Abstract
Nanofiber mats formed in electrospinning process have many advantages, such as extensive application prospects in the thermal insulation, filtration, water protection and so on. In this study, poly (vinyl alcohol) nanofiber mats prepared by electrospinning were chemically cross-linked with glutaraldehyde. The poly (vinyl alcohol)/ glutaraldehyde nanofiber mats were characterized by water durability test, mechanical, differential scanning calorimeter, thermogravimetric analysis and X-ray diffraction, respectively. Water durability test showed that cross-linked with glutaraldehyde results in improvement in water stability. Differential scanning calorimeter and thermogravimetric analysis showed that cross-linking improved the thermal stability. X-ray diffraction patterns suggest a decrease in the degree of crystallinity.
Introduction
Recently, electrospinning has become an important method in fabricating of nanofibrous structures. Compared with traditional nonwoven fiber mat, nanofibrous mats formed in this process have many advantages [1], such as light weight, high specific surface area and good protection. Nanofibrous mats have extensive application prospects in the thermal insulation, filtration, UV protection and so on.
Poly (vinyl alcohol) (PVA) is the major materials used to prepare nanofibrous products in electrospinning [2–7] owing to its excellent physical properties, biodegradability, good mechanical properties in the dry state, and many other advantages such as low price and easy to get, etc. However, its disadvantage of being strongly hydrophilic, due to a large number of hydroxyl groups present in the molecules, limits the PVA applications. Therefore, in order to extend the area of applicability it is imperious to improve the water resistance of PVA nanofibrous mats [8].
The water resistance of PVA is been studied by some researchers presently and much attention has been paid to the industrial field applications such as architectural coatings and adhesives. Water-insoluble PVA nanofibrous mats was rarely reported in the previous literatures. In this paper, glutaraldehyde (GA) was employed to cross-linked PVA nanofibrous mats based on other tests. Twelve modified solutions were obtained by using different ratios of GA. The effect of different concentration-modified solutions on PVA nanofibrous mats has been systematically studied. The water stability, mechanical properties, thermal stability and changes in the degree of crystallinity were also investigated.
Experimental section
Materials
Poly (vinyl alcohol) (88% hydrolyzed) was purchased from ACROS ORGANICS, New Jersey, USA. Glutaraldehyde (GA) (50% aqueous solution) was purchased from J&K Chemical®. Hydrochloric acid (HCl 36.5% aqueous solution) was purchased from Pinghu chemical reagent factory. Acetone (99.5% concentration) was purchased from Changshu Chemical Co, Ltd.
Electrospinning of PVA nanofiber mats
Preparation of electrospinning solutions
The PVA solutions were prepared by dissolving PVA powder in distilled water and heating to 85–90℃ under constant stirring for 2–3 h to get homogenous solutions and then cooled down to room temperature for later use. Here, 10% PVA solution was prepared.
Preparation of nanofiber mats
The electrospinning was performed at room temperature by homemade electrospinning device. The applied electric field was 60 kV, and the distance between the spinnerets and the collector was 32 cm.
Cross-linking of PVA nanofiber mats
The cross-linking process was carried out referring to the method reported by Wang et al. [9,10]. Electrospun PVA mats were cut into 5 cm × 5 cm samples and immersed in a given concentration of GA (GA is used to cross-link the hydroxyl group in PVA, and the concentration varies from 0.02% to 30%) and trace HCl (as the catalyst of GA reaction with PVA) in acetone solution for 0.5 h. Acetone was used as a reaction medium to prevent PVA from dissolving into water during the cross-linking process. The cross-linked PVA mats were taken out of the modified solutions and washed in the distilled water several times and then dried for 24 h before use.
Water durability measurements
The cross-linked PVA nanofiber mats were immersed in boiling water for 2 h, followed by carefully wiping the surface water with a filter paper and weighed immediately (W1). Then they were dried in a vacuum at room temperature for 48 h and weighed again (W2). The initial weight of each sample was W0. The weight loss (WL) and the swelling degree (WS) of the samples were calculated using the equations (1) and (2):
Mechanical properties
A mechanical property (tensile strength, samples width = 5 mm, length = 30 mm) was determined by the XQ-2 fiber tensile tester. The samples were conditioned for 24 h under constant temperature and humidity before tested.
