Abstract
The development of electric circuit fabrication on flexible polymer substrates has attracted significant interest as a pathway to low-cost, comfortable movement and large-area electronics among direct printing techniques. The term electronic textiles, or e-textiles, are used to denote the class of fabric structures that integrate electronic elements with textiles and can sense changes in its environment and respond to it. This new class of wearable electronic systems is being designed to meet new and innovative applications in the military, public safety, healthcare, space exploration, sports and consumer fitness fields. In this paper the inkjet printing technique (office thermal inkjet printer) was used as a simple method to chemically deposit silver Nanoparticles (85–300 nm) to the flexible surface. It is done by ejection of silver nitrate and ascorbic acid as a reducing agent to attain nanometals on the surface. Ink concentration and repeating the printing sequence of each ink determines molar ratio ejection of ink and is found to be necessary to be evaluated in order to attain the highest level of conductivity. The main purpose of this work is to gain the highest conductive printed tracks by using statistical design of experiment method on the fabric surface by applying inkjet technique. By optimizing the mentioned parameters, the highest conductivity of printed flexible tracks on the surface of paper is gained 8.0635 × 105 S/m and the result for polyester fabric is obtained 1.2417 × 104 S/m whereas metallic silver wire has a conductivity of 6.173 × 107 S/m.
Introduction
Revolutionary changes have been occurring at an unprecedented rate in many fields of science and technology [1]. The invention of electronic chips, computers, the internet, wireless communication, rapid advancements in nanotechnology and many other developments have transformed the entire world and affected nearly every human being on this planet [2].
Between these innovations, applying nanoscience causes novel properties to appear, which are not obtained in larger sizes.
Flexible printed circuit boards continue to be a high-growth technology in the area of electrical interconnectivity and smart structures. Over traditional rigid printed circuit boards (PCBs), wire and wire harnesses, flexible circuit boards provide considerable weight, space and cost savings [3].
Human researches are going on to develop artificial structures in order to make them smart. Based on the manner of response, these materials can be divided into passive smart which can only sense the environmental conditions or stimuli, active smart to sense and react to the conditions or stimuli and very smart materials in order to react and adapt themselves accordingly [4]. Consequently, three kinds of components: sensors, actuators and controlling units can be integrated into cloths in order to make them smart. In medical application, by integrating sensors into cloths without disrupting the outward aesthetics of self-presentation and dress, we enable an intuitive approach to sensing body signals and movements [5]. This long-term monitoring systems have been developed to automatically generate an activity diary of healthcare-monitoring applications, allowing the annotation of medical data with different user activities and contexts, without requiring user intervention [6].
The convergence of textiles and electronics (e-textiles) can be relevant for the development of smart materials that are capable of accomplishing a wide spectrum of functions, found in rigid and non-flexible electronic products nowadays [7].
The key element for creating smart textiles is applying electrically conductive fibers, yarns or inks that can consequently send signals throughout the flexible segments such as sensors, actuators or computer chips that are embedded in the garment [8]. There are different methods for printing conductive inks on the surface of flexible substrates such as: Gravure (based on using engraved metal rollers for each color of ink) [9], Flexographic (based on roller printing method which the rollers are covered in rubber or a composite molding and carry the design in relief), Rotary and Flat screen-printing technologies (based on using patterned screen mesh with photosensitive polymer; ink passes through this mesh based on existing pattern and deposit on the surface) [10], Pad printing (also called Tampography is accomplished using an indirect offset (gravure) printing process that involves an image being transferred via a silicone pad onto a substrate) and Ink jet printing [11].
Between highly conductive metals such as Ag, Cu, Au and Al, silver particles are preferred to be used as a precursor in conductive inks [12]. Besides the increasing of possibility for miniaturization of electronic elements by reducing the dimensions of elements, using silver nanoparticles in preparing conductive inks was demanded [13]. As the size of low-dimensional materials decreases in the nanometer size range, electronic, magnetic, optic, catalytic and thermodynamic properties of the materials are significantly altered from those of either the bulk or a single molecule. Especially, metal nanoparticles have very unique properties that directly relate to their dimensions and to the fact that a large ratio of the atoms in the particle are in the surface of the particle [14].
