Abstract
Bismuth vanadate-coated cotton fabric was synthesized by a chemical bath deposition method at low temperature (≤100℃) and characterized by using scanning electron microscopy, X-ray photoelectron spectroscopy, X-ray diffraction and UV-V is diffuse reflectance spectroscopy. Its photocatalytic activities were evaluated by decomposition of C.I. reactive blue 19 in aqueous solution under visible light irradiation. The as-prepared composite possesses excellent photocatalytic activity for the degradation of liquid contamination especially in static system due to its large specific surface area. The reduction of total organic carbon (about 48.0% after 4 h of irradiation) showed that the mineralization of C.I. Reactive Blue 19 over the bismuth vanadate-coated cotton fabric is realizable. Moreover, the preparation of the composites is convenient for potential practical application. The formation mechanism of bismuth vanadate on the fabric was also discussed preliminarily.
Introduction
To date, TiO2 has been well-proven as the best photocatalyst because of its low cost, high efficiency in decomposition of organic compounds and photostability [1]. However, the major drawback of pristine TiO2-based photocatalyst is the large band gap of 3.2 eV that responds only to ultraviolet (UV) light, which takes up only ca. 4% of the sunlight energy while visible light accounts for ca. 43% [2]. Bismuth vanadate (BiVO4) as a promising eco-photocatalyst appears in three main crystalline phases: monoclinic scheelite, tetragonal zircon and tetragonal scheelite structures. Recently, monoclinic BiVO4, relative to other semiconductor materials, has attracted much attention due to its narrow band gap (2.4 eV) and high photocatalytic activity for oxygen evolution in aqueous solutions under visible light irradiation [3,4]. It has been used for a wide range of applications including gas sensors, solid-state electrolytes, positive electrode materials for lithium rechargeable batteries and nontoxic yellow pigment for high-performance lead-free paints [5]. At present, most BiVO4-concerned research on the degradation of organic compounds are only concentrated on its powder applications. It is advantageous that suspended particles can be well-devoted to water purification for the resulting highly active surfaces. Nevertheless, technical problems may arise from undesirable particle agglomeration and the necessary particle-fluid separation. Therefore, the continuous support media with high surface areas such as fibers and fabrics is proved here to be a promising composite component for the catalyst recycling.
The development of functional textiles based on the immobilization of multi-phased materials on fabric surface has recently received growing interest from both academic and industrial fields [6]. In fact, BiVO4-fiber composite [7] has been made for degradation of organic dyes, which shows the similar photocatalytic activities of pure BiVO4. However, compared with synthetic fabric, cotton fabric provides the large surface with strong adsorptive capacity, low cost and is easy to shape, which is suitable for loading BiVO4. And the strong affinity of cotton fabric for organic contaminants makes it easier for BiVO4 to degrade those contaminants. Although many traditional methods have been proved as effective techniques for the synthesis of BiVO4 crystallites, such as aqueous process [8−10], hydrothermal process [4,11,12], organic decomposition method [13], solution combustion synthesis method [14], ultrasonic spray pyrolysis [15], flame spray pyrolysis [16] and solid-state reaction method [17−19], none of them is available to cotton fabric since the temperature is too high to sustain for finishing the phase transition of BiVO4. Fortunately, the method of chemistry bath deposition (CBD) [3], providing a mild condition for textile fabric, has been raised for BiVO4 particles growth showing an advantage over the traditional methods due to (a) good adherence characteristic of the grown layers; (b) the simplicity of the technique; (c) the low temperature needed and (d) the possibility of reusing the precursor solutions [3,4,20]. The CBD process involves a simple and repeatable anchoring reaction of the BiVO4 micro-nano phase on the surface of cotton fabric.
Herein, we demonstrate a facile method for preparing BiVO4/cotton composites at relatively low temperature in one-step process by CBD method. In fact, this method is important because of its potential application in industry. Our study performed accurate characterization of the structure and properties of the composite materials. The photocatalytic activities of monoclinic BiVO4-modified cotton fabric were determined by measuring the photocatalytic degradation of adsorbed C.I. reactive blue-19 (RB-19). And in order to prove the mineralization of the dye, the removal capacity of chemical oxygen demand (COD) and total organic carbon (TOC) in the degradation of RB-19 were investigated using BiVO4-coated cotton fabric. In addition, the BiVO4 developing mechanism on the fabric surface provides an understanding of details in the composite materials.
