Abstract
Novel nanoencapsulated phase change materials containing polyethylene glycol as the core material and urea formaldehyde as the shell material were prepared with an in situ polymerization method. The prepared nanocapsules were coated onto the cotton fabric using the pad-dry-cure method, and different ratios of nanocapsules to binder agent were evaluated. The morphology and characteristics of the polyethylene glycol and nanoencapsulated phase change materials were measured using scanning electron microscope, transmission electron microscope and Fourier transform infrared spectroscopy. The nanocapsules were found to have a regular spherical shape with a size of 141 nm. The thermal behavior of the textile fabrics was investigated by differential scanning calorimetry and thermal gravimetry analysis. The tensile strength, water absorption and abrasion resistance of the treated and untreated fabrics were also studied. The differential scanning calorimetry results found that the treated fabrics had latent heat storage energy of 0.198, 0.213 and 0.219 J/g, where the measurement was related to the nanocapsules/binder ratio. The thermal gravimetry analysis results showed that the treated fabric with a nanocapsules/binder agent ratio of 3:3 had good thermal stability.
Introduction
Clothing has a very important and controllable role for the body, with comfort being one of the most important functions of clothing. Clothing systems include many parameters that have dynamic interactions between each other. Transmission of heat and moisture is the most important factor for maintaining the thermal equilibrium between the body and the environment, and thus, achieving clothing comfort [1,2]. According to Goldman [3], the four primary factors affecting clothing comfort are function, feel, fit and fashion. These factors include thermal and moisture management, water absorbency and so on. Clothing can be accepted as a protective barrier between the body and the environment, and has a role in the human thermoregulation mechanism that provides thermal comfort in adverse environmental conditions.
According to the British Standard BS EN ISO 7730, the thermal comfort is defined as the condition of the mind that expresses satisfaction with the thermal environment. Persons who work in uncomfortably hot environment without appropriate clothing for such extreme conditions are most likely to be exposed to higher risks at work. The most comfortable human skin temperature is 33.4℃. The body is unable to sense changes in skin temperature that are within 1.5–3.0℃ of this ideal temperature. However, if the change in skin temperature is more than ±4.5℃, the human body feels discomfort. In addition, a core body temperature of 37℃ is required, and a rise or fall in temperature of 1.5℃ from the core temperature can be fatal [4–6]. To support the human thermoregulation process, especially in hot environments, smart textiles have been developed [7]. Smart materials and structures can be defined as materials that sense and react with environmental conditions, or stimuli, such as those from mechanical, thermal, chemical, electrical, magnetic or other sources. According to the functional activity, the smart textiles can be classified as passive smart, active smart and ultra-smart textiles. Passive smart textile materials can only sense the environmental conditions or stimuli, whereas active smart textile materials can sense and react to environmental conditions or stimuli. A thermoregulating textile fabric is an active intelligent textile that can appropriately respond to external temperature changes and environmental stimuli.
The thermoregulated textiles can be produced by incorporating phase change materials (PCMs) into the fabric to enhance the comfort of the wearer [8]. PCMs have properties that allow absorption, storage or release of heat energy when the material undergoes phase changes from solid to liquid or vice versa [9]. PCMs are classified into two main types: organic and inorganic. Organic PCMs are further classified as paraffin and non-paraffin PCMs. Inorganic PCMs are further classified as salt hydrated or metallic [10–12]. Polyethylene glycol (PEG) is one of the most commonly used PCMs for the manufacturing of thermoregulated textiles [7].
PEG is considered a thermal storage material due to its: (i) high heat of fusion; (ii) congruent melting behavior; (iii) non-corrosive behavior and (iv) wide melting point temperature range [9]. PEG is the most commonly used PCM that is incorporated directly into the textile materials [13]. This incorporation is carried out by a micro- or nano-encapsulation technique to prevent leakage of the applied PCMs.
Nanoencapsulation is the process of enclosing nano-sized particles of the solid or liquid state to produce nanocapsules or nanoparticles. The term nanocapsule is used if the size of the particles is <1 µm. For the particles in the size range of 1–1000 µm, the term microcapsules or microparticles is used [14]. Micro- or nano-encapsulation can be achieved by either physical or chemical methods. Physical methods include spray drying, fluidized bed and centrifugal extrusion processes. Chemical methods include in situ polymerization, interfacial polymerization and complex coacervation [15].
