Abstract
Nerve guides are the bridging devices used to bridge the proximal and the distal ends of the dissected peripheral nerves. Biocompatible nerve guides comprising polycaprolactone/polyvinyl pyrrolidone polymer were developed using centrifugal spinning technique. The developed nerve guides were loaded with triterpenoids isolated from Centella asiatica. Drug-releasing studies were carried out at various drug-loading percentages in the phosphate buffer releasing medium. The release studies confirm that the drug can be released into the medium to the extent of 50%. Cell culture studies also confirm that the triterpenoids-loaded nerve guides are biocompatible, provide an oriented substratum for cell adhesion and proliferation which is necessary for guided tissue regeneration.
Introduction
Peripheral nervous system is the vast communication network that transmits information from the brain and spinal cord to other parts of the body. Hence, nerve regeneration is a complex biological phenomenon, and injury to the peripheral nerves leads to malfunctions of the body parts. In case of short nerve injury, end-to-end fascicular suturing is preferred and gives satisfactory results only if the nerve ends are adjacent to each other and tension free suturing is possible. However, in extensive injury, auto grafting is considered as a gold-standard approach and it is limited by availability of nerves, donor site morbidity, painful neuroma formation, secondary surgery, functional loss, potential mismatch of tissue structure and size. In order to overcome these limitations, bioengineering strategies have been emerged as a potential key player for developing artificial nerve guides needed to bridge the gap between proximal and distal nerve stumps [1–5].
In order to accurately mimic natural regeneration in the damaged nerves, the developed nerve guide should be biocompatible, biodegradable, easily available, porous for oxygen and nutrient diffusion and possess oriented substrate for supporting cell growth [6–8]. Among these properties, substrate orientation plays a vital role for cell adhesion, oriented cell proliferation and nerve impulse transmission. Studies have reported that electrospun aligned fibres of polycaprolactone (PCL)–chitosan, PCL–gelatin, PCL–collagen, PLGA, poly acrylonitrile-co-methylacrylate (PAN–MA), poly L-lactic acid (PLLA) combinations are suitable for nerve regeneration. When compared to randomly oriented fibers, aligned fibers support the orientation of cells and improve the neurite outgrowth and contact guidance [9–12].
Based on the requirements of an ideal nerve guide, researchers developed numerous fabrication techniques such as magnetic polymer fiber alignment, injection molding, phase separation, solid free-form fabrication, ink-jet liquid polymer printing, micro patterning, lyophilizing, film rolling, braiding, extrusion, salt leaching and electrospinning [13]. The production of the aligned fibers for the growth of neural cells is a major limitation in all the above processes. Numerous attempts have been made by researchers to produce aligned nanofibers by electrospinning using approaches such as a wire drum collector, high-speed rotating drum, patterned electrodes, collection on a liquid medium followed by drawing, and two-pole air gap electro spinning. Even though attempts have been made to produce aligned fibers by electro spinning, the setup for the same is complex and getting continuous aligned fiber strands is still a challenge to the research fraternity [14].
Our research group has developed a high voltage-free spinning system for producing aligned fibres. This lab scale spinning system works based on centrifugal force. Ultrafine production by centrifugal spinning process starts by placing the polymer solution into the rotating polymer reservoir with a narrow orifice provided. While rotating the polymer holding reservoir at high speed, the fibres were produced. This in-house-built centrifugal spinning technique possesses advantages such as high production rate, good safety and attainment of high degree of fibre alignment. The design features of the machine and the effect of process variables on fibre quality have been discussed elsewhere [15,16]. In the present work, biocomposites comprising PCL/polyvinyl pyrrolidone (PVP) blends were developed.
Centella asiatica (CA) (also called as Gotu kola and hydrocotyle asiatica) is a perennial herbaceous creeper distributed widely in shady, moist and marshy areas. CA possesses wide range of therapeutic potentialities such as neuroprotective, wound healing and pain-relieving properties. Being a rejuvenating herb, it is generally used to enhance intelligence, memory and commonly called as nerve tonic [17–19]. CA possesses several active constituents such as asiaticoside, medacassoside, centelloside and asiatic acid. In addition, CA contains other components such as volatile oils, flavonoids, tannins, amino acids and sugars. Among these, asiaticoside and asiatic acid serve as a potential candidate in wound healing and neuroprotective activities [20–22]. It is reported in literature that asiatic acid shows an excellent neuroprotective activity in both in vitro and in vivo. Rats treated with CA extract after nerve damage recovered more quickly and showed increased axonal regeneration and rapid functional recovery [23]. Wanakhachornkrai et al. studied the neuritogenic effect of CAECa233 on human neuroblastoma cell lines. They observed that CAECa233 promotes the neurite outgrowth and can be efficiently used for the treatment of neuronal injury and neurodegenerative disease [24].