This study explores the suitability of cellulose surfaces such as paper and fabric as a substrate for printing electrically conductive structures. Printing technique offers benefits such as non-contact, speed, flexibility, creativity, cleanliness, competitiveness, eco-friendliness, low consumption of energy and chemicals, digital control over the ejection and positioning process, etc. [15]. In direct deposition at least two fluids, which react on the substrate to give the desired material for a range of applications, emerge from separate printing heads. Thus multiple printing steps are used to deposit different reagent solutions that react together to form solid products [16].
The present report is the result of our continuous research on inkjet deposition techniques to gain the highest conductive printed tracks by using statistical method, Design of Experiment (DOE), via multiple printing steps for deeper understanding in which parameters control the flow of inks by the use of ink-jet printers on the surface of paper which is similar to the fabric but smoother than it. This property provides more appropriate situation for measurement and comparison conductivity of surfaces. In the final stage, the result has been applied for fabric and a successfully conductive pattern has been obtained by this technique.
Experimental work
Conductive inks
Polymer composites containing nanosilver particles were printed with the Ink-Jet method in order to obtain a line or ‘points’ being electricity conductors or contacts in electronic assembly [17]. Khanna et al. [18] in 2008 has worked based on thermal decomposition method for silver nanoparticles synthesized in order to use in Ink-Jet application. As shown in Figure 1, this method is based on decomposition of silver salt (silver alkanoate) of fatty acid at high temperature in inert atmosphere. Silver salt is obtained as the result of the reaction of silver nitrate and fatty acid dissolved in sodium hydroxide water solution [18].
Schematic of the thermal decomposition synthesis of the stable silver nanoparticles with fatty acid coated.
By thermal decomposition of silver alkanoate in nitrogen atmosphere at 250℃, stable silver nanoparticles with narrow size distribution could be obtained [19]. In another reaction, fatty acid dissolve in non-polar and slowly evaporating solvent (for example 1-octadecene) at 210℃ [20]. The both reactions give silver powder composed of strongly aggregated ligand-stabilized AgNPs with narrow size distribution. Preparation of silver ink requires dissolving of the synthesized silver powder in non-polar organic solvent (usually n-tetradecane). Printed tracks become conducive after sintering at 240–250℃ for 30 min [21]. Print quality and efficiency depends on viscosity of the ink used, the proper amount of which is reported to be 2.3–4.4 mPas [22].
The most important challenge concerning inkjet processes is to formulate suitable inks. Conductive inkjet inks are comprised of a liquid vehicle (water or an organic solvent) which determines the basic properties of the ink and a dispersed or dissolved metal precursor of highly conductive metals such as Ag (σ = 6.3 × 107 S/m), Cu (σ = 5.96 × 107 S/m), Au (σ = 4.42 × 107 S/m) and Al (σ = 3.78 × 107 S/m) that among them silver particles are preferred because of the highest electrical conductivity and resistance to oxidation. In Nanoparticles-based inks, the metallic dispersion in the ink should be stable to aggregation and precipitation in order to obtain ink with reproducible performance [12].
The idea of reducing metallic salts to produce metallic patterns was originally expressed by Thomas Edison who proposed to pattern a dielectric with silver nitrate solution and reduce the salt to metal. Between different options accessible for metal precursor such as sulphate, nitrate, acetate or carbonate salts of silver, he applied silver nitrate to fulfill some necessities which restrict the choice of metal precursor in such procedures such as high solubility in solvent, protracted solution stability, acceptable pH, low corrosive nature, positive standard electron potential (Silver nitrate has a high standard electron potential (2.35 times higher than copper)), fast reaction in the lowest possible amount of solvent and irreversible oxidation of metal after reduction [16].
According to previous experiments, silver nitrate AgNO3 (99.5% purity, Merck) is the metal salt of choice and for reducing solution ascorbic acid is selected. Although there are several numbers of reducing agents such as sodium borohydride and formic acid, the unique necessities of ink-jet printing demand a reducing system with very low toxicity, non-corrosive nature and acceptable water solubility, excellent reducing power whilst working in atmospheric condition and with no risk of damage to the substrate that the best one is introduced ascorbic acid C6H8O6.
The main purpose of this research is to deliver aqueous metal salt solution and aqueous solution of reducing agents by using ink-jet technology in order to print highly conductive patterns with good adhesion and good resolution on paper.