Thermal analysis
The thermal behavior of PVA and PVA/GA mats was investigated by differential scanning calorimeter (DSC204F1, German) and thermogravimetry (TG) (TG209F1, German). The DSC cooling and heating rates were maintained at 10℃ min−1 from room temperature to 700℃. The TG heating from room temperature to 1000℃ under a nitrogen flow of 120 ml min−1 with a heating rate of 20℃ min−1.
X-ray diffraction
The PVA nanofiber mats were cut into small pieces. The samples were determined with a D/max-2550-X-ray power diffractometer with Ni-filtered Cu Kα radiation at 40 Kv and 200 mA. The scanning range (2θ) was 5–60°.
Results and discussion
The qualities of PVA nanofiber mats
In the process of electrospinning, different polymer concentrations have different effect. Compared with other polymer concentrations, the optimal concentration of PVA for producing nanofiber mats was 10%. Apparent characteristics of the samples are shown in Figure 1.
Apparent characteristics of poly (vinyl alcohol) (PVA) nanofiber mats.
Nanofiber mats after cross-linking
When concentration of modifying solution (GA) increased from 0.02% to 30%, there were obvious changes in performance of PVA nanofiber mats. In general, when the concentration of modified solution (GA) was below 1%, the nanofiber mats became thinner and broadly contracted. The reaction of PVA nanofiber mats with modified solution failed to form compact network on the surface of the mats. The PVA nanofiber mats became thinner, transparent and compact after dried as a result of the PVA nanofiber mats can easily be dissolved in the water in its modified solution. When the concentration of modified solution was 15%, it could form compact network on the surface of the mats, however, there was so much water in modified solution that this water could swell or even dissolve the mats. The change of mats’ stiffness (Figure 2) also explained that with the increasing of concentration, the degree of cross-linking increased, so the mats became more compact and stiff.
Photographs of (a) 0.02% GA-modified PVA mats, (b) 5% GA-modified PVA mats, (c) 30% GA-modified PVA mats. PVA: poly (vinyl alcohol); GA: glutaraldehyde.
Water resistance of cross-linked PVA nanofiber mats
Ten percent concentration of PVA nanofiber mats

PVA: poly (vinyl alcohol); W1: weight of samples after immersion in water; W2: dried weight of samples after immersion in water; W0: initial weight of sample before cross-linked with GA; WL: weight loss; WS: swelling degree.
PVA can be dissolved in hot water quickly and in cold water slowly, but if the PVA mats were completely cross-linked with GA, the hydroxyl groups in PVA macromolecules reacted with GA and the whole PVA mat formed a linked macromolecule, thus the mat could not be dissolved in water. If the mat was partly cross-linked with GA, some of the mat which was not cross-linked can be dissolved in water, the weight loss occurred.
After the mat is cross-linked with GA, the whole PVA cannot be dissolved in water, but the hydroxyl groups in PVA are hydrophilic and it can form hydrogen bonds with water, thus it can absorb a lot of water and it can swell.
Figures 3 and 4 show the weight loss percentage and swelling degree of samples, respectively, with various concentrations of GA from 0.02 to 30%. It can clearly see that with the concentration of GA increased, the weight loss percentage and swelling degree of samples became smaller in general.
Weight loss percentage of PVA nanofiber mats at different GA concentrations. PVA: poly (vinyl alcohol); GA: glutaraldehyde. Swelling degree of PVA nanofiber mats at different GA concentrations. PVA: poly (vinyl alcohol); GA: glutaraldehyde.

Mechanical properties of nanofiber mats
The mechanical properties of each PVA nanofiber membrane sample

PVA: poly (vinyl alcohol).
aTest time is five.
