Abstract
Introduction
Cardiovascular disease (CVD) is a major cause of morbidity and death among kidney transplant recipients and contributes significantly to poor graft outcomes.1–4 The rates of cardiovascular events and cardiovascular mortality in kidney transplant recipients are 50 times and 10 times higher than in the general population, respectively. 5 Moreover, approximately half of recipients die with a functioning graft 6 and about 30% of these deaths are attributable to CVD. 7
The high risk of CVD is related to several traditional and non-traditional factors, including diabetes, hypertension, dyslipidemia, anemia, obesity, smoking, homocysteinemia, hyperparathyroidism and proteinuria.5,8,9 Proteinuria is frequently observed after kidney transplantation and is prevalent among 11 to 45% of recipients with a functioning graft.1,10,11 Proteinuria is a known indicator of glomerular damage and is reported to be an independent risk factor of further disease progression. It is believed that excess proteins filtered by the glomerulus causes an interstitial inflammatory response that leads to lesions and scarring of the tubular interstitium. 12
Previous studies in the general population have shown that proteinuria is an independent predictor of CVD.13–16 It also results in a greater risk of poor graft outcomes and mortality in kidney transplant recipients.1,15–20 Specifically, recipients with a urine protein/creatinine ratio greater than 1.0 during the third month posttransplant were approximately twice as likely to have a cardiovascular event, sixteen times more likely to have graft failure, and three times more likely to die with a functioning graft compared to those without significant proteinuria.3,21
The relationship between proteinuria and CVD in kidney transplant recipients is clinically relevant due to the increased CVD incidence in this population and the potential to use proteinuria as a clinical marker and modifiable risk factor for CVD. This study aimed to ascertain the association of posttransplant proteinuria with the risk of 1) cardiovascular events and 2) graft failure in a cohort of kidney transplant recipients in a time-dependent analysis. For each outcome, an additional objective was to determine how this relationship was modified by other risk factors and assessed whether kidney function acted as a time-varying confounder affected by prior exposure.
Methods
Design
This was a longitudinal observational cohort study using existing health records. Approval for this study was obtained from the University Health Network Research Ethics Board.
Setting
The study was conducted at a large metropolitan center that follows solid organ transplant recipients. Kidney transplant recipients are followed by transplant nephrologists in the transplant clinic.
Population
All adults (age > 18 years) who received kidney transplants from 1 January 2000 to 31 December 2011 were initially included. Patients were followed from one-month post-kidney transplant to the date of the first major cardiac event, graft failure, death, loss to follow-up, or 31 December 2012. Those younger than 18 years, multi-organ transplant recipients, re-transplant and allografts with primary non-function were excluded from the study.
Data collection
Data for this study were retrieved from the Comprehensive Renal Transplant Research Information System (CoReTRIS) database. This in-house research database includes an extensive set of recipient, donor, transplant, lab and follow-up data for all kidney recipients since Jan 1, 2000. 22 Patients sign a consent form to have their information in this repository for any study deemed appropriate.
The main exposure variable in our study was total protein measured on a random spot urine sample. The dipstick test was used by transplant nephrologists to monitor urinary protein excretion on a routine outpatient basis during regularly scheduled blood work visits (ie, 2 times per week for 4 weeks, weekly for 8 weeks, every 2 weeks for 12 weeks, monthly for 6 months, and every 2 to 3 months thereafter).
Urinary protein concentration was categorized into the following 5 levels by the dipstick test: negative (< 0.15 g/d), trace (0.15 to 0.30 g/d), 1 + (0.30 to 1.00 g/d), 2 + (1.00 to 3.00 g/d), and 3 + (≥ 3.00 g/d). We defined urinary protein as either negative (< 0.15 g/d) or positive (> 0.15 g/d) for our primary analysis. The dipstick test results were dependent on urine concentration, therefore evaluating outcomes as a function of any proteinuria compared to no proteinuria was more informative.
We assessed random dipstick urine protein measurements at months 1, 3, 6, 9 and 12 in the first-year posttransplant, every 6 months in the second and third-year posttransplant, and yearly thereafter. Timed urine collections for protein measurement and protein adjusted for creatinine in a spot urine sample were not routinely available for analysis.
