Abstract
Conducting polyaniline-coated polyester textiles have attracted much attention due to their wide range of surface resistivity. The surface resistivity depends strongly on the polymerization condition. In this work, polyaniline coated on textile by in situ chemical polymerization method. The influence of substrate modification, synthesis condition, and especially redoping procedure on the surface resistivity of polyaniline-coated textile was investigated and an optimum condition specified. The obtained results indicate that polymerization in an ice-water bath and in a monomer:oxidant:HCl molar ratio equal to 1:1:7 for about 4–6 h decreases the surface resistivity significantly. Furthermore, the experimental results show that etching the surface of textile and redoping procedure with concentrated HCl vapor can decrease the surface resistivity, in a short time.
Introduction
Conducting polymers are a group of synthetic materials that can be synthesized by various methods. Polymer’s properties depend on the production method. Chemical and electrochemical polymerization are the most common and efficient ways to synthesize these polymers [1].
Today conducting polymers and their nanocomposites due to their variable electrical conductivity are very interesting and have been used as bio [2], gas [3,4] and chemical [5,6] sensors, solar cell [7,8], organic light-emitting diode [9,10], electromagnetic interference shielding [11–13], corrosion protection of metal [14,15], antistatic and static dissipative layer [16,17], etc.
Polyaniline (PANI) is one of conducting polymers that is low cost, easily synthesizable that has good environmental stability, and different oxidation forms with wide range of conductivities from insulating to metallic regime [13,18–21].
Along the importance of conducting polymers and coating on various types of substrates, we can produce usable, low cost, lightweight, flexible, and antistatic-coated products by applying textile as substrate.
Due to specific physics of textiles, special coating methods should be used. The most common way to synthesize conducting polymer-coated textiles is in situ chemical polymerization. In situ chemical polymerization of PANI on textiles was first reported by Milliken Research Corporation in 1989 [22]. The other methods are electrochemical and admicellar polymerization of different kinds of conducting polymers on cotton, nylon, and polyester fabrics as substrate [16,20,23].
Parvinzadeh Gashti et al. used UV irradiation to coat wool with polypyrrole–silver nanocomposite [24]. In another work, polypyrrole–silver nanocomposite was coated on cotton fabrics by in situ chemical oxidative polymerization [25]. Conductive surface on fabrics can be obtained by knife-over-roll coating method by using liquid dispersion of conducting polymer [26,27].
In the present work, we use a simple and efficient method to produce PANI-coated polyester textile with a wide range of surface resistivities in an acidic medium.
In order to obtain minimum surface resistivity, we study the influence of polymerization condition like polymerization time and temperature, monomer-to-oxidant ratio, dopant concentration, redoping agent types, and etching the surface of substrate. The mentioned conditions influence the conductivity of samples, because of their effective role in chain formation, morphology, crystallization, adherence, and uniformity of PANI layer [1].
The effect of some polymerization condition on the surface resistivity of PANI-coated fibers and textile has been investigated by some research groups [23,28–31]. However, the effect of surface etching and different redoping agents on the surface resistivity has not been reported yet.
Finally, we propose the optimum synthesis condition to achieve highly conductive PANI-coated polyester textile.
Experimental procedure
Materials and method
HCl (37%), H2SO4 (98%), H2O2, ammonium peroxydisulfate (APS), and toluene-4-sulfonic acid (PTSA, monohydrate) were purchased from Merck Co. of analytical grade and used without further purification. Aniline monomer was double distilled under vacuum before use.
In this research, we coated polyester substrates with PANI by in situ chemical polymerization method to produce low cost, light, and easy shaping conducting textile with a more simple method that can be used industrially.
Typical coating procedure contains three steps as follows:
Preparation of substrate
At first, substrates were washed with distilled water several times and prepared for surface modification via etching.
Etching method
The etching bath consists of 8 vol% HCl, 8 vol% H2SO4, and 4 vol% H2O2. The substrate was immersed in a 45℃ etching bath for 90 s and then thoroughly washed with distilled water. To remove surface abrasives and ions, the substrate was plunged in 20 vol%
Polymerization
As a typical method of preparing PANI, 2 ml of aniline monomer was dissolved in 100 ml aqueous HCl solution and 5 g of APS as oxidant agent was dissolved in 50 ml distilled water and were stirred separately for 1 h. The concentration of HCl was determined with respect to the monomer molar ratio.
To diffuse monomer to the polyester fibers, the substrates were soaked in monomer solution for about 90 min.
