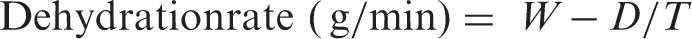Abstract
In this study, two common nonwoven fibrous dressings, based on carboxymethylcellulose and alginate, and their silver-treated forms were evaluated in order to compare their performance characteristics including fluid handling capacity, the mechanical properties, and the thermal behavior. The effect of immersing the dressings in the two fluids (solution A and distilled water) on pH was also studied. The results show some clear differences between the untreated and silver-treated dressings, for example the fluid handling and the swelling values of the dressings were reduced due to silver treatment. More considerably, changes were observed in the tensile properties which were decreased when these dressing were treated with the silver. There was no considerable effect on the thermal characteristics of the dressings upon the silver treatment. Statistically significant differences were observed in the pH over time for all tested specimens and also for both test fluids.
Introduction
In recent years, there has been a very marked increase in the interest and utilization of smart and bioactive fibers for wound management. These fibers have considerable scientific and commercial attraction. In general, wound dressings are classified in a number of ways: (a) functionality (antibacterial, absorbent, adherence, occlusive, debridement); (b) material type (alginate, carboxymethylcellulose (CMC), collagen, hydrocolloid); (c) physical form (nonwoven, foam, film, composite, gel); and (d) application period (primary, secondary, island). Besides these classifications, the wound dressings can be divided into two types, i.e. traditional and modern (advanced) wound dressings [1–4].
Acute and chronic wounds frequently become colonized with wide variety of bacteria, including Methicillin-resistant Staphylococcus aureus (MRSA), and these are a potential source of infection in the healthcare environment. Recent studies have shown that nanocrystalline silver dressings could be used as an effective barrier for wound infected or critically colonized with MRSA. This implies that their use could reduce the incidence of hospital acquired infections (HAIs). The data, both in vitro and in vivo, have demonstrated that the nanocrystalline silver dressing used had a bactericidal effect, killing MRSA in the laboratory and on patients [5–7]. The crystalline structure of nanocrystalline silver is metastable, and in a high-energy form of elemental silver has a smaller-sized crystal (about 15 nm) than normal silver within the same area, resulting in an increased surface area. Unlike normal silver, which does not dissolve in water, nanocrystalline silver dissolves to provide a concentration in solution of around 70–100 parts per million (ppm), which is three to five times higher than the levels required to kill MRSA. These physical properties of nanocrystalline silver allow its sustained release and maintain the desired levels. Different technologies are used to deliver silver from the dressings into the wound bed, and research has shown that in order to be most effective at killing bacteria, the silver has to be released into the wound bed at levels in excess of 20 ppm. However, not all dressings are designed to release silver into the wound bed; some take away the bacterial burden along with exudate, and then kill the bacteria within the dressing [8–10].
The pH value of the wounds is one of the important parameters for healing process due to pH and biochemical reaction speed interaction [11]. It has been reported that acidic exudate helps rapid wound healing. Changing the wound environment from alkaline to acidic can increase the rate of healing for chronic wounds (venous leg ulcers) [12–14]. Silver is the most important topical antimicrobial agent in wound management and numerous dressing products have been developed using silver as an antimicrobial agent. Silver-treated materials have been proven to possess bactericidal properties and are considered to be generally safe and effective in controlling the growth of bacteria. The silver kills the microbes on contact through multiple mechanisms such as inhibiting cellular respiration, denaturing nucleic acids, and altering cellular membrane permeability. Silver ions have been incorporated in different materials such as alginate, carboxymethylcellulose, hydrocolloid, and foam dressings for the treatment of acute and chronic wounds [15–17]. In addition, some procedures rely on the lower pH and antimicrobial efficiency interaction. In general, the wounds exhibit a pH higher than 7.4, which delays the healing process. Pathogenic bacteria require a higher pH value to proliferate. It has been proven that infected wounds have a high pH [18,19]. The lower pH environment has an inhibiting effect on the growth of some bacteria [20]. Recently, Slone et al. [19] determined the pH value of silver-treated alginate dressing and its antimicrobial efficiency. They emphasized the potential of a lower pH in wound treatment with the advantage that it can be applied as a topical antimicrobial isolate on chronic wounds. They analyzed 25 bacteria and yeasts on chronic wounds treated with silver containing alginate dressing. They reported that the silver alginate dressing showed a broad spectrum of antimicrobial barrier activity against all wound isolates. Furthermore, at a pH of 5.5, the antimicrobial barrier activity of silver alginate dressing significantly increased when compared with a pH of 7 [21].