Our primary endpoint of interest was a major adverse cardiac event occurring at least one-month posttransplant. Major adverse cardiac events were defined as the composite of acute myocardial infarction, cerebrovascular accident, revascularization by coronary artery bypass graft, percutaneous coronary intervention or coronary angioplasty, or all-cause mortality. Data on major adverse cardiac events were obtained through a comprehensive review of electronic patient charts and direct reports from patients during clinic visits. Total graft loss, which composed of graft failure or death with function, was used as a secondary endpoint.
Data Analysis
Baseline recipient, donor and transplant characteristics were evaluated using summary statistics. Extended Kaplan-Meier survival curves and the log-rank test 23 were used to assess primary and secondary study outcomes in relation to the presence or absence of proteinuria (ie, negative vs positive dipstick results for urinary protein) and increasing urinary protein concentration (ie, negative, trace, 1 + , 2 + , and 3 + ).
Stata/MP Version 11.0 (StataCorp, College Station, TX) was used to perform all statistical analyses and results were considered statistically significant if a two-tailed P-value of <0.05 was achieved. The association between any proteinuria and major adverse cardiac events was explored using multivariable time-dependent Cox proportional hazards models. A similar analysis was also conducted using the secondary end point of total graft loss (including death with function and allograft failure). Additionally, the relative hazard for each outcome by proteinuria level (ie, trace, 1 + , 2 + , 3 + ) was examined.
The study origin was chosen to be one-month posttransplant (model 1) because urinary protein originating from the native kidneys usually resolves within the first month posttransplant. 24 Additional analyses using 6 months posttransplant (model 2) and 12 months posttransplant (model 3) as times of origin were conducted to increase the likelihood that any observed proteinuria originated from the kidney transplant rather than the native kidneys.
Clinically relevant covariates were determined a priori and adjusted for in all models, including (a) recipient factors (ie, age, sex, estimated glomerular filtration rate calculated using the chronic kidney epidemiology collaboration formula, mean arterial pressure, time on dialysis before transplant, smoking status, history of cardiovascular disease, and history of diabetes mellitus); (b) donor factors (ie, sex and deceased vs living donor); and (c) transplant factors (ie, angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker treatment and transplant era categorized into tertiles of transplant years). Multiple imputation using the imputation by chained equations command in Stata/MP 11.0 was used for missing covariate data.
Results
A total of 1870 patients were identified in the CoReTRIS database as having undergone kidney transplantation during the study period. The final study cohort consisted of 1106 patients after applying our exclusion criteria (Figure 1). Baseline recipient, donor, and transplant characteristics (one-month posttransplant) are summarized in Table 1. The mean age of the study cohort at time of transplant was 49.8 (36.5, 63.1) years, with 61.9% male and 66.6% white. The level of missingness for each characteristic is described in Supplemental Table 1, with the percentage of missing data ranging from 0% to 8.95% across the characteristics. Patients were followed for a cumulative total of 5728.65 person years at risk. During this time, 144 primary outcome events were recorded out of which 57 patients that had a myocardial infarction, cerebrovascular accident or revascularization procedure, and 87 patients died. The median follow-up time was 4.41 years after the first month posttransplant. The proportion of patients with proteinuria at the study time points is displayed in Figure 2. Additional analyses were performed using 6 months and 12 months posttransplant as study origins, during which 131 events were observed in 1083 patients and 118 events in 1059 patients, respectively.
Study population flow diagram.
