Abstract
Water repellency was conferred to cotton and polyester fabrics by an hybrid organic–inorganic finishing obtained by hydrolysis and subsequent condensation of octyltriethoxysilane (OTES) under acidic conditions, in combination with melamine based crosslinking agent
Keywords
Introduction
Textile finishing by chemicals is widely used to improve the properties of natural and synthetic fibres and fabrics [1,2]. In particular, during the last few years, the increasing demand toward functionality of materials, hydrophobic and superhydrophobic surfaces (with water contact angles higher than 150°) has attracted considerable interest with respect to both academic research and industrial applications [3–5]. In general, low surface energy and extended degree of roughness are two critical factors in manufacturing hydrophobic surfaces; moreover, to provide the surface with such a property, two main directions can be basically followed: top-down approaches (including plasma treatment, lithographic and template-based techniques) and bottom-up approaches, such as chemical or layer-by-layer (LBL) deposition, hydrogen bonding, sol–gel method and colloidal assemblies [6]. Among bottom-up approaches, methods such as coating with paraffin wax, treating fibre surface with pyridinium compounds, silicone resin or fluorocarbon have been widely employed to obtain water repellency onto textile surfaces. The use of fluorochemicals is at present the most favourable treatment, because they strongly decrease the wettability of the textile substrate by forming a water-repellent and oil-repellent film on its surface. However, disadvantages of fluoroalkyl compounds are the high cost of materials, and a potential risk for human health as well as environmental concern [7–9]. In particular, perfluorooctanoic acid (PFOA) and perfluorooctane sulphonate (PFOS) are being replaced by other lower fluorocarbon alternatives [10,11], even if short fluorocarbon chains should be also considered potentially dangerous, due to the high strength of carbon–fluorine chemical bond [12–14]. Commercially available non-fluorinated chemistries made available by chemical manufacturers include the acrylic- and urethane-based, as well as other conventional chemistries such as paraffin, silicone and stearic acid-melamine. Unfortunately these non-fluorinated chemistries are not able to provide the desired fabric attributes as well as to meet their defined performance requirements, especially in situations where extremely low surface tension is needed. For this reason, non-fluorinated surface modification using sol–gel method has been proposed as an alternative approach.
The sol–gel process begins by combining organic and inorganic metallic solutions in which their compounds go through hydrolysis, condensation, polymerization and transformation to a gel [15]. In this novel approach, particular interest is focused on the self-organization of organosilane molecules which creates ordered hybrid materials performing textile properties [16]. Currently, hydrophobic properties have been obtained by introducing nano-sized particles onto the textile surface and the sol–gel technique has been reported as a promising tool for preparation of water-repellent coatings especially versatile for application on paper, textiles or wood [17–19]. In this way, it is possible to realize hydrophobic surfaces by either roughening the surface due to micro- or nano-structures or lowering the surface free energy chemical finishes on top of rough structure. However, high costs and poor washing durability of hydrophobic properties are an important limitation for sol–gel method in surface application, which is mainly induced by low compatibility and adhesion between fibre surface and silica sol. To avoid these limitations, new investigations have been made and multifunctional molecules, such as polycarboxylic acids or silane coupling agent, have been proved to be good chemicals to improve adhesion and reduce hydrophilicity of substrates, especially cellulose fibres [6,20–22].
The preparation of water-repellent textiles via sol–gel technique using non-fluorinated additives both for cotton and poly(ethylene)terephthalate (PET) fabrics is very interesting as it allows to test the chemical interactions of the finishing onto very different textile substrates and to explore new routes to increase the crosslinking. Cotton has always been the principal fibre for clothing due to its attractive characteristics such as softness, comfort, high resistance to heat and high resistance to alkaline conditions. However, its high absorbency as a result of the abundant hydroxyl groups on cotton diminishes the stain-resistance and water repellency of cotton textiles. The water repellency of PET has been also investigated because, although PET polymer is hydrophobic, the PET fabric soaks up liquid due to capillary force of fabric texture and additional finishes are required to impart superhydrophobicity to PET fabrics.
In this study, cotton and polyester fabrics were treated using mixtures of octyltriethoxysilane (OTES) and melamine based crosslinking agent,
Experimental
Materials
Name, code and chemical structures of the sol–gel precursors and of the cross-linker.

