Abstract
Nano-zirconium-oxide (nano-Zr-oxide) and nano-silver-oxide (nano-Ag-oxide) were in situ synthesized and deposited into cotton gauze fabrics by reduction of zirconium oxychloride or silver nitrate solutions, in the presence of fabric samples using sodium hydroxide-hydrogen peroxide mixture solution at pH 9.5. The resulted homogenous distribution of nano-Zr-oxide and nano-Ag-oxide inside the fabric were characterized by scanning electron microscopy coupled with energy dispersive X-ray (SEM-EDX), transmission electron microscopy (TEM) for fabric cross-section. The formed nano-Zr-oxide and nano-Ag-oxide in the prepared solution was characterized by TM, X-ray diffraction (XRD), and UV visible spectroscopy. Antibacterial activity of prepared samples was evaluated using reduction rate % in bacterial count (RBC %) against gram-negative bacteria (Salmonella typhimurium) and gram-positive bacteria (Staphylococcus aureous), while antifungal activity of prepared samples was evaluated according to clear inhibition zone diameter against filamentous fungus (Candida albicans), as well as nonfilamentous fungus (Aspergillus flavus) the data show that, the reduction rate of colony count of treated fabric samples with nano-silver oxide against gram positive and gram negative bacteria were 99.9% and 97% respectively. Gave 99.9% and 97% reduction in colony count against gram-positive and gram-negative, espectively, while sample fabrics treated with nano-Zr-oxide gave 98% and 95% reduction rate in colony count against gram-positive and gram-negative, respectively. Antifungal activity of fabric treated with nano-silver oxide was greater than that of fabrics treated with nano-Zr-oxide. Skin irritation test of treated fabric samples was evaluated and the data show that, there is no skin irritation was observed on the skin of tested rabbit. Skin wound of tested rabbit was healed after 4 days sample application. All prepared samples were durable to wash and killed the bacteria even after 30 laundering wash cycles.
Introduction
Due to the outbreak of infectious diseases caused by different pathogenic bacteria and the development of antibiotic resistance, specialists are on the lookout for newantibacterial compounds. At present, nanoscale materials have emerged as novel antibacterial agents due to their high surface area to volume ratio and unique chemical and physical properties [1,2]. Moreover, nanotechnology is emerging as a rapidly growing field with its application in science and technology for the purpose of manufacturing new materials at the nanoscale level [3]. Different types of nanomaterials like copper, zinc, titanium [4], magnesium, gold [5], alginate [6], and silver (Ag) have come up. Among these, silver nanoparticles have proved to be the most effective as they have good antimicrobial efficacy against bacteria, viruses, and other eukaryotic microorganisms [7]. Silver ion or metallic silver as well as silver nanoparticles can be exploited in medicine for burns treatment, dental materials, textile fabrics, water treatment, sunscreen lotions etc. and posses low toxicity to human cells [8]. The role of nanocrystalline silver dressings in wound healing management had been investigated [9]. The topical delivery of silver nanoparticles promotes healing of burn wounds with better cosmetic appearance and provides an effective therapeutic direction for scarless healing of wounds [10].
In addition, mesoporous zirconium (Zr) oxide nanoparticles have opened many new possibilities for applications in drug delivery, as well as for protein encapsulation. Over the past few decades, preparation techniques for mesoporous structures have attracted the attention of many researchers [11–13]. Furthermore, zirconium oxide is an important ceramic material because of its high thermal and chemical stability [14]. Zirconium oxide films have a high dielectric constant [15–17], low thermal conductivity [18–21], and excellent wear resistance [22–24]. The uses of Zr compounds continue to be widely and extensively used in deodorant and antiperspirant preparations. In the public health arena, Zr compounds have been studied or used in controlling phosphorus pollution and in the reclamation of poison and bacteria-contaminated water. Experimental and clinical studies support the general consensus that Zr compounds are biocompatible and exhibit low toxicity [25].
Furthermore, the development of new clothing products based on the immobilization of nanophased materials on textile fibers have recently received growing interest from both academic and industrial sectors [26]. A wide range of nanoparticles with various structures can be immobilized on the fibers, which brings new properties to the final textile product. These textiles can be widely used for hygienic clothing, wound healing, and medical applications in hospitals and other places where bacteria present a hazard [27].
