Abstract
In the present study, highly aligned poly(ɛ-caprolactone) fibrous network produced by centrifugal spinning method was used as base substrate for electrophoretic deposition of chitosan. Two major parameters, namely the material and process parameters were varied to study its effect on add on % of chitosan in the matrices. It was found that the molecular weight and pH of the acidic chitosan solution widely influence the amount of deposition and pattern of deposition. The use of ultrasonic radiation in the electrophoretic deposition cell led to matrices with rough topography. In electrophoretic deposition process, the inherent gas bubbles formation results in formation of porous chitosan structure on the surface of centrifugal spun matrices making it suitable for biomedical applications.
Introduction
Electrophoretic deposition (EPD) is one of the attractive biomaterial fabrication techniques used to construct micro- and nanoscaled architectures [1–3]. This technique is based on migration of charged particle under the influence of electric charges resulting in ordered deposition of metals, polymers, ceramics, glasses, and their composites over the substrates [2–9]. EPD of polymers on different substrates are carried out for various applications such as biological microelectromechanical systems (bioMEMS), biosensors, enzyme immobilizations, and microfluidic devices [10–17]. The deposition of polymers by EPD is also carried out for improved cell adhesion [18], antimicrobial activity, and drug delivery properties [3]. At present, pH responsive polymers such as chitosan and alginates are coated on metal implants by EPD for bone tissue engineering. In the present work, chitosan polymer was chosen as model polymer for deposition on fibrous substrate.
Chitosan is the second most abundant natural biopolymer, obtained by partial deacetylation of chitin which is predominantly present in the exoskeleton of shellfish and crustaceans [19–21]. Chitosan coated as thin films possesses diverse properties making them suitable for extensive applications such as water purification, filtration, surgical dressings, drug encapsulation, and food processing [22–25]. Another interesting property of chitosan is its pH sensitivity and is attributed to presence of abundant primary amine groups at C2 position of glucosamine residue [25]. At low pH, chitosan is readily soluble in water due to the protonation of primary amine groups. Once the pH is elevated above its pKa (6.3), deprotonation of the amine group takes place and it leads to the sol–gel transition. This property can be advantageously used for the deposition of chitosan on substrates using electrophoresis principle. In EPD of acidic chitosan, when an electrical potential is applied between the anode and cathode, hydroxyl ions generated at the cathode deprotonate the chitosan and it gets deposited as a gel over the cathode. Further increase in duration of applied voltage leads to increased deposition of the hydrogel film due to electrophoretic motion of charged chitosan macromolecules [2,15,16,27]. The deposition of chitosan macromolecules by EPD principles has been attempted on substrate such as titanium, gold carbon, and stainless steel [6–10,28]. However to our knowledge, no attempt has been made to deposit chitosan on ultrafine fibrous material by EPD principle. It has been reported in recent work that the coating of chitosan on poly(ɛ-caprolactone) PCL fibers by drop casting method demonstrated better functional properties compared to that of blending for wound dressing applications. It has been further reported that drop casted chitosan on PCL fibers exhibited good platelet aggregation, antibacterial, antiadhesive, and anti-inflammatory activities [29].
In the present study it is proposed to deposit chitosan on ultrafine fibrous web by electrophoresis principle to obtain uniform coating of polymer. The ultrafine fibrous web is produced by centrifugal spinning. Centrifugal spinning is a facile and emerging technology [30–34] for the production of ultrafine fibrous web and the detailed experimental setup of centrifugal spinning is reported elsewhere in our previous work [30,31]. In short, the polymeric solutions are converted into fibers by applying centrifugal force resulting in highly aligned fibrous web. The aligned ultrafine fibers are produced without application of high voltage in short duration which is very difficult to achieve in electrospinning process. In the present study, EPD of chitosan on centrifugal spun PCL substrate was carried out by varying material and process parameters. The developed web was further analyzed for its morphology using scanning electron microscope (SEM) and the results obtained are discussed in the following sections.
Experimental
Materials
PCL (MW = 70,000–90,000) and chitosan with low molecular weight (20–300 cP) (product number 448869), medium molecular weight (200–800 cP) (product number 448877), and high molecular weight (800–2000 cP) (product number 419419) were purchased from Sigma Aldrich (USA). Acetic acid, chloroform, and sodium hydroxide (AR Grade) were procured from SISCO research Laboratories Pvt Ltd (India). Universal pH indicator solution was purchased from Rankem Pvt Ltd (India). Regulated direct current (DC) power supply unit (0–30 V, 0–2 A capacity with accuracy of ±0.05%) was supplied by Kpos Pvt Ltd (India) and ultra sonicator bath was supplied by PCI analytics Pvt Ltd India. All chemicals were used as received.
