Abstract
The flame retardant functionality was imparted in cellulosic textile using banana pseudostem sap, an eco-friendly waste plant product. The extracted sap was applied in three different pH conditions, viz. acidic, neutral and alkaline to the pre-mordanted bleached and mercerized cotton fabrics. Flame retardant characteristics of both the control and the treated fabrics were analysed in terms of limiting oxygen index, vertical flammability and heat release related properties using a cone calorimeter. The thermal degradation and pyrolysis mechanism were studied using thermogravimetric analysis and differential scanning calorimetry. The elemental analysis was carried out with X-ray fluorescence, and the quantification of phosphorus and nitrogen was also done. Besides, the charring mechanism of both the control and the treated fabric was analysed in detail, and a char structure model has been proposed. The treated fabrics were also evaluated in terms of colour and other physical properties.
Introduction
Cellulosic cotton textile catches flame readily that is quite difficult to extinguish. This poses a serious risk to health and life of a living being and damages potential to textile products. Significant efforts have been made in the past to improve the flame retardant property of cotton textiles using various synthetic chemicals, and many of them are available in the market. The most simple and common chemical is borax and boric acid mixture [1]. Phosphorus-based flame retardants along with nitrogenous compound are the most effective formulation reported so far, due to their synergistic effect. Consequently, for the past 50 years, flame retardants based on the composition of phosphorus, nitrogen and halogen, such as Tetrakis phosphonium salt and N-alkyl phosphopropionamide derivatives, are widely dominating the commercial scenario [2]. However, as such formulations need to be applied in an acidic condition, the cotton fabric loses its tensile strength and become stiffer. Besides, such a treatment is expensive, non-eco-friendly due to the involvement of a large amount of chemicals, high temperature curing process and for toxic formaldehyde emission during treatment [3]. Although antimony in combination with halogen could impart good flame retardant property, it is still not very successful due to the negative impact of halogen compounds in the environment [2]. Although some intumescent-based flame retardants, such as silicate [4], composition of the nano zinc-oxide and polycarboxylic acid [5], have been developed to make the process environmentally friendly, such treatment cannot satisfy the handle, strength and the fire resistant durability requirements of the fabric. Recently, researchers have also used plasma treatment with various polymerization gases to impart the fire retardancy property to the cellulosic fabric. Plasma process is water free and eco-friendly, but it is very costlier and the imparted flame retardant property is not durable to washing and ageing [6]. Hence, there is a need to develop more cost-effective, environmentally friendly and sustainable fire retardant products, which when applied to cotton fabrics will maintain its quality and flame retardant durability to a great extent. This apart, the increased awareness on human health and hygiene in recent years is also driving the demand of those cellulosic textiles, which are finished with natural products, such as natural dyes for colouration, enzyme for bio-polishing, neem and aloe vera extract for antimicrobial finishing [7,8]. A very few researches have so far been reported on imparting fire retardancy to cellulosic fabric using natural products [9–11]. Recently, the researchers have reported that DNA from herring sperm and solomon fishes can be applied to the cotton fabric to make it thermally stable [9]. Attempts have also been made to impart fire retardant to cotton fabrics with whey proteins, casein and hydrophobins due to their phosphate, disulphide and protein content, as they can influence the pyrolysis by an early char formation [10, 11]. However, a limited application of waste plant bio-molecules has been made for imparting flame retardant finishing to any textile and/or polymeric material till date to the best of our knowledge. As some of the plants contain phosphorus, silicate and other minerals and mineral salts, they offer immense potential to be utilized to impart flame retardancy to cellulosic and non-cellulosic textiles. In our earlier publications, we have reported a detailed study of imparting fire retardant to cellulosic cotton fabric, using spinach juice [12] and wastage banana pseudostem sap (BPS) [13]. BPS is a 100% pure liquid extracted from the pseudostem of banana plants. It is stable up to a wide range of pH. However, it turns to colourless at an acidic pH, at alkaline pH it remains with khaki colour. When BPS was applied on the mordanted cotton fabric at alkaline pH, treated fabric showed uniform khaki colour and BPS treatment does not appear as a stain on the fabric. Our previous publication [13] provided information on the application of BPS in alkaline condition to the cotton fabric and data on the fire retardant durability and the elemental composition of sap and treated fabric by Energy dispersive X-ray (EDX) and Secondary ion mass spectrometry (SIMS) analysis. It showed the presence of sodium, magnesium, silicate, iron, proteinaceous nitrogen, nitrate, organophosphate and other metallic constituents in the BPS responsible for imparting fire retardant property. Further, as far as the wash durability is concerned, oxygen index value of the BPS-treated fabric reduced from 30 to 24 after single ISO 2 washing. As the wash durability of the end product is low, it can be used only for the textiles which are not washed regularly. In the present study, an attempt has been made to apply the BPS in different pH, and the fire retardant properties, heat, temperature generation and pyrolysis mechanism have also been assessed in detail by multiple thermal characterization techniques. Besides, the charring mechanism has been proposed in detail, whichalso provides knowledge regarding the total phosphorus, nitrogen content of the treated fabric, colour and strength parameters. This developed process could use flame retardant finishing of home furnishing products such as home window curtain, railway curtain, hospital curtain, table lamp and as a covering material of non-permanent structure, such as in book fair, festival and religious purpose, where large quantity of textile is used and has chance of fire hazards. BPS is abundantly available in India as well as in other countries, and it is normally considered as waste material. It is eco-friendly and produced from renewable source. Therefore, the application of BPS in cotton textile for colouration and functionalization will give the advantages of value addition using natural product.