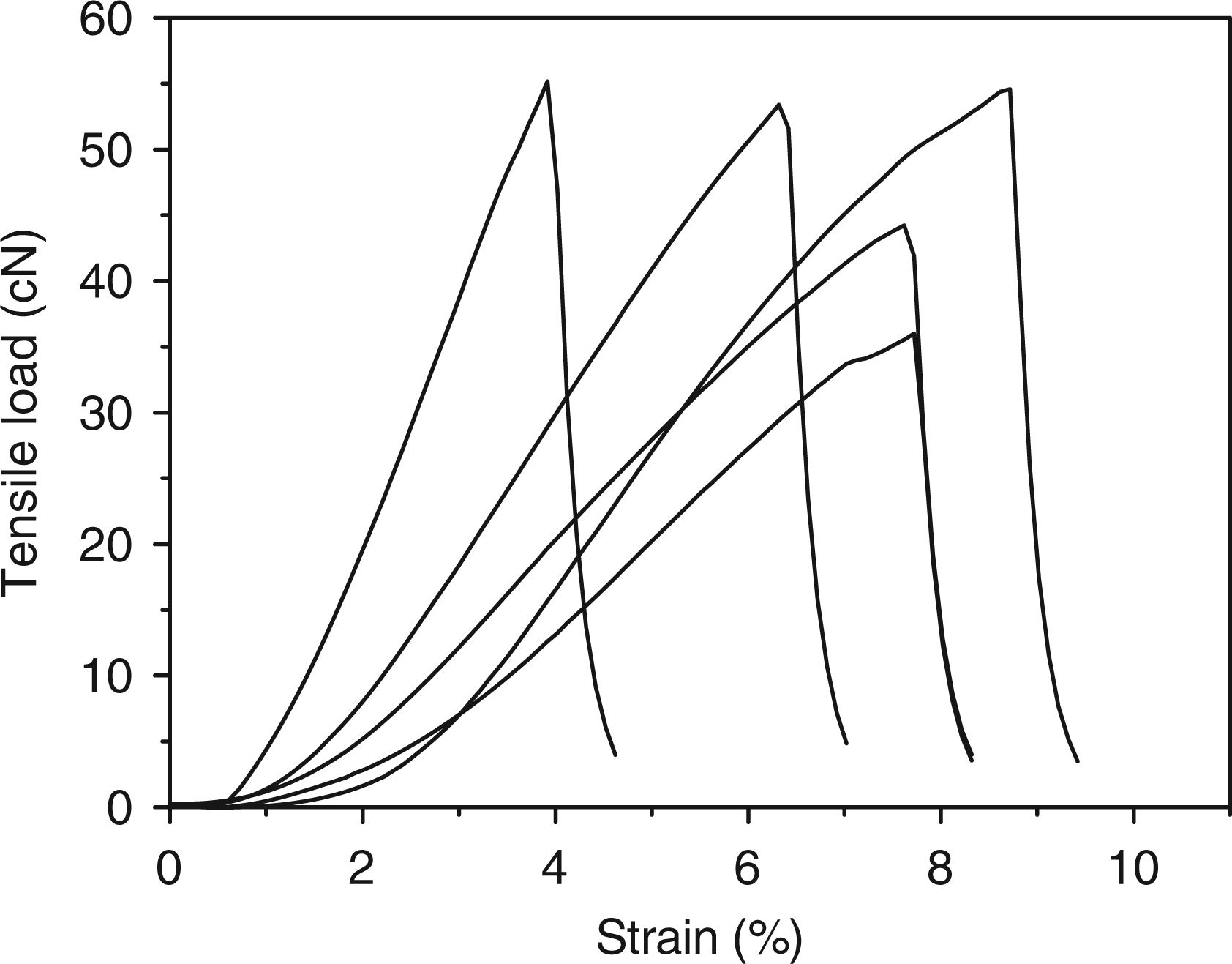
The stress and elongation curves of PVA nanofiber mats before and after cross-linking are shown in Figures 5 and 6, respectively. The results indicate that the modification enhance the stiffness of electrospun PVA mats.
Stress-elongation curves of unmodified PVA mats. PVA: poly (vinyl alcohol). Stress-elongation curves of modified PVA mats. (5% GA-modified PVA mats). PVA: poly (vinyl alcohol); GA: glutaraldehyde.
