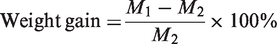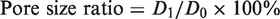Abstract
Polymer chains were introduced to esterified hydroxyapatite particle surface with methacrylic acid for modification and subsequently modified particles were immobilized onto PET filter fabric via heat fusion method to impart adsorption property. The polymerization process of methyl methacrylate on esterified hydroxyapatite surface was optimized and effects of treating time, temperature, particle size, and concentration on immobilization ratio and pore size of filter fabric were discussed. FTIR were applied to characterize the structure of modified hydroxyapatite and treated PET filter fabric. TG was used to determine average chain length of grafted polymer on particles. The results showed that polymer chain with length of 13.57 (mole ratio to hydroxyapatite) was introduced to particle surface and modified particles were embedded onto PET filter fabric with a hydroxyapatite immobilization ratio of 63.6% and pore size of 102.3 µm under optimum conditions. Cadmium adsorption amount of 118.4 mg/g was achieved at the cadmium concentration of 392.8 mg/L, implying that PET filter fabric has adsorption ability toward heavy metal ions.
Introduction
Filter fabric is a traditional industrial textile with mature manufacturing techniques and large variety of types [1]. PET filter fabric is one of the most widely used industrial filter fabrics for water treatment with excellent mechanical properties. However, traditional filter fabric mainly aims to separate solid particles from aqueous solution or gas stream without any other functionality. It is of great necessary to develop functionalized filter fabrics [2]. Hydroxyapatite (HAp) is a promising heavy metal adsorption agent in water treatment because of its huge specific surface area and special crystal structure [3,4]. As HAp is often in the shape of power and difficult to realize its functionality directly, many works have been conducted to prepare HAp–polymer composite due to its poor machining property. As chitosan with natural hydroxyapatite derived from Thunnus Obesus bone (chitosan/HAp), chitosan grafted with functionalized multiwalled carbon nanotube, in addition to HAp (f-Multiple-Wall-Carbon-Nano-Tube-g-chitosan/HAp) scaffolds, was prepared for the first time via freeze-drying method in Jayachandran Venkatesan's work [5]. Besides, Peter X. Ma prepared highly porous poly(l-lactic acid)/hydroxyapatite composite scaffolds for bone tissue engineering [6]. There are many methods for preparing inorganic particle incorporated composites. Inorganic–organic films were fabricated by evaporation of solvent with different percentage of cellulose acetate and HAp in Khalil Azzaoui's work [7]. Alginate bioencapsulating nanohydroxyapatite composite was synthesized in Kalimuthu Pandi's work for defluoridation studies in batch mode [8]. Most of these methods need lots of chemical reagents or complicated processes. Heat fusion is a physical method for treating materials with seldom any reagents and simple procedures [9]. In the present work, hydroxyapatite particles were surface grafted with polymer chain and subsequently immobilized onto the surface of PET filter fabric via heat fusion method to impart heavy metal adsorption property.
Experimental
Materials
PET filter fabric with pore size of 188.5 μm was supplied by Dalian Hualong filter fabric Co. Ltd. and HAp was purchased from Nanjing Aipurui nanomaterial Co. Ltd. Aqueous ammonia, sodium hexametaphosphate, methacrylic acid (MAA), methyl methacrylate (MMA), hydroquinone, toluene-p-sulfonic acid, ammonium persulfate, acetone, cadmium nitrate, and cadmium nitrate standard solution were provided by Tianjin Kermel chemical reagent Co. Ltd. Ultrasonic processor (FS-600, Shanghai Shengxi ultrasonic facilities Co.Ltd.) and nano-microfluidics dispersion (M-100P-UL-CE, MFIC company) were used for dispersion. Atomic adsorption spectrophotometer (HG9600A) was from Shenyang Huaguang precision instrument Co. Ltd. ZETA potential diameter analyzer (Delsa Nano C Particle) was provided by Bechman Coulter Inc.
Modification of HAp
Esterification of HAp with MAA was firstly carried out with 40% of MAA, 0.5% of hydroquinone, and 8% of toluene-p-sulfonic acid in acetone under 65℃. Subsequently, certain amount of MMA was added into esterified HAp dispersion and stirred for 1 h at a speed of 180 r/min. Ammonium persulfate was quickly added and kept stirred under fixed time and temperature after sealed up. Weight gain of particles was applied to determine the results of polymerization according to equation 1. PMMA polymer chain introduced onto HAp particle surface was used to connect particles onto PET filter fabric after melting under high temperature. Polymerization ratio and chain length of PMMA chain was discussed.