Experimental procedure
Materials
Bleached plain woven cotton fabric weighting 140.0 g•m−2 was used throughout this work. Triple distilled water was used in all experiments. Bismuth nitrate pentahydrate (Bi(NO3)3·5H2O, 99%), sodium metavanadate (NaVO3, 98%), ethylenediaminetetraacetic acid (EDTA, 99%) and chemicals were of analytical purity from Sino-pharm Chemical Company. The aqueous solution of sodium hydroxide (NaOH, 2 mol/L) was prepared prior to use.
Preparation of BiVO4 suspension
Two solutions were prepared. Solution A: Bi(NO3)3·5H2O (4 mmol) and EDTA (1.5 mmol) were added to a buffer phosphate solution (50 ml) with vigorous stirring (200 r/min) for 20 min. Solution B: NaVO3 (5 mmol) and water (5 ml) were added to another buffer phosphate solution (50 ml) with NaOH (10 ml) and mixed well by a magnetic stirrer (200 r/min). The two solutions were then mixed together and stirred vigorously for 30 min.
Synthesis method
The cotton fabric was treated with acetone for 30 min in ultrasonic surroundings to remove impurities like wax, fat, etc. and dried at room temperature. For the impregnation, the cotton fabric pretreated, as described above, was then immersed in the BiVO4 suspension at 90℃ for 10 h. The extracted fabric was then placed in boiling water for 1 h to remove the unattached BiVO4 from the cotton surface and dried in the oven at 50℃. A second sample was obtained by treating the cotton fabric in the same way, except the BiVO4 impregnating phase. And the conventional powdered BiVO4 particles were prepared by a hydrothermal process according to the literature [21].
Characterization techniques
The morphology of the pure and BiVO4-coated cotton fabric was obtained on a JSM-5600LV scanning electron microscopy (SEM). The chemical compositions of the samples’ surface were examined by X-ray photoelectron spectroscopy (XPS) on a Kratos XSAM800 instrument. The X-ray diffraction (XRD) patterns of the samples have been collected by means of a D/Max-2550 PC diffractometer in θ-2θ configuration to identify the crystal phase and structure, using monochromatized Cu Kα radiation (λ = 0.154056 nm). The UV-V is reflectance spectra were carried out at room temperature on a Lambda 950 UV-V is spectrometer to detect UV absorption intensities.
Evaluation of photocatalytic properties
C.I. RB-19 shows a maximum absorbance at 593 nm that is used to monitor the photocatalytic degradation of RB-19 presented in Figure 1 and Table 1. This dye was chosen due to its high chemical resistance to degradation by conventional procedures, which is attributed to its anthraquinone chromophore. The photoactivity of the BiVO4-coated cotton fabric was evaluated by the degradation of RB-19 under visible-light irradiation, which was carried out on at 295 K, using a 400 W metal halide lamp (λ > 420 nm). Experiments were performed as follows: 0.5 g of the as-prepared BiVO4-coated fabric was immersed in 200 ml RB-19 solution which was prepared with a concentration of 15 mg/L and pH ≈ 5. The photochemical reactor magnetically stirred during the reactions. The concentrations of RB-19-detected each 10 min periods were monitored with a Hitachi U3310 UV-V is spectrophotometer in terms of the absorbance at 593 nm during the photodegradation process. The ratio (C/C0) of RB-19 concentrations after reaction for some time (C) and at the initial (C0) was used to evaluate the photocatalytic performance. COD of RB-19 was estimated after the treatment using the K2Cr2O7 oxidation method. TOC was measured with a Shimapzu TOC-VcpH analyzer.
The chemical structure of C.I. reactive blue 19. General characteristics of reactive blue-19 (RB-19).

Wash fastness
The wash fastness of the treated cotton fabric was tested using the method of ISO 105 C06 A1S [22]. SDC multifiber strip fabric as an adjacent material was used. The samples were sequentially washed five times.