One of the important properties to be taken into an account while designing the fibre-based tubular nerve guide is fibre orientation. The extracellular matrix of peripheral nerve possesses highly aligned architecture. Aligned fibres can be effortlessly produced by our in-house-built centrifugal spinning process. In the present study, it is aimed to utilize the aligned fibers produced through centrifugal spinning system for developed tubular nerve guides. A small lab scale experimental set up has been designed for developing tubular nerve guides. The aim of the present contribution was to develop a nerve guide by incorporating CA extracts in PCL/PVP biocomposites. The developed nerve guides were characterized using various analytical tools. The drug-releasing behaviors and in vitro cell culture studies were also studied.
Materials and methods
Materials
PCL (Mn 70,000–90,000) and PVP (Mw 1300 KD) were obtained from Sigma Aldrich India. Chloroform, methanol, hexane and dichloromethane (RANKEM, India) used in this study were of analytical grade and used without further purification.
Preparation of herbal extract
CA leaves from the plant were collected from Koyambedu market, one of the biggest markets in Chennai city, Tamil Nadu, India. The collected leaves were cleaned, dried and finally ground into fine powder. This powder was initially extracted with hexane followed by dichloromethane. The obtained solid residue after complete removal of solvents was extracted with methanol in the ratio of 1:20 at 50℃ for 4 h. After evaporating methanol by using rotary evaporator, a dark greenish brown powder was obtained and subjected for further studies [25].
Confirmatory test for triterpenoids – Salkowshi test
The obtained crude extract of CA was subjected to “Salkowshi Test” to determine the presence of triterpenoids. In a test tube, little amount of extract was mixed with 2 ml of chloroform. Then 3 ml of concentrated sulphuric acid was added to the thoroughly mixed solution [26].
Fabrication of an aligned nerve guide
Centrifugal spinning was carried out using 12% w/v solutions of PCL/PVP in the ratio of 80:20 in chloroform. Prior to centrifugal spinning, the solutions were stirred for an hour; 1%, 2% and 3% crude extracts of CA with respect to polymer concentration were dissolved in 1 ml of methanol followed by 9 ml of chloroform and stirred continuously overnight. After obtaining complete homogeneity, PCL and PVP were added and stirred for an hour. The prepared solutions were carefully injected into the pot type rotating spinneret head with the help of a syringe. Centrifugal spinning was carefully carried out under a constant 2000 r/min. The formed ultrafine fibres were collected in an aluminum foil wrapped over a round-bottomed collector. The collected aligned fibres were carefully peeled off from the foil (40 mm × 30 mm) and wrapped over a Teflon mandrel having an inner diameter of 1.5 mm and 50 mm length. The Teflon mandrel was connected to the rotating shaft, which is attached to the cylindrical glass container. Inside the container a beaker filled with 5 ml of chloroform was kept. Then the nerve guide was removed from the mandrel at an interval of 5, 10, 15 and 20 min. The effect of time on the morphology and stability of the nerve guide was analyzed. Based on the above trials, the optimized time was selected for developing CA crude extract-loaded nerve guide. The schematic diagram of the in-house developed experimental set up used for developing the nerve guide is shown in Figure 1.
Schematic diagram of an in-house-developed nerve guide fabrication set up.
Scanning electron microscopic analysis
Surface morphology of the developed nerve guide was observed under a scanning electron microscope (SEM) (S3400NSEM, HITACHI) at an accelerating voltage of 15 kV. Cells seeded on PCL/PVP and CA-loaded PCL/PVP ultrafine fibrous web collected over Thermanox® cover slips were fixed with 2% glutaraldehyde. The cells were again re-fixed with 1% osmium tetroxide after washing thrice with 0.1 M sodium cacodylate buffer. The samples were then dehydrated with the increasing gradients of acetone–water mixtures followed by treatment with increasing gradients of acetone–hexamethyldisilazane mixtures. The samples were snap frozen with liquid nitrogen and freeze-dried in a lyophilizer. Prior to scanning under the SEM, the samples were sputter coated with gold using a fine coater (E 1010, HITACHI).
