Abstract
Objective:
Physician-modified endografts (PMEGs) are widely accepted as a viable option in the urgent management of patients with complex abdominal aortic aneurysms (CAAAs). While PMEGs have emerged as the predominant repair modality for CAAAs in the United States, their acceptance in Europe remains limited—particularly in the context of elective repair. The aim of this study was to present the initial experience with elective cases at pioneering European centers offering this treatment.
Methods:
This is a retrospective observational study based on an international multicenter registry (REgistry of MOdified Treo endografts in Europe—REMOTE). Data were collected from 4 European centers on patients who underwent elective PMEG repair using the Treo device for CAAAs between August 2023 and February 2025. Demographic characteristics, clinical status, procedural details and follow-up data were recorded. Primary outcomes included technical success, overall and aortic-related mortality, and adverse events.
Results:
Twenty-eight patients (20 males; mean age 76.3±6.8 years) underwent elective PMEG repair with the Treo device. Most procedures incorporated at least 3 target vessels (68.3%). The average time required for device modification was 47.4±20.3 minutes. Technical success was achieved in 96.4% of cases. The single technical failure was associated with a lost accessory renal artery, with no acute kidney injury occurred nor endoleak observed. There were no in-hospital deaths or major adverse events. At short-term follow-up (mean 5.9±4.8 months), overall mortality was 3.6% (n=1, non–aortic-related). Freedom from reintervention was 92.9% (n=26). Aneurysm sac shrinkage was observed in 25% of patients (n=7), with the remainder showing stability. Primary and secondary target vessel patency rates were both 98.8% (n=81), with one renal stent occlusion noted on follow-up imaging.
Conclusion:
This multicenter retrospective observational study demonstrated that elective PMEG repair of CAAAs using the Treo device is associated with a high-technical-success rate and a low incidence of clinical complications, supporting the safety and feasibility of this approach in the elective setting.
Clinical Impact
REMOTE provides a multicentre European snapshot of elective PMEG-FEVAR on a single abdominal platform (Treo), showing high technical success with major clinical complications being uncommon, supporting feasibility in the elective setting. This could change practice by enabling experienced centres to offer a structured elective PMEG pathway and reduce decision-to-operation time from the 1–4 months often seen with CMD logistics to days (or even hours), thereby lowering interval-rupture risk and patient anxiety. For clinicians, this means a more predictable workflow that can shorten the learning curve and broaden access where CMD availability/cost is limiting.
Introduction
Physician-modified endografts (PMEGs) are increasingly employed as an alternative to custom-made devices (CMDs) to treat complex abdominal aortic aneurysms (CAAAs).1,2 Initially reserved for symptomatic and ruptured cases, the growing experience with PMEGs has facilitated their adoption in elective settings as well. 3 In a recent study of the Vascular Quality Initiative, PMEGs were identified as the predominant endovascular repair modality of CAAAs outside of investigational device exemptions, surpassing CMDs. 4 Although this was not a choice but a result of the limited availability of CMDs for American vascular surgeons, it helped us gain insight into the safety and durability of such interventions. The situation in Europe is very different, where the access to CMDs is more widespread and much less restricted, hence the acceptance and experience with PMEGs remains limited. Current guideline of the European Society for Vascular Surgery does not recommend PMEG as a standard technique for the elective treatment of CAAAs. 5 This is unlikely to change in the near future, as a recent debate on whether the guideline requires interim revision concluded that further data are needed to justify broader adoption, stating that the existing evidence largely stems from a limited number of single-center studies conducted at specialized academic aortic centers.6,7
While preparations are made to initiate a European collaboration regarding a prospective study on PMEGs to trigger changes in the guidelines, a snapshot of the current status might be of interest. Thus, the aim of this multicenter registry report is to demonstrate the initial European experience from pioneering centers offering PMEGs as an elective treatment option.