Results and discussion
SEM image
Figure 2 shows the SEM images of the cotton fabric before and after surface coating by BiVO4 photocatalyst. From Figure 2(a) and (b), it can be seen that the pure cotton fabric has the smooth folds running parallel to the elongation direction of the fiber. Otherwise, Figure 2(c) shows that large compact particles of irregular shape aggregate on the surface of cotton fabric through low magnification, which refer to BiVO4 proved in section 3.2. There are a fraction quantity of small irregular particles adhering to the large particles, which is similar to the literature reported in [23]. And at higher magnification in Figure 2(d), it clearly shows that treated fibers are mostly covered by plate-like micro-nano-sized, about 200–1000 nm in size, BiVO4 particles with a sharp brim. The results indicate that BiVO4 has chemical affinity to cotton fibers by CBD method.
SEM images of (a) pure cotton fabric, (b) enlarged view of pure cotton fabric, (c) BiVO4-coated cotton fabric and (d) enlarged view of the BiVO4-coated cotton fabric.
XPS spectra
The XPS spectrum is a useful tool for identifying the chemical states of various hybrid species on the samples’ surface. Figure 3 shows the high-resolution XPS spectra of pure cotton fabric and BiVO4-coated cotton fabric. Results indicated that the modified cotton fabric contained Bi, O, V and C element, and the pure cotton fabric just contained O and C elements. The spin-orbit split of Bi 4f 7/2 and Bi 4f 5/2 signal peaks are well demonstrated by two curves at approximately 158.8 and 164.0 eV, respectively, corresponding to Bi4+ in the sample (Figure 3(b)) [24]. The V2p orbit of 2 p 3/2 and 2 p 1/2 show peaks at 516.5 and 523.6 eV, respectively, which are attributed to V5+ of BiVO4 particles (Figure 3(c)) [25]. In this respect, the observed O1s peak at 531.8 eV can be attributed to the OH on the surface of cotton fabric, which covers the narrow peak of the lattice oxygen in crystalline BiVO4. However, an unapparent hump can be still observed at 529.9 eV for the lattice oxygen, since the board peak shows slight asymmetry (Figure 3(d)).
The high-resolution XPS analysis of (a) BiVO4-coated cotton fabric and (b) Bi 4f, (c) V 2 p and (d) O 1 s for BiVO4 on the surface of cotton fabric.
XRD patterns
The crystal structure of BiVO4, BiVO4-coated fabric and pure cotton were examined by XRD as shown in Figure 4. The XRD patterns revealed sharp peaks, indicating the high degree of crystallinity. Curve (b) reports two broad peaks and one intense peak at 14.73°, 16.45° and 22.619°, respectively, which comprise the typical XRD pattern of cotton fabric [26]. According to the pattern of pure cotton fabric, new peaks were observed obviously to the characteristic peaks of BiVO4 from curve (c). The XRD pattern of modified cotton fabric matches closely with the characteristic peaks of monoclinic scheelite BiVO4 structure according to the database of the Joint Committee on Powder Diffraction Standards (JCPDS 14-0688). Major characteristic peaks at 18.7°, 28.8° and 30.5° were observed. In order to eliminate the interference of the characteristic peaks of cotton fabric, the BiVO4 particles left in the solution were collected for the XRD pattern described in curve (a). The presence of the tetragonal zircon-type phase for the samples reported here is ruled out since the characteristic strong peak at 2θ = 24° was not observed in any of the samples prepared [3]. Curve (a) conforms to the peak positions of the standard monoclinic phase BiVO4. This formation of single-phase monoclinic BiVO4 in each samples implied that the temperature of 90℃ was appropriate.
XRD patterns of (a) pure BiVO4, (b) pure cotton fabric and (c) BiVO4-covered cotton fabric; the black vertical lines superimposed to XRD patterns represent the peak positions of the standard monoclinic phase BiVO4 (JCPD file no: 14-0688).