Fourier Transform Infra Red spectroscopy analysis
Infrared spectra of C-spun PCL/PVP and CA-loaded PCL/PVP fibrous mat were obtained using FTIR spectrophotometer (Perkin-Elmer, USA model). Samples of each fibrous mat was ground and mixed thoroughly with potassium bromide at the ratio of 1:5 and the mixture was made into pellet. The IR spectra of the pellets were recorded at the wavelength range of 500 to 4000 cm−1 with the resolution of 4 cm−1.
Thermal analysis
The thermal behavior of C-spun PCL/PVP and CA-loaded PCL/PVP fibrous matrices was examined by thermo gravimetric analyzer (TGA) (TA Q50, USA). TGA measurements were conducted over a temperature range of 0℃ to 700℃ at a heating rate of 20℃/min under nitrogen purge.
In vitro drug release studies
A CA extract-loaded nerve guide (0.5 g) was first placed in a vial containing 20 ml of releasing medium containing 10% v/v of methanol. The addition of methanol was to facilitate the dissolution of triterpenoids in the releasing medium. In this case, 4 ml of sample was taken from the medium after appropriate intervals for about 24 h and then the same volume of fresh release medium was added as a replacement. A calibration curve was obtained for the crude extract of CA concentration at a peak absorption wavelength of 204 nm, and a linear equation was derived by a curve-fitting method. In the assessment of drug release behavior, a cumulated amount of the released drug was calculated. Drug-releasing kinetics were carried out on the drug-releasing behaviors from the nerve guide using first-order, Higuchi, Hixson–Crowell and Peppas model.
Cell culture studies
Glial C6 cell lines were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum, 4 mM/L – glutamine, 4500 mg/L glucose, 25 mM HEPES, streptomycin (100 µg/ml), penicillin (100 units/ml), gentamicin (30 µg/ml) and fungizone (2.5 µg/ml). The cells were maintained at 37℃ in a humidified 5% CO2 incubator (Binder CB150, Germany) in 25 cm2 flasks. Prior to cell seeding, C-spun PCL/PVP, CA-loaded PCL/PVP fibrous matrices were sterilized under UV light for 2 h. C6 cells were then seeded into the fibrous matrices in culture medium, following which, cell morphology, cell compatibility and cytotoxicity were assessed.
Cell proliferation
The proliferative potential of C6 glial cells on C-spun PCL/PVP and CA-loaded PCL/PVP matrices was assessed by MTT (3 -(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) assay. A uniform number of (12 × 103) cells was seeded into a 24-well culture dish containing the scaffolds and allowed to proliferate for 24 h. The growth as an index of cell proliferation was measured spectrophotometrically (Lambda 45, UV/Vis Spectrometer, Perkin Elmer, Singapore) at 570 nm after treating with MTT (0.5 mg/ml). The kinetics of the cell proliferation (6 × 103 cells/well) was studied by increasing the incubation period up to 3 days.
Result and discussion
Effect of time on ideal nerve guide formation
Nerve guides serve as an important platform for bridging the proximal and distal ends of the injured peripheral nerves. Ideally, nerve guides also aid in the directional growth of regenerated axons. The nerve guides are generally tubular in nature to simulate the natural architecture of the peripheral nerves. Tubular nerve guides are prepared by molding and wrapping process depending upon the scaffold preparation techniques. In-house-developed C-spin technique offers a unique advantage in terms of higher degree of fibre alignment and ease of production.
The SEM images of centrifugal spun PCL/PVP fibrous matrix are shown Figure 2(a). From the figure, it can be observed that the obtained fibres were highly aligned. The degree of fibre alignment was analyzed by fast Fourier transform (FFT) approach as cited in the literature by using image analyzing software namely Image J [27]. The FFT image of the centrifugal spun PCL/PVP matrices showed sharp and narrower peak (Figure 2(b)). This is further confirmed by the alignment plot, which also possesses a sharp peak which is shown in the Figure 2(c).