Development of centrifugal spun fibrous web
The PCL solution (15%) was prepared by dissolving PCL in chloroform and the solution was stirred for an hour without any air bubbles. PCL solution was filled into the pot type spinneret head using syringe and schematic diagram of centrifugal spinning is shown in Figure 1 [31]. The rotation speed of pot type spinneret was fixed at 5000 r/min based on various preliminary trials. The produced microfibrous web was collected in the round bottom collector. The collected aligned microfibrous sheet was kept in vacuum to remove excess solvent present on the fibers and used for further study.
Schematic diagram of centrifugal spinning system.
EPD of chitosan on centrifugal spun web
Chitosan with different molecular weight solutions was prepared by dissolving chitosan flakes using acetic acid as a solvent for desired concentrations (0.25, 0.5, 0.75 w/v). The required pH (3.5, 4.5, 5.5) was adjusted using NaOH drop wise into the chitosan solution. Graphite sheet was used as an electrode (both anode and cathode). Centrifugal spun microfibrous web was cut into sample size 20 mm × 20 mm having an average thickness of 0.11 ± 3 mm. The centrifugal spun web was placed on the cathodic surface with the help of adhesive tape. The chitosan solution (50 ml) was filled in the electrophoretic cell with the dimensions of 5 cm × 3 cm × 5 cm (length, width, height) and distance between the electrode was kept as 5 cm. Desirable voltage with respect to time was supplied by regulated DC power supply unit. The effect of ultrasonic radiation on the deposition of chitosan macromolecules on PCL matrices was also studied with frequency of 33 kHz and power of 60 W. After electrodeposition the microfibrous web was dried at room temperature for 48 h.
Characterization of chitosan-coated PCL web
The weight add on % was calculated by precise LC–GC (Model AS 220/X) weighing balance and it was calculated using the below formula.
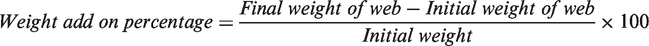
The surface morphology of the web was observed by using SEM after sputtering the samples with gold at an acceleration voltage of 10 kV.
Results and discussion