After that in an ice-water bath (2 ± 1℃), oxidant solution was added gradually to the cooled monomer solution containing substrate, during 5 min under mild stirring. Then the mixture was left 1–8 h at rest to complete the polymerization. During polymerization process, color and viscosity of the solution were changed. Finally, PANI-coated textile was brought out and washed with distilled water to remove additional surface precipitates. In this step, the coated textiles are dark green.
The resulting precipitate (PANI powder) was vacuum filtered and washed with distilled water and ethanol several times to remove unreacted materials and finally PANI-coated textile and powders were dried at 50℃.
Redoping
The resulting coated textiles were redoped with different types of redoping agents, i.e. concentrated HCl vapors (for 5 min), 1 M HCl, and 1 M PTSA solutions (for 24 h) at room temperature. The effect of redoping agents on the surface resistivity of the coated textiles was investigated.
To understand the effect of etching and synthesis temperature, some of the samples were prepared without etching or at room temperature.
Instrumental analysis and measurements
The UV–Vis absorption measurements were carried out with a Jasco model 7800 double beam spectrophotometer with 1 cm quarts cuvettes. The images were recorded using the TESCAN SEM system. The operating voltage of the system was 15 kV. Fourier transform infrared (FTIR) measurements (in ATR) were recorded on a Brucker spectrometer.
According to the special physics of textile such as spaces between fibers and the cylindrical shape of coated fibers, electrical parameter measurements should be done via special methods. The surface resistivity of samples was measured by using a homemade setup based on the American Association of Textile Chemists and Colorists test method 76-1995 at room temperature [32]. The resistance (R) between two electrodes of spacing (l) and width (w) was measured by a digital multimeter (APPA 305) and then using R = Rs l/w the surface resistivity (Rs) was calculated in ohm/sq.
Results and discussion
Surface structure of coated textiles
SEM images of PANI-coated polyester fiber and textiles are shown in Figure 1.
SEM images of PANI-coated (a) polyester fiber, (b) and (c) polyester textiles surface.
As shown in Figure 1, textile was coated with PANI so completely and uniformly without any defects on the surface.
The effect of etching
The substrate was chemically etched in acidic media for cleaning and increasing absorption of monomer on the surface and diffusion of monomer into fibers. It is a common procedure in plastic plating in order to increase catalyst absorption. Inserting substrate into the etching bath that contains oxidative agents produces some microscopic holes on the surface and hence increase the mechanical adherence of the substrate. Etching solution can also make the polyester fiber hydrophilic, and the hydrogen atoms of some of the hydrocarbon molecules in the plastic are believed to be replaced by –OH groups. The attachment of the –OH groups to the surface molecules makes the plastic surface hydrophilic [33].
Figure 2 shows decreasing the contact angle for water droplet from 100° to 84° that confirms the hydrophilicity of polyester surface after etching.
(a) Pure polyester textile, (b) etched polyester textile.
As shown in Figure 3 the smooth surface of polyester textile becomes rough through etching procedure.
(a) Pristine polyester textile, (b) etched polyester textile.
The effect of etching on the surface resistivity of samples that were prepared under different polymerization time and HCl ratio and redoped with concentrated HCl vapor for 5 min is shown in Figure 4. The results confirm the effective rule of surface modification in order to absorption of monomer with respect to unprepared surfaces and improving the conductivity of the coated textile.
Surface resistivity of different polymerization time and monomer:oxidant:HCl molar ratios in an ice-water bath. WET: with etching procedure; WOET: without etching procedure.
The effect of temperature
Synthesis temperature is one of the most important parameters determining electrical conductivity of conducting polymers. Compared to room temperature synthesis, slow polymerization in an ice-water bath allows better polymerization inside the fabrics, orderly growth on the surface, and smaller defects in the structure and finally improves the electrical conductivity. As a result of agglomeration of polymer on the surface due to faster polymerization at higher temperature, the surface resistivity increased and the faster saturation occurred in this case. The effect of synthesis temperature on the surface resistivity of samples with 7 and 9 HCl molar ratios and concentrated HCl vapor as redoping agent is shown in Figure 5. It is obvious that corresponding to the previous works [31,34,35], in all cases the surface resistivity has increased by increasing the synthesis temperature and polymerization at an ice-water bath brings low surface resistivity coated textile.
Surface resistivities in different polymerization temperatures.
The effect of polymerization time
Polymerization time is a key factor affecting the conductivity of fabrics. Monomers should have enough time for polymerization and taking long chain with high molecular weight and conductivity.