Fang et al. [16] successfully prepared nanoencapsulated PCMs with
Sánchez et al. [8] successfully prepared polystyrene microcapsules for textile applications and showed that the addition of the coated microcapsule to the textile substrate resulted in good thermoregulating properties. The development of thermoregulated textiles has resulted in more research studies over the past two decades, but the research has remained focused on the selection of a PCM, composition of the shell, methods of micro- or nano-encapsulation and methods of adding micro/nano-encapsulated PCMs to the textile material. To date, no study has reported on the preparation of nanocapsules using UF as the shell layer and PEG as the core material, or their incorporation into the fabric. The aim of this study was to determine the thermoregulation properties of a cotton fabric incorporated with nanocapsules containing UF as the shell material and PCMs, such as PEG, as the core material. SEM and differential scanning calorimetry (DSC)/thermal gravimetry analysis (TGA) were used to characterize the morphology and thermal properties of the textile fabrics, respectively. In addition, this article determined the tensile strength, water absorption and abrasion resistance of the textile fabric after incorporation of the nanocapsules.
Experimental methods
Materials
PEG (Mw – 6000) was used as the core material (Hi-Media, Mumbai), and urea (Hi-Media, Mumbai) and formaldehyde of 37–41 wt% (Merck, Mumbai) were used as the shell layer. Sodium dodecyl sulfate (SDS) supplied by Hi-Media was used as the emulsifier. Sodium hydroxide and polyvinyl alcohol were used as the pH controller and stabilizing agent, respectively. Hydrochloric acid was used as the activator.
Preparation of the nanocapsules
PEG/UF nanocapsules were prepared by the in situ polymerization method. This method involves two steps: In step 1, 16 g of PEG, 2.9 g L−1 of SDS and 100 mL of water were emulsified mechanically at 80℃ with a stirring rate of 2500 r/min for 45 min. Subsequently, 1.8 g L−1 of PVA was added to the mixture to stabilize the emulsion. In step 2, 24 g of urea and 14.8% formaldehyde were added to water. The mixture was stirred and adjusted to pH 8.5–9 with an aqueous solution of 10% sodium hydroxide. Then, the mixture was continuously stirred at 70–75℃ for 1 h to prepare the pre-polymer solution. Droplets of the pre-polymer solution were added to the emulsion where the emulsion mixture was stirred at a rate of 1000 r/min. The pH was reduced to the range of 5–5.5 by adding dilute hydrochloric acid into the mixture. The mixture was agitated continuously at a stirring rate of 600 r/min for 1 h, and the temperature was slowly reduced to 35℃. The resultant nanocapsules were filtered, washed and dried in an oven at 70℃ for 8 h to remove water.
Coating the nanocapsules onto the cotton fabric
Fabric particulars.

Parameters for the prepared coating composition.

Tensile testing
The strength of a fabric is defined as its ability to resist tensile stress. The strength and elongation at breaking point of the fabric were measured using an Instron 3360 series tensile tester in accordance with ISO 13934. The size of the test samples was 60 mm × 300 mm. All samples were conditioned for 24 h prior to testing.
Water absorbency testing
This method was used to measure the water absorbency of the samples by calculating the time taken for the fabric to absorb a drop of water as per standard AATCC 79 (2010). In this test, the sample is cut to a size of 200 mm × 200 mm and mounted on an embroidery hoop such that the side of the sample to be tested is face up and free from wrinkles. In this test, the nozzle of a burette is placed 10 ± 1.0 mm above the specimen surface and one drop of distilled water is allowed to fall on the cloth. Then, the time taken for the drop of water to be completely absorbed, i.e. the time taken for the drop of water to lose its reflectivity, is calculated.
Abrasion resistance test
The abrasion resistance test was carried out using an abrasion resistance tester (Martindale method) in accordance with EN ISO 12947. The size of the test samples that were used was 38 mm in diameter. The test specimen was placed face down into the base of the specimen holder. The test was carried out until two separate threads were completely broken down.