Two main types of modern fibrous wound dressings are currently available on the market. Those based on alginate fibers, produce weak gels on contact with wound exudates. The other, based on carboxymethylated spun cellulose fibers (CMC), form a much stronger and more cohesive gel following hydration. CMC-based dressings are mainly used for heavily exuding wounds due to their unique absorbency properties. CMC fiber-based dressings have strong initial absorption but they have undesirable lower lateral wicking [22]. The aims of this study were twofold. Initially, it was proposed to compare CMC and alginate fiber-based modern dressings regarding the fluid handling capacity, the mechanical properties, the thermal behavior, and the pH. Second, the effect of silver treatment on the above-selected properties of these fibrous dressings was investigated.
Materials and methods
The study was conducted on carboxymethylcellulose (CMC) and alginate-based fibrous nonwoven dressings, and their silver-treated forms. Aquacel® (CMC), (ConvaTec, Skillman, NJ, USA) and Algisite* M (alginate) (Smith & Nephew, London, UK), and their silver-treated forms Aquacel® Ag and Algisite* Ag, respectively. The dressing specimens were purchased from the UK market. Prior to all the testing, the dressing specimens were conditioned for 48 hours in 65% relative humidity and 20°C atmosphere. As a preliminary test, solution A was used which consisted of sodium chloride (2.298 g/L), calcium chloride dihydrate (0.368 g/L), and distilled water. The solution A was prepared to simulate serum and wound fluid. Dressing mass (g/m2) and thickness (mm) were determined in accordance with BS EN 12127:1998 and BS EN ISO 9073-2:1997, respectively. The following experimental tests were performed in this study on the dressing specimens.
Absorbency, dehydration rate, swelling, and vertical wicking
The absorbency of wound dressings was determined using BS EN 13726-1:2002 section 3.2 free swell absorptive capacities. For this test, 5 × 5 cm2 dressing specimens were prepared. All the prepared dressing specimens were weighed and recorded before testing and placed in Petri dishes. The solution A was warmed to 37 ± 1°C, and 40 times the mass equivalent of the specimen was dispensed slowly and gently onto the specimens in the Petri dishes. The Petri dishes were then placed in an incubator for 30 minutes at 37 ± 1°C (body temperature). After 30 minutes of conditioning, the dishes were removed from the incubator and suspended by one corner using tweezers to allow excessive solution to drip off for 30 seconds and reweighed for wet mass. The dehydration behavior was assessed by measuring the difference between the mass of wet and dry specimens. The specimens were dried in an incubator for 24 hours at 37 ± 1°C [23].
The calculations of the dehydration rates were made using the following equation:
The swelling of fibrous dressings was measured using Labophot-2 optical microscope at a magnification of 200 × . The image Proplus software was used to obtain images of the fibers and the diameters were determined. The solution A and distilled water were introduced into the fibers without removing fibers from the microscope and was allowed to swell for 1 minute. Five replicate tests were carried out for each fiber types. The image of the swollen fiber was captured and the diameters compared for both dried and wet fibers. The vertical wicking test specimens were prepared to 15 mm width and 100 mm length along machine direction. The vertical wicking tests were performed five times for each dressing specimen. Eosin B was added into the solution A to determine the fluid wicking distance. The specimens were slowly immersed into the solution vertically up to 10 mm length and left for 60 seconds [23–25].
Tensile properties
The dressing specimens were prepared in 15 cm length × 5 cm width. The specimens were taken from the machine direction of the dressings. The specimens were then mounted in the grips of an Instron universal tester with 100 mm gauge length and 300 mm/minute extension rate. For each dressing types, five measurements were carried out and the mean values were calculated.