Method for conductive coating
There are various methods for making conductive patterns by the suitable conductivity in order to use on the flexible surfaces and make smart plates: electroless plating(submerging the substrate in an electroless plating solution) [23], evaporative deposition (the coating metal is evaporating and the substrate is exposed to the vaporized metal where it condenses on the surface and changes to a solid forming the coating) [24], sputtering (the coating material is throwing out atom by atom and is collected on the surface of the fabric) [25], coating conductive polymer and printing conductive inks [11].
Between mentioned methods, inkjet printing is one of the most potential techniques by which the circuit components can be ultimately coated onto the substrate in one step. Conductive printing depends on ink penetration to the substrate and is optimized when it remains mostly on the surface, not being dispersed into the internal structure of the substrate [26].
In our experimental work, digital printer (HP Deskjet 1280) has been chosen to print metallic salt and reducing agent solutions in separate runs. This printer uses two ink cartridges that come in black and tri-colour (cyan, magenta, yellow). Microsoft Word was employed as the print controlling software. Normal Speed and Resolution for black cartridge is 6 ppm and 600 × 600 dpi, respectively. Available inkjet printers have droplet volumes ranging from 2 to 40 picolitres depending on the nozzle size and the driving force. Ejected droplets spread on the surface by∼20–200 µm, depending on the droplet volume and the wettability of the surface [16]. Because of simple structure for filling the cartridges with the ink in thermal inkjet printers we preferred to use this head instead of piezo-based types that have a greater range of ink compatibility [27].
Dearden in 2005 used this method for printing silver conductive ink on the surface and high-temperature (around 200℃ and long heating time about an hour) treatment had been done after printing process in order to achieve conductive tracks. The homogeneous surface of printed silver traces change into the locally crystallized structure after the heat treatment [28]. This is because of rapid evaporation of solvent which limits the movement of silver particles and results in localized crystal growth. To form a conductive printed pattern, conductive particles must be sintered to create continuous connectivity and thus percolated paths. Sintering is a process of welding particles together at temperatures below their melting point [12].
The conventional approach to sinter metal NPs is heating. High surface-to-volume ratio of nano-sized particles decreases their melting temperatures. There are various sintering methods: thermal [29], photonic, microwave, plasma, electrical [30] and chemically triggered.
As the curing temperature increases above 150℃, the resistivity of printed media stabilizes at the low level around this curing temperature. The resistivity largely decreases after a certain period of curing time, but extended time above that point does not improve the conductivity [28].
In the previous literatures, usually the substrate was first printed by reducing ink and after an intermediate drying at room temperature for 5 min; the silver nitrate solution was overprinted for achieving the better conductivity on the substrate. Metal formation occurred in-situ as a consequence of the redox reaction between reducing agent and metal salt solutions leaving metallic layers composed of aggregated metal particles. It is necessary to replicate the printing sequence more than once to attain connectivity between the metallic particles formed on the surface. It is noticeable to have intermediate drying process at 50℃ between these runs to decrease the effect of printer leading rollers and holders on the final quality. The thickness of the deposited layer may be increased by repeating the inkjet deposition cycle [16].
It is observable that the different concentrations of each ink will cause different levels of electrical conductivity on the substrate and also change the number of printing layers of reducing agent and silver salt solutions could build up silver patterns having various packing density and conductivity.
Therefore, two important parameters that must be considered for reaching the highest conductivity in the printed tracks are ink concentration and number of the printing layers of both inks simultaneously.
Experimental design
In order to examine the main factors and their interactions for the conductivity properties of the cellulose flexible surface full factorial design was used. To design an experiment means to choose the optimal experiment design to be used simultaneously for varying all the analyzed factors. By designing an experiment one gets more precise data and more complete information on a studied phenomenon with a minimal number of experiments and the lowest possible material costs [31].
Full factorial experiments are the experiments in which the effects of more than one factor on response are investigated.
Factorial designs are mostly used to examine the effects of experimental factors and the interactions between those factors, that is, how the effect of one factor varies with the level of the other factors in a response. Also the number of experiments geometrically increases with the increasing number of factors and levels [32,33].