Characterization
The morphology and particle size of the samples were analyzed using SEM and TEM. The particle size was measured using a TEM (CM200; Philips), operated at 20 kV with 2.4 Å resolution. The sample morphology was analyzed using a JSM 6390. The SEM equipment was also furnished with an energy-dispersive X
Results and discussion
Surface and elemental analyses of the nanocapsules
SEM and TEM were used to analyze and identify morphological changes in the nanoencapsulated PCMs. Figure 1(a) and (b) shows the SEM images of the PEG and nanocapsules. Figure 1(b) shows that the PEG is encapsulated in the shells of UF to form regular spherical-shaped nanocapsules and the surfaces are mostly smooth. TEM analysis determined that the particle size of the nanocapsules was 141 nm (Figure 2(a)) and also confirmed that the PEG was encapsulated in the shells of the UF to form spherical shell-structured nanocapsules (Figure 2(b)). The UF shells provided the mechanical strength for the nanoencapsulated PCM and prevented leakage of the melted PEG. The EDX analyses were performed to identify the elements present in the PEG and nanoencapsulated material. The EDX results shown in Figure 3(a) demonstrate that the core material dominantly consists of C and O. It can be observed from Figure 3(b)that the presence of N atom confirms that the PEG core material encapsulated by UF shell material. The chemical composition of core material was 55 wt% C and 38 wt% O, whereas the nanocapsules was 38 wt% C, 27 wt% O and 35 wt% N. Compared with EDX spectra of PEG, C was slightly reduced after encapsulated the core material.
SEM images of PEG and nanocapsules: (a) PEG and (b) PEG in UF shell. TEM images of nanoencapsulated PEG: (a) size of nanocapsules and (b) morphology of nanocapsules. EDX analysis of PEG and nanocapsules: (a) PEG core material and (b) nanoencapsulated PEG.


FTIR analysis of the nanocapsules
The FTIR spectra of the PEG and nanoencapsulated PCMs are shown in Figure 4(a) and (b), respectively. As shown in the figure, the PEG spectra bands occur at with bands 2881, 1107 and 839 cm–1.The absorption peaks at 2881 cm–1 are characteristics of the aliphatic C–H stretching vibration, whereas the absorption peak at 1107 cm–1 is associated with C–H bending vibrations and the peak at 839 cm–1 corresponds to the CH2 rocking vibration. Figure 4(b) shows that the spectra bands of the nanoencapsulated PCMs occur at 3346, 2953–2883, 1653 and 761 cm–1. The absorption peak at 3346 cm–1 is presumed to be due to –OH stretching, overlapped with the N–H stretching vibration; the absorption peak at 1653 cm–1 corresponds to the CH stretching vibration. These additional bands in the nanoencapsulated PCMs are associated with the characteristics of the peaks of the UF polymer shell material.