Dispersion of modified HAp
Modified HAp particles were suspended in sodium hexametaphosphate aqueous solution with pH adjusted to 8–10 for ultrasonic dispersion and subsequently dispersed with microfluidics under 15,000 psi. Different dispersion conditions were applied to obtain modified particles with different sizes.
Immobilization of modified HAp
Modified HAp dispersion was uniformly sprayed onto PET filter fabric surface and carried out with heat fusion method at temperature of 240℃, particle concentration of 10 g/L for 120 s after predried under 130℃ for 5 min. The sequence of the treatment is illustrated in Figure 1. Immobilization ratio of particles was calculated with equation (2). Heat fusion is known to be a welding process used to join two different pieces of a thermoplastic, involving heating both pieces simultaneously and pressing them together. In this study, the thermoplastic PET filter fabric and the thermoplastic polyester chains on the surface of HAp particles are pressed together upon heating using heat fusion method.
Process of heat fusion method.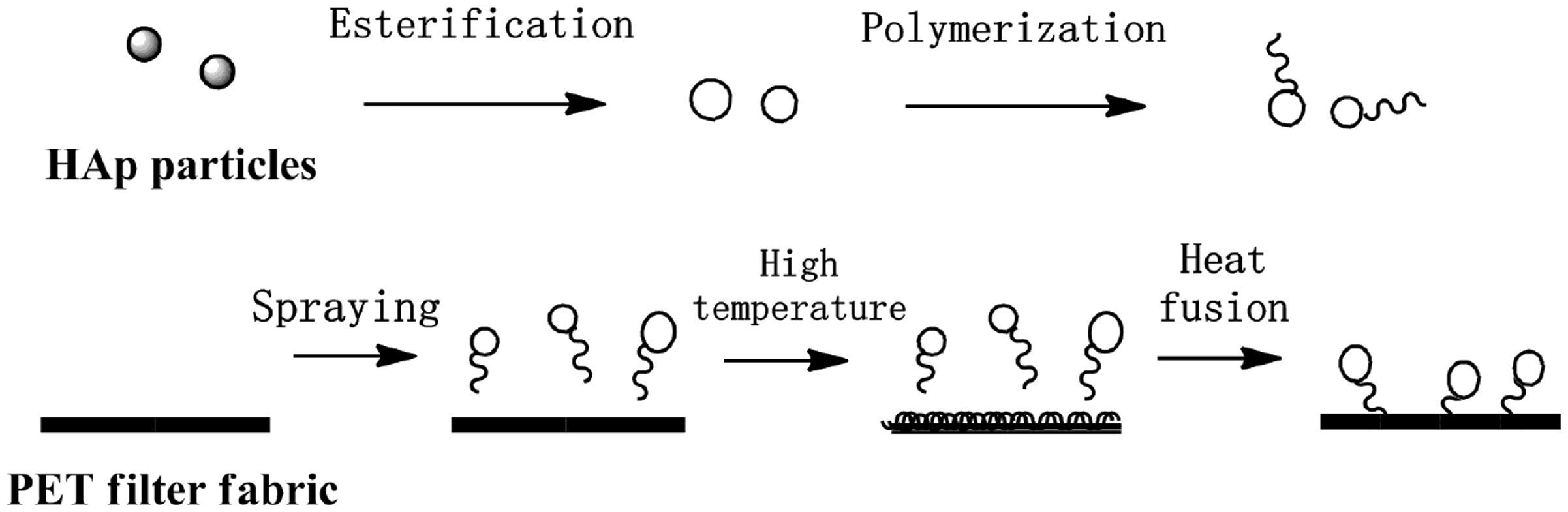
Pore size test. Pore size of PET filter fabric was tested with bubble pressure test method [10] and the pore size could be calculated according to the equation (3) and pore size ratio (ratio of the pore size of treated filter fabric to pristine PET filter fabric) could be obtained from equation (4)
Characterization
Infrared spectrophotometer (Nicolet 6700, Thermal Fisher Scientific Inc.) was applied to characterize the structure of samples. The treated samples under optimum conditions were observed using scanning electron microscope (SEM) (JSM-6460LV) from Japan Electron Optics Laboratory Co. Ltd. Simultaneous Thermal Analysis (STA PT1600) was provided by Linseis Inc.