Investigation of thermal degradation
The experimental conditions were heating rate 10°C/min; nitrogen purges rate 30 cm3/min; fiber sample weights in the range of 25–35 mg.
The pH value evaluation
The dressings were fully immersed in the fluids in order to determine the pH value. The pH measurements were taken over a test period of 7 days, which is the recommended maximum period of usage for most of the dressings investigated in this study. The pH measurements were performed using Hanna HI 8424 pH-meter at room temperature, and the pH meter was calibrated for each and every group of measurements with the buffer solutions pH 4.01 and pH 7.01. Analysis of variance (ANOVA) was employed to determine the significance of the pH change in general and Student t-test for assessing the statistical significance of the differences between the control fluids and each applied dressing with the fluids.
Results and discussion
Absorbency, dehydration rate, swelling, and vertical wicking
The dressings were investigated as described above, and the test results of the various specimens are given in Table 1. The mass per unit area and thickness of the alginate dressings were higher than the CMC dressings. A number of factors can affect the fluid absorption of the dressings [26,27]. The CMC fiber-based dressing’s absorption of solution A (22.1 g/g) was similar to the silver-treated CMC dressing’s absorption (20.9 g/g), and the alginate dressing’s absorption was 17.2 g/g, which was also similar to its silver-treated counterpart (16.1 g/g). There were small differences (∼5%) between the untreated and silver-treated nonwoven dressings but these are not as noteworthy for moderately exuding wounds as they are for heavily exuding wounds. The differences between CMC and alginate fibers were found to be considerable due to higher absorbency of CMC fiber. As seen in Figure 1, the mass and thickness of the dressing did not directly affect its absorption behavior. The alginate dressings had higher mass per unit area and thickness, but for both cases, silver-treated and untreated alginate dressings had lower absorbency compared to the CMC fiber dressings. Similar results have been reported in previous studies [23,26,28]. The dehydration rate of the silver-treated dressings was greater than the untreated dressings and also alginate-based dressings had higher dehydration rates compared to the CMC-based dressings. The dehydration rate ranged from 0.022 to 0.040 g/min. All the dressings dried out within 8 hours.
Thickness and fluid handling properties. Test results of nonwoven dressings. Under 5 g/cm2 pressure.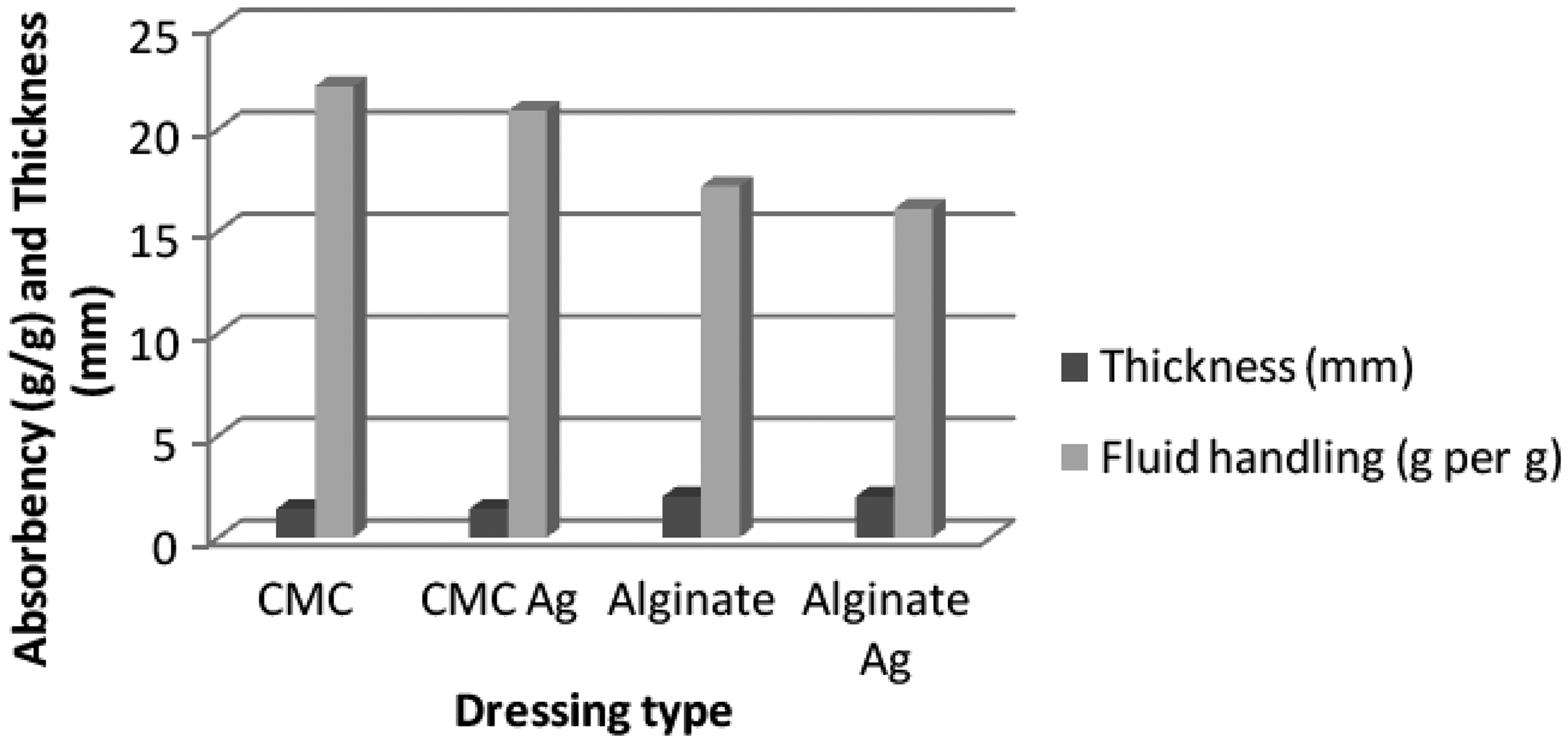