(a) SEM image of centrifugal spun PCL/PVP matrices (b) FFT image of centrifugal spun PCL/PVP (c) alignment plot of centrifugal spun PCL/PVP.
Wrapping of C-Spun fibrous matrices over the Teflon mandrel yielded tubular geometry with enhanced orientation of fibres. However, a perfect tubular architecture is necessary for promoting the directional growth of axons. In order to achieve uniform tubular structure, the wrapped C-spun fibrous matrices were exposed to chloroform vapours at different time intervals and the images of the nerve guides are shown in Figure 3. From this figure it can be seen that, the exposure to chloroform vapour plays a critical role in altering the surface morphologies of the nerve guide. From Figure 3(a) and (b), it is observed that when the nerve guide is exposed to chloroform vapours for 5 min, fibre morphology was not significantly altered and the desired tubular architecture was not achieved. The developed tube collapsed on releasing from the mandrel. With increasing the time for 10 min, the fibres started to coalesce and grouping of fibres was observed as seen in Figure 3(c) and (d). The handleability of the tube was not good and also collapsed on releasing from the mandrel. From Figure 3(e) and (f), it can be observed that, at 15 min of exposure, the nerve guides of desired architecture in terms of handleability were achieved. The fibrous morphology was retained in the nerve guide. When the time was further increased to 20 min the fibres coalesced completely with adjoining fibres resulting in the formation of a film, which is evident from Figure 3(g) and (h). Hence, for further studies nerve guides exposed to chloroform vapours for 15 min were used. The nerve guides were also prepared by loading the crude extract of CA, commonly called as nerve tonic which enhances the peripheral nerve regeneration. Presence of triterpenoids in the crude extract was confirmed by the confirmatory test namely Salkowshi test. Formation of brown ring (green colour in the upper layer and deep red in the lower layer) in the interface confirms the presence of triterpenoids in the crude extract of CA and the results are shown in Figure 4. CA extract-loaded nerve guides exposed to chloroform vapours for 15 min show similar morphology to that of control nerve guides and the SEM images of CA-loaded nerve guide are shown in Figure 3(i) and (j). The nerve guides were loaded with different concentrations of CA extract (1%, 2% and 3%) and the releasing profiles were analyzed.
(a, c, e and g) Exposure of the nerve guide to chloroform vapours for 5, 10, 15 and 20 min. (b, d, f and h) Surface morphology of the nerve guide to chloroform vapours for 5, 10, 15 and 20 min. (i) CA extract-loaded nerve guide exposed to chloroform vapours for 15 min. (j) Surface morphology of CA extract-loaded nerve guide exposed to chloroform vapours for 15 min. Phytochemical screening of the crude extracts of CA for triterpenoids. Formation of brown ring (indicated by arrow) at the junction confirms the presence of triterpenoids.

FTIR analysis
The FTIR spectrum of C-Spun PCL, PVP, PCL/PVP, PCL/PVP/CA-loaded ultrafine fibrous web is shown in Figure 4. FTIR for PCL is shown in Figure 5(a) and it showed characteristic peaks of C = O stretching vibrations at 1731 cm−1, CH2 bending nodes at 1471, 1397 and 1365 cm−1 and COO– vibrations at 1739 cm−1. The C–O–C stretching vibrations yield peaks at 1169, 1108 and 1049 cm−1. The spectrum of PVP is shown in Figure 5(b). The peak at 1654 cm−1 appeared due to C = O stretching vibrations of PVP. The CH2 bending nodes were observed at 1376, 1424, 1441 and 1463 cm−1 and that of the C–N vibrations occurred at 1291 cm−1. The spectrum of PCL/PVP (80/20) is shown in Figure 5(c).The spectrum showed characteristics of both PCL and PVP. The spectrum of CA-loaded PCL/PVP ultrafine fibrous web is shown in Figure 5(d). The presence of the absorption band at 918, 970 confirms–(CH2), 1060,2907 and 3274 cm−1 ascribed to – C–O, –C = O, C–H and R–OH of asiatic acid along with the characteristic peaks of PCL/PVP. Two peaks at 2917 cm−1 and 2844 cm−1 indicate the presence of C–H stretching which confirms the presence of hydrocarbons [28,29].