Materials and methods
Material and BPS application
A 200 GSM (areal density) plain woven bleach cotton fabric of 30 EPI (ends/inch) and 40 PPI (picks/inch) was procured from the local market and was used for the flame retardant finishing. BPS was supplied by Navsari Agricultural University, Gujarat, India, for the treatment. Supplied BPS was characterized by atomic absorption spectroscopy analysis, which showed the presence of 0.2 ppm phosphorus, 7.4 ppm potassium (outside the range limit of the machine), 0.9 ppm magnesium, 4.5 ppm manganese and 4.9 ppm iron. The supplied sap had a pH of 7.5 (neutral), which was made both alkaline (pH 10) and also acidic (pH 5) with the addition of soda ash and acetic acid, respectively. The bleached cotton fabric was first mordanted with 5% tannic acid and 10% alum. Thereafter, the mordanted fabrics were impregnated separately in the BPS solution of three different pH, viz. acidic, neutral and alkaline, maintaining material to liquor ratio of 1:10. The fabric was then treated for 30 min followed by drying at 110℃ for 5 min.
Determination of percent add-on
Before any physical and chemical characterization, both the treated and the control cotton fabrics were conditioned at 65% RH and 27℃ for 48 h. After the application of BPS on cellulosic cotton textile, the add-on, i. e. the increase in sample weight, was determined by gravimetric principle from the bone dry weights of the sample before and after the treatments, and the results have been expressed in percentage over the initial weight of the sample.
Thermal characterization
Flammability assessment
The burning behaviour of both the control and the treated samples was evaluated by standard methods. For the limiting oxygen index (LOI) analysis, IS 13501 test method was used. For LOI analysis, sample size was 140 × 52 mm, with an ignition time of 30 s was maintained as per BIS method [14] which is equivalent to ASTMD 2863-13 test method. In vertical flammability, the different parameters were measured as per IS: 1871 method A (equivalent to the ASTM D6413), where a fabric sample was ignited by 38 mm flame for 12 s [15]. The maximum temperature produced during the burning of a sample was measured using infrared thermometer, Fisher Scientific made (Model No. 15077968 FB61354 225PE) in non-contact mode. Based on the data, the temperature generation profile curve was plotted for both the control and the treated fabric.
Cone calorimeter analysis of treated fabric
The cone calorimeter available at the Laboratory of Ahmedabad Textile Industries Research Association, India, was used to test the 100 × 100 mm cellulosic cotton specimens in accordance with the procedure of ASTM international test method, E 1354-10 a. The specimens were tested at horizontal orientation with heat flux of 35 kW/m2. Three replicates of each sample of 2 g weight were tested. Before testing, all the samples were conditioned at 65% RH and 27℃. The data recorded included those specified in the ISO5660-1 standard. The parameters reported in the paper included the following measurements, consisting of total heat release (MJ/m2), effective heat of combustion (MJ/kg), heat release rate (kW/m2), average mass loss rate for the period of 10–90% mass loss (g/s-m2), average mass loss rate (g/m2s), specific extinction area (m2/kg), total smoke production (m2/m2), carbon monoxide and carbon dioxide generated (kg/kg) during the burning.
Thermogravimetric analysis in N2 atmosphere
Thermogravimetric (TG) curves of both the control and the treated fabrics were obtained by using a Thermo Gravimetric Analyser, METTLER TOLEDO TG-50/MT5 at a heating rate of 10℃/min at N2 atmosphere.
Differential scanning calorimetry analysis in N2 atmosphere
The differential scanning calorimetry (DSC) of both the control and the BPS-treated cotton fabric was measured by using METTLER TOLEDO DSC30 thermal analyser, where the temperature was maintained at 10℃/min.
Scanning electron microscope analysis
The char analysis of both the control and the treated samples was analysed using scanning electron microscope (SEM) PHILIPS- XL30. The samples were coated with a thin layer of conducting material (gold/palladium) by using a sputter coater, and the same were examined under the SEM with an accelerating voltage of 12 kV.
X-ray fluorescence analysis
X-ray fluorescence (XRF) analysers from Bruker were used to qualify and quantify the presence of elemental substituents in the material. Here, BPS has been dried first and then used for the evaluation of elemental analysis of BPS.
P and N content analysis
The amount of Phosphorous (P) in the treated fabrics was measured according to the soil chemical analysis method by using stannous chloride [16] and the amount of nitrogen (N) by Kjeldahl method [17]. As far the phosphorus analysis is concerned, one stock solution was prepared by potassium dihydrogen phosphate and sulphuric acid. Besides, a specific weight of treated cotton fabric dissolved in the mixture of sulphuric acid, nitric acid and perchloric acid. Then, Chlor molybdic acid was added in different concentration of stock solutions and also in the solution containing the above dissolved fabric. All the solutions are reduced by stannous chloride. Then, a curve of the absorbance, vis-à-vis the wavelength, was plotted, from which the concentration of phosphorus was determined.
As far as the N2 analysis is concerned, initially 0.3 g fabric sample mixed with 10 ml 98% concentrated H2SO4 and 3 g catalyst (1 part CuSO4 + 5 part K2SO4). The mixture was digested at 420℃ for 2 h till it reduced to a clear solution. Then, mixture was then cooled in room temperature and thereafter mixed with alkali and boiled. During the boiling, the released NH3 was trapped in boric acid, which was back titrated against 0.1 N HCl. The concentration noted in the titration was taken as a measure of nitrogen.
Colour parameters
It was observed that after application of the BPS in the cotton textile, its colour changed from white to khaki at alkaline pH and turned to colourless at acidic pH. Here, colour parameters such as K/S, L.a and b were measured using a Perkin–Elmer double-beam spectrophotometer, Lambda (35 model) equipped with an integrating sphere. The colour depth of the BPS-treated fabric was determined in terms of K/S from the reflectance data using the Kubelka–Munk equation as follows
Strength parameters
Tensile and tear strength were evaluated according to the ASTM D5035 and ASTM D1004, respectively.
Results and discussion
LOI and vertical flammability
Flammability parameters of the control only mordanted and mordanted followed BPS-treated cotton fabrics.

Total burning time = Burning with flame time + burning with afterglow time (after flame stopped).
Sample catches flame after 6–7 s of continuous contact with flame.