In terms of swelling properties, the dressings demonstrated two different characteristics with respect to the applied test fluids. The silver-treated dressings had lower swelling ratio in distilled water compared with the untreated dressings. For the application of solution A, totally different pattern of swelling behavior was observed as the silver-treated dressings exhibited higher swelling ratio than the untreated specimens. This may be due to the reaction between silver and solution A. When the solution A was applied onto the silver-treated CMC and alginate fibers, the swelling ratio of these fibers were much higher than their untreated counterpart. Silver-treated alginate had the lowest swelling ratio when tested with water. These results also show that CMC fiber-based dressings show significantly higher swelling properties than the alginate fiber-based dressings (Figure 2).
Solution A and distilled water swellings of dressings.
For both fiber types, the silver treatment affected the vertical wicking characteristics and treated dressings had slightly lower vertical wicking compared to the untreated dressings (Table 1). In a previous study, the vertical wicking distance of CMC dressings was found to be 12.5 mm [23], which was confirmed by the results of this study.
Tensile properties
The CMC dressing’s tensile strength was 51.7 N and its silver-treated form had a 22.8 N tensile strength, while the alginate dressing’s tensile strength was 5.4 N and the value for its silver-treated form was 4.1 N. It can be observed that the silver treatment has a noticeable impact on the tensile strength of both types of dressings. In both cases, the tensile strength values of silver-treated dressings were much lower than their untreated counterparts. Furthermore, the CMC dressing exhibited significantly higher tensile strength values than the alginate dressing (Table 1 and Figure 3). The breaking extension of the dressings was increased by the silver treatment. For both cases, the alginate-based dressings have higher breaking extension compared to the CMC fiber-based dressings. Wasif and Laga treated cotton fabric with nano-silver and they too found that silver treatment decreased the tensile strength of the fabrics [29]. There are two scenarios that could explain this decrease. The first scenario is that the tensile strength could be affected because of the interaction between the metal ions and the polymer structure. The metal ions could cause the fibers to become more brittle, and this can have a negative impact on the physical properties of the fiber. The second scenario could be that the presence of silver could make the fabrics more dry, which also could cause a reduction in the tensile behavior. These findings require further in-depth study, in order to establish the true impact of silver and how it can cause a decrease in fabric tensile properties.