Nanosol preparation and coating process
A volume of 9.7 mL (0.03 mol) of OTES was hydrolysed with 2 mL (0.0002 mol) of HCl (0.1 N), 2 mL of deionized water and ethanol to a final volume of 100 mL under vigorous stirring for 12 h at room temperature to obtain the silica sol (OSi).
Contact angle (CA), advancing (θ

UD: undetectable; ---: pure.
Analysis and measurements
Hydrophobic properties of the cotton and PET fabric surfaces were first estimated by contact angle measurement, performed using a CCD camera (Imaging Source model “DMK 41BU02.H”), in combination with a Navitar®1-50486 optical lens. Typical spatial resolution was 4 µm/pixel. The contact angles were measured using the standard sessile drop method, expanding and contracting quasi-statically a drop on a horizontal surface and measuring advancing and receding contact angle (θA and θR, respectively) and their difference (Δθ). Drop images are post-processed by fitting with a circle the drop contour in the vicinity of the contact point, to measure the contact angle. Values of contact angle reported in the present paper represent the mean value over at least five measurements, and the corresponding standard deviation was about 4°. All measurements were carried out at room temperature. The contact angle hysteresis was calculated as the difference between advancing contact angle and receding contact angle. Since the irregular surface of a textile reduces the accuracy of (large) contact angle values, while for lower contact angles their porous structure immediately absorbs the liquid drop, the wettability of textile surfaces was also monitored by indirect methods such as drop test, water/alcohol test and water uptake [24]. Water/alcohol repellency of the untreated and treated cotton/PET samples was measured according to a modified AATCC 193-2009 Test Method. AATCC 193 test method evaluates the treated fabric resistance to wetting by a series of 12 water/alcohol solutions with different surface tensions (1 being 98:2 water–isopropyl alcohol ratio and 12 being 100% isopropanol). Drops of standard test liquids are placed on the fabric surface and are observed for wetting and wicking. The water uptake (
The results were the average of 10 readings. Water uptake was calculated according to Equation (1):

To evaluate durability of water repellency, water contact angle and infrared spectra of treated textile fabrics were collected after one and five washing cycles performed according to AATCC Test Method 61-2006, at 40℃ in presence of 50 stainless steel balls and of standard reference detergent. After drying, water contact angle and infrared spectra were performed.
FTIR spectra were acquired by means of a Thermo Avatar 370 spectrometer equipped with an ATR accessory. A diamond crystal was used as internal reflectance element on the ATR accessory. Spectra were recorded at room temperature, in the range from 4000 to 700 cm−1, acquiring 32 scans per set data of 4 cm−1 resolution.
Results and discussion
Surface characterization
The ATR-FTIR spectroscopy was employed to examine the change of surface functionalities after deposition and curing of hybrid sols on textile fabrics and the results are shown in Figure 1. In order to find evidence of any relevant change in the fabric surfaces due to the treatment with the hybrid sols, analysis on both fabrics was performed studying the coatings realized at the highest concentration (100 g/L) of MF, using the 1:4 molar ratio. Generally, the spectra of treated cotton (Figure 1a) and polyester (Figure 1b) samples are very similar to the untreated ones used for comparison. At 3500–3000 cm−1 cotton samples show the characteristic stretching vibration bands of cellulose hydrogen bonded O–H groups [26], while for polyester samples it is possible to detect a very strong peak at 1728 cm1 due to the stretching vibrations of C=O groups [27].
ATR-FTIR spectra of treated and untreated cotton (a) and PET (b) fabrics. ATR-FTIR: attenuated total reflectance Fourier transform infrared; PET: poly(ethylene)terephthalate.