Materials and Methods
The REgistry of MOdified Treo Endografts in Europe (REMOTE) was an international, multicenter, retrospective, observational study that included all consecutive patients who underwent endovascular repair of a CAAA using a fenestrated PMEG based on the Treo device (Terumo Aortic) designed to preserve the visceral arteries. Four European centers participated, enrolling patients treated from August 2023 to February 2025. The study was conducted in compliance with the Declaration of Helsinki and was approved by the local ethics committee of the coordinating center (129/2024). In the participating centers, the study was either approved by the respective institutional review boards or granted a waiver in accordance with local regulations. This study followed the reporting guidelines from the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement for cohort studies. 8 Thorough discussions were completed with all potential patients with regards of the differences of the PMEG technique compared with CMDs, including but not restricted to the risk associated with the interval rupture until the operation, the costs of the repair, and the additional risk they might have with the PMEG technique: lack of long-term data, standardization and quality control issues. All patients who gave informed consent to undergo PMEG FEVAR (fenestrated endovascular aneurysm repair) over CMD FEVAR were included in the analyses.
Inclusion criteria were any elective endovascular repair of a juxtarenal or pararenal CAAA using the PMEG FEVAR technique based on the Treo bifurcated main body or cuff (Terumo Aortic, Inchinnan, UK). At least, one visceral or renal artery (celiac trunk, superior mesenteric artery, or renal arteries) had to be preserved using a fenestration. Bridging stents from any manufacturer were permitted. Graft infections and infective native aortic aneurysms were excluded.
Definitions of outcomes and variables were used in accordance with the reporting standards of the Society for Vascular Surgery. 9 Retrospective data collection was performed using an electronic data collection sheet. Gathered data included demographic and anamnestic data, maximal aneurysm diameter, anatomical classification of aneurysm (juxtarenal or pararenal), suspicion of infected native aortic aneurysm, number and diameter of target arteries, procedural details, including femoral access strategy, spinal drainage, proximal landing zone, neck length, upper extremity access, fusion imaging, endograft diameter and length, fenestration positioning method, diameter reduction ties, fenestration technique, fenestration diameters, reinforcement (no reinforcement, snare loop, guidewire tip, and nitinol ring), suture for reinforcement, strut modification, type and parameters of bridging stents, flaring, technical success, total operating time, time of modification, total contrast volume, fluoro time, dose-area product, and air kerma.
Main outcomes were technical success, in-hospital mortality, and adverse events. Technical success was defined as successful arterial access and deployment of all devices, successful catheterization and stenting of all planned target vessels without occlusion on final angiogram, and freedom from type I or III endoleak on first computed tomography angiography (CTA) within 30 days.
The technical details of the PMEG procedure were left to the discretion of the operator. While heterogeneity was observed among the participating centers in the steps of modification, each center applied its own methods rather uniformly across the treated patients. The punch card technique was used in one center routinely for translating the fenestration plan to the actual device as already published. 10 In brief, a proportionally scaled fenestration diagram was created and printed on a printable transparency using a standard office printer prior to the operation. A hole was created using a paper punch at the middle of each desired fenestration, after which the punch card was sterilized. At the beginning of the back-table modification, the aortic component was partially deployed in a tube that was formed from the punch card. With the gentle rotation of the semi-deployed device within the transparent tube, a configuration was searched where all desired fenestrations aligned with a strutless area of the aortic component. The position of the fenestrations was marked with a sterile pen through the holes of the punch card, and the preparation was continued in a standard fashion.
Closed-ring reinforcement rings, Hungarorings were also used routinely by one center. 11 These rings are similar to the reinforcement rings of CMDs in regards of tensile strength, maintaining the diameter of the fenestration during the flaring maneuver. 12 This might add to the durability of the repair.
In-hospital adverse events were defined as all-cause mortality, myocardial infarction, respiratory failure (prolonged [>24 hours from anticipated] mechanical ventilation or reintubation), acute kidney injury (>50% increase in baseline creatinine or new-onset dialysis), bowel ischemia (requiring surgical resection or not resolved with medical therapy), stroke, and paraplegia. Overall survival, freedom from aortic-related mortality, freedom from branch-related occlusion or reintervention, freedom from branch-related endoleak, as well as primary and secondary target vessel patency were also assessed. Significant sac growth or shrinkage was defined as a >5 mm change in the maximal diameter of the aneurysm sac on follow-up imaging compared with the preoperative value.