FTIR of C-spun fibrous matrices. (a) PCL. (b) PVP. (c) PCL/PVP. (d) CA extract-loaded PCL/PVP.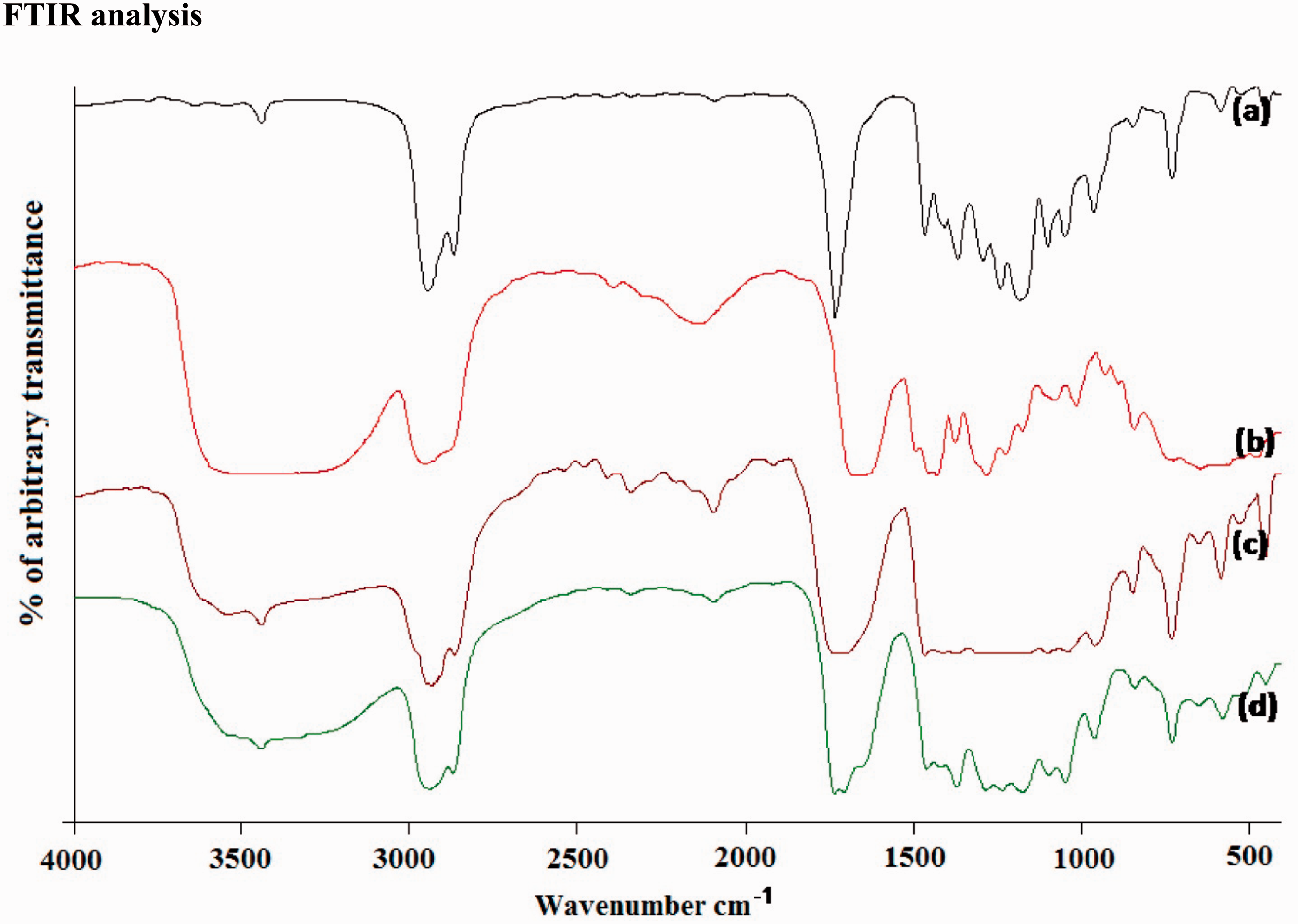
Thermal properties
The thermogram of control (PCL/PVP) and the herbal drug loaded (1%, 2% and 3%) nerve guides fabricated by C-spin technique is shown in Figure 6. The weight loss in the polymer takes place in two stages. At the first stage, the bound and unbound moisture present in the sample are removed. The removal of moisture and the degradation starts from 110℃ to 360℃. In case of herbal-loaded samples, the amount of weight loss percentage increases up to 11–12% at a temperature of 360℃. The increase in weight loss can be attributed to the removal of moisture. Moreover, the polymer also starts to degrade at this temperature. It can be seen from the thermal profiles that the thermal stability decreased on the addition of herbal drug to the matrices. This may be due to the increase in the disordered region in the polymeric matrices. Similar results have been obtained by Elakkiya et al. for curcumin loaded electrospun Bombyx mori nanofibres [30]. The DTG thermogram of the sample used for the study is shown in Figure 7, which indicates that the maximum degradation rate shifts to lower temperature on the addition of herbal drug. This also further confirms the increase in the disordered region in the polymeric nerve guides.