Tensile strength properties of dressings.
Thermal properties
The effect of silver treatment on the thermal stability of CMC and alginate fibers was studied by using differential scanning calorimetry (DSC). The results illustrated in Figure 4 show that the untreated and treated fibers demonstrated similar thermal behavior for both types of fibers. The differences between CMC (endothermic peak 32.14°C and exothermic peak 288.17°C) and CMC Ag (endothermic peak 31.34°C and exothermic peak 293.01°C) were not found to be noteworthy. Both alginate fiber forms had higher endothermic peaks than the CMC and CMC Ag, on the other hand, exothermic peaks of the CMC and CMC Ag fiber were higher in comparison to alginate and alginate Ag.
DSC curves of various fibers used in the dressings. ANOVA plot for the dressings that immersed into the solution A (A) and the distilled water (B) with 95% confidence interval.

The pH value evaluation
Mean pH values, standard deviation (SD), and t-value of the solution A after the dressing immersion over a 7-day testing period.

Mean pH values, SD, and t-value of the distilled water A after the dressing immersion over a 7-day testing period.

The ANOVA statistical test results for the solution A after the dressing immersion.

The ANOVA statistical test results for the distilled water dressing immersion.

It should be noted that the immersion of alginate Ag dressing in both fluids resulted in appreciably higher pH values compared to the rest of the specimens (Figure 5). CMC fiber-based dressings slightly reduced the pH value of the solution A and distilled water. However, the differences may not be clinically important, particularly for high-exuding wounds. The higher pH values of the fluids due to the immersion of alginate-based dressings could have been a result of the presence of sodium ions in the alginate fiber structure.
The pH is an important parameter for the human body due to its effect on the speed of biochemical reactions. It has been demonstrated that pH has an essential role to play during the healing process and an acidic environment is more beneficial for the wound healing process. According to our results, the silver-treated alginate dressings for both test fluids had the highest alkaline values over a 7-day test duration, which could have a negative impact on wound healing time [30].
Conclusions
The aim of this study was to evaluate the effect of silver treatment on the properties of different types of wound dressings. The efficiencies of the CMC-based dressings and the alginate-based dressings were highlighted in this experimental study. From the test results, the following conclusions can be drawn.
It was noted that the absorbency of both types of dressings was reduced by 5% after the silver treatment. This appears to be mainly due to the water repellent influence of silver coating applied to the dressings – the silver particles do not allow fluid to imbibe fully into the fiber’s structure. The CMC-based dressings were thinner and lighter than the alginate-based dressings, although they have significantly higher absorbency values for both the untreated and the silver-treated dressings.
The swelling properties of the CMC fiber are superior to the alginate fiber due to its higher absorbency behavior. The swelling ratio with distilled water was lower for the silver-treated specimens, on the other hand, the silver-treated dressings had significantly higher swelling ratio compared to the untreated dressings with solution A. The swelling differences due to the different test fluids have occurred due to the interaction of silver and the Na+ ions in solution A, which leads to somewhat higher swelling ratio of the dressing.
In case of vertical wicking results, the alginate-based dressings had more than two times higher values compared to the CMC-based dressings. This is an expected outcome because the CMC-based dressings have higher absorbency and swelling over a certain time. The silver treatment decreased the vertical wicking around 10% in both types of dressings.
The alginate dressing demonstrated poor tensile behavior. In all cases, it has 10 times lower tensile strength value than the CMC dressing. The silver treatment had a negative influence on the tensile properties. The breaking extension of the alginate dressing was higher than the CMC dressing.
The statistical analysis results clearly demonstrated that the pH values of the fluids are not constant due to the complicated nature of the dressings’ chemical structures and designs. The silver-treated alginate had significantly higher influence on the pH values of the fluids compared to the rest of the specimens. CMC fiber-based dressings slightly reduced the pH value of the solution A and the distilled water, while the alginate-based dressings had an alkaline effect on the fluids.
Footnotes
Funding
This work is supported by The Turkish Council of Higher Education in collaboration with Marmara University, Turkey and the University of Bolton, UK.