In both spectra, the presence of octyl silicates is confirmed by very intense asymmetric and symmetric C–H stretching vibrations. The existence of these intense peaks (more intense for PET than the cotton) due to the bands at 2920 cm−1, 2850 cm−1 (ν(CH2)) and 1460 cm−1 (δ(CH2)) is consistent with the presence of alkyl groups in the octyl-modified silica film. Moreover, the broad bands at 1546 cm−1, 1479 cm−1 and 1335 cm−1 are ascribed to the triazine ring and exogenous C–N stretching vibrations, whereas that at 813 cm−1 is due to the triazine ring out-of-plane bending. The presence of the SiO2 matrix onto textile fabrics is usually confirmed by the Si-O-Si bending mode absorption at around 786–749 cm−1 [28]: unfortunately this weak band is overlapped by the triazine ring out-of-plane bending, observed at 813 cm−1 for cotton fabric, or by the characteristic absorption bands of pristine polyester sample. During the condensation and annealing process probable crosslinking reactions occur between the melamine based resin, the OTES hydrolysed sol and textile fabrics according to the possible interactions displayed in Figure 2. The change of the cellulose and polyester surface properties after the modification with alkoxysilane and its derivates was ascertained by water-repellent measurements.
Schematic representation of possible interactions of MF and OTES on the fabric surface (MF: OTES: 1:1 and 1:4 molar ratios). MF: 
Water-repellent properties
The wettability of the films was characterized by the contact angle. The testing methods for contact angle can be categorized into two groups: one is the static drop micro-observation (CA), while the other is the dynamic testing method. The first method was used to determine the static contact angle, while the dynamic testing method was used to explore the dynamic process of wetting, by measuring the advancing and receding contact angles both onto cotton and PET samples (Table 2). Untreated cotton sample is hydrophilic and the fabric was thus immediately wetted. The presence of 20 g/L or 60 g/L of melamine based resin did not modify these results. When cotton was treated with only silica precursor, it showed a slight water repellency (about 80°). Conversely, in case of MF-OTES sols prepared with 60 g/L or 100 g/L of resin (MF2 and MF3, whatever the molar ratio) the water CA values increased up to 120°–130°, while MF1 concentrations (regardless the OTES molar ratio) was unable to confer water-repellent performances. Untreated PET samples already showed a CA value of about 50°. The presence of only silane coating rose up this value to 130°. In combination with melamine, PET samples showed an increase of water CA to values higher than 130° as well, up to static contact angles close to superhydrophobicity (150°), which was detected for 1:4 ratios in main cases. For both textile samples, these results suggest an additive effect between melamine and silica precursor, probably due to the geometrical configuration that the molecules assume when combined, thanks to the structure of the triazine ring able to influence the orientation of the OTES alkyl chain.
A survey up-to-date literature reveals that contact angle hysteresis is determined by the balance of surface forces, by the roughness of a surface or by the properties of the liquid used [29].
In this paper, the surface roughness is supposed to be one of the main reasons causing the hysteresis, especially for the receding contact angle, while the introduction of long non-polar alkyl chains onto the textile surface is believed as the main reason for the increase of advancing contact angle. The effect of surface roughness on wettability was explained by the Cassie-Baxter model, which basic assumption is that the water droplet suspends above the asperities [30].
Results reported in Table 2 show that, for cotton samples, both advancing and receding contact angles slightly increase with the enhancement of MF:OTES molar ratios, principally depending by the initial MF concentration. The introduction of a great number of alkyl chains onto the basically irregular cotton surface, in fact, modifies the advancing contact angle due to its effect on the free energy surface. At the same time, the increased amount of finishing applied produces a rougher cellulose surface, thus increasing the receding contact angle as well. Finally, the hysteresis values show a constant behaviour as a consequence of both the advancing and decreasing contact angle increments. Similarly, the advancing and receding contact angles for polyester fabrics are also increased after treatments. In fact, the receding contact angles in PET samples rose up to about 17° after finishing with the different formulations, but this behaviour was balanced by a similar increase in advancing contact angles, which rose up to 21°. Also in this case, this can be attributed either to the grafting effect of the finishings applied either to their chemical interaction with the textile substrates or to the surface roughness. The surface free energy and the initial smooth surface of the PET fabrics are changed by the application of the finishings and the creation of a new rough surface, depending on the quantity of the film applied, with a decrease in the free energy connected to the introduction of the alkyl chains.