Based on the mentioned effective parameters and applying DOE, subsequent design has been considered for the experimental tests:
In Response Surface Methods (RSM) that finds the ideal process settings and achieve optimal performance, we have chosen ‘Central Composite Design’ that in this way each numeric factor is varied over five levels.
Experimental layout using a full factorial experimental design.

After printing process, to obtain proper electrical conductivity, sintering process is essential. This reaction takes significant role in electrical resistance decreasing because it can improve the yield of the redox reaction by increasing the kinetic energy. Therefore, the samples have been exposed to the temperature of 150 ℃ for minimum 1 min [32].
Conductivity analysis
When there are more than two electrons in an atom the electrons are arranged into shells at various distances from the nucleus. It is possible for an atom to lose an electron and electrons that move from one atom to another are called free electrons and such random motion can continue indefinitely by an electric pressure or voltage. Current is the rate of movement of charge and Charge carrier species can be many, e.g., electrons, holes, protons and ions. Conductors are materials that contain electrons that are loosely connected to the nucleus and can easily move through the material from one atom to another. All metals are conductors and some examples include copper, aluminum, brass, platinum, silver, gold and carbon.
For a continuous current to flow between two points in a circuit a potential difference or voltage, V, is required between them. The flow of electric current is subject to friction. This friction, or opposition, is called resistance R and is the property of a conductor that limits current. The unit of resistance is the ohm; 1 ohm is defined as the resistance which will have a current of 1 ampere when 1 volt is connected across it. According to Ohm’s law: R = potential difference/current [34].
Materials display an extremely wide range of resistivity, superconductors have resistivity <10–25 Ωm, typical metal conductors ∼10–8 Ωm, semiconductors 10–4…1010 Ωm and insulators 1010…1020 Ωm [35].
Since the conductance is generally proportional to the area of the electrodes and inversely related to the distance between them, it is defined as one reciprocal ohm and the SI unit of Conductance is the Siemens G = 1/R [36].
An ammeter is an instrument used to measure current and must be connected in series with the circuit. Since all the current in the circuit passes through the ammeter it must have a very low resistance [34].
The conductivity of the films provides a measure of the underlying microstructure since any discontinuities or anomalies arising from the deposition process would lower conductivity.
A four-contact method was used in preference to the four-point probe method usually employed in the electrical assessment of films [37]. A circuit diagram of the four-contact apparatus which is designed and built is shown in Figure 2.
Circuit diagram of a four-contact measuring apparatus.
For this measurement, four-contact-probe holder is positioned over the printed surface. The holder is fabricated with four copper probes which could make contact with the surface of each sample. To keep away from any gap between probes and the printed surfaces, a weight of 500 g was placed over the probes’ holder to press them against the printed surface. Then by supplying a constant current of 100 mA to the outer probes, the voltage between the inner probes is measured using a voltmeter. To minimize measurement errors mainly caused by inconsistent contact between the copper probes and the printed surfaces, each sample was tested three times and the average value was used in conductivity calculations. In this way, an average value of resistivity over the entire width of the film strip is obtained [16].
The electrical conductivity (σ) is a measure of how well it passes electrical current and may be determined from the resistivity (ρ) which is the inverse of conductivity.
Resistivity is determined through the following relationship

The definite sample length applied in the resistance calculations is 20 mm which is the distance between the two inner probes of the four contact device.
The thicknesses of the deposited metallic layers are measured using the cross-sectional scanning electron microscopy (SEM Philips XL30) image as shown in Figure 3.
Cross-sectional SEM images from printed sample.
Results and discussions
SEM technique was used to investigate the morphology of the deposited pattern. As it is clearly visible (in the highly magnified image), the inkjet deposition process has been capable of producing nano-sized silver particles ranging from 85 to 300 nm on the substrate (Figure 4).
Scanning electron microscope (SEM) images from the surface of printed sample.
Design of experiment results from conductivity.

Inkjet printing of solutions with different concentrations of ascorbic acid and silver nitrate is shown in Table 2. It also presents that the varied number of printing for each ink could lead to different levels of conductivity in deposited patterns. This technique produces layers with conductivities comparable to that of bulk metals, such as copper (5.813 × 107 S/m), silver (6.173 × 107 S/m) and graphite (7 × 104 S/m).