The major parameters that affect the EPD of chitosan on the matrices are material and process parameters. The material parameters, namely molecular weight of chitosan, concentration of chitosan, and the process parameters, namely voltage, time duration of voltage, and pH of the cell were also altered to study its influence on weight add on % and topography of deposition. The schematic diagram of EPD of chitosan on centrifugal spun fibrous matrices is shown in Figure 2. In our previous attempt quicker sol–gel transition of chitosan and large amount of gas evolution was observed and in the present study utmost care was taken to overcome the above limitation by carefully selecting the electrode and solvent for the chitosan [35].
Schematic diagram of EPD of chitosan on centrifugal spun PCL fibrous matrices.
Influence of chitosan molecular weight and concentration
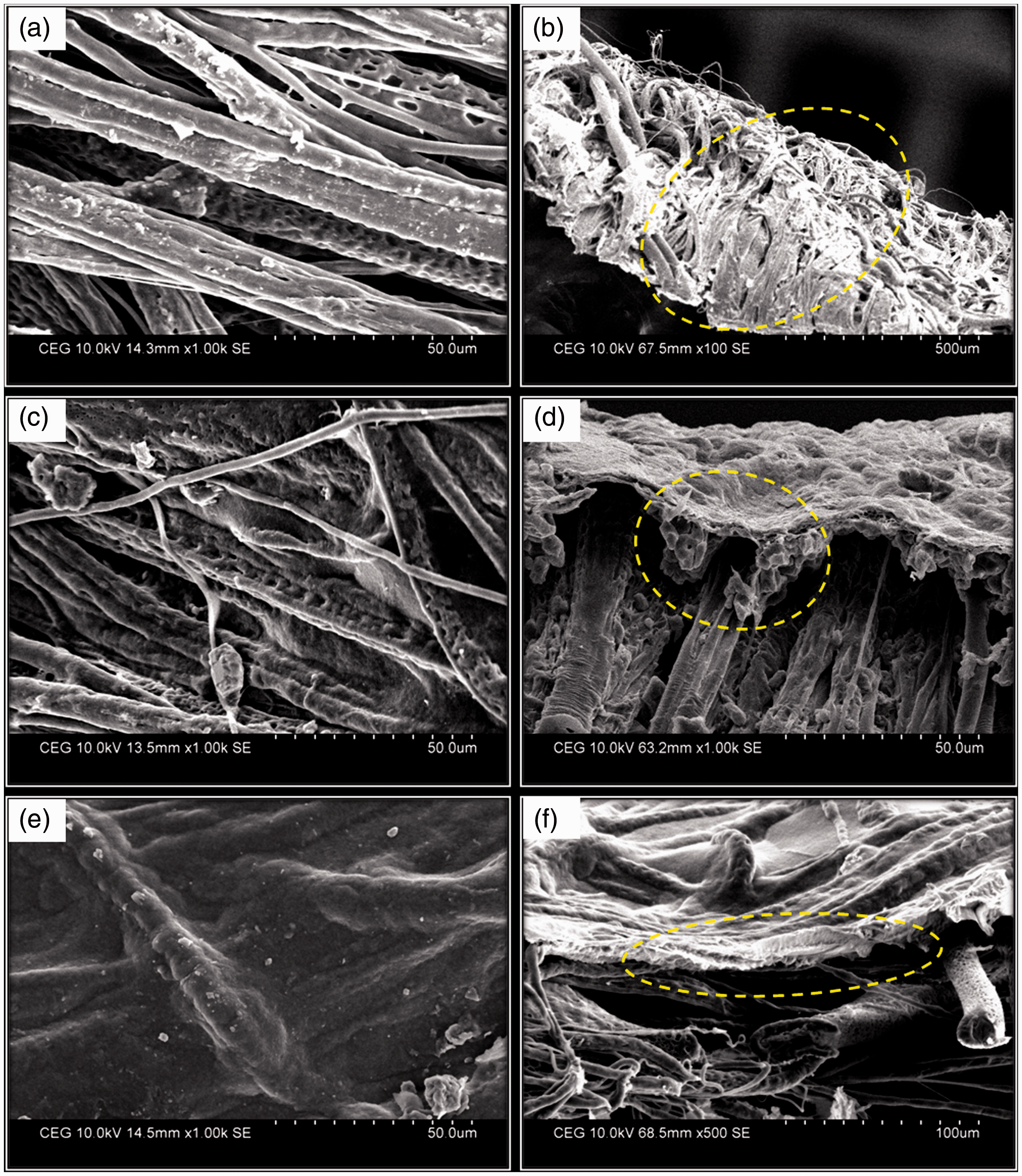
The chitosan concentration used in the electrophoresis cell was altered from 0.25 to 0.75 w/v for three different molecular weights of chitosan with voltage of 5 V, pH of 5.5, and time duration of 5 min. The data for weight add on % as a function of chitosan concentration and molecular weight are shown in Figure 3. From the figure, it can be seen that for all molecular weight taken for study, the deposition of chitosan macromolecules on matrices increased with increase in concentration of chitosan. However, the handle ability of the deposited mat was good when 0.25 and 0.5 w/v chitosan concentration was used. Moreover with increase in molecular weight, add on % was found to be higher for high molecular weight chitosan and topography of deposition was significantly different for all molecular weights. The topography of deposition was analyzed using SEM and they show different surface profiles for the various samples (Figure 4). Better infiltration and coating of chitosan macromolecules on ultrafine fibrous web was observed in low (Figure 4(a) and (b)) and medium molecular weight (Figure 4(c) and (d)) chitosan compared to that of high molecular weight chitosan. In the case of high molecular weight chitosan, Figure 4(e) shows the surface topography of the web and it was coated with chitosan uniformly without any space, whereas Figure 4(f) shows the cross section view of the matrices in which chitosan can be observed on the surface alone and confirms the poor infiltration.