TG thermogram of C-spun control and drug-loaded nerve guides. DTG analysis of C-spun control and drug-loaded nerve guides. (a) PCL/PVP. (b) PCL/PVP/CA (1%). (c) PCL/PVP/CA (2%). (d) PCL/PVP/CA (3%).

Drug release studies
The delivery of therapeutic agents in a sustained controllable manner to the injured site is highly beneficial for healing. The inherent higher degree of fibre alignment achieved in the fibrous mat by C-spin technique aids in nerve regeneration as they mimic the native architecture of peripheral nerves. The cumulative release behavior of natural bioactive molecules from the matrices for various samples is shown in Figure 8. The release was carried out for a period of 24 h and the results showed interesting trends. The release profile shows that there is an initial burst release followed by controlled release for all the samples taken for the study. The initial rapid release can be attributed to the release of triterpenoids along with water soluble PVP molecules. In a biodegradable drug delivery system, the drug release is associated with diffusion as well as degradation of the basic polymeric template. The samples taken for the drug release profile studies are composed of PCL/PVP in the ratio of 80:20. As PVP is a water soluble polymer, the bioactive molecules associated with the polymer get released on contact with the releasing medium. Similar trend is observed for various concentrations of bioactive substances in the nerve guide. However after 15 h duration, the release of drug from the nerve guide slows down and the drug is held by the hydrophobic PCL backbone by entrapment. The cumulative release percent for various drug-loaded samples at 24 h is 40, 46 and 50%.
Cumulative triterpenoid release profiles of different percentages of CA extract-loaded nerve guides.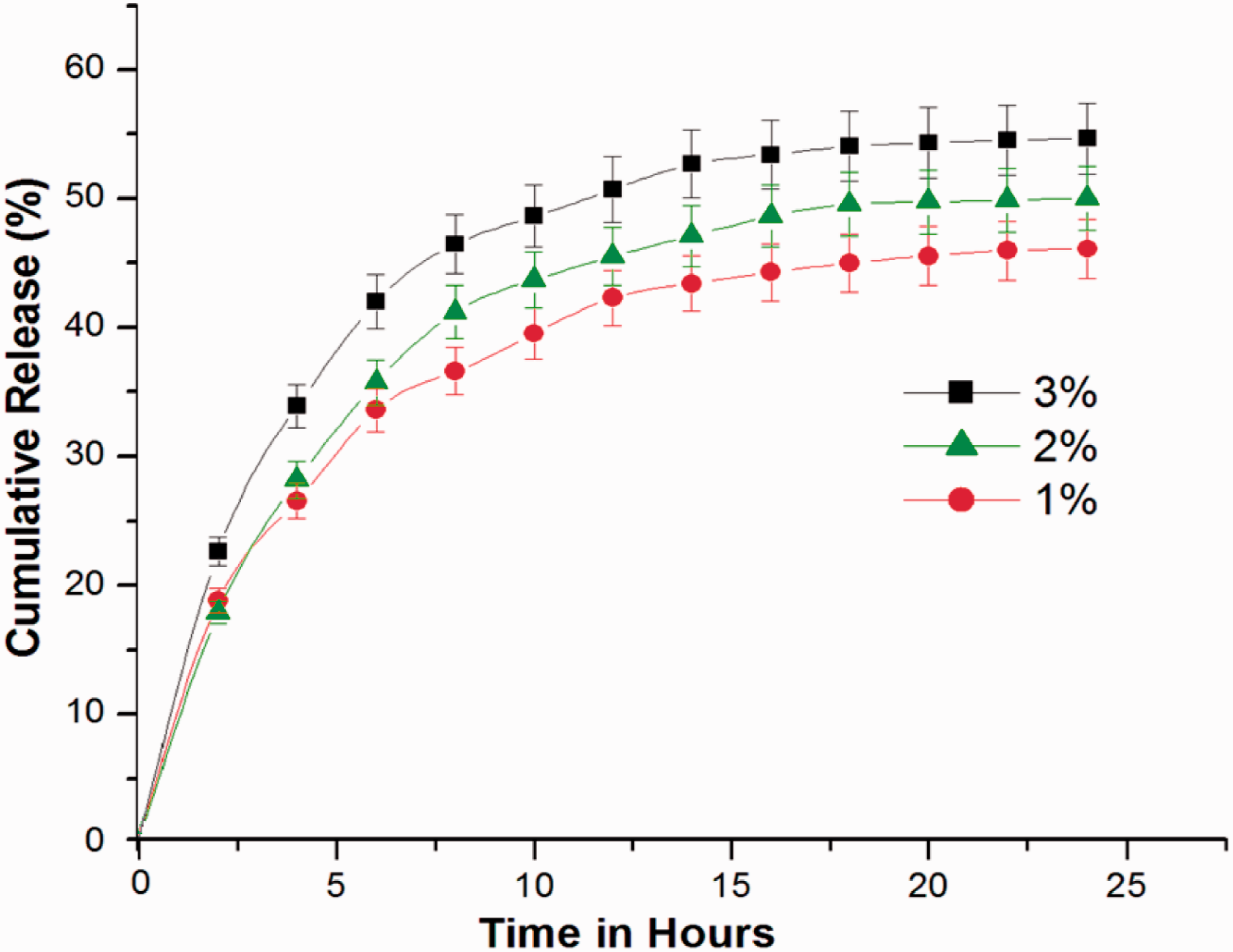
Diffusion constant (n) and regression coefficient of triterpenoids released from the nerve guides calculated by Peppas equation.