Being pure cellulosic in nature, the cotton fabric showed a lower LOI value of 18 that catches flame readily with flashing. It was observed from Table 1 that there was no change in the LOI value of the mordanted sample. After application of BPS at different pH, the LOI value of cotton textile was found to increase. Regarding the mordanted fabric treated with acidic BPS solution (pH 5), the LOI value was found to increase to 28, which is almost 1.5 times higher than that of the control sample. As the pH shifted towards the neutral, the LOI also remained unchanged like in the acidic condition with a value of 28. As far as the alkaline pH (pH 10) was concerned, the LOI value increased to 30, and in the vertical flammability measurement, the fabric showed flame for only 4 s and then self-extinguished. The sample took 900 s for the complete burning, whereas the control sample caught the flame readily in 1 s and burnt fully within 60 s. The fabric C showed a slow thermal decomposition without any flame, and the amount of temperature generated during the thermal decomposition was also less (200℃). This will required a serious hazard to happen several hours. It may be the presence of alkali in the treatment formulation had provided more thermal stability and also helped to pick up more amount (5% add-on) of the BPS in the treated sample compared to the fabrics A and B. To represent the vertical burning phenomena of the BPS-treated sample C, the control sample D, we have proposed a model presented at Figure 1. As far as the vertical flammability of the sample A and B is concerned, they catch flame quickly than fabric C. However, all the samples take more than 4 min for the complete burning. Another interesting phenomenon was observed that although the LOI value of fabric C is higher than those of the samples A and B, the extent of afterglow, smouldering and smoke formation in fabric C was more than that noted with the sample A and B. We have also tried the BPS treatment without mordant at different pH on the cotton fabric; however, they showed lower add-on (1–2.3%) and lower oxygen index value of 23–25.
Vertical burning model of the control (D) and the BPS-treated fabric (C).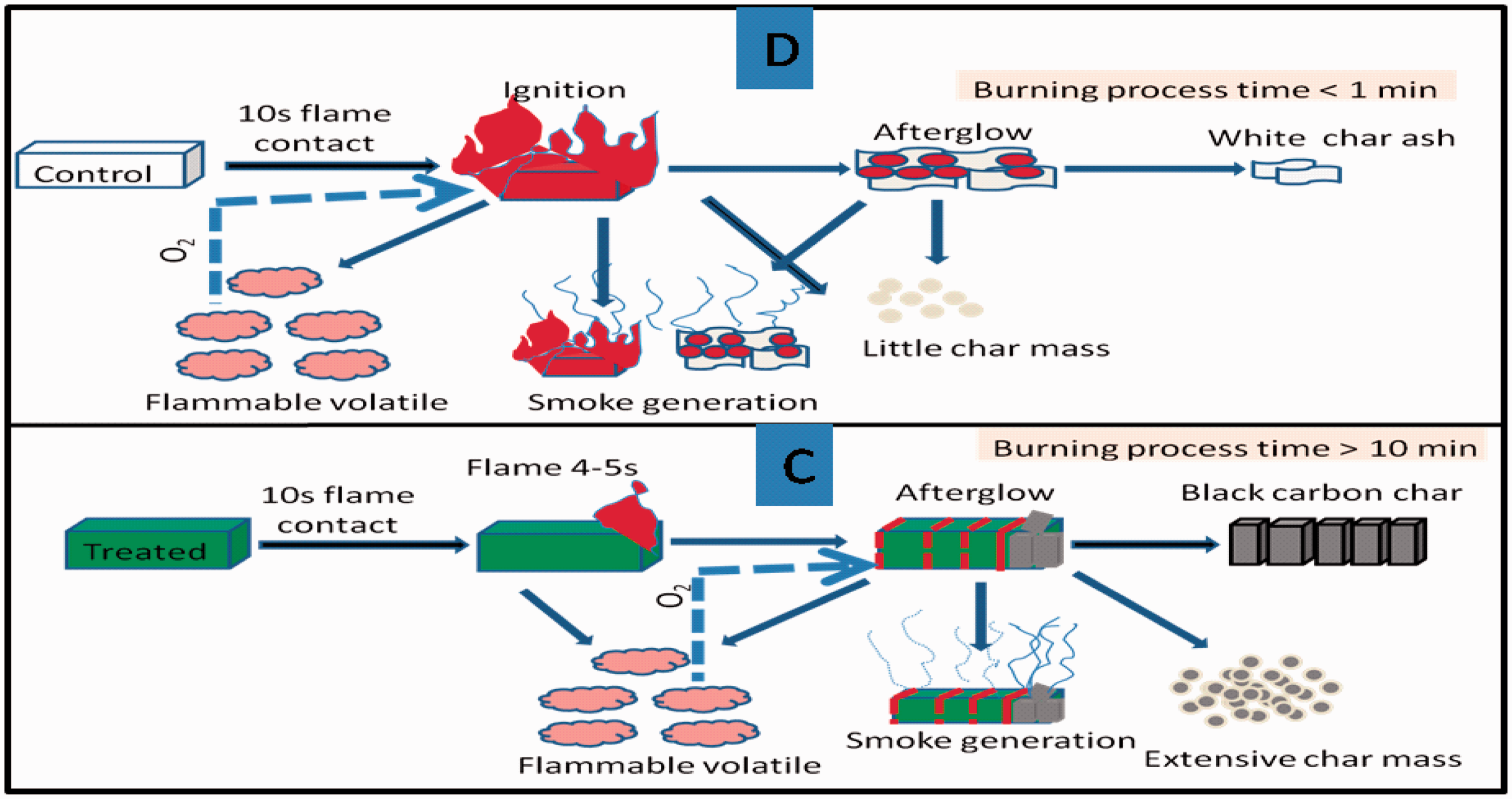
Thermogravimetric analysis
Figure 2 represents the thermal stability curves of the control and the BPS-treated (at different pH) cellulosic cotton fabric in N2 atmosphere. Curve D showed a sharp fall at 350℃, signifying cellulosic degradation, i.e. the pyrolysis occurred at that temperature. The amount of char remained after the pyrolysis is also very less. In contrast, TG and derivative thermogravimetric (DTG) curves of only BPS showed steep humps at temperature below 100℃ represent the evaporation of moisture from the extracted BPS. After that main degradation had occurred at 200℃, so that less flammable gas was produced. With addition the amount of carbonaceous char remained at high temperature 500℃ is also 38%. It may be due to the presence of more inorganics (such as sodium, potassium, silicon, magnesium, iron) in the extracted BPS [13]. Connected to it, curve A and C (BPS-treated cotton fabric) showed initial humps at below 100℃, which may be due to the evaporation of moisture [18]. DTG of curve A showed an earlier pyrolysis at 310℃ (shifted 40° from control), which signifies that the BPS treatment has aided to dehydrate the cellulosic fraction of the fabric earlier, so that the less flammable gas will be evolved and also, the amount of the char mass remained at higher temperature will be more compared to the control one. As far as the DTG of the curve C is concerned, it has also showed an early cellulosic degradation like curve A. However here, the amount of char mass production is more compared to the curve A. In addition, mass loss rate is also lower compared to the curve A. Both of these phenomena proved the fact that the alkaline pH BPS-treated cotton fabric is thermally more stable compared to the acidic pH BPS-treated cotton fabric. It may be due to more pick up of the BPS by the cotton fabric at alkaline PH. The additional alkali used during the treatment might have helped to improve the thermal stability of the fabric [19]. The char production observed from the TG curves can also be correlated with the real life vertical burning pictures as shown in Figure 2. Curve D and the vertical burning series D showed negligible amount of residual char after complete burning, whereas the vertical burning series C showed more amount of residual carbonaceous char after the burning, which can be correlated with the TG and DTG curve of C.