FTIR spectra: (a) before encapsulation of PEG and (b) encapsulation of PEG by UF shell layer.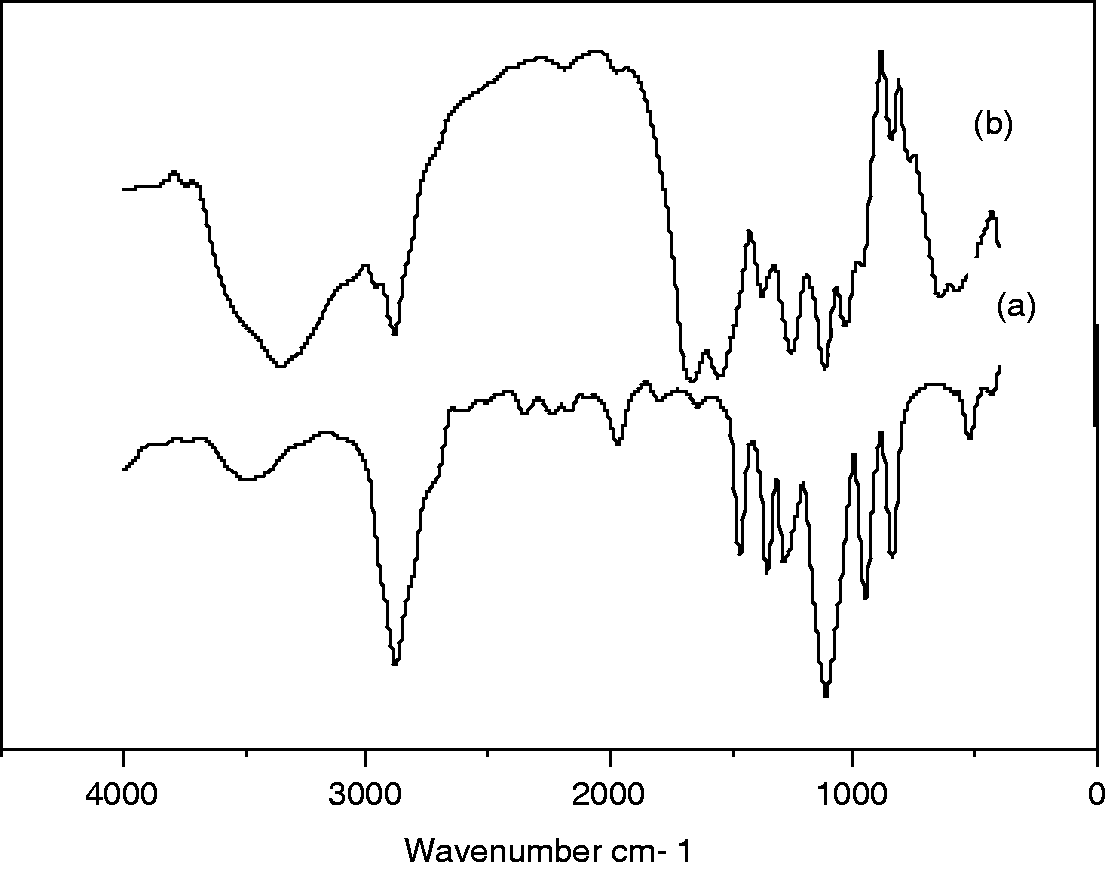
Thermal properties of the nanocapsules
The melting point and latent heat measurements of the PEG and nanoencapsulated PCM were obtained from DSC measurements and are shown in Figure 5. Figure 5(a) shows that the peak temperature and latent heat measurement of the PEG were 63℃ and 190.3 J g–1, respectively. However, the peak temperature and latent heat of the nanocapsules were 41.9℃ and 17.8 J g–1, respectively, as shown in Figure 5(b). These results show that the latent heat of the nanocapsules is lower than that of the PEG core material alone, due to the low thermal conductivity of the shell layer.
DSC curves of: (a) PEG and (b) nanoencapsulated PEG.
The thermal stability of the nanocapsules can be determined using TGA curves (Figure 6) that were obtained by heating the nanocapsules within a temperature range of 0–400℃ at a rate of 10℃ min–1. Figure 6(a) shows that the PEG started to lose its mass at temperature >350℃. At 400℃, the PEG showed complete loss of its mass. However, as shown in Figure 6(b), the nanoencapsulated PEG showed a slight loss of mass at temperatures between 150℃ and 240℃ and demonstrated a 78% mass was retained after 360℃, showing that the nanoencapsulated PEG has good thermal stability appropriate for textile application.
TGA curves of: (a) PEG core material and (b) nanoencapsulated PEG.
Morphology of the treated fabrics
Figure 7 shows the morphology of the untreated cotton fabrics. On the SEM image (Figure 7(a)), some protruding fibers can be observed on the surface of the untreated cotton fabric. Figure 7(b) shows that the grooves and fibrils can be observed on the surface of the untreated cotton fiber. In Figure 8(a), SEM images show nanocapsules distributed over different locations on the cotton fabric. The coated nanocapsules have a smooth and regular surface that coated the surface of the fabric, as observed in Figure 8(b).
SEM photographs of untreated samples. SEM photographs of treated samples.

Tensile test
Tensile strength properties of treated and untreated fabrics.

