Abstract
In this study, we have extracted bioactive compounds from seabuckthorn (SBT) leaves. Total flavonoid content of SBT leaf extract estimated as rutin equivalent was found to be 116.98 ± 3.06 mg/g of extract. Further, we pre-treated aramid (Nomex IIIA) fabric with triethylene tetramine to increase the wickability of the fabric for better extract uptake. Surface etching on the amine treated fabric morphology was determined by scanning electron microscope analysis. Then, SBT leaf extract was coated using citric acid as cross-linking agent on to aramid fabric by pad-dry-cure method for improved wash durability. Universal attenuated total internal reflection Fourier transform infrared characterization of the SBT coated fabric showed distinct hydroxyl peak at 3305 cm−1, carbonyl peak at 1728 cm−1, and –C–O–C peak at 1083 cm−1. There was no significant difference in the char produced by SBT coated fabric and control samples as estimated using flammability tester. Antimicrobial activity of the SBT coated fabric was assessed by both qualitative (agar diffusion method) and quantitative (percentage reduction test) methods using test organisms. An inhibition zone for Escherichia coli and Staphylococcus aureus was found to be 10.2 mm and 12.3 mm, respectively, by agar diffusion method. Quantitative assessment by percentage reduction test showed a reduction percentage of 96.00% and 93.00% for S. aureus and E. coli, respectively. Results suggested that SBT might be a valuable ingredient for the development of antimicrobial aramid textiles.
Introduction
In the development of fabrics, functional aspects such as antibacterial and UV protection are playing important role [1,2]. Because of their large surface area and ability to retain moisture, textile goods provide an excellent environment for the growth of the microorganisms. Antimicrobial finishing of textile materials aims to protect the users from pathogenic microorganisms or from unpleasant odour generators and to ensure a proper functionality of textiles by preventing rotting or aesthetic changes [3]. An ideal antimicrobial treatment should provide protection against a wide range of bacterial and fungal species, it should be durable to washing, dry cleaning, and hot pressing, it should be compatible with textile chemical finishing processes such as dyeing and it should be cost effective [4]. A number of chemicals such as, triclosan, organometallics, iodophors (substances that slowly release iodine), phenols and thiophenols, metallic salts, antibiotics, heterocyclics with anionic groups, nitro compounds, ureas and related compounds, formaldehyde derivatives, and amines [5,6] have been developed and quite a few are also available commercially [7]. Although the synthetic antimicrobial agents are very effective against a range of microbes and give a durable effect on textiles, they are cause of concern due to the associated side effects, action on non-target microorganisms, and water pollution [8]. Hence, there is a great demand for antimicrobial textiles based on ecofriendly agents [9–11] which not only help to reduce effectively the ill effects associated due to microbial growth on the textile material but also comply with the statutory requirements imposed by regulating agencies.
Natural products such as chitosan, natural dyes, and tulsi leaf extract have been used as antimicrobial finishing on textile materials [12–17]. There is a vast source of medicinal plants with active antimicrobial ingredients. Although there are many natural products rich in antimicrobial agents, the study on their use in textiles is very limited and not well documented. The relatively lower incidence of adverse reactions of herbal products as compared to modern synthetic pharmaceuticals, coupled with their reduced cost, can be exploited as an attractive ecofriendly alternative to synthetic antimicrobial agents for textile applications [9,11]. Recent developments on plant based bioactive agents have opened up new avenues in this area of research.
Seabuckthorn (SBT; Hippophae rhamnoides L., Elaeagnaceae) has been domesticated in several countries such as, India, China, Nepal, Pakistan, Myanmar, Russia, Britain, Germany, Finland, Romania, France, etc. at an altitude of 2500–4300 m [18]. It has been recognized as a versatile nutraceutical crop with diverse uses, from controlling soil erosion to being a source of horse fodder, nutritious foods, drugs, and skin-care products [19]. Different parts of this plant are used in traditional medicine for the treatment of diseases, such as flu, cardiovascular diseases, mucosal injuries, and skin disorders [20,21]. All parts of this wonder plant are considered to be a good source of a large number of bioactive compounds, including carotenoids, tocopherols, sterols, flavonoids, lipids, vitamins, tannins, minerals, etc. [21,22] which contribute to its wide usage as a natural antioxidant and antimicrobial agent [23–25]. Although SBT has been known to possess a numerous beneficial properties, its application on textile for antibacterial property enhancement is not reported.
The present investigation primarily aims at developing an eco-friendly natural antimicrobial finish from SBT leaf for textile application. SBT leaf extract was applied to Nomex IIIA (NIIIA) fabrics and screened for their antimicrobial activity. The fabric coated with SBT was characterized by Fourier transform infrared spectroscopy (FTIR). An extensive study was conducted to assess the antimicrobial effect of SBT leaf extract by employing standard test methods.