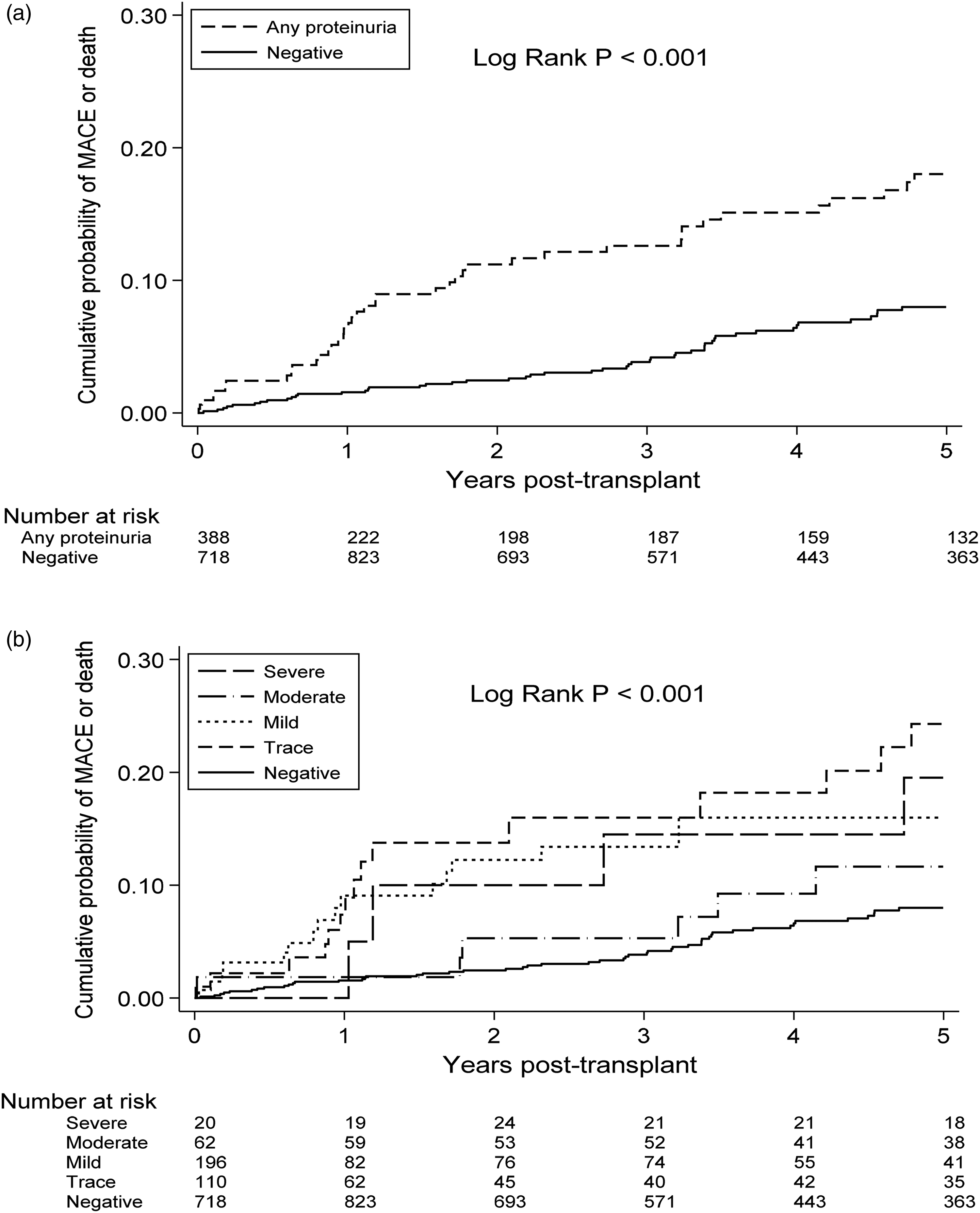
Kaplan-Meier curves for Major adverse cardiac events or death, by urine protein, (a). Risk of major cardiovascular event by present of any proteinuria. (b). Risk of major cardiovascular event by severity of proteinuria.
Recipient, Donor, and Transplant Characteristics.

The cumulative incidence of major adverse cardiac events at 5 years posttransplant was 18.0% in patients with any proteinuria compared to 8.0% in patients with no proteinuria (log rank P < 0.001) on Kaplan-Meier analyses (Figure 2a). When urinary protein was separated into negative, trace, 1 + , 2 + and 3 + proteinuria levels, the corresponding incidence of major adverse cardiac events at 5 years posttransplant was 8.0%, 24.3%, 16.0%, 11.6% and 19.5% (log rank P < 0.001) (Figure 2b).
Effect of proteinuria on major adverse cardiac events
The association between proteinuria and major adverse cardiac events or death was explored using 3 time-dependent Cox proportional hazards models with varying study times of origin. The results of these models are summarized in Table 2. Any urinary protein detected on a dipstick test (ie, trace, 1 + , 2 + or 3 + ) from one-month posttransplant resulted in a 2.00 times higher hazard of major adverse cardiac events or death (P < 0.001) compared to no proteinuria (Table 2a). When 6 months posttransplant and 12 months posttransplant were used as study origins, any urinary protein resulted in a 1.89 (P < 0.001) and 1.74 (P < 0.01) times increased hazard of major adverse cardiac events or death compared to no proteinuria, respectively (Table 2a). The effect of any urinary protein was only found to be significant with time-fixed variables when the study origins were 6 months posttransplant or 12 months posttransplant (Table 2b).