Thermogravimetric analysis plots of control (D), BPS-treated cotton fabric in acidic pH (A), alkaline pH (C) and only BPS. BPS: banana pseudostem sap.
DSC analysis
The TG analysis has showed an early pyrolysis and extensive char formation of the BPS-treated fabrics. To support the same, DSC analysis was also carried out for both the control and the treated cotton fabric (Figure 3). It can be observed from the figure that initially at below 100℃, both the control and the treated fabric showed steep endotherm corresponding to the evaporation of water from the fabric samples [18]. It was also observed from the figure that curve A exhibited an exotherm with the peak at 374℃ [20, 21]. On the other hand, curve B showed an exothermic peak observed at 340℃. It means that similar to that in the TG analysis, the exothermic peak of BPS-treated cellulose B, which was also accompanied by an endothermic shoulder, shifted towards lower temperature (peak shifted by 34℃) compared to the control one (A). The same phenomenon was also observed in the DSC curve of cellulosic fabric finished with different fire retardant agent [20].
DSC analysis of the control cotton fabric (A) and the alkaline pH BPS-treated (B) fabric.
Temperature generation analysis
As far as the vertical flammability is concerned, we have reported that the alkaline BPS-treated fabric showed flame for 4 s and then self-extinguished, and the observed afterglow burnt the total fabric in a much slower rate compared to the control fabric. Here, the temperature data have been noted during flaming as well as during afterglow. Associated with it, Figure 4 showed the temperature generation curves of the control and the BPS-treated fabric during vertical burning of the samples in the real life situation. In case of the control cotton fabric, the initial temperature generation was high around 400–450℃ as the fabric was burnt with flame and flashed, whereas in case of the treated fabric, only 300℃ temperature was generated during the first 20 s of ignition due to the presence of the flame for 4 s. In the second phase, the control fabric showed an afterglow for 30 s with a temperature around 350℃. Thereafter, the temperature of the fabric was gradually dropped due to the complete damage of the fabric and ash formation after burning. In contrast, the BPS-treated fabric showed a very slow rate of damage and a decrease in temperature of afterglow with the increase in burning time. This less amount of temperature generation is very much beneficial for the end users in practical real life situation, as it will generate less heat and fire hazards. The less temperature generation may be due to the fact that the BPS treatment forms a thick insulated, coated layer on the treated fabric surface as seen in the SEM micrographs [13], which acts as an intumescent that may have thickened on heating and absorbing the heat energy through endothermic reaction.
Temperature generation profile during burning of control and alkaline pH BPS-treated cotton fabric. BPS: banana pseudostem sap.
Cone calorimeter analysis
Summary of cone calorimeter results at heat flux of 35 kW/m2.

HRR: heat release rate; THR: total heat release; EHC: effective heat of combustion; MARHE: maximum average rate of heat emission.