Materials and methods
Fabric sample
NIIIA fabric for treatment was purchased from DuPont, USA. Specifications of fabric are blend of 93% Nomex, 5% kevlar, 2% nylon; count: warp – 2/40 s, weft – 2/40 s.
Collection of plant material
The SBT leaves were collected from the hilly regions of the North-West Himalayas (the region lies between latitude 32–36° North and longitude 76–79° East) in the month of September, where the plant grows widely under natural conditions. Plant material was characterized by an ethnobotanist at the Defence Institute of High Altitude Research (DIHAR), Leh, India.
Preparation of extract
Fresh leaves were cleaned thoroughly with distilled water, dried under shade in a clean and dust-free environment. Maceration method was used to prepare the 70% ethanol extract of SBT leaves by soaking the powdered dry leaves with 70% ethanol (1:5 w/v) at room temperature (25 ± 1℃). After 24 h, the supernatant was decanted and the residue was re-soaked in fresh solvent. The process was repeated three times for complete extraction. The supernatant was pooled, filtered through muslin cloth, and centrifuged at 5000 g (Remi PR 24) for 10 min at 4℃. Ethanol content of the hydroalcoholic extract was evaporated using rotary evaporator (Buchi R-124 Labortechnik AG, Postfach, CH-9230, Flawil, Switzerland) at 40℃. Finally, the supernatant solution was lyophilized (Lyophilizer, HITOSICC, Heto-Holten A/S, Denmark) and the dried extract was stored in airtight dark bottle at 4℃.
Determination of total flavonoid content
One millilitre aliquot of appropriately diluted sample solution was mixed with 2 mL of distilled water and subsequently with 0.15 mL of a 5% NaNO2 solution. After 6 min, 0.15 mL of 10% AlCl3 solution was added and allowed to stand for 6 min, then 2 mL of 4% NaOH solution was added to the mixture. Then the mixture was thoroughly mixed and allowed to stand for another 15 min. Absorbance of the mixture was measured at 510 nm versus prepared blank. Rutin was used as standard compound for the quantification of total flavonoid. All values were expressed as milligram of rutin equivalents per gram of sample [26].
Pre-treatment of fabrics
Fabrics (size: 30 cm x 30 cm) were pre-treated at 80℃ for 60 min with 70% triethylene tetramine at the liquor ratio of 15:1. After treatment, the samples were washed in water at 100℃ for 30 min, dried in the oven at 80℃ for 30 min, and conditioned at 25℃ at 65% RH for 60 min in the environment chamber [27].
Scanning electron microscopy analysis
Scanning electron microscopy (SEM) analysis was carried out with a SEM QUANTA 200 3D microscope (FEI Co. Netherlands), in low vacuum mode, in humid atmosphere, at a pressure of 60 Pa, at a distance from the scanning pin of 15 mm, with scanning speed of 0.1 ns per pixel, at scanning depth of 3–4 µm with a Wolfram filament at 20 kV tension and a large field detector [4].
Assessment of hydrophilicity
The hydrophilic behaviour of the treated and untreated fabrics was analysed using wicking test (BS 4554). Fabric strip of 0.5 cm × 8 cm dimension was suspended vertically with its lower edge having contact with distilled water. A spontaneous rise in the height of water in the fabrics due to capillary force was observed. The height in the rise of water for a given time is taken as the direct indication of wicking rate of the fabrics. The rise of water level in the fabric was measured at 20, 40, 60, 120, 180, and 240 s. The wicking height measured at 240 s was considered for the assessment of hydrophilicity of the fabrics [28].
Coating onto fabrics
SBT extract was applied on the fabric to a wet pick up of 80% using the “pad-dry-cure” method. The NIIIA fabric (size: 30 cm × 30 cm) was immersed in the solution containing SBT (5% w/v) and 2% citric acid binder for 5 min and then it was passed through a laboratory padding mangle (RGE make), which was running at a speed of 20 r/min with a pressure of 1.5 kgf cm−2 to remove excess solution. After padding, the fabric was air-dried and then cured for 3 min at 140℃. The treated samples were then evaluated for antimicrobial activity.
Bacterial strains
Escherichia coli MTCC 25922 as a representative of Gram-negative bacteria and Staphylococcus aureus MTCC 25293 as a representative of Gram-positive bacteria were used for this study as both are reference strains used for antimicrobial susceptibility testing according to AATCC standard method. The strains were cultured on nutrient agar (Hi-Media, Mumbai, India) and incubated aerobically at 37℃ overnight.
FTIR characterization
The functional group and absorbency of the SBT-coated fabric was determined using Universal attenuated total internal reflection FTIR (UATR-FTIR) spectrophotometer. The spectra of the NIIIA and SBT-coated NIIIA fabric samples were recorded using Perkin Elmer (Spectrum 100) spectrophotometer in the range of 4000–650 cm−1. UATR consists of Zn-Se single crystal that collects the spectra from the fabrics of dimensions 10 mm × 10 mm placed on to it. Pressure was applied to the samples to ensure a good contact between the sample and crystal to prevent the loss of incident IR radiation. The spectra were obtained for each sample at a resolution of 4 cm−1 with 32 scans.