By increasing the polymerization time, most of monomers available on the surface of fibers as well as those which are diffused in the fibers are polymerized and hence the conductivity of samples enhances. However, by increasing the polymerization time beyond an optimum value, the surface resistivity of coated textile saturates or even increases. It is due to saturation of polymerization on the surface and breaking some of polymer chains according to hydrolysis in reacting media [36].
The optimum polymerization time depends on the synthesis conditions. As shown in Figure 4, at an ice-water bath, for 1 and 3 HCl molar ratios, the optimum polymerization time is 6 h while in presence of higher dopant agent ratios it is about 4 h.
The thickness of PANI layer on the textile fibers depends on the polymerization time and it was approximately less than 2 µm in this work.
The effect of monomer-to-oxidant ratio
The ratio of monomer to oxidant is another important factor that is studied here. Theoretically, the molar ratio of APS oxidant to aniline monomer in chemical polymerization in an aqueous solution was determined by Gregory as 1.25 [37]. It means that each aniline molecule consumes 2.5 electrons by an average for oxidation and formation of emeraldine base. In this study, we have examined 0.5, 1, and 1.25 APS:aniline molar ratios for different HCl ratios and polymerization times. All the samples were redoped with concentrated HCl vapor. In Figure 6, the surface resistivity of samples with different monomer:oxidant:HCl molar ratios is shown. As it is evident, to obtain the lowest surface resistivity for PANI-coated polyester textile, the monomer:oxidant molar ratio should be equal to 1.
Surface resistivity of samples with different monomer:oxidant:HCl molar ratios.
The effect of redoping agent
The fundamental mechanism of doping is transferring charges between organic polymer and dopant due to oxidation or reduction. The produced charges can move along the polymer chain and hence make it conductive. Based on dopant type and degree of doping, the conductivity of polymers can vary from that of insulators to metals [1,30].
Surface resistivity of samples with different redoping agents (ohm/sq).

Monomer:oxidant:HCl molar ratio.
The table data implies that using suitable HCl molar ratio as doping agent and redoping procedure can decrease the surface resistivity by about two orders of magnitude. Figure 6 also confirms our results which will be illustrated later. Finally, we propose concentrated HCl vapor as an effective and economical way for fast decrement of the surface resistivity.
It should be noted that the surface resistivity of all samples was measured five times and the average value was used in this work.
FTIR spectra
FTIR spectroscopy measurements (in ATR) were carried out using a Brucker spectrometer.
The FTIR spectra of pure and PANI-coated polyester textile are shown in Figure 7.
FTIR spectra of (a) pure polyester and (b) PANI-coated polyester.
In the spectra, the band observed at 1713 cm−1 belongs to the carbonyl groups of ester. The bands at 1457 and 1575 cm−1 in Figure 7(b) correspond to C–N and C = N groups in the benzenoid and quinoid structures in PANI, respectively [20]. The results confirm the presence of PANI in the structure.
UV–Vis absorption spectra
Figure 8 shows the UV–Vis absorption spectra of synthesized PANI powder and redoped PANI powder dissolved in dimethyl formamide (DMF) solvent. As shown in Figure 8, the spectra show three characteristic absorption peaks in the wavelength regions 250–320, 320–450, and 450–750 nm due to UV–Vis spectroscopy of PANI and redoped PANI samples.