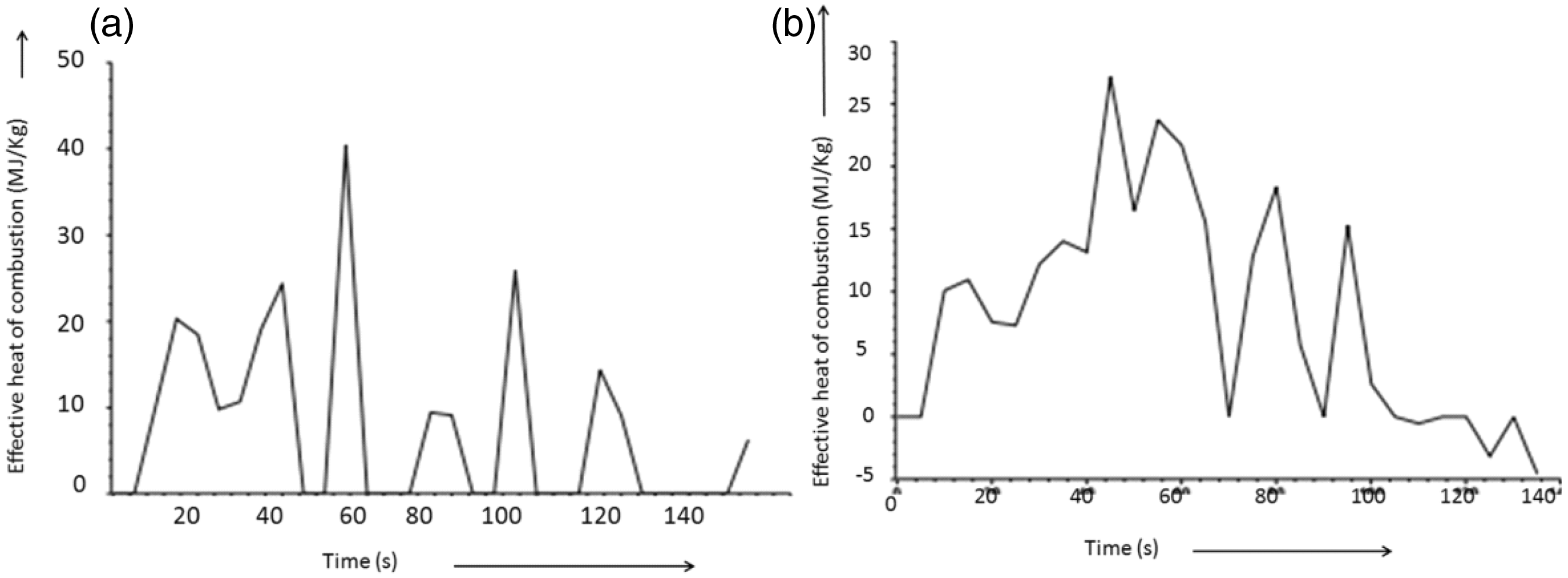
Table 2 showed that the length of time observed for ignition of the control and the treated sample is 7 s and 14 s, respectively. In addition, flame came from the control and the treated sample after 20 s and 33 s, respectively. It showed that the BPS treatment on the cellulosic cotton fabric increased the sample ignition time as well as delayed the flame out time. Further as far as the treated fabric is concerned, the amount of mass remain at the flame out condition is 26.05% higher than the control fabric. Heat release related results showed that the mean heat release rate and the total amount of heat release of the control cotton fabric are 31% and 29% higher compared to the BPS-treated cotton fabric. These phenomena also have been represented in the Figure 5. From the figure, it was clearly observed that BPS treatment lowers the heat release rate. Figure 7 showed that even after 80 s, the rate of heat release has been continually increasing for the control fabric, whereas for the treated fabric, the rate of heat release has been constant after 80 s. Similar phenomena also have been observed in the temperature generation profile curve of the treated fabric. Curve showed that after 80 s of burning, temperature generated from the BPS-treated fabric remains almost constant 160℃, which may indirectly support the constant heat generation during burning in cone calorimeter after 80 s. Heat stability after 80 s might be because of the more amount of carbonaceous insulated char mass formation as observed from the DSC and the TG curves. Here, char mass may help to absorb the generated heat during the burning cycle in cone calorimeter. BPS treatment also reduced the average effective heat of combustion as shown in Figure 6. Regarding the heat of combustion, control fabric generates average combustion heat of 21.45 MJ/kg, whereas BPS-treated fabric generates only 12.24 MJ/kg heat which is almost half of the control fabric. In addition, heat of combustion peak observed for the control fabric is 40.44 MJ/kg, whereas it was 27.12 MJ/kg for the BPS-treated fabric. Maximum average rate of the heat emission is also 17.81% less for the treated fabric compared to the control fabric. Connected to the heat release results, total fuel load, i.e. the amount of energy released by complete combustion of per unit mass of oxygen consumed, is 11.79 MJ/kg for the BPS-treated fabric, whereas for the control fabric, fuel load is more than 25.77 MJ/kg (54.2% more). Like TG analysis, here also the average specific mass loss rate of the BPS-treated fabric is 45% lower in contrast to the control cotton fabric. However, the BPS treatment causes an increase in total average smoke, carbon monoxide and decrease in the carbon dioxide formation during burning. Connected to the smoke production average surface extinction area (m2/kg) observed for the control fabric is 5.18, whereas for the treated fabric, it was reported 19.78. It might be due to the fact that in case of treated fabric, delayed ignition was observed and also sample takes longer time for total ignition. This phenomena might also be correlated with the observed afterglow during the vertical burning of alkaline pH BPS-treated fabric, after self-extinguishing of flame. Regarding the smoke formation, it has been reported in the literature that phosphate-based flame retardants increase in smoke emission and also rise in carbon monoxide with decrease in carbon dioxide formation [22]. Connected to it, researchers recently reported that the cotton fabric treated with the mixed formulation of diammonium phosphate (15%) and urea (5%) delivered 144 m2/kg smoke in contrast to the 73 m2/kg smoke generated after burning of cotton fabric [23]. As some amount of halogens such as chlorine are present in the BPS, it may also tends to increase the amount of smoke formation as reported in the literature [24].