BiVO4 developing mechanism
In the synthesis of BiVO4-coated cotton fabric by chemical bath deposition reaction, Bi(NO3)3 and NaVO3 are used as raw materials. At the beginning of reaction, Bi3+ and VO3- will be produced when Bi(NO3)3 and NaVO3 are dissolved in the buffer phosphate solutions A and B, respectively. While the two solutions containing Bi3+ and VO3- mentioned above are mixed, BiVO4 will be produced immediately. Widely accepted, BiVO4 will produce with a quick kinetic growth process when high content of free Bi3+ and VO3- ions collide, which results in the large particle size of BiVO4 [27]. And the crystal growth speed was strongly dependent on the concentration of Bi3+ and VO3- ions. However, if the EDTA were introduced into the reactive system, the Bi3+ in the buffer solution would react with EDTA to form stable chelate complexes via the electrostatic attractions, which greatly reduces the free Bi3+ concentration in the solution. Meanwhile, with addition of the NaVO3-contained buffer solution, the Bi3+ would be released slowly from chelate complexes to shape the BiVO4 particles. In the subsequent chemical bath deposition process, the chelate complexes would be dissociated gradually to provide a small amount of free Bi3+. Because of the release rate limitation of Bi3+, the small particle size generates in a slow kinetic growth process. With the increase of reaction temperature, the amount of free Bi3+ setting free from the dissociation of the chelate complexes would increase gradually, which leads to a quicker kinetic growth process with a little larger particle size obtained [27]. Therefore, the kinetic process of BiVO4 formation could be controlled by changing the concentration of EDTA, reactive temperature and time. With the synthesis of BiVO4, some of the particles adsorb constantly to the surface of cotton fibers and root on them in a stable state. Based on the above points, possible deposition mechanism of BiVO4 particles is suggested as follow Scheme 1.
UV-V is diffuse reflectance spectroscopy
UV-V is diffuse reflectance spectroscopy (DRS) was used to characterize the electronic states of the as-prepared samples. The UV-V is absorption of the bare cotton fabric before deposition of BiVO4 and the BiVO4-coated cotton fabric obtained with CBD process were measured in the wavelength range 300–700 nm using a UV-V is spectrometer and the results are shown in Figure 5. It is observed from curve (b) that in addition to strong absorption in the UV-light region, the BiVO4-coated cotton fabric also possessed strong absorption in the visible-light region. The prolonged absorption tail until ca. 650 nm in each UV-V is spectrum might be a result of the presence of crystal defects generated during the growth of monoclinic BiVO4 which cover the surface of cotton fabric [28]. The absorption edge upward shifted with respect to the presence of the monoclinic phase BiVO4 mentioned in XRD analysis, of course, is absent in the curve (a) corresponding to the pure cotton fabric.
UV-V is absorption spectra for (a) pure cotton fabric and (b) BiVO4-coated cotton fabric; inset is the (ahν)2 versus hν curve.
As a crystalline semiconductor, the optical absorption near the band edge follows the formula αhν = A(hν-Eg)n/2, where α, ν, Eg and A are absorption coefficient, light frequency, band gap and a constant, respectively [29]. Among them, n depends on the characteristics of the transition in a semiconductor. The value of n, for BiVO4, is 1, demonstrating that it is a direct band gap material. The energy of the band gap of BiVO4 photocatalyst could be thus obtained from the plots of (αhν)2 versus photon energy (hν), as shown in inset of Figure 4. Based on the intercept of the tangents to the plots of the absorption edge, the values can be estimated that BiVO4 coated on the surface of cotton fabric has a band gap of 2.4 eV, which conforms to those of BiVO4 materials reported previously [30,31].