Cox Models for Effect of Urine Protein on major Adverse Cardiac Events or Death.
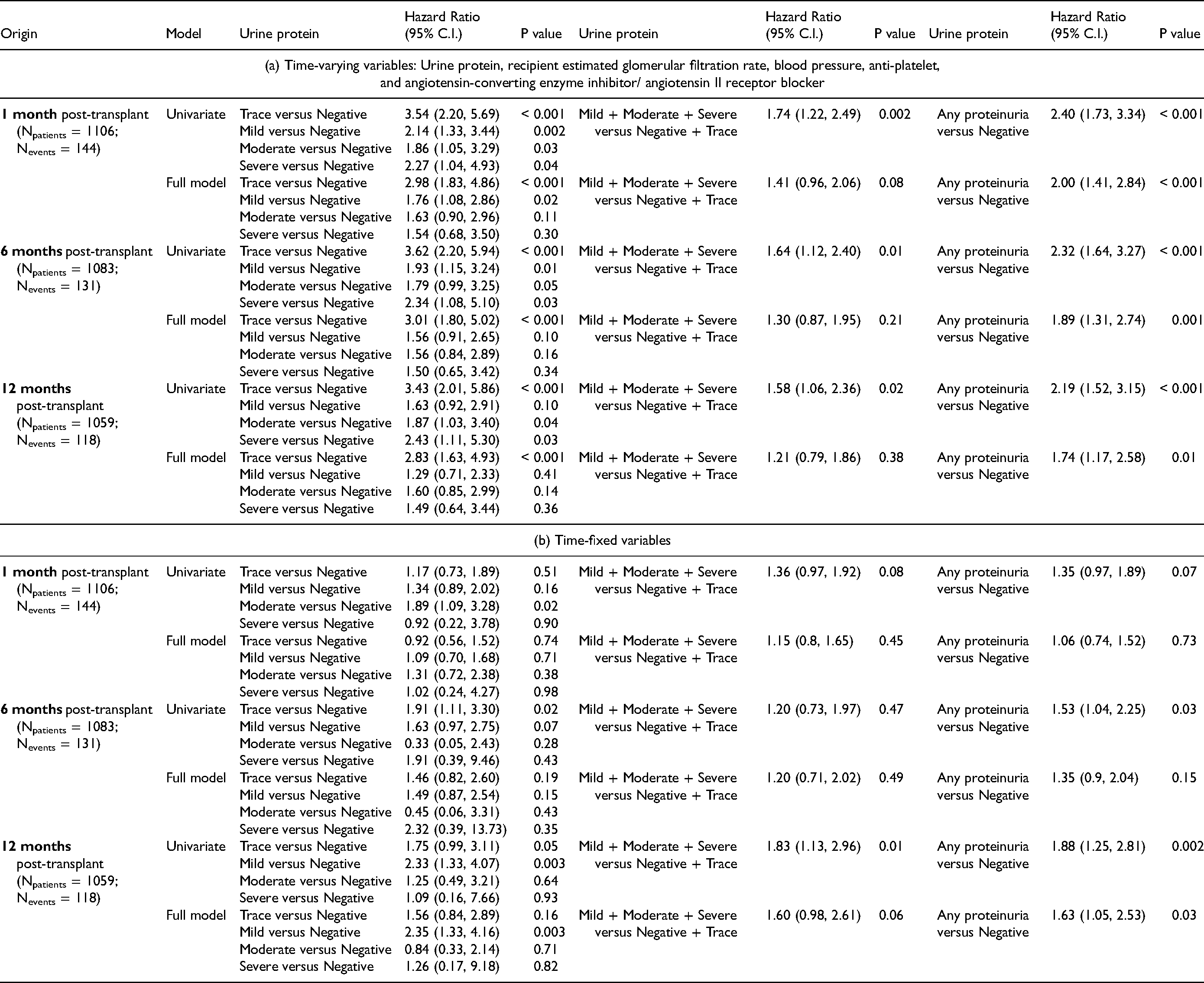
Only trace and 1 + proteinuria were associated with a significant hazard of major adverse cardiac events or death, when trace, 1 + , 2 + and 3 + proteinuria levels were evaluated independently (Table 2a). The hazard ratio of major adverse cardiac events or death was found to be 2.98 (95% confidence interval [95% CI] 1.83, 4.86), 1.76 (95% CI 1.08, 2.86), 1.63 (95% CI 0.90, 2.96) and 1.54 (95% CI 0.68, 3.50) for trace, 1 + , 2 + and 3 + proteinuria, respectively. Similarly, only trace proteinuria was associated with a significant hazard of major adverse cardiac events or death when the study origin was defined as 6 months or 12 months posttransplant. The association between urinary protein and major adverse cardiac events alone was not found to be significant (Supplemental Tables 2a, 2b).