45˚ flammability test (ASTM D1230)
Dried NIIIA fabric sample of about 2 × 6 inches of fabric surface was inserted in a flammability tester at an angle of 45°. A standardized flame (16–18 mm) as per ASTM standard [29] was applied to the surface near the lower end of the fabric for 12 s, and the distance of flame to proceed up the fabric after 12 s was recorded.
Qualitative and quantitative antimicrobial activity assessment
Antibacterial activity was evaluated by both qualitative and quantitative test methods. The following are the descriptions of test methods employed for this study. Qualitative assessment was made by agar diffusion method (AATCC 147-2001). Treated and untreated control fabric samples placed in intimate contact with AATCC bacteriostasis agar, which has been previously inoculated (Mat culture) with inoculums of test organisms. After incubation, a clear area of uninterrupted growth underneath and along the side of the test material indicates the antibacterial effect of the fabric.
Quantitative assessment was done by percentage reduction test (ASTM E2149). Specimens of the test material were shaken in a known concentration of bacterial suspension and the reduction in bacterial activity in standard time was measured. The efficiency of the antimicrobial treatment was determined by comparing the reduction in bacterial concentration of the treated sample with that of control sample expressed as a percentage reduction in standard time.
Wash durability analysis
Washing was carried out as per test no: 1 of IS: 687-1979 by using a neutral soap (5 gpL) at 40 ± 2℃ for 30 min, keeping the material: liquor ratio at 1:50, followed by rinsing, washing and drying. After drying, the test samples were assessed for antimicrobial activity using ASTM E2149 procedure up to 15 laundering cycles [30].
Results and discussion
Extraction of SBT, determination of total flavonoid content and application on to NIIIA fabrics
In this study, SBT was extracted using 70% ethanol by maceration method and the yield of the extract was found to be 28.03%. Dry add-on percentage of the NIIIA fabric coated with 5% SBT extract solution with wet pick up of 80% using the “pad-dry-cure” method was found to be 5.1%.
As a part of chemical composition analysis, total flavonoid content of the extract was determined by the colorimetric method. The total flavonoid content was found to be 116.98( ± 3.06) mg rutin equivalents/g of extract. In our previous study, some of its flavonoid constituents, kaempferol, quercetin, isorhamnetin, and quercetin 3-O-galactoside (Figure 1) were found in the range of 0.2–2.41 mg/g of extract as quantified by reverse phase high performance liquid chromatography [23–25]. The ratio of kaempferol, quercetin, isorhamnetin, and quercetin 3-O-galactoside contents in the SBT extract was found to be 1.0:1.3:4.3:12.1, respectively. Flavonoids form a class of benzo-γ-pyrone derivatives including flavones, flavanes, flavonols, anthocyanidines and catechins. They possess a wide spectrum of biological activities such as anticancer, antibacterial, antifungal, antiviral, spasmolytic, hypoglycaemic, antihistaminic and radioprotective potential [31–33]. Some of these properties derive from the free radical-scavenging activities of flavonoids. Therefore there are many reports relating to the reactivities of flavonoids with active oxygen species. Recent interest in these substances has been stimulated by the potential health benefits arising from their antioxidant and antimicrobial activities.
Structure of the quantified phenolic compounds in Seabuckthorn leaf extract: (a) quercetin 3-O-galactoside, (b) quercetin, (c) kaempferol and (d) isorhamnetin.
Effect of amine treatment on the hydrophilicity of the fabric
The effect of amine treatment on the hydrophilicity of the fabrics was determined by measuring the wicking height at different interval of time (20–240 s). Graph is plotted using wicking height and square root of time as shown in the Figure 2. It is clear from the figure that, wicking height of the treated fabric considerably improved in comparison with the untreated fabric. This indicates that the amine treatment enhanced the formation of polar species on the surface of the fabric due to etching making it hydrophilic. The existence of surface etching on the amine treated fabric morphology was determined by SEM. The SEM micrographs of the untreated and treated fabrics are shown in Figure 3a and b, respectively. SEM micrograph shown in the Figure 3b confirms surface etching on amine treated fabric. Similar results were reported by Chyung-Chyung and Cheng-Chi [27] who showed that the amine treatment for Nomex yarns enhanced the wicking height and dye uptake.
Wicking rate of treated and untreated fabrics. (a) SEM picture of NIII A fabric (control). (b) SEM picture of amine treated NIII A fabric.