Adsorption property test
Certain amounts of treated materials under optimum conditions were suspended in cadmium solution for adsorption under 25℃ for 24 h. Removal ratio and adsorption amount of Cd2+ were decided by the decrement of concentration of Cd2+ in solution obtained with atomic adsorption spectrophotometer and calculated with equations (5) and (6) below
Results and discussion
Modification of HAp
Effect of monomer amount on polymerization
Figure 2 presents the relationship between amount of monomers and weight gain of particles. It could be found that weight gain of particles increases with amount of monomers and reach maximum of 30.5% when mole ratio of HAp and MMA was 1:15. Further increase of monomer amount leads to decrease of weight gain. It is believed that both copolymerization and homopolymerization occurred in the free radical polymerization process and copolymerization dominates in the reaction in low concentration of monomers. But when monomer concentration is high enough, homopolymerization would prior to copolymerization and homopolymers would hinder process of copolymerization, leading to decrease in weight gain [11].
Effect of monomer amount on polymerization.
Effect of initiator amount on polymerization
It could be learned from Figure 3 that weight gain of particles increased from 8.83% to 30.5% with initiator amount increasing from 4% to 12%, which is thought to be that large amount of radicals could be produced in a short time and transferred to unsaturated double bonds of esterified particles for polymerization. As initiator amount further increases above 12%, chances of chain termination greatly increase and radicals would transfer to monomers rather than double bonds on the surface of particles, leading to domination of homopolymerization and decrease of weight gain to 24.2% with initiator amount of 20% [12].
Effect of initiator amount on polymerization.
Effect of time on polymerization
As is observed from Figure 4, long time is beneficial for increasing of weight gain because monomers could be grafted onto particle surface in great extent within sufficient time. Excess time makes small difference to weight gain as radicals lose its activity after complete reaction.
Effect of time on polymerization.
Effect of temperature on polymerization
According to Figure 5, weight gain of particles increase quickly with temperature rising and slow down when temperature is above 60℃. Decomposition rate of initiator increased with temperature and more radicals could be produced in a short time, accelerating the copolymerization process. However, more radicals could also lead to more chances of homopolymerization, resulted in low increasing speed of weight gain.
Effect of temperature on polymerization.
Structural analysis
FTIR was adopted to characterize the chemical structure of particles and results are shown in Figure 6. In comparison with the three curves, adsorption band around 2934 cm−1 and 2971 cm−1, which are attributed to ethyl group and methyl group in curve of esterified Hap, suggested that HAp was successfully modified with MAA. Besides, adsorption peak at 1735 cm−1, which can be ascribed to ester group, indicated that polymer chains are introduced to HAp particle surface.
FTIR spectra of untreated and modified particles.
Thermal property test
Inorganic particles would keep its weight constant at temperature below melting point after dried. If organic substances exists on inorganic particles, it often decomposes under high temperature and leads to weight lose. In another words, amount of organic substances on inorganic particle surface could be calculated according to weight lose of particle under high temperature. Figure 7 presents the relative mass change of HAp, esterified HAp, and polymerized HAp with temperature. Slight decrease of mass of HAp is believed to be impurities. Esterified HAp and polymerized HAp start to lose weight from 190℃, which is supposed to be caused by decomposition of polymer chain. On the basis of that, amount of monomers on esterified HAp could be calculated with equation (7).
TG of untreated and modified particles.
The calculation of average chain length is described using the data of weight loss in Figure 7 as an example. According to equation (7), 0.41 mol of monomers react with 1 mol of HAp, indicating an average chain length of 0.41 of esterified HAp and an esterification efficiency of 41% as one HAp particle could connect with one monomer at most. Average chain length of polymerized HAp could be calculated with equation (8) in the same way and it could be obtained that 13.57 mol of polymers were connected to 1 mol of HAp particle surface with a polymerization efficiency of 34.3% when the molar ratio of HAp and MMA was 1:15.