It is obvious that the relative quantity of precursors also plays an important role in determining the hydrophobicity of the treated fabrics. The data on all the treated fabrics show that an increase in the quantity of alkylsilane leads to higher water contact angles. A possible hypothesis for the detected improved water repellency, especially for 60 and 100 g/L MF concentration for both fabric substrates, could be related to a better orientation of the alkyl chains of the precursor, which is realized when MF cross-linker is used, and to an enhancement of the textile surface roughness. The rise in molar ratio and in MF concentration could bring an improvement of the active sites between the silica finishing and the substrate. In particular, a 1:4 molar ratio could create a multiple-linking system, in which the influence of the MF hydrocarbon chains steric hindrance is balanced by an increase in the number of links between the finishing and the sample substrate. Moreover, the surface free energy of the fabrics decreased by the application of the finishing, and in particular by the introduction of long alkyl chains, which realized rougher surfaces both for cotton and PET samples, depending on the quantity of the film applied. This new morphology of the fabric surfaces influenced the results in terms of wettability, especially when considering the contact angle hysteresis.
Drop test and water/alcohol test results for finished cotton and PET samples.

Table 3 also reports the water/alcohol test results for cotton and polyester fabrics treated with MF-OTES. Water/alcohol test method, by evaluating the wettability of the sample to a selected series of water/alcohol solutions with different surface tensions, is used to determine the effectiveness of a protective finish that is capable of imparting a low energy surface to all types of fabrics. Pure cotton fabrics showed total wettability (grading zero) to water/alcohol solutions, even when coated with pure OTES and pure MF: the only exception was the finishing with 100 g/L MF, which showed a slight enhancement to grading one. Conversely, PET samples showed a one-grade wettability when uncoated, which rose up to grading 3 by the deposition of pure silica onto their surfaces. The deposition of pure MF onto PET fabrics appeared less significant and not affected by the MF initial concentration. The MF1-OTES treated cotton samples (20 g/L MF) showed the same wettability as the untreated cotton fabric: the deposition of the films with different MF:OTES molar ratios does not change the repellency of the substrate. Nevertheless, the increase of the MF concentration reduces the wettability, and the best results have been obtained for 100 g/L of MF with 1:4 molar ratio. The MF:OTES mixture treated PET samples showed the best results for both the 60 g/L and 100 g/L MF concentration samples, independently from the MF:OTES molar ratios. The water/alcohol test confirm once more that for PET and cotton samples an additive effect of the mixture on the repellency properties does exist.
In Figure 3, the water uptake results for cotton and polyester fabrics treated with MF-OTES are reported. Both cotton and PET untreated samples show a similar wetting behaviour, and their weight percentage increased by about 225% after their immersion in water. However, since the water uptake of the fabric depends on the nature of the substrate, PET surface, due to its surface energy and capillary structure, was only wetted, while cotton fibre also absorbed water. The deposition of repellent hybrid coatings improved the repellency of all samples, when compared to reference. In particular, deposition of only silica coatings resulted in a decrease of water uptake percentage to 174% for cotton and 30% for PET treated samples. Water uptake results of pure melamine finishing onto textile fabrics depend on the concentration of MF, and a decrease in water uptake percentage to 110% and 70% for cotton and PET, respectively, takes place (Figure 3a). Treated cotton water uptake was not only influenced by the MF and precursor separate applications, but also by the MF:OTES molar ratios, and repellency improved with the increasing of the MF and the precursor concentrations, giving the best results (about 1% water uptake) for 1:4 molar ratio and 100 g/L of MF (Figure 3b). The weight increase for MF:OTES treated PET samples was less than 10% (wt.%) and results were quite similar for all molar ratios and MF concentrations, but for 20 g/L of resin with a molar ratio equal to 1:1 (Figure 3c). In particular, the best results (around 1% water uptake) were observed for the sample treated with MF3-OTES (100 g/L of resin) solution.
Water uptake test results for cotton and PET samples treated by pure chemicals (a), and for cotton (b) and PET (c) samples treated by all MF concentrations with different MF:OTES molar ratios. MF: 
This test once more confirms the effectiveness of the deposition of a mixture of OTES and melamine to get the best water repellency performances both for cotton and PET substrates, when compared to the single finishings deposition. Thus, it is likely that MF was preferentially consumed in the growth of siloxane networks in a horizontal (in-plain with textile surface) direction rather than in a vertical (out of plane). Consequently, the neighbouring silica alkyl chains at the outermost surface can be considered to be separated by condensed MF species acting as spacer moieties. These findings suggest that the alkyl chains at the surface are able to move freely, providing low surface energy on textile fabric as a consequence of the addition of melamine based resin as a molecular spacer.