CA: Centella asiatica.
Regression coefficients of different mathematical models fitted to the release of triterpenoids from the nerve guides.

CA: Centella asiatica.
Cell viability and morphology
Centrifugal spun ultrafine fibrous mat serves as a suitable matrix for cell adhesion and proliferation owing to its physical property (higher degree of fibre alignment) mimicking the properties ECM present in the peripheral nerves. Biocompatibility of the developed C-spun control and bioactive molecule loaded matrices (3%) was subjected for cell culture studies for 24, 48 and 72 h. The results suggested that the scaffolds were biocompatible and they exhibit higher proliferation compared to the control sample and it is shown in Figure 9(a). From this figure, it can be seen that third day after cell seeding, there was a significant increase in cell proliferation compared to first day which is due to the oriented architecture and the release of loaded bioactive molecules. Soumyanath et al. investigated the neurotrophic activity of the extract of CA in both in vitro and in vivo and found that the multiple components present in the extract of CA accelerate nerve regeneration of damaged nerves [23]. Higher degree of fibre alignment offered by C-spin technology can provide guidance cues for glial cell growth and subsequent axon regeneration. Schnell et al. studied the effect of fibre alignment on glial cell culture and found that aligned PCL, PCL/collagen nanofibres can enhance the glial cell growth, glial cell migration and promote axon regeneration [32]. The representative SEM of glial cells after 24 h is shown in the Figure 9(b) and (c). From this figure, it is apparent that the cells adhered well on the surface of C-spun matrices and the direction of cell growth was parallel to the direction of fiber alignment. The alignment of the cells along the fiber morphology suggests that better infiltration of cells can be achieved by using C-spun matrices.
