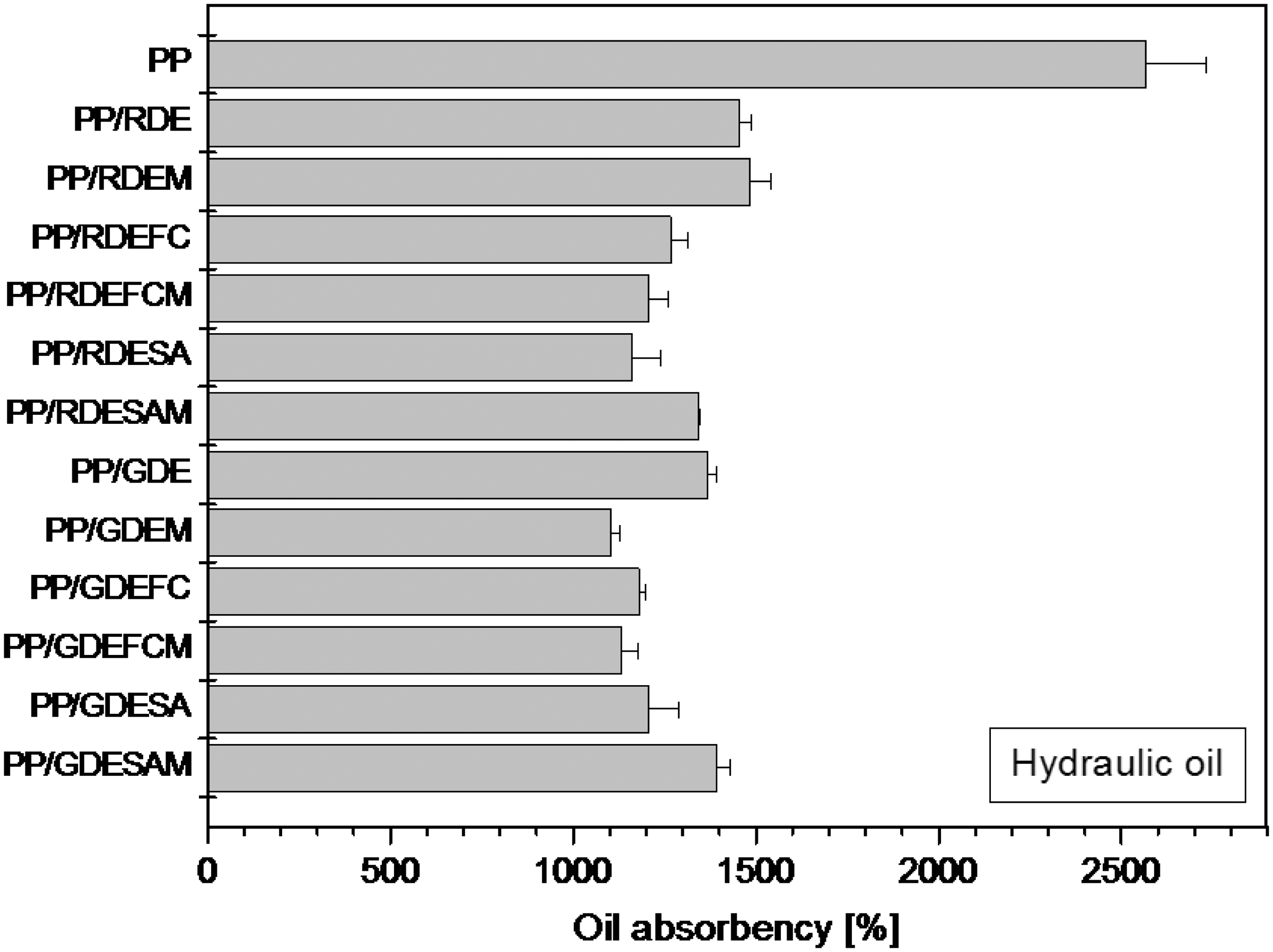

Reduction of oil absorbency in DE containing PP composite nonwovens could be attributed to the fiber structure, pore size, and void content. Incorporation of DE led to thicker fibers having a so-called rigid sheath layer on the fiber surface resulting in a rigid and brittle structure. This structure probably acted as a barrier and led to a substantial reduction in oil absorbency.
ANOVA test results for the polypropylene/diatomite (98/2 w/w) nonwoven structures.