Washing fastness evaluation
This study was principally focused on the formulation of a hybrid coating and on its water-repellent behaviour. For this reason, in this first step, wash fastness evaluation was only related to five washing cycles, with the only aim to investigate the different interactions between the two more diffuse textile fabrics and the chemical finishes. Anyways, laboratory washing fastness tests resulted in stronger treatments, compared to domestic laundering. Generally, one washing cycle is approximate to five times of commercial laundering [31]. According to the open literature to evaluate the durability of films coated onto textile fabrics [27,32], ATR-FTIR spectra have been collected after five washing cycles for both MF:OTES = 1:4 (MF = 100 g/L) treated samples (Figure 4) to investigate the washing fastness of the repellent films coated onto cotton and polyester fabrics. The existence of the octyl-surface groups was coupled with the permanence of the hydrophobicity of the samples also after washing cycles. Generally, the presence of the alkyl chain and of the melamine based resin is evident in all the spectra treated for both the fabric surfaces, thus confirming the presence of the films applied.
ATR-FTIR spectra of cotton (a) and PET (b) fabrics treated by 100 g/L of MF, using the MF:OTES = 1:4 molar ratio, before and after five washing cycles, respectively. ATR-FTIR: attenuated total reflectance Fourier transform infrared; MF: 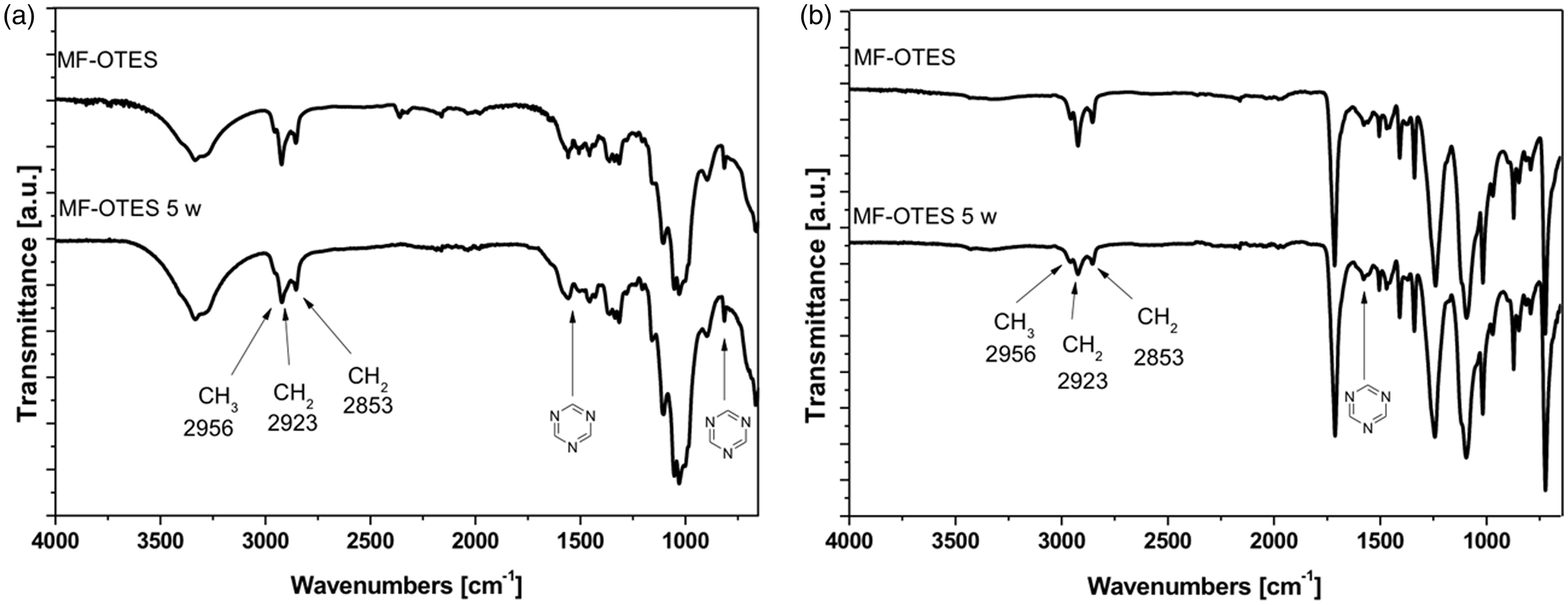
Particularly, the existence of several peaks in the 2956–2850 cm−1 range, assigned to the C–H symmetric and antisymmetric stretching vibrations of –CH3 and –CH2 groups, and the bands at 1546 cm−1 and 813 cm−1, ascribed to the triazine ring, are consistent with the presence of alkyl groups in the hybrid film onto washed treated cotton samples (Figure 4a).