Moreover, coefficient of variation has been considered for one test to evaluate the accuracy of this method. For this purpose three runs are supposed to have the same situation (all parameters are the same), which is shown in Table 2 (Run: 6-7-8) and the statistical evaluation has been done as follows

Divisions of concentrations of ascorbic acid effect on the conductivity of printed tracks.

Divisions of concentrations of silver nitrate effect on the conductivity of printed tracks.

Divisions of printing number of ascorbic acid effect on the conductivity of printed tracks.

Divisions of printing number of silver nitrate effect on the conductivity of printed tracks.

The amount of p value (probability factor) in this test is in 0.05 levels. (If the amount of p value is less than 0.05 with a confidence level of 95%, the effect of the related parameter will be significant.)
The amount of significance for each division shows the significance between the parameter on that division, for example in Table 3, 9577.80, 76451.45, … is the amount of conductivity at different concentrations of ascorbic acid and in Table 6, 129319.40, 170877.68, … is the amount of conductivity at different printed number of silver nitrate layers calculated by the ANOVA program.
Significant 1.000, 0.228, 0.749 … shows similarity of the values in one group i.e. sig. 1.000 indicates that the values in one group are exactly the same. Sig. 0.749 implies that although the values are categorized into one group, there are some differences between them. Sig. 0.000 means the values should be categorized into different groups.
As it is observed, the optimum quantity for each considered factor can be achieved in order to attain the maximum conductivity from this method. These values are as follows
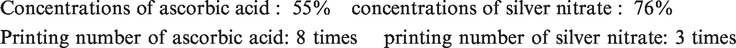
Taking into account the concentration of each ink and the output of each printing head (5.4 g m–2 silver nitrate ink and 5.8 g m−2 ascorbic acid ink), the achieved results for number of requested printing layers reveals almost twice molar consumption of ascorbic acid compared to silver nitrate (2:1, respectively) for attaining the highest level of conductivity.
A sample of printed paper with the mentioned specification in printing procedure is shown in Figure 5.
A sample of printed paper.
In constant amount of ascorbic acid and silver nitrate concentrations, if we consider the constant value for printing number of silver nitrate and change the number of printing sequences for ascorbic acid, the amount of conductivity will be decreased (as its obvious in the result of experiments in runs : 11, 12, (6, 7, 8)). More than required amount of ascorbic acid will not take part in any reaction and causes extra amount of this material to remain on the surface that occupies space between nanosilver particles which reduces conductivity. Also less than the required amount will result in less reduction of Ag which again causes less conductivity. This can also happen when the amount of printing sequences for ascorbic acid is constant and printing number of silver nitrate changes. In this case extra amount of silver nitrate will remain on the surface and decreases conductivity or less amount of this material will cause less reduction of Ag particles and less conductivity (in runs : 9, 10, (6,7, 8))
These phenomena are obvious with the constant number of printings and various ink concentrations which makes less conductivity based on described reasons in above.
The statistical analyses of ANOVA for Response Surface Quadratic Model indicate model terms are significant. In this case, the effect of A2, B2, C2, D2 on conductivity are significant in model terms. Based on this model, proper coefficient for each factor is estimated in Table 4 for using in final equation in terms of actual factors

Studentized residuals versus predicted values by the model for this set of experimental conditions are shown in Figure 6.
Studentized residual values by model.
Whenever the predicted and actual values are near to the line with slope 45’, we can use that model for other cases. As we can see in Figure 5, in this model these two groups of data are near to line x = y.