Thermal properties
DSC was a valuable technique for investigating the curing reaction of cross-linked polymers. Figure 7 presents the DSC thermograms over the temperature range 50–300℃ for PVA powder, PVA mats and modified PVA mats. As shown in Figure 7, there are two obvious endothermic peaks in curves (b) and (c) at 95℃ and 86℃, respectively, which maybe related to the moisture of the samples. The second endothermic peaks appear around 200℃, and the melting temperature of the modified PVA mats is higher than PVA powder and unmodified PVA mats. The increase in Tm of the modified PVA mats is a result of the decrease in PVA chain mobility due to the chemical cross-links. PVA is a semi-crystalline polymer [11], chemical cross-linking can change its structure, different cross-linking agents and cross-linking density will produce different effect on Tm. The hydrogen bonds were disturbed by cross-linking agents, which will cause to change the stiffness of the chains. Meanwhile, the cross-linking agents will also lead to form new chains. Much aldehyde groups and ether bonds were formed in 5% PVA/GA, and the chains have high cross-linking density, high stiffness, which can explain the modified PVA mats have higher Tm.
DSC thermograms of (a) PVA powder, (b) unmodified PVA mats, (c) 5% modified PVA mats. DSC: differential scanning calorimeter; PVA: poly (vinyl alcohol).
The TG and differential thermogravimetry (DTG) curves are presented in Figure 8. The PVA powder has three main thermal degradation steps [12]: the first one, around 100℃, due to the release of volatile compounds, mainly water, which were present in the hydrophilic material. The second stage, at 330℃, was regarded as the elimination of acetic acid to form a polyene. The third stage, around 450℃, corresponded to the breakdown of the polymer backbone of the main chain of PVA.
TG (A) and DTG (B) curves of (a) PVA powder, (b) unmodified PVA mats and (c) 5% modified PVA mats. DTG: differential thermogravimetry; PVA: poly (vinyl alcohol); GA: glutaraldehyde; TG: thermogravimetry.
The curve (c) also has three degradation steps, the trend of which is similar to curves (a) and (b). In comparison to unmodified PVA mats, the first stage of weight loss was slightly reduced in the modified PVA mats, which indicated a decrease in membrane hydrophilicity. Also, the degradation happened at higher temperatures, 382℃ and 452℃, which suggested that the membrane thermal stability increased by cross-linking PVA mats with GA. This method of modification provided a useful protocol for increasing thermal stability by using cross-liking PVA mats with GA in different concentration.