Water contact angle (CA) of the treated cotton and polyester samples as a function of the number of laundering cycles.

UD: undetectable; ---: pure.
The results show that the contact angles of water recorded in the range of 110–120° indicate the characteristic of surface hydrophobicity of cotton and PET fabrics. The results of washing fastness tests showed that MF:OTES = 1:4 (MF = 100 g/L) cotton finishing retained its water-repellent property as reflected in the water contact angle, that changed from 130° (unwashed) to 114° (5 time washed).
Polyester finishing shows worst repellent properties because the initially high contact angles (140°) changed to 102° after only one washing, and repetitive laundries strongly deteriorated the water-repellent properties to the same extent up to 93° after five washing cycles.
This behaviour could be due to the presence of less reactive functional groups on the surface of polyester able to react with the chemical finishes, when compared to the cotton ones.
Anyway, further investigation on physical–chemical interactions between polyester and the proposed hydrorepellent finishing will be carried out. According to the ‘Thermosol’ process, developed by the DuPont company for the continuous dyeing of polyester fabrics, the effect of the drying heating to a high temperature about 190–205℃ to improve adhesion of hybrid finishes onto polyester fabric will be tested. During this process the fibre molecular chains open up at these elevated temperature and the finishes are supposed to diffuse into the polymer. Finally, on cooling, the finished could be trapped within the fibre yielding treated fibres that could have good fastness properties. With the aim of increase the number of reactive functional groups on the surface of polyester and thus to improve the adhesion of coating, the effect of plasma as a surface activation treatment before the application of hybrid coatings will be also investigated. However, even if wash fastness tests are very important for fabrics to be used in clothings, this could not be the case for non-conventional applications where a high level of coating adhesion would not be required.
Conclusions
From the presented data it can be concluded that the surface free energy of the fabrics decreased by the introduction of long alkyl chains, which realized rougher surfaces both for cotton and PET samples, depending on the quantity of the film applied. This new morphology of the fabric surfaces influenced the results in terms of wettability, especially when considering the contact angle hysteresis. Furthermore, an additive effect between the cross-linker (MF) and the silica precursor when combined together has to be emphasized. In fact, the 60 g/L and 100 g/L MF concentrations and the highest silica molar ratio showed the best contact angle (up to 130° for cotton and 150° for PET) and in general the best performances, in particular for cotton samples. A possible hypothesis for the improved water repellency could be related to the mobility/better orientation of the surface tethered alkyl chains, improved by the triazine ring, and to an enhancement of the textile surface roughness. Because of the nature of the textile surfaces, the modified silica sol system was most likely covalently bound to the cotton fabric as indicated by the high water contact angle even after five home launderings. These performances could not be obtained by PET due to the low interaction between finishes and surface. The reported one-step coating system is simple and reproducible for the preparation of transparent water-repellent surfaces over a large area. The proposed coating might provide a novel, easy alternative to conventional methods for the manufacturing of hydrophobic textile fabrics, highlighting that perfluorination is not strictly necessary. Anyway, further investigations must be done to characterize the surface morphology by using AFM on the nanoscale, to reveal the surface roughness of the fibres and to confirm the hypothesis submitted in this preliminary study. Moreover, the influence of the silica based coating on water repellency after highest washing cycles and on the mechanical properties of the natural and the man-made fabrics should be investigated.
Footnotes
Acknowledgements
The authors would like to thank Prof Marco Marengo and Dr Carlo Antonini (University of Bergamo, Italy) for the contact angle measurements.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