The photocatalytic performances
To study the photocatalytic activities of the BiVO4-coated cotton fibers, RB-19 was chosen as a model pollutant. Figure 6 displays the temporal evolution of spectral changes during the photodegradation of RB-19 under visible-light illumination. An apparent decrease of RB-19 absorption at 593 nm is observed by addition of BiVO4-coated cotton fabric. Accompanying the decrease of absorption of RB-19 during the photodegradation, its major absorption band presents a distinct blue-shift, indicating the decomposition of the dye chromophore with time. However, after prolonged irradiation, the increase absorption at about 450 nm suggests that organic aromatic intermediates such as quinones and phenols are transformed, which is in good accordance with that in the literature [32]. Figure 7 represents the variation of RB-19 concentration (C/C0) with irradiation time over different photocatalytic conditions. The photocatalysis test demonstrates that as a blank experiment in the absence of photocatalyst, the self-degradation of RB-19 is extremely slow, 10% of RB-19 is photolyzed after 70-min irradiation. However, with BiVO4-coated cotton fibers as photocatalytic system, 90% of the RB-19 was decolorized after 70 min, revealing a high photocatalytic activity under visible-light irradiation. Such photocatalytic performance was similar to that obtained over the BiVO4 catalysts reported by other researchers [12,33]. The adsorption of RB-19 on the BiVO4-coated cotton fibers in the dark was also checked. After 70 min, the concentration of RB-19 changed a little, which was possibly caused by the color absorption of cotton fibers. This result suggests that the degradation of RB-19 by BiVO4-coated fibers is mainly caused by photodegradation but not adsorption. While for the conventional powdered BiVO4 particles, the photodegradation rate of RB-19 could only reach up to 60% in the same time. This may be for the lower adsorption of RB-19 on the BiVO4 particles than that on the BiVO4-coated cotton fabric. So the BiVO4 coated on the surface of cotton fabric could react with RB-19 more efficiently. And this phenomenon also indicated that the cause of RB-19 degeneration was more from hvb+ on BiVO4 rather than ·OH radicals.
UV-V is spectra taken over time during the photodegradation of RB-19 (15 mg/L) mediated by BiVO4-coated cotton fabric. RB-19 concentration changes with irradiation time over (a) BiVO4-coated cotton fabric with visible-light irradiation, (b) BiVO4 particles with visible-light irradiation, (c) cotton fabric with visible-light irradiation, (d) BiVO4-coated cotton fabric without visible-light irradiation and (e) visible-light irradiation.

Photocatalytic activity is influenced by many factors in which the transport properties of photo-induced charge carriers is one key factor. The photo-induced charge carriers transport properties in the semiconductor always play a main role in improving the photocatalytic activity. For BiVO4 system, photo-oxidations occurring in aqueous media, the mechanism may involve direct reaction of the organic chemical (dye) with surface hvb+ of semiconductor, indirect reaction with ·OH radicals or a dual mechanism involving both surface hvb+ and ·OH radicals [34,35]. The pathway of photocatalytic degradation can be described through the following equations:

In view of these results, the transfer properties of photo-induced charge carriers have significant effects on its photocatalytic activity. For the oxidation process of BiVO4, the more photo-induced holes accumulate on the surface of BiVO4 nanoparticles, the higher photocatalytic efficiency it will have.
To investigate the recyclability of the BiVO4-coated cotton fabric, sample fabric after photocatalytic reactions was cleaned by ethanol and water solutions for the subsequent photoreaction cycle. The stability of the BiVO4-coated cotton fabric was evaluated by recycled runs in the photocatalytic degradation of RB-19 under visible-light irradiation. The decreases of the absorption maximum of RB-19 are shown in Figure 8. In the first three runs, the activity of composite materials debased slightly because there were some intermediates adsorbed on the surface [24]. After being cleaned, dried and then recycled, the BiVO4-coated cotton fabric exhibit activity as high as the first run that the photocatalytic degradation of RB-19 was up to 94%. This reveals that the composite does not suffer from photocorrosion during the photocatalytic degradation of RB-19, which is similar to the result in the literature [36].
Cycling runs in photocatalytic degradation of RB-19 in the presence of the BiVO4-coated cotton fabric under visible-light irradiation. Mechanism of BiVO4 particles deposition on the surface of cotton fabric.