Total heat release of BPS treated (A) and the control (B) cotton fabric. Effective heat of combustion of control (A) and BPS-treated (B) cotton fabric. SEM micrographs of char residues of the control (B) and the treated (A) fabric after vertical flammability test.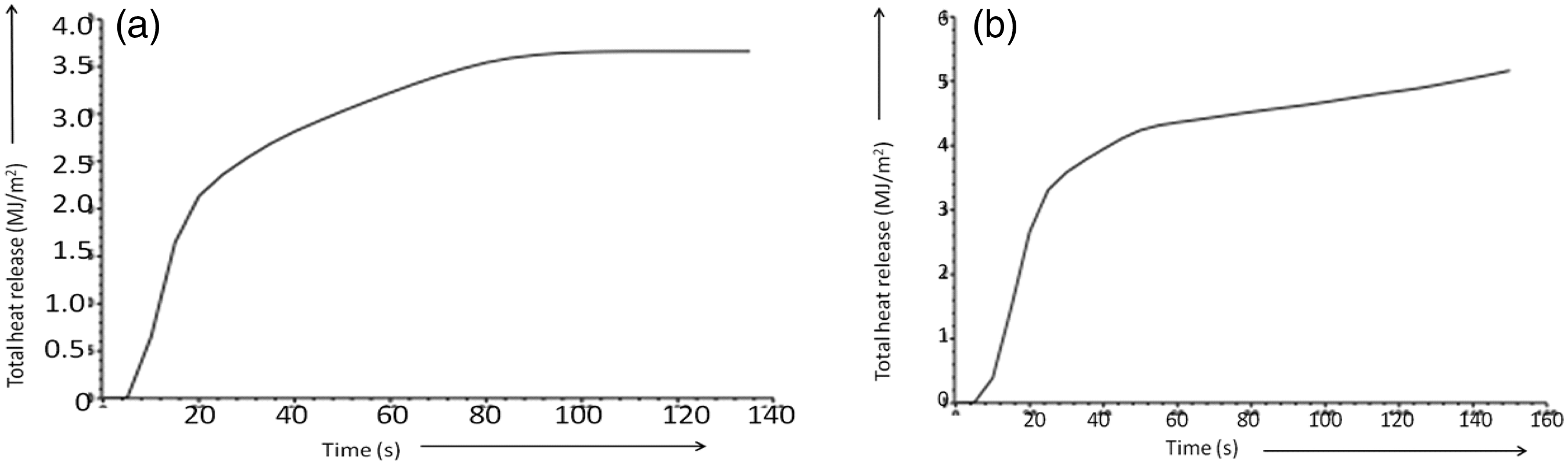

Char analysis
The char residue of the samples after combustion was further investigated, and their SEM micrographs are presented in Figure 7. It was already noted that the residual remains were negligible for the control fabric (B) after the complete burning, whereas in case of alkaline BPS-treated fabric (A), both the weight and the volume of the residuals were much higher. The same has clearly been shown by the black colour insulated carbonaceous char mass formation. Further, structural integrity of the fabric (A) was found to be maintained after the complete burning, in contrast with the complete lightweight white ash formation noted in the SEM micrograph of the control fabric (B). As far as the char morphology is concerned, from the SEM pictures of the char residues and from the char models (Figure 8), it has been proposed that the treated fabric had an intact char structure of closed cells containing gas pockets, gas bubbles, while freezing into the gas pockets, expands and thickens the alkaline BPS coating that quickly solidifies. The solidified structure of the residual mass of the treated fabric looked like an ant nest or honeycomb (as seen in SEM micrograph, Figure 8(A)) which restricts the formation of the flammable liquids by forming a heat insulating foam-like layer on the polymer substrate. On the other hand, as far as the char structure of the control fabric is concerned, it showed an open dry leaf net like channels by which gas and flammable volatiles can easily escape by capillary action into the hotter flammable region resulting in the burning of the cellulosic cotton polymer continuously, with high temperature and heat generation.
Model char structure of the control (A) and the BPS-treated cotton fabric (B). BPS: banana pseudostem sap.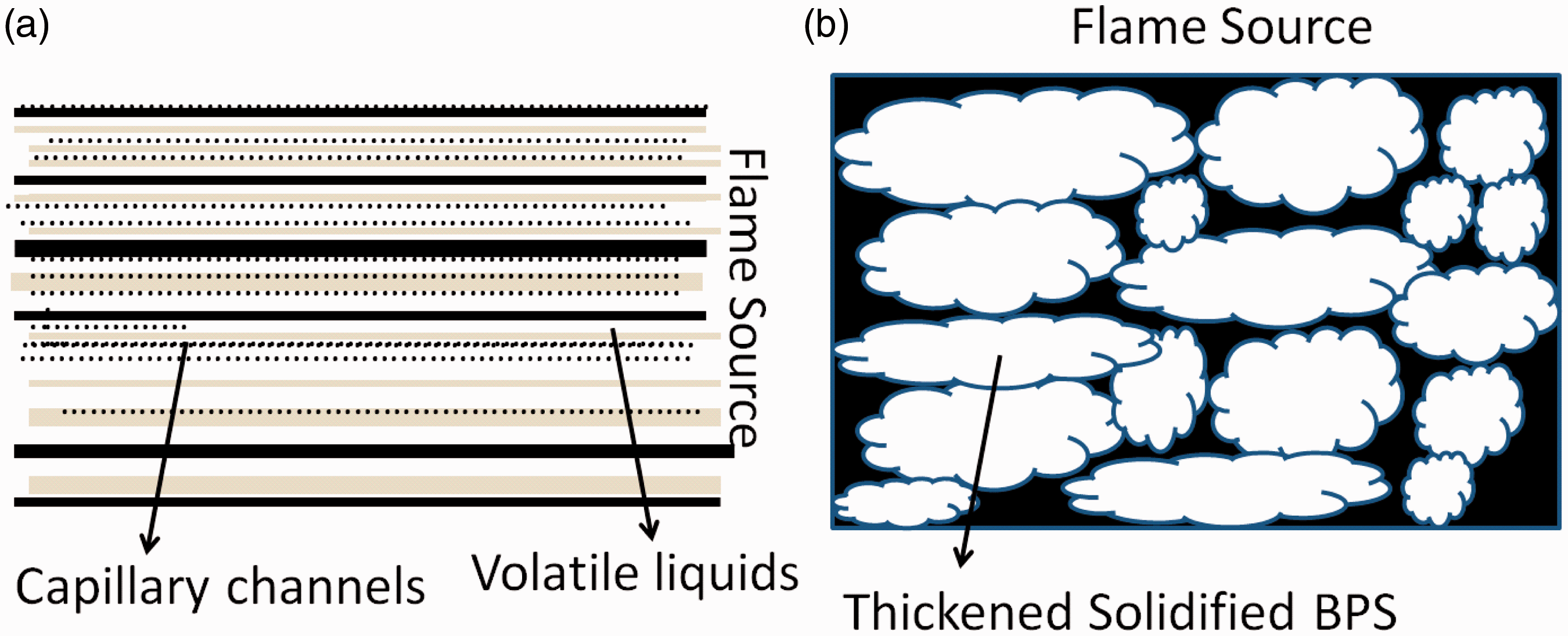
XRF analysis
The SEM pictures of the treated fabric showed a thick insulated BPS coating on the fibre surface as shown from our earlier reported work [13]. To determine the presence of different elements responsible for the fire retardant behaviour of the treated fabric, EDX analysis was carried out. The analysis showed the presence of different elements such as calcium, phosphorus, chlorine, potassium, aluminium [13]. EDX analysis showed the percent weights of magnesium, 5.8; phosphorus, 0.7; chlorine, 1.9; potassium, 1.7 and aluminum, 0.2 in the surface of the alkaline pH BPS-treated fabric [13]. In the present study, to reconfirm the results of the EDX analysis, XRF studies of both the extracted BPS and alkaline pH BPS-treated fabric were carried out and reported in Figure 9. The results showed that the BPS contain elements such as phosphorus, chlorine, silicon and trace amount of aluminium, whereas the BPS-treated fabric contains elements such as chlorine, phosphorus, silicon, calcium and aluminium with chlorine and phosphorus being predominant. From the EDX analysis, it was found that in the treated fabric, the amount of the aluminium present is 0.2%. However, extracted BPS contains very less amount (0.04%) aluminium [13]. It may be due to the addition of aluminium by the mordanting process in the fabric before the treatment.