Effect of proteinuria on graft failure
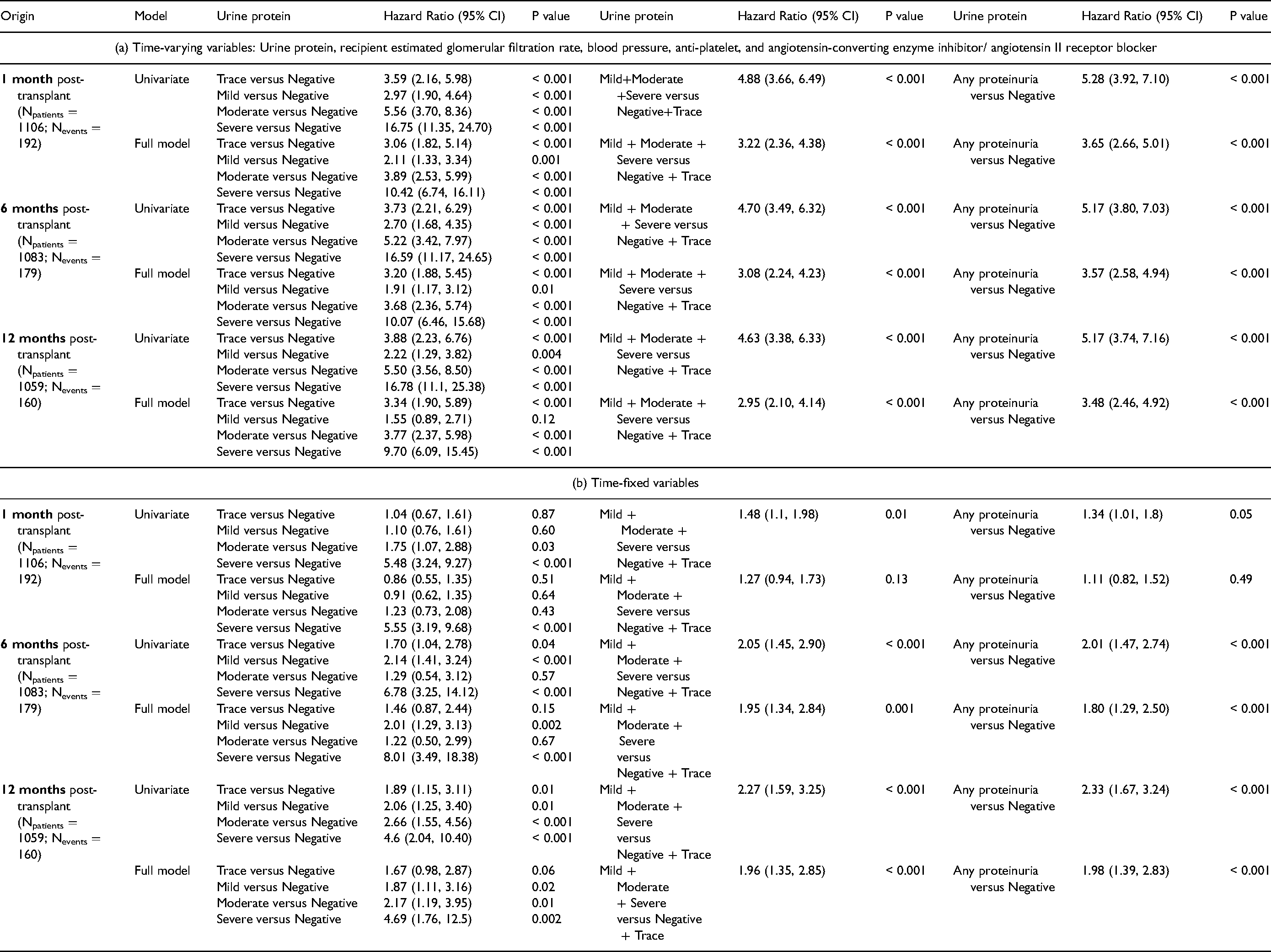
The relationship between proteinuria and graft loss was also examined using adjusted time-dependent Cox proportional hazards models, the results of which are summarized in Table 3. Any proteinuria detected by dipstick from one-month posttransplant resulted in 3.65 times increase in total graft loss (P < 0.001), where total graft loss was defined as either graft failure or death (Table 3a). Any proteinuria from one-month posttransplant was not associated with a significant hazard of total graft loss using time-fixed variables (Table 3b). Proteinuria was associated with a hazard ratio of 6.04 for death-censored graft failure (P < 0.001) (Supplemental 3a) and hazard ratio of 2.30 for death with graft function (P < 0.001) (Supplemental Table 3b) when evaluated individually. Notably, the hazard of graft failure significantly and monotonically amplified with an increased level of urine protein concentration as measured by urine dipstick. The effect of proteinuria on death-censored graft failure and death with graft function was not significant when using time-fixed variables (Supplemental Table 3c, 3d). The hazard ratio of death-censored graft failure was 2.22 (95% CI 0.73, 6.75), 2.85 (95% CI 1.29, 6.26), 6.41 (95% CI 3.23, 12.74), and 19.71 (95% CI 10.12, 38.39) for trace, 1 + , 2 + and 3 + urinary protein compared to negative, respectively (Supplemental Table 3a).
Cox Models for Effect of Urine Protein on Total Graft Failure.
Discussion
Cardiovascular disease is the leading cause of death in kidney transplant recipients and the primary cause of death with a functioning graft. 4 Traditional risk factors, such as diabetes, hypertension, dyslipidemia, obesity and smoking, and non-traditional risk factors, such as proteinuria, act in concert to increase the risk of CVD in this patient population.5,8,9 Older data has shown that proteinuria was associated with a relative risk of 2.45 for CVD, 4.18 for graft loss and 1.92 for death. 25 In our study, we similarly observed that any proteinuria measured by dipstick was associated with an increased hazard for CVD (HR = 2.00), total graft loss (HR = 3.65), graft failure (HR = 6.04) and death (HR = 2.30). We found that any level of proteinuria was independently associated with an increased risk of major adverse cardiac events, total graft loss, graft failure or death. Results were consistent whether 1 month, 6 months or 12 months posttransplant were used as study origins.