Immobilization of modified HAp
Effect of time on immobilization ratio and pore size ratio
Effect of treating time on pore size ratio and immobilization rate with heat fusion method is shown in Figure 8. It could be found that pore size ratio decrease quickly with time in the first 120 s and then descend in a slow speed, while immobilization rate increases gradually and finally reach equilibrium with approximately 64%. It is believed that abundant particles could be embedded onto fabric surface stably with a certain period of 120 s. Longer time has little impact on immobilization rate and would result in continuous decrement of pore size ratio because of heat.
Effect of time on immobilization ratio and pore size ratio.
Effect of temperature on immobilization ratio and pore size ratio
It could be learned from Figure 9 that immobilization ratio and pore size ratio undergo two stage of change with temperature. In the first stage, immobilization ratio increases quickly and reaches equilibrium at about 64% under 200℃ and further increase of temperature over 240℃ leads to continuous increment of immobilization in the second stage, while pore size ratio performs the contradictory tendency with temperature. In the process of heat fusion, polymer chains on particle surface begin to melt before 190℃ and adhere to fabric surface, resulted in increase of immobilization ratio until equilibrium. As temperature is above 240℃, PET fibers begin to melt and particles could be embedded onto fabric surface more easily, leading to further increase of immobilization ratio. Decrease of pore size ratio is thought to be caused by high temperature and embedded particles, which block up in gaps between fibers.
Effect of temperature on immobilization ratio and pore size ratio.
Effect of particle amount on immobilization ratio and pore size ratio
Figure 10 shows that immobilization rate increases with concentration, while pore size ratio presents an opposite trend. Nearly 11% of decrease of pore size ratio without particle is ascribed to high temperature that melted the PET fibers and narrowed down the gaps. Another 60% of decrease of pore size ratio could be attributed to nearly 90% of immobilization ratio as embedded blocked on the pores and resulted in change of pore size.
Effect of particle amount on immobilization ratio and pore size ratio.
Effect of particle size on immobilization ratio and pore size ratio
Different sizes of particles in average are obtained with different dispersion conditions and it could be learned from Figure 11 that small particle size is favorable for immobilization which is thought to be that decreased particle size helps decreasing the steric hindrance as particles are mainly connected to PET filter fabric surface via melted polymer chain. Decrease of pore size ratio is believed to be caused by immobilized particles.
Effect of particle size on immobilization ratio and pore size ratio.
Effect of chain length on immobilization ratio and pore size ratio
As is observed from Figure 12, length of polymer chain introduced to particle surface is significant in improving immobilization ratio of particles as longer polymer chain have increased possibility of adhering to fabric surface when molten. Immobilized particles lead to decrease of pore size ratio.
Effect of chain length on immobilization ratio and pore size ratio.
Structural analysis
Chemical structure was characterized with FTIR and results are shown in Figure 13. Existence of peak at 564 cm−1, 601 cm−1, and 1026 cm−1, which are attributed to PO43− in FTIR, suggested that modified HAp particles are embedded onto PET fabric surface successfully. Moreover, we could learn that heat fusion treatment does not damage the structure of PET fabric.
FTIR spectra of treated and untreated PET filter fabric.
Morphology analysis
Morphology of untreated PET filter fabric and fabric embedded with HAp was observed with SEM and shown in Figure 14. It could be found that untreated PET fiber had smooth surface with few impurities unevenly distributed on it, while quantity of particles were attached to fiber surface and surface of fibers became rough with pores.
SEM graphs of untreated (a) and treated (b) PET filter fabric.
Adsorption property test
Results of adsorption property of treated PET filter fabric.

Note: HAp, hydroxyapatite.
Conclusion
PMMA chains were introduced onto HAp particle surface successfully under 60℃ for 3 h with initiator amount of 12% and ratio of HAp to monomer of 1:15 via radical polymerization. Modified HAp particles could be embedded onto PET fabric surface under 200℃ for 120 s with the particle concentration of 10 g/L and HAp particle size of 309.5 nm and length of polymer chain of 13.57 (mole ratio to HAp). Immobilization ratio of 63.6% and pore size of 102.3 µm could be achieved under optimized conditions. Results of FTIR and SEM indicated that modified particles were uniformly embedded onto PET fabric surface, and adsorption property test showed that PET filter fabric had good adsorption property with Cd2+.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.