Continuous variables are expressed as mean ± SD or median (interquartile range) according to the distribution, while categorical variables are expressed as numbers (percentages). Time-to-event variables were analyzed using Kaplan-Meier curves. Data analysis was performed using STATA (version 18.0; StataCorp LLC, College Station, TX, USA).
Results
Twenty-eight patients who underwent PMEG FEVAR with the Treo device were included. Patient characteristics are shown in Table 1.
Patient Characteristics.
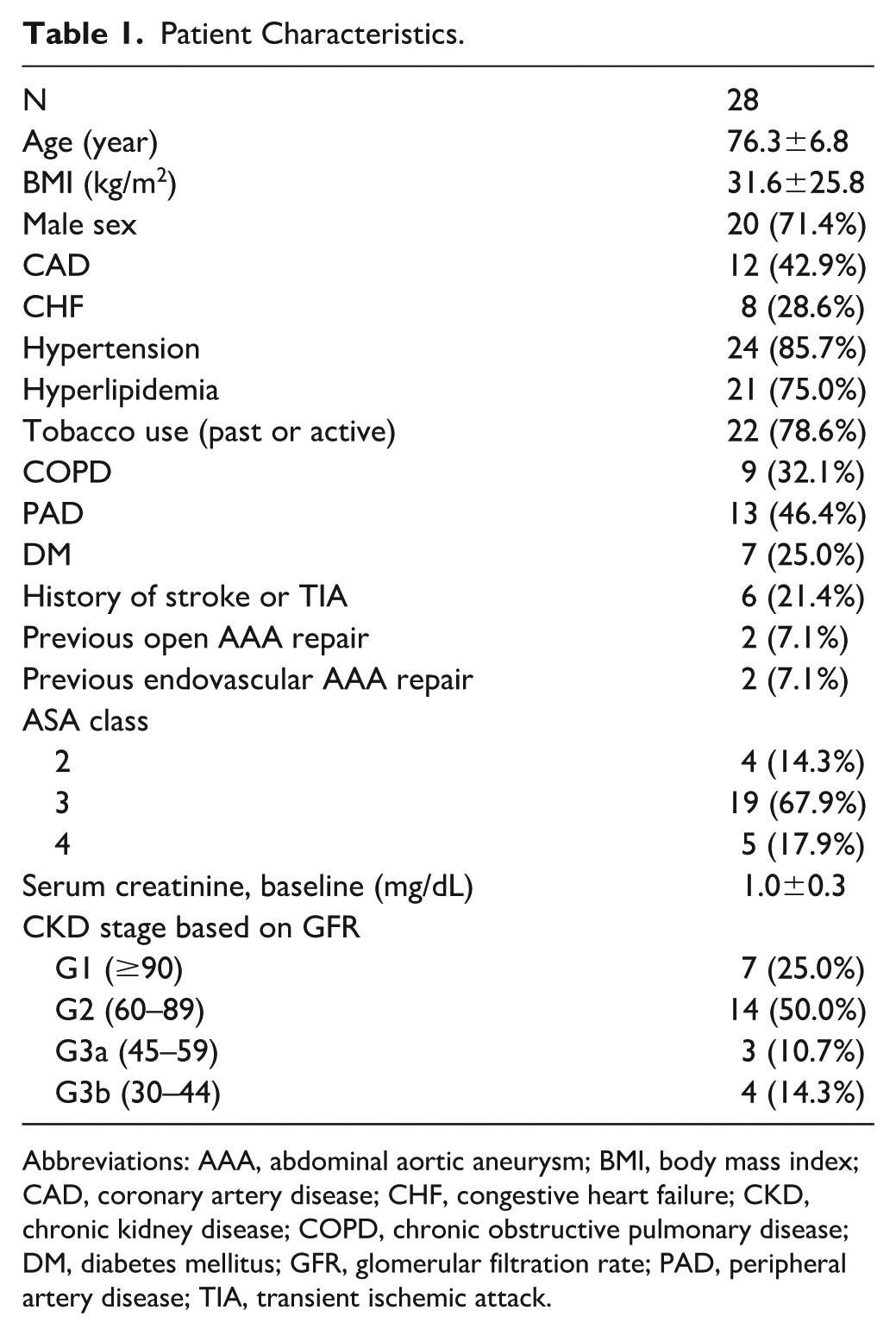
Abbreviations: AAA, abdominal aortic aneurysm; BMI, body mass index; CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; GFR, glomerular filtration rate; PAD, peripheral artery disease; TIA, transient ischemic attack.