The aim of this work is to develop a new method for enhancing antimicrobialand wound healing acceleration properties to cotton gauze bandages by immobilization of nano-zirconium-oxide (nano-Zr-oxide) and nano-silver-oxide (nano-Ag-oxide) into cotton gauze fabric using reduction method. The prepared nanoparticles were characterized by using UV-visible spectroscopy and transmission electron microscopy (TEM), as well as XRD in its colloidal solutions. Prepared cotton gauze fabrics containing nano-Zr-oxide and nano-Ag-oxide were characterized using TEM of cross-sectional samples, scanning electron microscopy-energy dispersive X-ray (SEM-EDX). Antibacterial activity of prepared cotton gauze fabric samples quantitatively against gram-positive and gram-negative bacteria as well as antifungal activity qualitatively against filamentous and nonfilamentous fungi were evaluated. Biological activity of prepared samples for skin irritation and wound healing were evaluated.
Experimental
Materials
Bleached and sterilized (100%) woven cotton gauze fabric 10 cm × 500 cm was purchased from Misr Company for Spinning and Weaving-Mahalh El Kubra, Egypt. Zirconium oxichloride and silver nitrate were purchased from Aldrich Co. and poly (vinylpyrrolidone) (PVP) as stabilizer was purchased from Sigma Co. All laboratory chemicals used were of analytical grade.
Method
Preparation of nano-Zr-oxide and nano-Ag-oxide/cotton gauze fabrics
Preparation of nano-Zr-oxide and nano-Ag-oxide cotton gauze fabrics were prepared according to reported method [28], the experimental technique was adjusted as follows: nanometal oxides deposition was performed by padding the cotton gauze fabric samples in an aqueous solution containing 50 mMole of metal salts solution (zirconium oxichloride or silver nitrate) and 1.5 wt% (w/v) of PVP as stabilizing agent. The fabric was then squeezed to a wet pick up 100%. Padded cotton gauze fabric was padded twice in the reducing bath containing 4 g/L sodium hydroxide and 10 mL H2O2 (35%) at pH 9.5 then squeezed to at wet pick up of 100%. The treated fabric was wrapped in polyethylene package, heated at 50°C for 20 min, and finally the fabric was thoroughly washed with water for 45 min at 50°C and dried at 60°C for 20 min.
Testing and analysis
Prepared nano-Zr-oxide and nano-Ag-oxide colloidal solutions were characterized using UV-visible spectroscopy, TEM (ZEISS-EM-10- GERMANY) as well as XRD.
Treated cotton gauze fabrics with nano-Zr-oxide and nano-Ag-oxide were characterized using TEM images of the cross section of treated cotton gauze fabric [29,30], and nano-Zr-oxide and nano-Ag-oxide in situ deposited into cotton gauze fabrics were examined qualitatively and quantitatively using energy dispersive X-ray spectrum (SEM-EDX), coupled with SEM (type JXA-840 an electron probe microanalyzer-JOEL).
Antimicrobial activity
Antibacterial properties of untreated and treated cotton gauze fabrics with nano- Zr-oxide and nano-Ag-oxide were quantitatively evaluated against gram-positive (Staphylococcus aureus) and gram-negative bacteria (Salmonella typhimurium) according to the standard test method used to measure the reduction rate in the number of colonies formed [31], as following: 1 g fabric sample was dipped into a test tube containing the bacteria culture solution in which the bacteria concentration was 2.5 × 105/mL the test tubes were shaken at 35°C for 1 h on a rotary shaker at 100 rpm and 1:100 dilutions of the test solutions were made. One milliliter of the diluted test solution was poured onto agar broth and incubated at 35°C for 24 h andthe number of colonies in the agar broth was calculated using the following equation:

Where A is the number of colonies before shaking and B is the number of colonies after 1 h shaking. In addition, the antifungal activity of the prepared fabric samples such as filamentous (Candida albicans) and nonfilamentous (Aspergillus niger) fungi have been evaluated according to AATCC test method 147.
Biological activity and wound healing
Cotton gauze fabrics treated with nano-Zr-oxide and nano-Ag-oxides were tested for skin irritant and wound healing accelerator on rabbit skin according to the method [32] described by organization for economic cooperation and development (OECD)-guidelines for the testing of chemicals (test no. 404). The treated fabric samples were applied in single strips to the skin of an experimental animal; untreated skin areas of the test animal served as the control. The degree of irritation/corrosion was observed and scored at specified intervals and was further described in order to provide a complete evaluation of the effects. The duration of the study should be sufficient to evaluate the reversibility or irreversibility of the effects observed.