Water absorbency test
The water absorbency measurements of the treated and untreated fabric samples are shown in Figure 9. The untreated fabric took more time to absorb water because it had more protruding fibers on the surface of the fabric. Sample S1 took the least amount of time for water absorption compared with all other samples due to coating materials reducing the number of protruding fibers during the drying and curing processes. The time taken for water absorption for Sample S2 was longer compared with Sample S1. This result is due to the higher amount of coating material added to the surface of the fabric, which reduces the number of pores on the fabric surface. In the case of Sample S3, larger amount of the coating materials on the surface of the fabric resulted in the time taken for water absorption is shorter compared with Sample S2 but longer than Sample S1. This result showed that the polar OH groups present in the cellulosic cotton make the untreated fabric more hydrophilic than the fabrics when the fabric is treated with hydrophobic nanocapsules.20 For Sample S4, the fabric took longer time to absorb water than all other treated fabrics due to an insufficient amount of binder agent relative to the amount of nanocapsules.
Water absorbency of treated and untreated samples.
Abrasion resistance
The abrasion resistance measurements of the tested samples are shown in Table 4 and Figure 10. The test was carried out until the fabric threads on the abraded surface broke down. Samples S1, S4 and the untreated fabric were abraded after 10,000 cycles, whereas Samples S2 and S3 were abraded after 11,000 cycles. Overall, Samples S2 and S3 showed better durability, which was the result of the higher amount of binder agent added in the coating composition.
Abrasion resistance properties of treated and untreated samples. Abrasion resistance properties of treated and untreated fabric.

Thermal properties of the treated fabrics
The DSC curves of the nanocapsules treated cotton fabrics are shown in Figure 11. Thermal properties of the treated fabrics, including the melting temperature ( DSC curves for samples: (a) S1, (b) S2 and (c) S3. Thermal properties of the treated fabrics.

Thermal stability
A thermogravimetry (TG) experiment was used to assess the thermal stability of the treated and untreated fabric samples. Figure 12 shows the TG curves observed and demonstrated that the weight loss percentage occurs between 20℃ to 900℃. The thermal degradation data obtained from the TGA curves are given in Table 6. All of the samples show two-step weight loss degradation, but there is only single-step weight loss degradation for untreated fabric. In all cases, changes in the weight loss percentage occurring at temperature <250℃ were a result of the removal of physically adsorbed water. The untreated cotton fabric containing cellulose was found to lose 70% of its weight between the temperatures of 300℃ and 400℃. In this stage, the rapid weight loss is due to the dehydration of cellulose. At temperatures >476℃, char formation occurs and produces CO2, carbonyl and carboxyl products. The weight loss of the treated Samples S2 and S4 between 300℃ and 400℃ was higher than that of the untreated fabric. The treated samples containing a nanocapsules/binder agent ratio of 3:1 and 3:3 had a higher decomposition temperature in the first stage than the other samples. In the second stage, the decomposition temperature for Sample S3 (nanocapsules/binder ratio of 3:3) was higher than that of the other samples due to the binding agent cross-linking with cellulose. With the exception of Sample S4, the amount of final residue was lower in the treated samples than the untreated fabric.
TGA curves for treated and untreated samples. Thermal degradation interval of treated and untreated fabric.

Conclusion
Nanoencapsulated PCMs were successfully prepared using an in situ polymerization method. The morphology of the nanocapsules was a spherical shape and 141 nm in size. The FTIR spectra confirmed that the PEG was successfully encapsulated within the UF shell. The resultant nanocapsules were found to have a lower latent heat storage energy capacity than the core material. Although lower thermal stability than the PEG was exhibited, the nanocapsules showed good thermal stability, which makes the coated fabric appropriate for textile application. The nanocapsules were fixed into the cotton fabric using the pad-dry-cure method. The treated fabrics Samples S1, S2 and S3 were capable of absorbing 0.198, 0.213 and 0.219 J/g of heat, respectively, which was related to the different nanocapsule to binder agent ratios used in each fabric sample. However, Sample S4 showed no peak or latent heat storage values, due to an insufficient amount of binder agent used during the fixation of the nanocapsules. Sample S3 was considered as the best thermoregulating fabric for adding the dynamic heat regulation function in addition to the passive thermal insulation effect of the cotton fabric.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