Effect of molecular weight and concentration of chitosan on weight add on (%). (a) Low molecular weight chitosan-deposited PCL fibrous surface, (b) cross section of low molecular weight chitosan-deposited matrices, (c) medium molecular weight chitosan-deposited matrices, (d) cross section of medium molecular weight chitosan-deposited matrices showing better infiltration, (e) high molecular weight chitosan-deposited matrices, and (f) cross section of high molecular weight chitosan-deposited matrices showing surface deposition.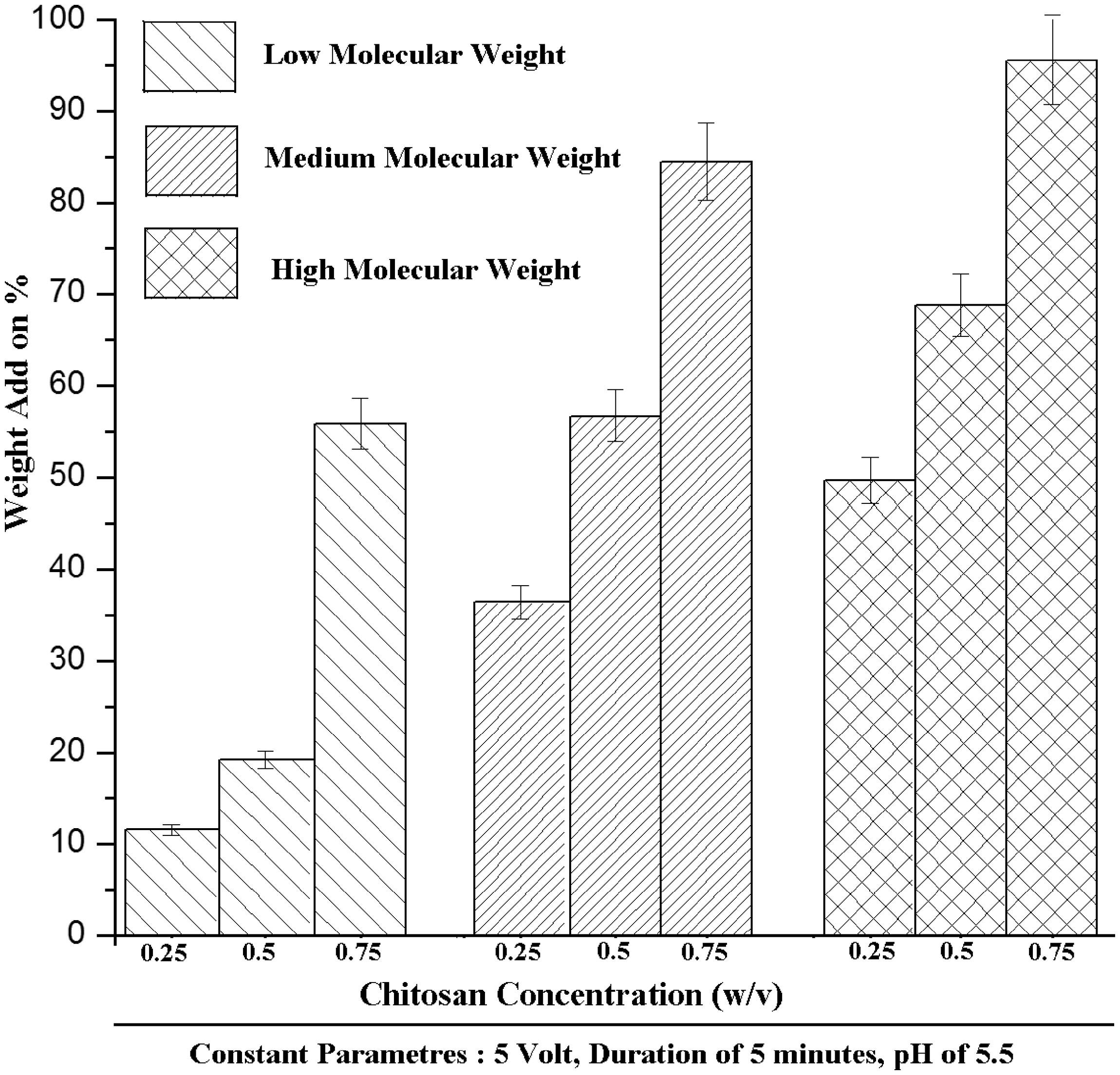
Influence of voltage and time
In order to study the net effect of chitosan deposition on the matrices with respect to voltage on the ultrafine fibrous web, the chitosan concentration was fixed at 0.5 w/v for medium molecular chitosan. The time of deposition was fixed at 5 min and the pH was maintained at 5.5. The weight add on % of chitosan shows significant variation with respect to varying applied voltage as shown in Figure 5. The weight add on % increased up to 10 V and further increase of voltage to 15 V led to decrease in weight add on %. These results can be better explained by the evolution of gases at cathode. The gas evolution is controlled at lower voltage but at elevated voltage of 15 V, gas evolution was found to be higher. This reflected in the weight add on % leading to decreased deposition at higher voltage. The SEM micrograph confirms (Figure 6(a) and (b)) the above statement and it can be seen that surface becomes rougher and lot of pores in the matrices can be noticed. Moreover with increase in voltage, chitosan macromolecules quickly attain the sol–gel state and are not able to infiltrate into the ultrafine fibrous web resulting in layered coating on the surface of fibrous web as shown in Figure 7.