Test procedure
Healthy young adult albino rabbits, New Zealand white strain, were used (three rabbits for every examined treated fabric). The average body weight ranged between 1150 and 1250 g. All animals were housed in metal cages and fitted with perforated floors; water and standard feed were given. Fur should be removed by clipping the dorsal area of the trunk of the animals about 6 cm2, at least 24 hours before the experiment. The untreated fabric samples and treated cotton gauze fabrics with nano-Zr-oxide and nano-Ag-oxides were applied to the clipping area of the tested rabbit skin, with the treated fabric samples attached to the skin with good skin contact. Then, the fabric samples were held in place with non-irritating tap. At the end of exposure period, which is normally 4 h, the treated sample fabrics should be removed. Clinical observation and grading of skin reaction was observed. All animals were examined for signs of erythema and edema and the response scored at 1 h and then at 24, 48, and 72 h till day 14.
Durability
The durability of prepared fabric samples to wash was determined according to AATCC Method 124. The fabric samples were laundered 30 cycles with a soap detergent in a washing machine with full water level at 40°C for 10 min and dried for 45 min in an oven at 40°C. The obtained results of the aforementioned antimicrobial analysis and test methods are the average of triplicate tests.
Result and discussion
Figure 1 shows the cotton gauze fabric samples before and after in situ deposition of nano-Ag-oxide (b) and nano-Zr-oxide (c) into cotton gauze fabrics. The dark grey color was formed with those fabric samples treated with nano-Ag-oxide as shown in Figure 1(b), whereas those fabric samples treated with nano-Zr-oxide appear white (like a blank) as shown in Figure 1(c). The amount of nano–Zr-oxide and nano-Ag-oxides determined by atomic absorption were 0.3 wt% nano-Zr-oxide and 0.5 wt% nano-Ag-oxide (based on weight of fabric).
Color of undeposited cotton gauze fabric (a), deposited silver oxide nanoparticle (b), and zirconium oxide nanoparticle (c) in situ cotton gauze fabric.
Transmission electron microscopy of nano-Zr-oxide and nano-Ag-oxide solution and the treated cotton gauze fabric
Figure 2(a) and (b) show the nano-Ag-oxide and nano-Zr-oxide suspended in the aqueous solution containing PVP as stabilizing agent. Figure 2(a) and (b) depict a very homogenous spherical shape and quite uniform particle size distribution. The particle size diameters obtained varied from 2 to 5 nm for both nano-Zr-oxide and nano-Ag-oxide. All metal oxides nanoparticles analyzed are very stable and no aggregation could be observed by TEM.
Transmission electron microscopy (TEM) of silver oxide nanoparticles solution (a) and zirconium oxide nanoparticles solution (b) prepared by reduction-oxidation method in presence of poly (vinylpyrrolidone) (PVP) as stabilizing agent.
To confirm the formation of nano Zr-oxide and nano-Ag-oxide into cotton macro and micro fibril, we carried out cross-sectional TEM analysis for the treated and untreated cotton gauze. The results are shown in the Figure 3.
Transmission electron microscopy (TEM) cross sections of untreated cotton gauze fabrics (a), nano-zirconium-oxide cotton gauze fabric (b), and nano-silver-oxide cotton gauze fabric (c).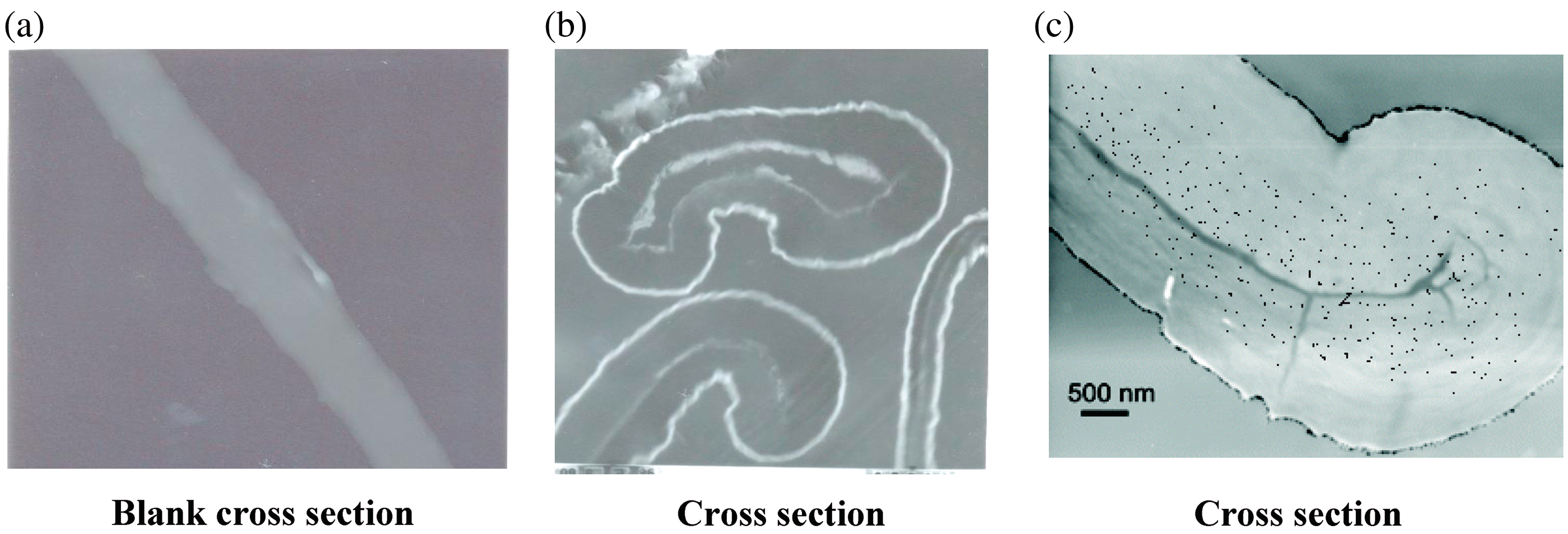