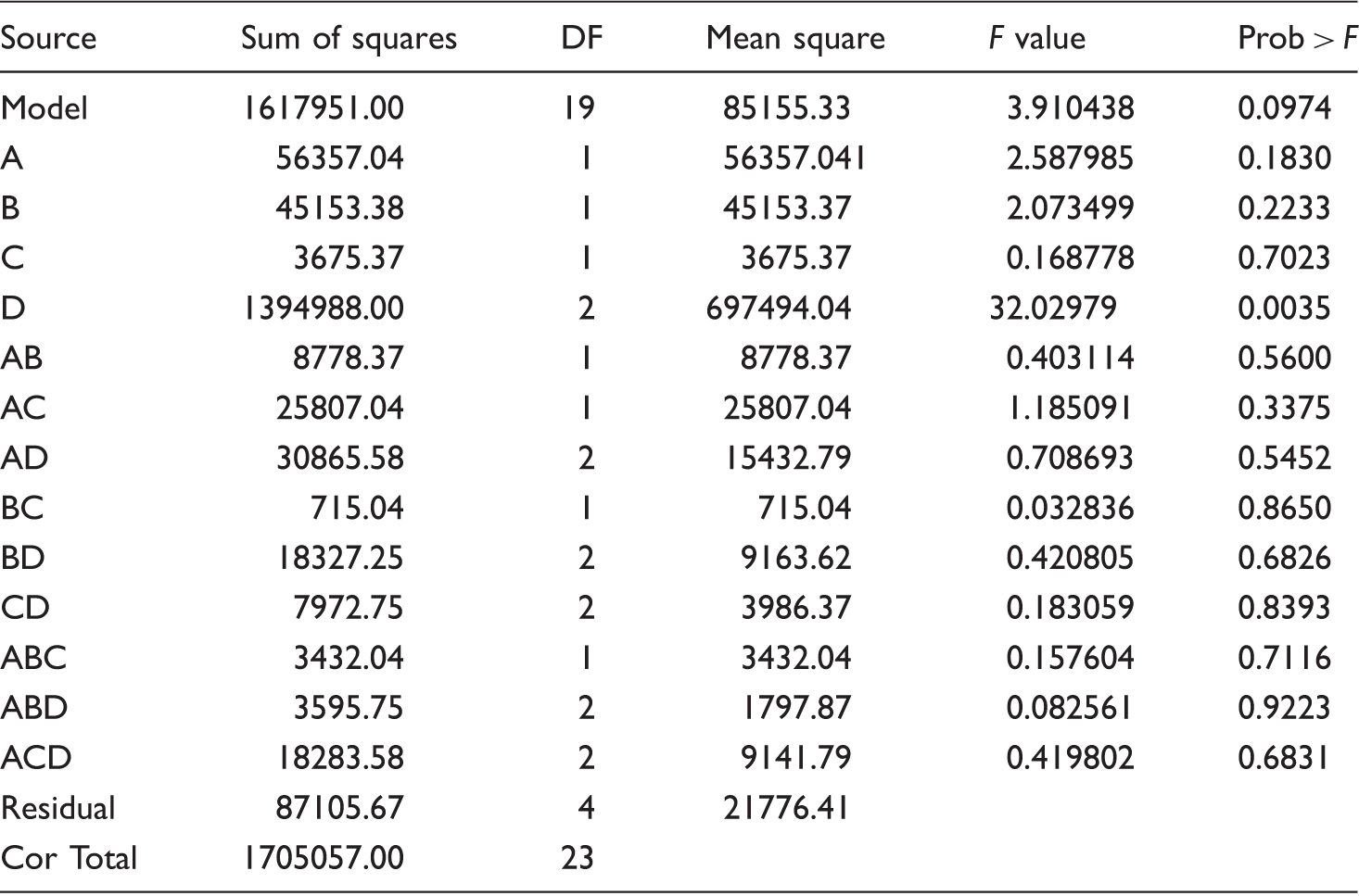
A: grinding; B: compatibilizer; C: stearic acid surface treatment; D: oil type.
ANOVA test results for the polypropylene/diatomite (98/2 w/w) nonwoven structures.

A: grinding; B: compatibilizer; C: fluorocarbon surface treatment; D: oil type.
Conclusions
It was determined that the porous structure of the DE disappeared completely in dry grinding while wet grinding destroyed the porous structure to a small extent. A total grinding time of more than 48 min did not change particle size distribution of the DE noticeably.
Application of a grinding process, a surface treatment, and usage of a compatibilizer was revealed to be dispensable for DE containing PP composite nonwoven structures since DE particle size already reduced during melt compounding and melt blowing processes to a large extent and more than 2 wt.% DE could not be embedded to the PP nonwoven.
It was shown that the addition of DE led to increased fiber diameters due to adversely affecting the melt blowing process and a rigid and brittle structure by DE accumulation on the PP surface. As a consequence, DE on the surface acted as a barrier and worsened the oil absorbency of meltblown nonwoven structures. Reduction in oil absorbency was more distinctive when more viscous oil was used. ANOVA tests delivered statistically insignificant differences in terms of oil absorbency property independent of grinding, surface treatment, compatibilizer usage, and oil type used.
Footnotes
Acknowledgments
The authors would like to thank Mr. Tuncay Saka from Organik Kimya A.Ş. for providing fluorocarbon chemical, Mr. Tayfun Tümer from Mikron’s A.Ş. for providing the coating facility Cyclomix, Ms. Menekşe Sarıhan from ERNAM (Erciyes University Nanotechnology Research Center) for SEM analysis, and Mr. Serkan Göğüş from Mogul Tekstil Sanayi ve Ticaret Ltd Sti. for pore size measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) (Project Number: 113M512).