Anatomical characteristics of our patient cohort are presented in Table 2. Three celiac artery occlusions (10.7%) were found along with a right renal artery occlusion (3.6%). Diameter or the visceral arteries were as follows: 7.0±1.0 mm for the celiac artery, 7.3±1.2 mm for the superior mesenteric artery, 5.3±0.8 mm for the right renal artery, 5.6±0.9 mm for the left renal artery, and 3.8±0.4 mm for the aberrant fifth branch. A total of 83 target arteries were incorporated to the repair, of which 82 (98.8%) were successfully preserved during the procedures.
Anatomical Characteristics.
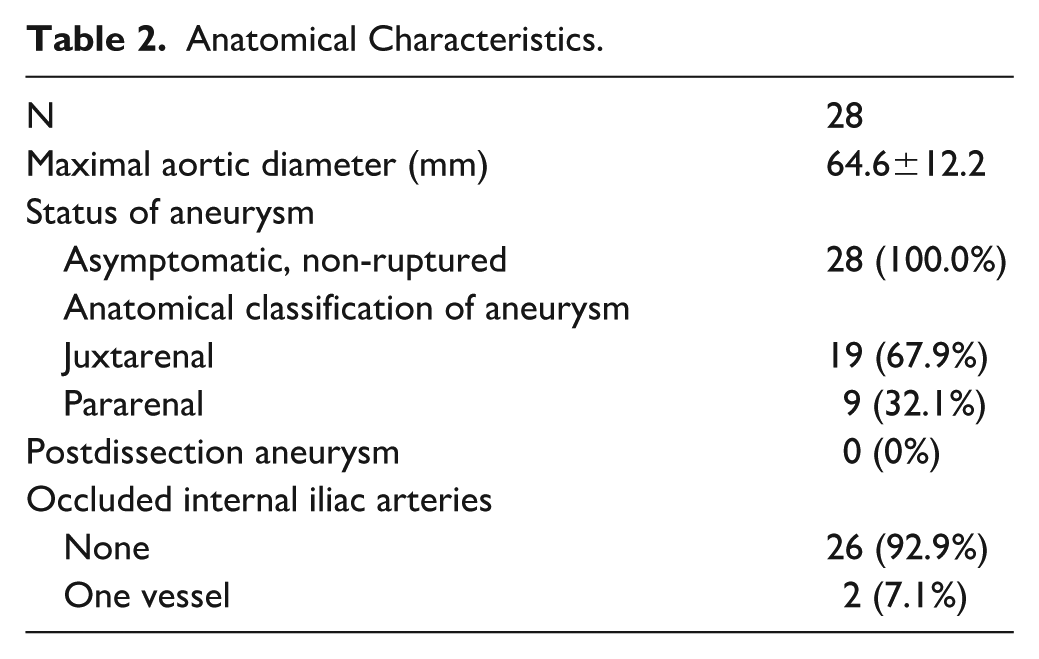
Procedural details are summarized in Table 3. Bilateral percutaneous femoral access was used in 26 cases (92.9%), whereas a unilateral cutdown was performed in 2 cases (7.1%). Main body was inserted from the left side in 15 cases (53.6%). Upper extremity access was used in 4 cases (14.3%), whereas fusion imaging with CTA in 16 (57.1%). Prophylactic or therapeutic spinal drainage was not performed. Preloaded catheters or guidewires were not utilized. Bifurcations (n=21, 75%) and cuffs (n=7, 25%) were both used. Fenestration positions on the device were identified using arc length measurements in 11 cases (39.3%), whereas the others were managed using a punch card (n=17, 60.7%). Fenestrations were created either with a scalpel (n=9, 32.1%) or with a cautery (n=19, 67.9%). Distance from proximal edge was 24.1±6.0 mm for the celiac artery, 35.4±14.1 mm for the superior mesenteric artery, 38.7±22.5 mm for the right renal artery, 41.3±22.3 mm for the left renal artery, and 45.0±31.1 for the fifth branch. Diameter of the fenestrations were 8.4±0.6 mm for the celiac artery, 8.4±1.0 mm for the superior mesenteric artery, 6.8±0.8 mm for the right renal artery, 6.9±0.9 mm for the left renal artery, and 5.0±0.0 mm for the fifth branch. All fenestrations were reinforced, either with a snare loop (n=12, 42.9%) or with a Hungaroring (n=16, 57.1%). Strut modification was avoided, scallops were not used. Diameter reducing ties were used in 16 (57.1%) cases. A total of 82 balloon expandable covered stents were used as follows: iCover (n=33, 30.2%), BeGraft Peripheral (n=31, 37.8%), Advanta V12 (n=7, 8.5%), BeGraft Plus (n=5, 6.1%), VBX (n=4, 4.9%), or BeFlared (n=2, 2.4%).