It is evident from Figure 3 that nanoparticles do not appear to form aggregates and are well distributed inside the fabric due to the stabilization of prepared nanometal oxides, within the cellulose network. Figure 3 also confirmed that the amount of nano-Ag-oxide deposited in situ cotton gauze fabric was more than the amount of nano-Zr-oxide. Figure 3(b) and (c) show nano-Zr-oxide and nano-Ag-oxide distribution in cross-sectioned cotton gauze fabric with an ultra-microtome and in the topmost layers have penetrated inside the fabric. The presence of nanometal oxides as in Figure 3(c) and the color of the loaded Ag-cotton gauze reported in Figure 1(b) suggests that nano-Ag-oxide exists inside and on top of the textile surface.
UV-visible spectroscopy
UV-visible absorption spectroscopy is one of the most widely used simple and sensitive techniques for the characterization of nanoparticles. The recorded absorption spectrum of the prepared colloidal solutions is presented in Figure 4(a) and (b).
UV-visible absorption spectra of silver oxide nanoparticles (a) and zirconium oxides nanoparticles (b) synthesized by reduction oxidation method in presence of polyvinylpyrolidon as dispersing agent.
Figure 4(a) shows a single strong peak with a maximum at 418 nm was observed, which corresponds to the typical surface plasmon resonance (SPR) of spherical silver nanoparticles [33]. In these UV-visible spectra, there were no peaks located around 335 and 560 nm, indicating the complete absence of particle aggregates [34]. Hence, silver oxide nanoparticles formed are highly stable and well dispersed.
Figure 4(b) shows UV-visible absorption spectrum recorded for nano-Zr-oxide in the wavelength range 250–700 nm. The spectrum shows sharp and prominent absorption band with maximum at 292 nm, which can arise due to the transition from valence bands to conduction band [34]. The weak absorption in the near UV and visible region is expected to arise from transitions involving extrinsic states such as surface trap states or defect states or impurities [35]. The enhancement intensity of UV emission at room temperature can be achieved by high crystal quality and quantum confinement in nanostructures. The broad emission band in the UV region is attributed to the singly ionized oxygen vacancies in nano-Zr-oxide. Hence, UV emission can arise as a result of the recombination of a photo-generated hole with an electron occupying the oxygen vacancy [34].
XRD of nano metal oxides colloidal solutions
Figure 5(a) shows XRD pattern of nano-Zr-oxide peaks appearing at 2θ: 30.02, 35.0, 50.41, and 60.0 the observed XRD peaks can be attributed to tetragonal (JCPDS file 17-0923) or cubic (JCPDS file 49-1642) zirconia. However, it is difficult to distinguish between the tetragonal and cubic zerconia phase owing to the peak broadening. The peak broadening is related to the small size of zerconia nanoparticles formed [36]. The XRD pattern of the sample shows signature for nano silver as shown in Figure 5(b). The peaks appearing at 2θ: 38.2° and 44.2° confirmed the presence of nano silver. The peak at 38.2° with 100% relative intensity peak counts which confirm the silver in nanosize. Figure 5(b) confirms silver nanoparticles formation cubic phase clearly identified from diffraction peaks at 38.2° and 44.2°crystallographic. The reflection peaks can be indicating the small size of the silver nanoparticles [33].