The results of X-ray diffraction (XRD) are shown in Figure 9 [13,14]. Figure 9 shows there is an intense peak appearing near 19.2° (2θ), as a result of hydrogen bond function. When the PVA electrospun to PVA membrane, as shown in Figure 9, the diffraction peak is obviously weak, due to the rapid solidification process of stretched chains under high elongation rate during electrospinning, and the most chains are under amorphous structure. The crystallinity analysis shows the degree of crystallinity of unmodified PVA mat is 43.53%, less than 51.85% of PVA powder. Compared with the unmodified PVA mat, there is also a diffraction peak appearing near 19.2°(2θ) of the modified PVA mat, but much weaker. This result indicates that the chemical cross-linking hinders the formation of PVA crystals [15], causing the degree of crystallinity to reduce to 30.13%.
XRD date of (a) PVA powder, (b) unmodified PVA mats and (c) modified PVA mats. PVA: poly (vinyl alcohol); GA: glutaraldehyde; XRD: X-ray diffraction.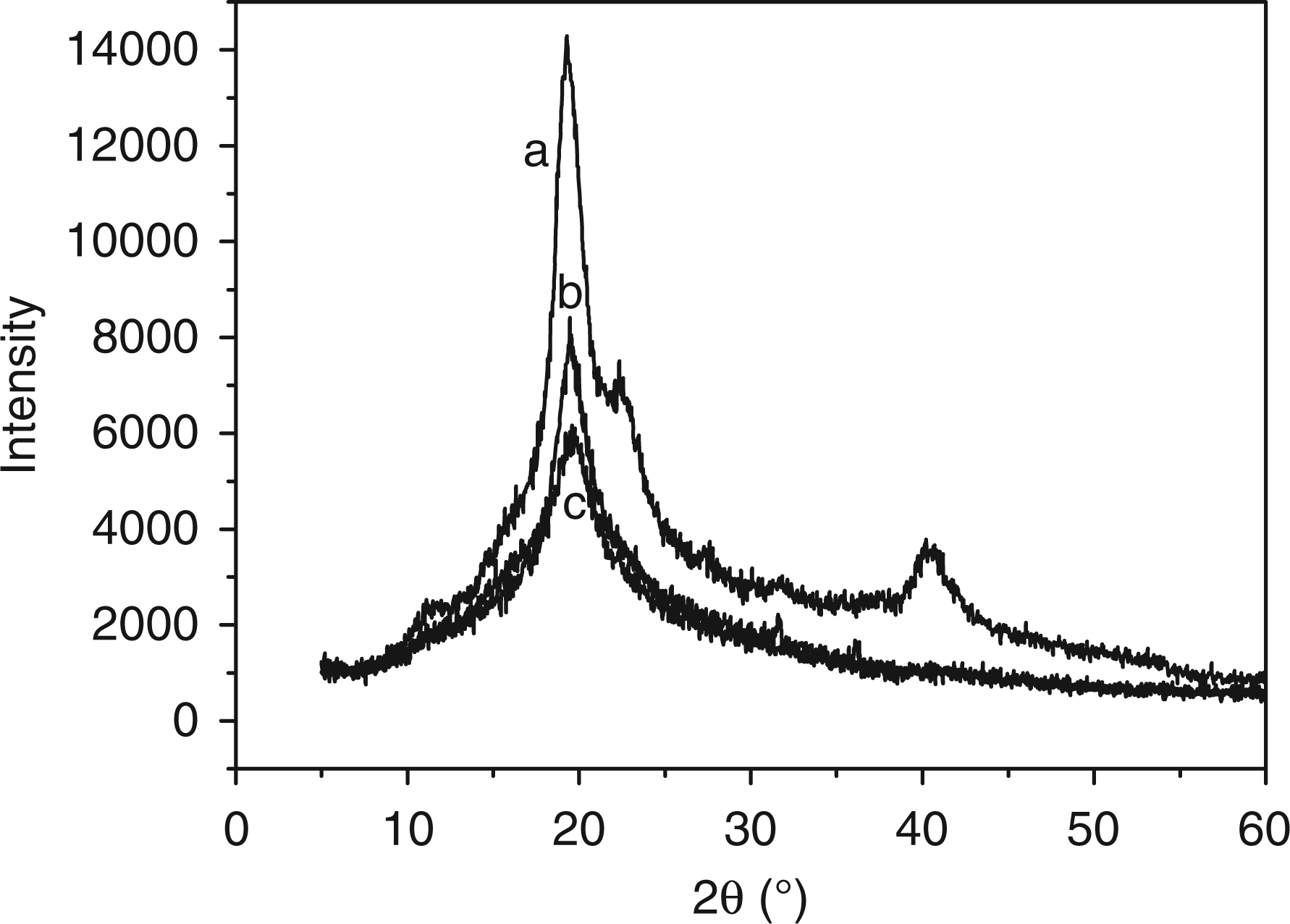
Conclusion
PVA nanofiber mats were cross-linked with GA. The chemical structure of the PVA nanofiber was changed by the addition reaction of hydroxyl groups in PVA and aldehyde groups in glutaraldehyde, thus the hydrophilicity of the PVA decreased. Water durability test found modified PVA nanofiber mats can effectively improve the stability in water. DSC and TG results showed the cross-linking between PVA and GA increased the thermal stability of the modified PVA mats and XRD showed a decrease in the crystallinity. The modified PVA mats, which have excellent stability in water and thermal stability, may lead to broaden its application. Enzyme and drug could be trapped in these mats independently to study sustained release. These hybrid cross-linked nanostructures have presented 3D pore size architecture for potential use in tissue engineering applications. The PVA nanofiber system has a great potential in decolorizing dyeing wastewater for environmental applications.
Footnotes
Funding
This work was partly supported by grants (50973014 and 11172064) from the National Natural Science Foundation of China and from the Foundation for the Author of National Excellent Doctoral Dissertation of P.R. China (200961), as well as sponsored by Shanghai Rising-Star Program in China (10QA1400100) and Fok Ying Tong Education Foundation (121071). Furthermore, it was also Supported by Program for New Century Excellent Talents in University and the Fundamental Research Funds for the Central Universities as well as “Shu Guang” project supported by Shanghai Municipal Education Commission and Shanghai Education Development Foundation.