(a) MTT assay of glial cells on PCL/PVP and CA-loaded PCL/PVP C-spun matrices. (b) SEM micrograph of glial cell attached on CA-loaded C-spun PCL/PVP matrices. (c) Magnified SEM images of a cell on C-spun CA-loaded PCL/PVP matrices depicting the orientation of cell in line with fiber orientation.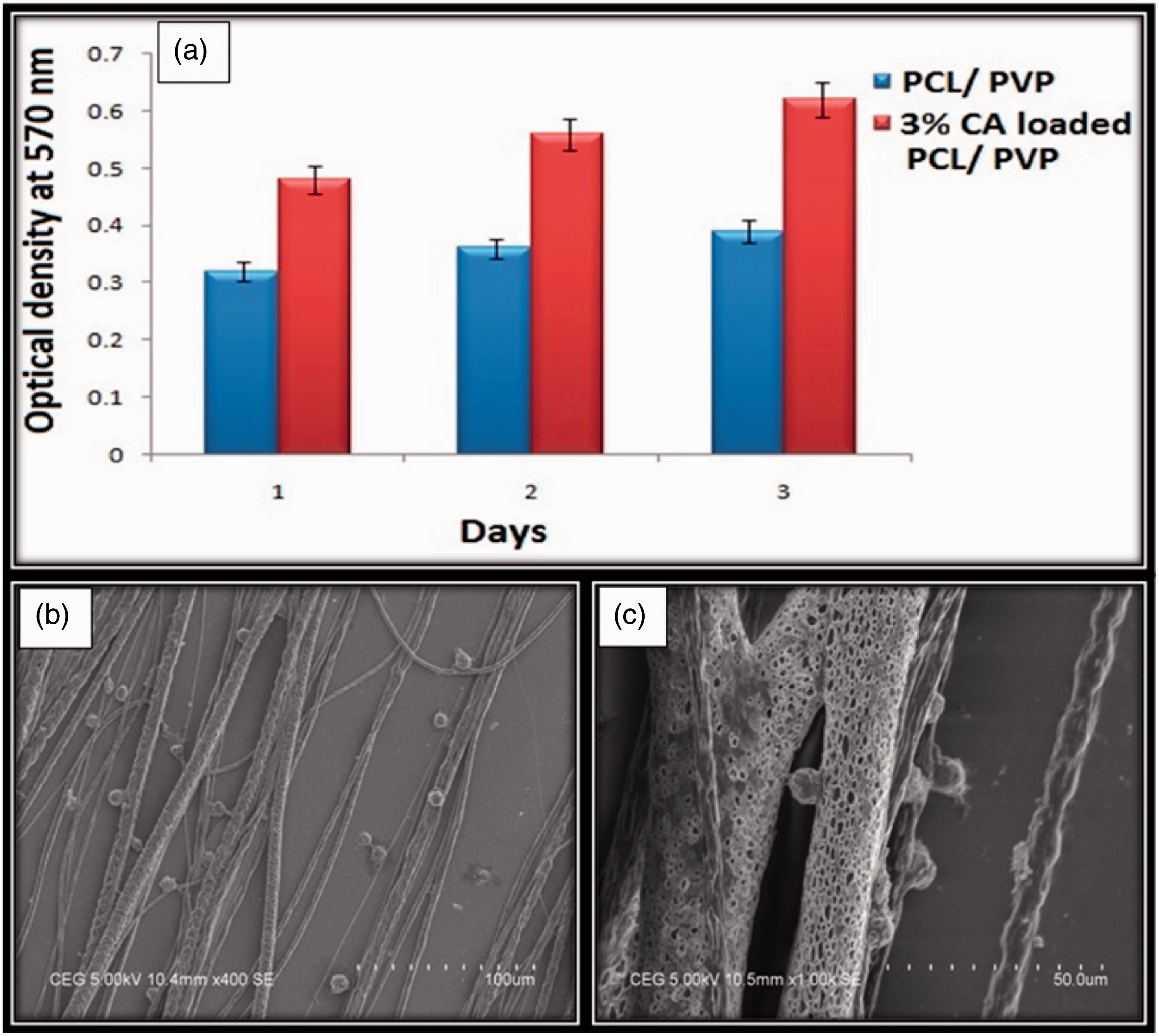
Conclusion
The in-house-developed centrifugal spinning system was used to develop highly aligned nerve guides. Herbal drug-loaded nerve guides have shown promising potential in tissue regeneration. In the present work, triterpenoids isolated from CA was loaded in the developed nerve guide. Higher degree of fibre alignment has been achieved. The drug release studies suggested that the herbal drug can be retained in the matrices even after a period of 24 h. Cell culture studies confirm that the developed matrices were biocompatible and can be used for guided tissue regeneration which plays a pivotal role in nerve tissue engineering.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge the Indian Council of Medical Research (ICMR) for funding (Proceeding no: 5/20-6(Bio)/11-NCD-I).