XRD of zirconium oxide nanoparticles (a) and silver oxide nanoparticles (b) prepared by reduction oxidation method in presence of polyvinylpyrolidon as dispersing age.
SEM-EDX for Prepared Fabric Samples
Characterization of prepared fabric samples by SEM coupled with energy dispersive X-ray was used to qualitative and quantitative confirmation of nano metal oxides in situ and on the surface of prepared fabric samples. Figure 6 shows the SEM and EDX for untreated cotton gauze and those treated with nano-Ag-oxide and nano-Zr-oxide. From Figure 6(a), (b) and (c), we can see that besides the above elements, there emerged two new peaks in Figure 6(c), one for carbon and the other for silver in the EDX pattern of the fabric, the strong peak for carbon was caused by cross-linked fabrics, while the peaks for Ag and Zr were very weak because the silver content in fabric was only 3.17% and Zr was 2.41% supposed to be completely reduced. From the above results, we can see that silver element existed on both the fabric surface and in situ the fabric, while the Zr element existed in situ only the fabric. These results indicated that Ag and Zr nanoparticles were successfully immobilized in the cotton gauze fabrics. It is well known that the surface area of particles and their surface energy increased linearly with the decrease of particles dimension. When the size of particles were reduced to nanometer scale, the surface energy of particles became very huge, and it is hard to disperse the aggregated nanoparticles or to remove the nanoparticles adhered to solid surface. In our method, this trouble was avoided because nano-Ag-oxide and nano-Zr-oxide were produced and stabilized using polyvinylpyrolidon.
Scanning Electron microscopy-EDX of untreated cotton gauze fabric (a), treated cotton gauze fabric with zirconium oxide nanoparticles (b), and cotton gauze fabric treated with silver oxide nanoparticles (c).
Antimicrobial activities
Antibacterial activities
Antibacterial activity of cotton gauze fabric with treated nano-zirconium-oxide and nano-silver-oxide

%RBC: reduction in bacterial count percent.
Antifungal activity
Antifungal activity of cotton gauze fabric treated with nano-zirconium-oxide and nano-silver-oxide

Biological activity and wound healing
Skin irritation test

Skin reaction for fabric sample treated with nano-silver-oxide

Skin reaction for fabric sample treated with nano-zirconium-oxide

Wound healing
Wounds heal by various processes such as coagulation, inflammation, matrix synthesis and deposition, angiogenesis, fibroplasias, epithelialization, contraction, and remodeling [46–50]. If the bacterial count is greater than 105 bacteria per gram of tissue, healing is impaired. The major focus of wound healing has been on the relationship between tissue destruction by a group of collagens enzymes known as metalloproteinases (MMPs) and tissue synthesis, which is stimulated by growth factors [51]. When the cotton fabric samples treated with nano-Zr-oxide and nano-Ag-oxide were applied to the wound of the tested rabbits, as shown in Figures 7–11, a significant effect on granulation tissue formation, infiltrating cells, and a minor effect on the degree of reepithelialization was seen [51,52]. Furthermore, the use of nano metal oxide is important in reducing the wound’s microbial load and alters the inflammatory events in the wound [49,53,54]. Zirconium oxide nanoparticles and silver oxide nanoparticles in particular provide added benefits by downregulating MMPs to levels that facilitate wound healing [55].
Preparation of the animals. Application of the treated textile with nano-silver-oxide at the clipping area. The patch was tied around animals to keep contact. Application of the treated textile with nano-zirconium-oxide at the clipping area. Skin of experimental animals showing no reaction at the end of the experiment.