Procedural Details.

Abbreviation: DAP, dose-area product.
Technical success was achieved in 27 cases (96.4%). One technical failure occurred (n=1, 3.6%) due to the inability to catheterize an accessory renal artery measuring <4 mm in diameter. In that case, 4 other target arteries—including both main renal arteries—were successfully cannulated. The unstented fenestration did not result in a type III endoleak on pre-discharge CTA, as it was oriented toward the aortic wall at the neck of a juxtarenal aneurysm. No significant increase in serum creatinine was observed during the postoperative course, and the aneurysm sac remained stable on the most recent follow-up imaging. There were no complications recorded during preparation (0%). Endoleak was present at completion angiography in 3 cases (10.7%), 2 of which was type II, the other being a type Ia, but it was left untreated and was closed on 30-day follow-up CTA. No in-hospital death, myocardial infarction, respiratory failure, stroke or spinal cord injury was recorded. Intensive care unit (ICU) stay was not needed in 26 cases (92.9%), whereas 2 patients underwent a single day of ICU observation (7.1%). A postoperative acute kidney injury (>50% increase in serum creatinine) was observed in 1 case (n=1, 3.6%) with no clinical sequela. There were 4 access complications (n=4, 14.3%): 1 in the upper extremity (brachial artery bleeding needing suture) and 3 in the common femoral artery (an occlusion and a bleeding that required open conversion at the end of the index operation and 1 symptomatic stenosis treated with transbrachial balloon angioplasty on the third postoperative day). After a hospital length-of-stay of 3 (3–5) days, patients were discharged to home in 26 cases (92.9%) or to a rehabilitation hospital in 2 cases (7.1%).
At a short-term follow-up of 5.9±4.8 months, overall mortality rate was 3.6% (n=1). Aortic-related mortality was 0%, the single mortality recorded was associated with a hemorrhagic stroke. Freedom from reintervention was 92.9% (n=26). Two reinterventions were performed, 1 renal bridging stent relining with an additional covered stent due to a type III endoleak, and 1 aortoiliac kissing stenting due to a high grade compression of the limbs detected on the 30-day follow-up CT imaging. Final sac diameter was 62.4±12.7 resulting in an average shrinking of −2.2±4.5 mm. Significant sac shrinkage was detected in 7 cases (25%), while the other 21 sacs were stable (75%). No significant sac dilatation was detected. There was no aortic rupture, conversion to open repair, migration, or integrity issue recorded. Graft infection was not detected.
Freedom from any branch-related endoleak and freedom from any branch lesion requiring reintervention were both 96.4% (n=27), both associated with a renal stent disconnection in a single patient that was successfully managed by a relining.
Primary and secondary target vessel patency was both 98.8% (n=81), with a single renal stent occlusion detected at follow-up imaging (Figure 1).