Evidence also suggests that proteinuria monotonically increases the risk of CVD, graft loss and death.4,14,20,26 A meta-analysis of general population cohorts by the Chronic Kidney Disease Prognosis Consortium found that albumin/creatinine ratios of 1.1 mg/mmol, 3.4 mg/mmol and 33.9 mg/mmol were associated with hazard ratios of 1.20 (95% CI 1.15-1.26), 1.63 (95% CI 1.50-1.77) and 2.22 (95% CI 1.97-2.51), respectively, for all-cause mortality compared to an albumin/creatinine ratio of 0.6 mg/mmol. Similar results were seen for cardiovascular death in the meta-analysis by Chronic Kidney Disease Prognosis Consortium. 14
We observed this dose-response between proteinuria and graft failure in our study sample. We did not find that proteinuria increased the risk of major adverse cardiac events in a dose-dependent manner. When proteinuria was categorized into trace, 1 + , 2 + , or 3 + urine protein levels, only trace and 1 + dipstick results were significantly associated with major adverse cardiac events after adjustment for all covariates. We believe that this unexpected result could have been due to the limited number of major adverse cardiac events in our study sample; only 144 major cardiac events or deaths were observed in 1106 patients over 5728.7 person-years. The dose-dependent relationship between proteinuria and graft failure may also have played a role by decreasing the observation time for major adverse cardiac events in patients with greater urinary protein.
In the present study, time-varying proteinuria was investigated by adjusting for drugs that inhibit the renin-angiotensin-aldosterone system (RAAS), which include angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers. Use of RAAS inhibitors were associated with a reduction in proteinuria in the kidney transplant and non-transplant population.4,5,20 The use of RAAS inhibitors was also associated with decreased risk of CVD and prolonged graft survival. 4 Thus, it was important to adjust for angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers in the multivariate model to account for the confounding factor present in real life scenarios.