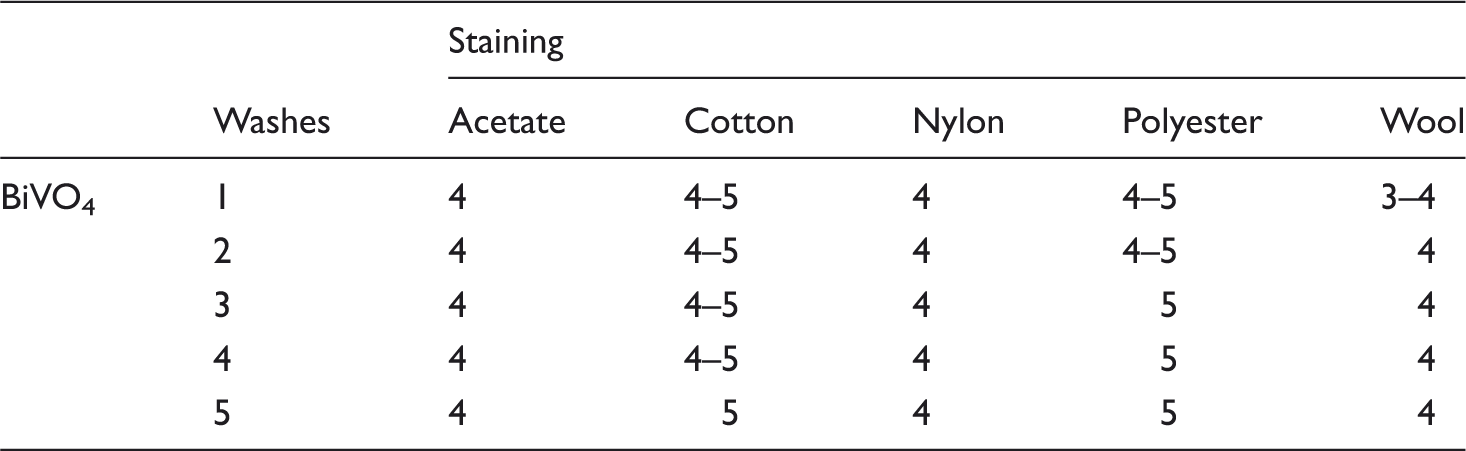
Wash fastness
Staining assessments for bismuth vanadate (BiVO4) on cotton fabric.
COD and TOC removal during the degradation of RB-19
Since the decolorization could not reveal the mineralization of the organic species, the COD and TOC, which reflect the general concentration of organics in solution and have been widely used to evaluate the degree of degradation or mineralization of organic species, were measured in the photodegradation process of RB-19 by the as-prepared BiVO4-coated cotton fabric under visible light. As is shown in Figure 7, 90% removal rate of color was achieved within 70 min. At the same time, the COD and TOC value of the dye decreased approximately by 52.5% and 23%, respectively. However, as time goes on, 48% of TOC was removed after 4 h, which indicates that the RB-19 mineralization by the BiVO4-coated cotton fabric is possible. It is well known that the disappearance of color is due to the fracture of dye chromophore. But it is quite difficult for the oxidation of aromatic ring in dye structure, especially anthraquinone structure, which leads to a gradual decrease of TOC [37].
Conclusion
Micro-nano-sized BiVO4-coated cotton fabric was prepared by chemical bath deposition technique at low temperature (≤100℃), which allows to deposit and to graft BiVO4 particles on cotton fabric. The adopted chemical bath synthesis conditions guarantee the stability of the solution for the industrial application. Under the selected conditions the cotton fabric was not damaged and BiVO4 covered the whole surface of the fabric uniformly. The particle size of BiVO4 crystallites is about 200–1000 nm. And its band gap absorption edge is determined to be 650 nm, corresponding to band gap energy of 2.4 eV. On the basis of the results presented before, we can conclude that the particles of BiVO4 constituted by monoclinic crystallites strongly adhering to the underlying support in mild temperature possess photocatalytic activities. It is a matter of fact that the BiVO4-coated cotton fabric shows high photocatalytic efficiency in decomposing the adsorbed RB-19 under solar-like light. The removal of COD demonstrated that the BiVO4-coated cotton fabric was capable of breaking stable conjugated aromatic structure. Moreover, the TOC reduction showed that the slow mineralization of RB-19 over the BiVO4-coated cotton fabric is feasible.
Footnotes
Acknowledgement
The author thanks the Instrument Analysis Center of Donghua University for providing the microscopy facility.
Funding
This work was supported by “the Research Innovation Funds of Shanghai Municipal Education Commission” and “the Fundamental Research Funds for the Central Universities.”