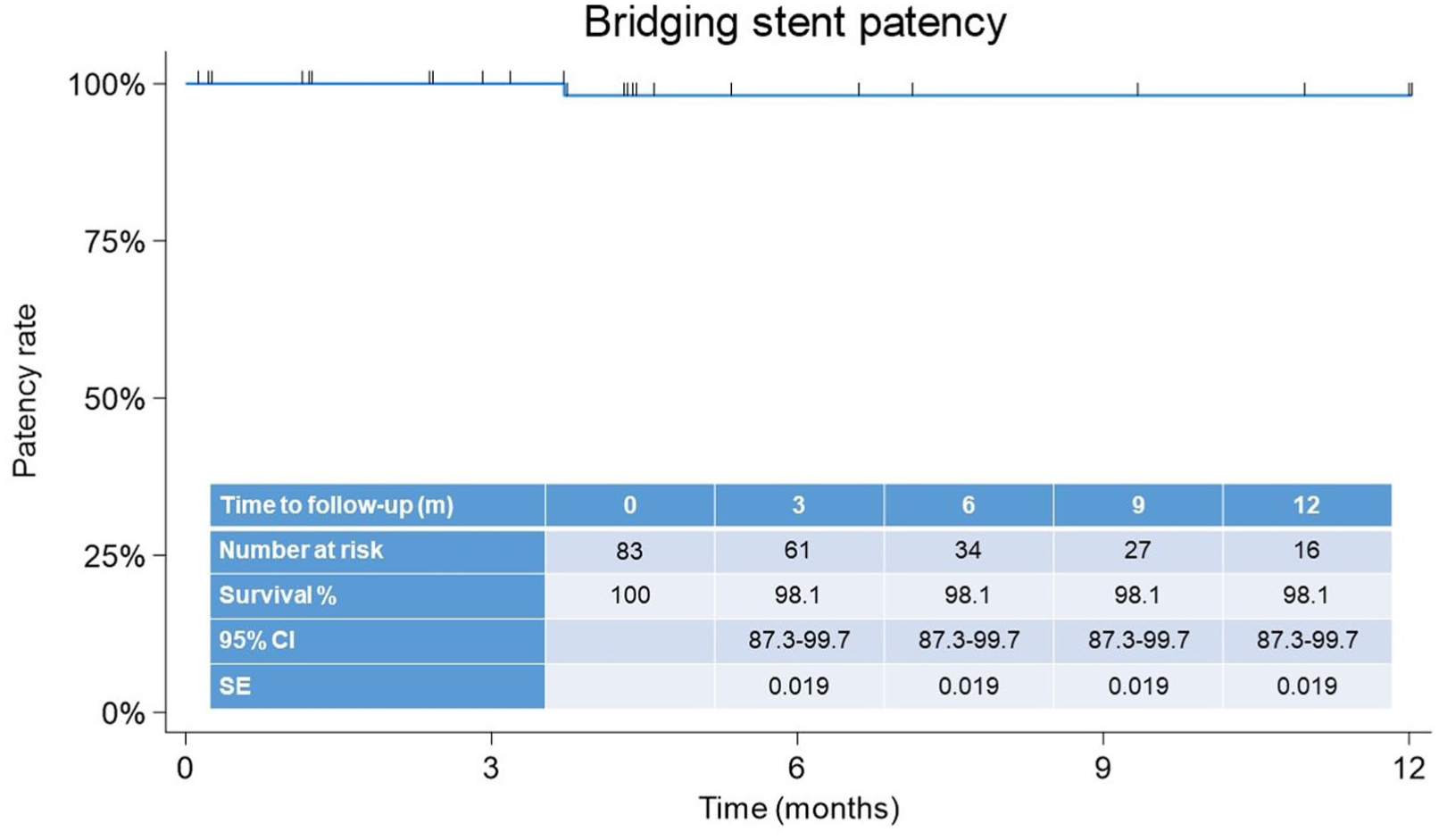
Bridging stent patency.
Discussion
This cohort represents the initial European experience with the PMEG technique in asymptomatic patients with CAAAs. An international multicenter study was conducted with 4 experienced centers across Europe and including a total of 28 patients. With a high-technical-success rate (96.4%), a low complication rate, and no in-hospital mortality, these results suggest that the PMEG technique may be a safe option in this clinical setting.