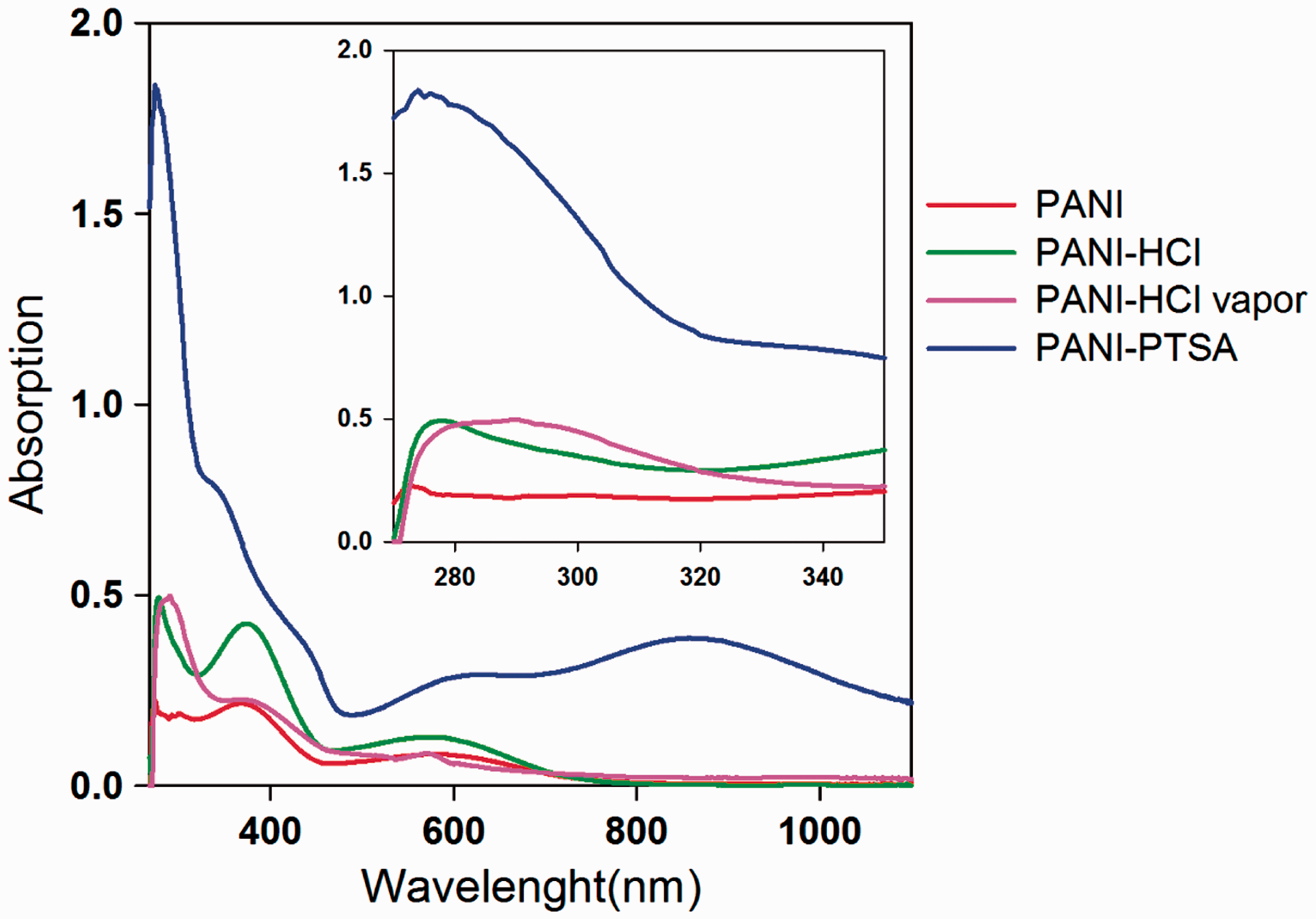
where h is the Planck’s constant, c is the speed of light, and λmax is the wavelength of the first absorption peak [30]. More conductive samples have lower band gap and therefore higher λmax. As shown in Figure 8, redoping procedure moves the characteristic peak to the higher wavelengths, and due to decrease in the band gap energy, the conductivity increases. The figure also implies that the band gap energy of samples is in the order of PANI> PANI-PTSA> PANI-HCl> PANI-HCl vapor and consequently PANI that was redoped with concentrated HCl vapor has the highest conductivity. The higher absorption in PANI-PTSA is related to great solubility of PANI redoped with organic acid in DMF. These results are well compatible with those obtained by measuring the surface resistivity and shown in Table 1.
Conclusions
In this research, most of effective synthesis conditions on the surface resistivity of polyester textile have been examined. We found that in addition to effective parameters that were studied by other research groups, etching the textile surface and redoping procedure after the polymerization have important impacts on reduction of the surface resistivity.
Etching in an acidic media can modify the surface of polyester textile by attachment of the –OH groups to the surface molecules and make the surface hydrophilic for better monomer absorption and polymerization. Redoping the PANI-coated polyester textile with different agents affects intensely the surface resistivity and can decrease it up to 100 times. It was also found that among the surveyed redoping agents, concentrated HCl vapor is the most effective one to decrease the surface resistivity of samples.
In conclusion, to obtain a low surface resistivity PANI-coated polyester textile, the polymerization should be done at a low temperature (ice-water bath), in a medium with monomer:oxidant:HCl molar ratio of 1:1:7 for about 4–6 h. By etching the substrate and redoping about 5 min by concentrated HCl vapor, we obtained surface resistivity as low as 467 (Ω□−1). Using concentrated HCl vapor as a new redoping agent, we proposed a simple way for immediate decrement of the surface resistivity of PANI-coated polyester textiles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