XRF analysis of alkaline BPS-treated cotton fabric (A) and only BPS (B).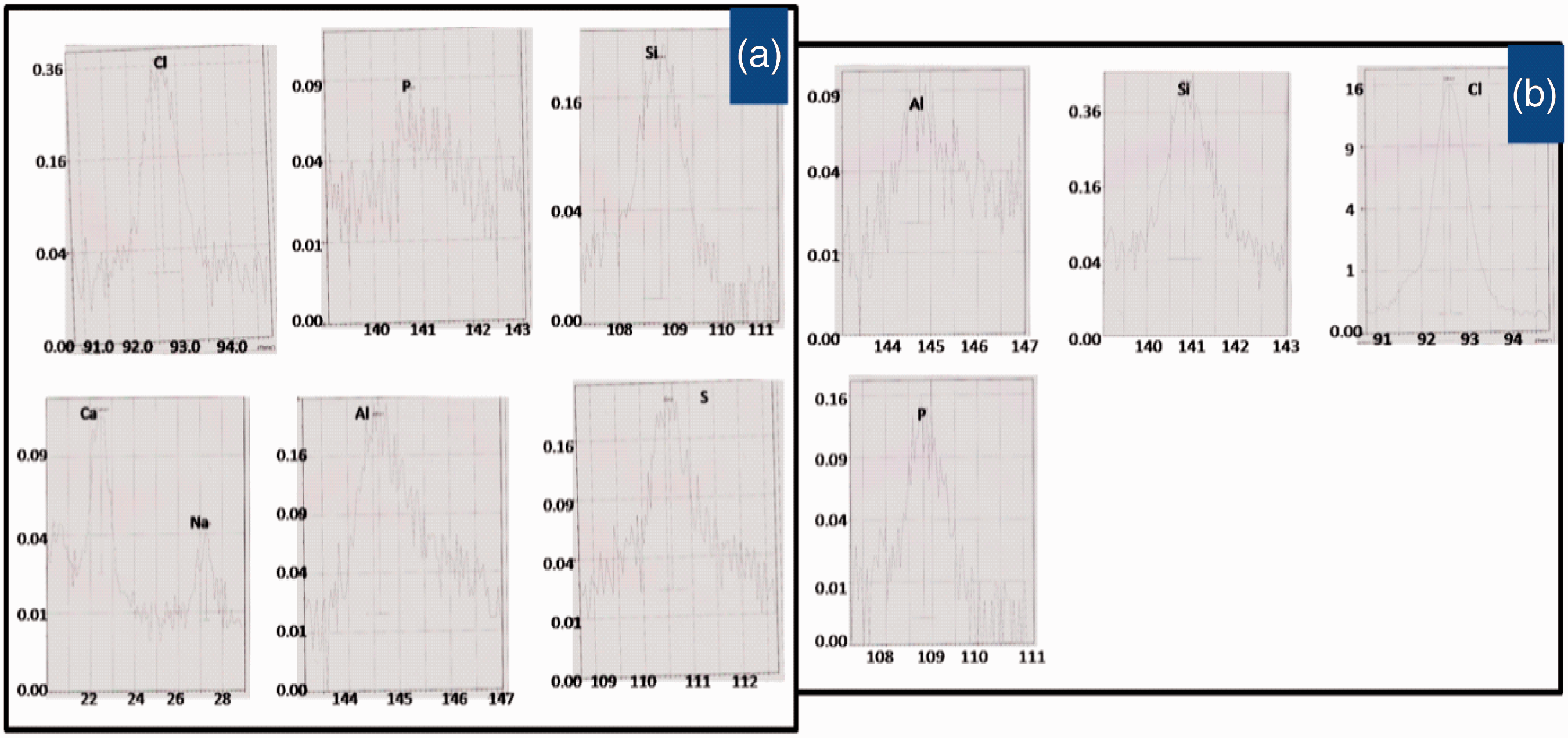
P and N content analysis
Content of nitrogen and phosphorus in control and treated fabrics.

LOI: limiting oxygen index; BPS: banana pseudostem sap.