There were some notable limitations to our study. We relied on the urine dipstick test for urinary protein quantification. The urine dipstick test is dependent on urine concentration, and thus, not ideal for quantifying urine protein. Other preferred methods directly measure the total urine protein excreted over a period of time (ie, 24-h urine collection) or adjust for urine concentration (ie, albumin/creatinine ratio) since they do not depend on urine volume. We addressed this limitation of dipstick testing by analyzing proteinuria as a binary variable (ie, any vs no proteinuria). We also validated dipstick measurements by comparing dipstick results to available albumin/creatinine ratio or 24-h urine protein data and found the results of these tests to be well correlated.
Multiparametric conventional dipsticks also have been shown to have a sensitivity of 100% and specificity of 58% (area under the receiver operating characteristic curve = 91%) for protein levels over 150 mg/L when a trace cutoff is utilized. 26 The high sensitivity and low specificity of the dipstick test when a trace cutoff is selected for proteinuria means that our results likely underestimate the true effect of proteinuria on CVD, total graft loss, graft failure, and death.
Dipstick tests can be used as a tool to detect the presence of proteinuria at early stages posttransplant. Dipstick test results were readily available at the necessary time-points for our analysis, while 24-h urine collection and albumin/creatinine ratios were less accessible. The dipstick test is used by transplant nephrologists at our center to monitor urinary protein on a routine outpatient basis during regularly scheduled blood work visits. Therefore, the dipstick test was chosen as a measure of urinary protein excretion for our primary analysis. The detection of proteinuria could have clinical utility in using it as an indicator for being at risk for major adverse cardiac events and negative graft outcomes. Since the dipstick test has limited specificity and sensitivity, more accurate and rigorous screening tests, including spot urine tests, can be used to differentiate risk above trace and the 1 + threshold.
Another limitation of this study was that it was confined to a single-center population, which decreases its generalizability. It was also an observational study and confounding cannot be ruled out, despite the multi-variable analyses.
Conclusion
Proteinuria can be readily identified with dipstick tests during routine follow-up visits due to the low cost and accessibility. Preliminary tests such as the dipstick test could have clinical utility as a clinical marker in detecting early-onset proteinuria to recognize the risk for major adverse cardiac events, graft loss, and mortality in kidney transplant recipients. Furthermore, the role of interventions to reduce proteinuria to mitigate the risk of adverse cardiovascular and graft outcomes in kidney transplant recipients requires further study.
Supplemental Material
sj-docx-1-pit-10.1177_15269248211046011 - Supplemental material for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients
Supplemental material, sj-docx-1-pit-10.1177_15269248211046011 for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients by Tanya Kuper, Olusegun Famure, Jamie Greenfield, Yanhong Li, Syed Ibrahim, Tanya Narang, Monika Ashwin and S. Joseph Kim in Progress in Transplantation
Supplemental Material
sj-docx-2-pit-10.1177_15269248211046011 - Supplemental material for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients
Supplemental material, sj-docx-2-pit-10.1177_15269248211046011 for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients by Tanya Kuper, Olusegun Famure, Jamie Greenfield, Yanhong Li, Syed Ibrahim, Tanya Narang, Monika Ashwin and S. Joseph Kim in Progress in Transplantation
Supplemental Material
sj-docx-3-pit-10.1177_15269248211046011 - Supplemental material for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients
Supplemental material, sj-docx-3-pit-10.1177_15269248211046011 for Time-Varying Proteinuria and the Risk of Cardiovascular Disease and Graft Failure in Kidney Transplant Recipients by Tanya Kuper, Olusegun Famure, Jamie Greenfield, Yanhong Li, Syed Ibrahim, Tanya Narang, Monika Ashwin and S. Joseph Kim in Progress in Transplantation
Footnotes
Acknowledgments
Many thanks to the Multi-Organ Transplant Student Research Training Program students for updating and auditing the Comprehensive Renal Transplant Research Information System (CoReTRIS) and Elizabeth Murakami for her administrative assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors of this paper received funds from the ASN Student Scholar Grant to carry out this study. Dr Kim was a member of advisory boards for Astellas Pharma, Genzyme, Novartis Pharma and Pfizer, and has received research grants from these organizations. He also previously worked as a consultant for Astellas Pharma.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