Effect of voltage, time, and pH on weight add on (%). EPD of chitosan on PCL matrices: (a) 10 V at 5 min, (b) 15 V at 5 min, (c) 10 min at 5 V, and (d) 15 min at 5 V. Schematic diagrams showing the effect of voltage on infiltration of chitosan in PCL matrices.


To evaluate the influence of duration of applied voltage on weight add on %, the voltage was fixed at 5 V and the other parameters (pH 5.5, duration of time 5 min, medium molecular weight chitosan (0.5 w/v)) were kept as a constant. It was observed that the weight add on % significantly varied with respect to the duration of voltage application (Figure 5). The weight add on % increases up to 10 min of voltage application and on further increase in duration of applied voltage the add on % decreased. The decrease in weight add on % is attributed to the gas evolution which results in creation of pores followed by disintegration of chitosan deposited on the matrices (Figure 6(c) and (d)).
Influence of pH
The effect of pH of chitosan solution in EPD was altered from 3.5 to 5.5 to study its effect on weight add on % (other parameters such as concentration of medium molecular weight chitosan (0.5 w/v), 5 V and 5 min duration are kept as a constant) and the results are shown in Figure 5. As reported in literature, higher deposition of chitosan macromolecules on the matrices was observed at pH 5.5 [36,37]. At lower pH, decreased deposition is observed due to decrease in ionization and lower net charge density as cited in literature. The SEM micrograph shows the deposition of chitosan studied at various pH (Figure 8). It can be seen from the figure that uniform deposition is obtained at pH 5.5 with pores (Figure 8(c)). The porous morphology obtained in the substrates will aid in transport of moisture and air across the web, and these features make the substrate as a suitable candidate for wound dressing application.
EPD of chitosan on PCL matrices at varying pH: (a) 3.5, (b) 4.5, and (c) 5.5.
Influence of ultrasonic radiation on chitosan deposition
Chitosan deposition on the PCL matrices was carried out at 0.25 w/v concentration with medium molecular weight chitosan (pH 5.5, 5 V, and 5 min) in the presence of ultrasonic radiation. Interesting topography of chitosan deposition on PCL matrices was observed on application of ultrasonic radiation and the SEM images are shown in Figure 9. The usage of ultrasonic waves in the EPD cell resulted in the formation of craters on the deposited chitosan thereby making the surface rough. The craters are formed due to quick destruction of hydrogen gases evolved from cathode and the surface of chitosan gets the impression of bubbles generated. The developed morphology will have significant effect when the matrices are used for wound dressing applications. It has been reported in literature [38] that surface roughness will aid in cell affinity and adhesion of cell. The developed substrate will definitely aid in achieving the above function.
EPD of chitosan on PCL matrices under ultrasonic radiation: (a) lower magnification and (b) higher magnification.
Conclusion
The present study demonstrated that the material parameters and process parameters have significant effect on the amount of deposition and topography of chitosan deposition on the matrices. The results indicate that pH of acidic chitosan solution plays a vital role in weight add on % compared to other parameters such as molecular weight, concentration of chitosan, voltage, and duration of voltage application. But the above-mentioned parameters have significant effect on the topography of the coating on the PCL fibrous matrices. Better infiltration of chitosan in the matrices was observed when low and medium molecular weight chitosan was used for EPD. At higher voltage and longer duration of applied voltage, higher amount of gases was evolved from the electrodes leading to poor adhesion of the chitosan on the PCL matrices. Based on the studies conducted, better deposition of chitosan on PCL matrices was observed for medium molecular weight chitosan of 0.5 w/v concentration when the EPD cell was maintained at pH of 5.5, 5 V for a duration of 10 min. The usage of ultrasonic radiation during EPD made the surface of the composite matrices rough. The observed results provide ample scope for tailoring the deposition of chitosan with different topography for various biomedical applications such as wound dressing and tissue engineering. The use of the ultrasonic radiation during electrophoresis deposition needs further detailed study for attaining different topographies of deposition.
Footnotes
Acknowledgement
Authors acknowledge CSIR (22(0566)/12/EMR-II), Government of India.