Model graphs
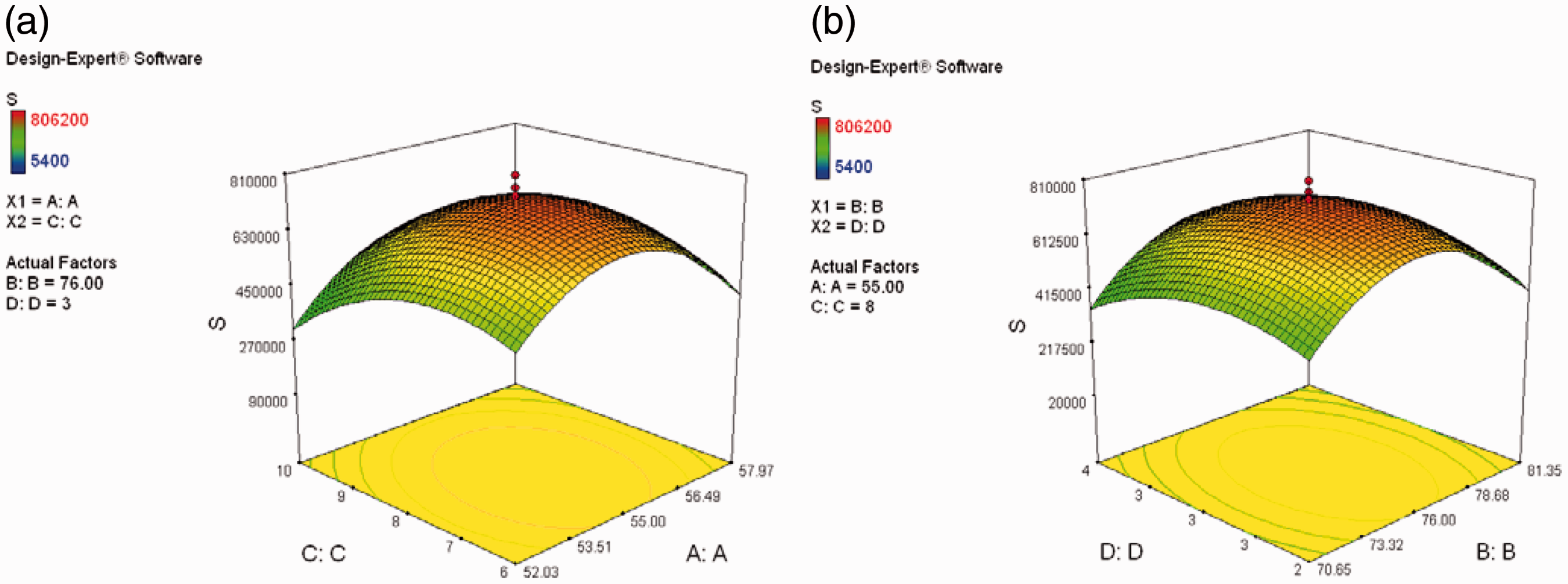
Three-dimensional graphs in Figures 7 and 8 show the relation between considered parameters and final conductivity.
Relation between (a) concentration and (b) repeating the print sequences of each ink with final conductivity. Relation between concentration of ink and repeating the print sequences with final conductivity for (a) ascorbic acid (b) silver nitrate.
The characteristic of printed fabric.

In comparison between the obtained conductivity from the Printed tracks on the surface of paper and fabric, it is obvious that the conductivity of printed tracks on the surface of fabric with the same condition in printing process is decreased because of the fabric surface characters. Fabric consists of yarns that are weaved in different patterns and makes rough surfaces in comparison with paper.
The thickness of printed ink on the surface is about 40 µm (because of hydrophobic property of polyester). Printed fabric with the mentioned specification in printing procedure is shown in Figure 9.
A sample of printed fabric.
Conclusions
A global market demand for high-quality and low-cost electronic components requires innovative fabrication technique that is faster and cheaper compared with traditional production methods. In this regard inkjet printing of metal salt solutions and reducing agents has been developed and deposited
Silver nanoparticles (85–300 nm) on the surface of paper and polyester fabric are performed.
The deposited layers by this method showed a level of flexibility, which is a useful way for printing on different flexible materials; such as paper, textiles etc.
The results showed that the conductivities of the prints are dependent on concentration and repeating the printing sequence of each ink in inkjet printing on papers and fabrics. The results achieved by DOE technique shows the concentration of 55% W/v solution of ascorbic acid and 76% of silver nitrate would be the best choice in order to make the highest possible conductivity. In this way also optimum replicate number of printing for ascorbic acid and silver nitrate is achieved 8 and 3 times, respectively. The highest conductivity which is obtained on the surface of paper is 8·0635 × 105 S/m and the result for polyester fabric is achieved 1.2417 × 104 whereas metallic silver wire has a conductivity of 6.173 × 107 S/m.
Although the goal of this research was to expand the quality of digital and controllable printed conductive tracks by nanosilvers on the surface of flexible materials to be appropriate for use in the electronic industry, continuing these research on other surfaces is essential.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