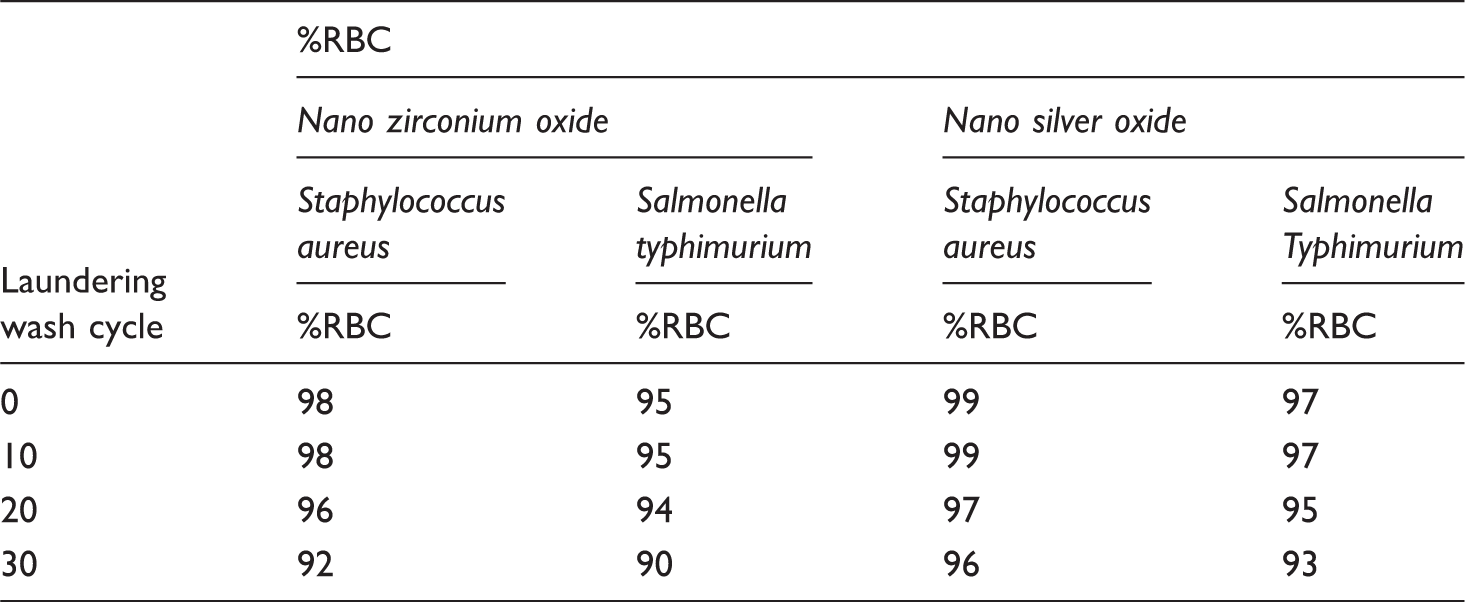
Washing durability
Antibacterial activity of cotton gauze fabric with treated nano-zirconium-oxide and nano-silver-oxide using different laundering wash cycles
Conclusion
Zirconium oxide and silver oxide nanoparticles were in situ synthesized and deposited into cotton gauze fabrics using reduction technique. The particle size diameters obtained varied from 2 to 5 nm. All metal oxides nanoparticles analyzed were very stable and not aggregate. Reduction rate of colony count percent (RBC) of cotton gauze fabrics containing nano–Ag-oxide against gram-positive bacteria (S. aureus) was 99% and gram-negative bacteria (S. typhimurium) was 97%. These values were more than those obtained with gauze fabrics containing nano-Zr-oxide which gave RBC equal to 98% and 95% against gram-positive and gram-negative bacteria, respectively. Results also revealed that both samples treated with nano-Ag-oxide and nano-Zr-oxide have more inhibitory effect against gram-positive than gram-negative bacteria. Antifungal activities of cotton gauze fabrics treated with nano-Ag-oxide gave clear inhibition zone diameter 16 mm and 14 mm against C. albicans and A. flavus, respectively. Cotton gauze fabric treated with nano-Zr-oxide inhibited C. albicans only with clear inhibition zone diameter equal to 10 mm and was not able to inhibit the growth of A. flavus. Results also revealed that, both samples treated with nano-silver and nano-Zr-oxide have more inhibitory effect against C. albicans than A. flavus. All animals applied with cotton gauze containing nano-Ag-oxide and nano-Zr-oxide showed no clinical signs of skin irritation during the time of experiment. The prepared cotton gauze fabrics were found to be non-irritant to the skin of tested rabbits and the wound healed after 4 days from application.
Footnotes
Notes
Published over 17 papers in well known international journal and conference in the field of medical textiles. Supervised 2 PhD and 2 M.Sc. thesis's in the field of technological innovations based on frontier sciences for development of textile printing, micro/nano-encapsulation to enhance the performance properties of cellulose containing fabrics as well as dyeing and enhancing multifunctional properties of Textile Fabric Using Nanotechnology'.