FTIR characterization
UATR FTIR, a surface sensitive technique was used to study the chemical modification on the surface of the treated fabrics. The IR spectrum of the control and coated fabric samples are shown in Figure 4. Main characteristic peaks of the base fabric at 1651 cm−1, 1593 cm−1 and 1253 cm−1 correspond to amide I, amide II combined with aromatic and amide III, respectively [34]. The SBT-treated NIIIA fabric has characteristic peak at wavelength of 3305 cm−1 for hydroxyl, at 1728 cm−1 for carbonyl and at 1083 cm−1 for –C–O–C group [35]. These characteristic peaks proved the coating of the SBT phenolic compounds onto the NIIIA fabric as the control NIIIA fabric does not show such characteristic peak. Further, there is much decreased transmittance at 1728 cm−1 control NIIIA fabric. The prominence of this feature may be ascribed to the relatively high content of carboxylic acid and hydroxyl containing compounds in SBT.
FTIR spectra of control and coated fabrics (4000–1000 cm−1).
Similar results were reported by Jain and Mittal [36] who showed that the plant extract containing flavonoids showed the characteristic hydroxyl and carbonyl groups at 3307 cm−1 and 1641 cm−1, respectively. It was also reported by Rajeev et al. [37] that the plant extract containing rich flavonoid content shows the bands at 3351 cm−1 and 1714 cm−1. Whereas the fabric without SBT coating does not have these characteristic peaks. Further, it was confirmed that peaks at ∼3500 cm−1 correspond to –OH stretching and peaks at 1740 cm−1 correspond to C = O groups by Chandana and Sanjeeb [38].
Flammability test
This test method is used to measure and describe the properties of fabrics in response to heat and flame under controlled laboratory conditions. Char length of the samples estimated using flammability tester for untreated control sample was found to be 22.17 (±0.76) mm and for treated sample 21.96 (±0.78) mm. Obtained result found no significant differences in the area of char produced by control and treated samples. It indicates that the treatment has no effect on the flammability parameters of the fabric.
Qualitative and quantitative antimicrobial activity assessment
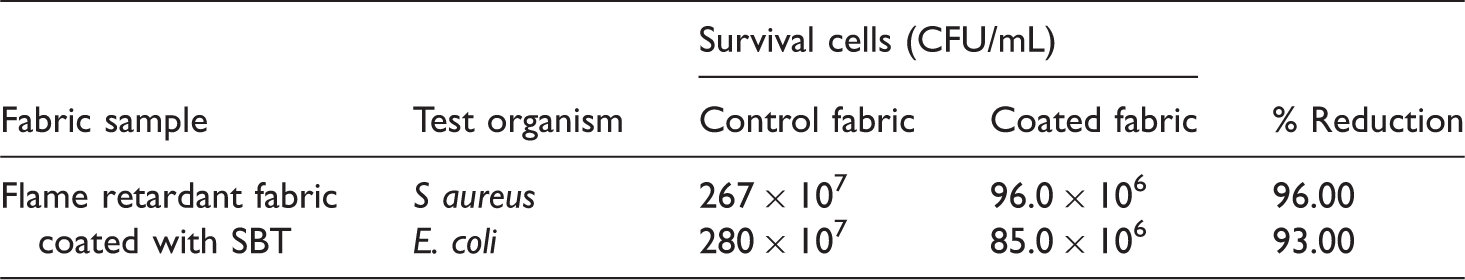
The results of agar diffusion method against the standard test organisms E. coli (Gram negative) and S. aureus (Gram positive) are given in Figure 5a and b. There was a clear zone of inhibition around the fabric tested with SBT against both the test organisms in contrast with control fabric which allowed the growth of organism. The agar diffusion showed a zone of inhibition which indicated that the antimicrobial activity of SBT coated sample exhibited a zone inhibition of 10.2 ± 1.1 mm for E. coli and 12.3 ± 1.3 mm for S. aureus.
Photograph showing zone of inhibition by disc diffusion method. (a) E. coli and (b) S. aureus.
Percentage reductions of both the test organisms.
Wash durability
Wash durability analysis of coated fabric.

Conclusion
In this work, we extracted bioactive compounds from SBT leaves using 70% ethanol by maceration method. We focused on studying the antimicrobial property of NIIIA fabric coated with SBT extract. The SBT-coated fabric showed a high degree of bactericidal activity against test organisms E. coli and S. aureus used in this study. In the FTIR spectra, it showed distinct hydroxyl, carbonyl and –C–O–C peaks. Wash durability study conferred that the SBT-treated fabrics sustained antibacterial activity until 15 washes. In future, we are interested in further increasing the durability of the SBT coated NIIIA fabric using various cross-linking agents, so that the antimicrobial property of SBT is retained in the fabric even after several washes and thus enhancing durability. We are also interested in studying the fungicidal activity of SBT. By this way, we desire to develop an eco-friendly antimicrobial aramid textile with a novel natural antimicrobial agent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