Mechanism of imparted flame retardancy
Cotton being pure cellulosic in nature has a low LOI value of 18 and does not show any protection from flame. Prior to bleaching and mercerization of cotton fabric followed by mordanting, applications of BPS varying in pH condition ensured a good add-on (4–5% uptake). This implies that cotton is exhibiting flame retardancy mainly because of the chemicals present in the BPS. More explicitly, the effect of flame retardancy imparted by BPS may be attributed to the presence of various mineral salts in it in the form of chloride, phosphate and silicate, such as sodium silicate, magnesium chloride, sodium chloride. The presence of these inorganic salt molecules was roughly observed in the Fourier transform infrared spectroscopy analysis and by the peaks of the elements in the EDX analysis of BPS [13]. The surface ion mass spectroscopy analysis has also confirmed and quantified the amount of phosphate, phosphite, chloride, potassium chloride and other metal elements in the pure BPS, which is responsible for fire retardant property [13]. Besides, in this paper, we have analysed the presence of different elements in the BPS and the alkaline pH BPS-treated fabric by XRF. We have also quantified the total amount of nitrogen and phosphorus in the control and the BPS-treated fabric at different pH. It has been found that the amount of phosphorus and nitrogen is more in the alkaline BPS-treated fabric, and it is showing more thermal stability rather than the acidic BPS-treated fabric. As these phosphates and chlorinated products can act as fire retardants for the cellulosic substrate, the flame retardancy effect in the BPS-treated cotton fabric is attributed to the combined effect of the presence of metal salts, silicate, phosphate, nitrate and bound and unbound water molecules. Their presence has helped in the formation of more char and non-flammable gases like CO2, H2O, etc. As far as the flame retardancy mechanism is concerned, BPS mainly acts in the condensed phase mechanism, possibly due to phosphate and other metallic constituent contents, which are aiding the pyrolysis of the treated fabric by forming a char at an earlier stage, as observed from the thermogravimetric analysis and DSC curves. SEM pictures of the treated fabric showed that when this treated cotton fabric is exposed to heat or flame, it favours the formation of a stable and protective char and the char model shows the presence of closed cells, which limits the exchange of oxygen and combustible volatile products, thus adding towards the flame retardancy of the BPS-treated textile. It is possible that the BPS coating also might have acted as an intumescent that swells on heating and increase its volume, thus protecting the underlying polymeric material from heat or flame action by char insulation and foaming.
Physical properties and colour parameters
Strength and the colour parameters of the treated fabric.
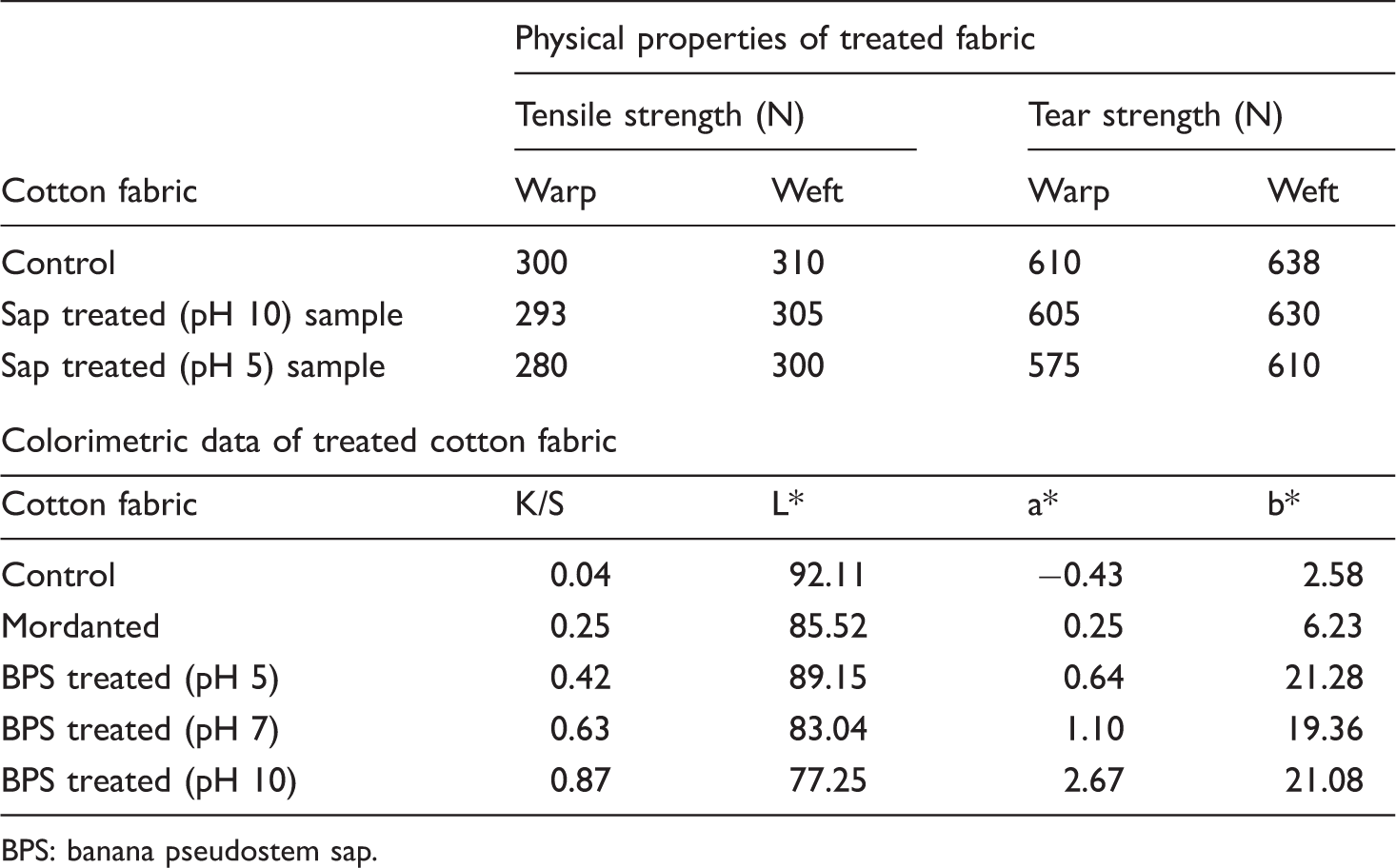
BPS: banana pseudostem sap.
Conclusion
The present study has investigated the flame retardancy effect of BPS on cellulosic cotton textiles at different pH. After application of BPS, the thermal stability of the fabric was found to increase and it also delivered more safety time either to extinguish the fire or to escape from fire hazards zone. Not only more time could be available, but also the total heat production and temperature generation in BPS-treated sample were lower. It was found that the BPS solution is more effective for thermal stability at alkaline pH. Flame retardancy property in the BPS-treated cotton fabric might have been attributed to the presence of (i) phosphate compounds and (ii) metal salts especially sodium and potassium chloride. Also the presence of approximately 14% inorganic materials in the BPS-treated cellulose textile might have helped in production of more char and non-flammable gases. TG curves showed the dehydration and char formation phenomena in the BPS-treated cotton fabrics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.