Current guideline of the European Society for Vascular Surgery recommends the PMEG technique for the treatment of urgent patients only, unless within a framework of a clinical trial. 5 However, being a technically rather demanding procedure, it might make sense to gain experience not only in urgent settings, but also in elective scenarios, which can help shortening the learning curve, thus improve the overall outcome. The inherent disadvantage of CMDs is the lengthy period between the therapeutic decision and the actual operation, associated with a non-negligible risk of interval rupture. Besides the shipping time of 6 to 8 weeks after the initiation of production, this period often includes 2 to 3 weeks of discussions with the planning center, waiting in the planning queue, as well as institutional administration of the custom device order, resulting in an actual decision-to-operation time of 3 to 4 months. With PMEGs, this could easily and instantly reduced to days, if not hours. This is getting recognized by more and more centers, making it a rather attractive approach, especially since recent studies shows that long-term outcome of such repairs seem to be non-inferior to CMD FEVAR.1–3
The use of the PMEG technique in elective settings remains controversial, with both ethical and medicolegal implications. In the participating centers, patients consistently preferred expedited treatment of their CAAA with PMEG, despite being offered and on-label CMD FEVAR. Timeliness seems to be the single most important decisive factor for the vast majority of the patients, who primarily feared rupture. In many countries outside of Western Europe, the limited budget available for endovascular aortic procedures places physicians in an ethical dilemma regarding the allocation of scarce resources. Restricting the use of the PMEG technique and offering CMD repair only will most likely result in less CAAA patients treated by endovascular means, thereby forcing physicians to prefer open surgical repair in patients whose anatomy would otherwise be suitable for FEVAR. Moreover, current regulatory changes in the approval process of implantable device will most likely further increase the price of CMDs, which is already astronomical at times. In health care systems with reimbursement structures based on fixed budgets, the ethical dilemma of allocation is inevitable.
Potential financial benefits of the PMEG technique over CMD FEVAR cannot be overseen. Available reports are contradictory, indicating that it is very much dependent on the surrounding health care system. 13 Nevertheless, the PMEG technique represents an appealing alternative to CMDs for many countries, as the latter is associated with costs up to nearly 4 times higher. 14
Although we are in the progress of defining the best practice for PMEG, heterogeneity regarding both the endograft platforms and the procedural steps persists, hindering a more widespread acceptance of this approach and also limiting the generalizability of the reassuring findings of recent reports regarding the long-term outcome of such repair.1–3,15–17
The platform of the PMEG technique in the current study was the Treo device, which offers several advantages for such use, for example, a long main body, an integrated bifurcation, widely spaced struts both circumferentially and longitudinally, and a low insertion profile with an active fixation at the hub-end of the semi-deployed device within the delivery system.
The long main body provides plenty of space for positioning multiple fenestrations along the device, while also maintaining a safety distance from the flow divider, which can cause issues during and after the deployment of the bridging stents if the lowermost fenestration is in the close proximity of the device’s bifurcation. In this series, only 3 renal fenestrations were positioned 15 to 20 mm proximal to the flow divider, while the vast majority of the others were positioned in the 30 to 50 mm range. This allows an easy cannulation from either side and causes no issue of the flared segment compromising the iliac limb’s patency. A long main body also offers the opportunity of using a supraceliac landing zone, which might be beneficial regarding late reinterventions due to landing zone dilations, without increasing the overall risk of the procedure. 18
Besides the obvious benefit of reducing the number of aortic components, for example, avoiding an unnecessary overlap zone, reducing procedural complexity and additional costs, an integrated bifurcation might also increase the safety of the repair. A recent refinement of the modular design CMDs includes distal components with a short dilator tip to reduce interference with the deployed and flared bridging stents, thus to reduce the need of on-table or in-hospital reinterventions. This issue is mostly absent if the fenestrated component has an integrated bifurcation with a lowermost fenestration more than 30 mm proximal to the flow divider for the majority of the cases.
Widely spaced struts allowed full avoidance of strut modifications in this cohort, a technique that is often necessary with other platforms due the interference of the struts and the fenestrations, which can compromise the durability of the device. However, the large longitudinal interstrut distance comes at a price of difficulties during reloading: unsupported segments between the stent rows often show bulging during reload, which might be challenging to manage especially if fenestrations are also present in the affected segment. Wide strutless areas within a stent row’s circumference are a consequence of the larger angles utilized at the peaks of the stent rows; this is a feature usually present on thoracic implants, making them generally better for such use than abdominal devices.
A low insertion profile of an implant is paramount to keep our repair minimally invasive. The 18–19F delivery system of the aortic component is significantly smaller than the thoracic components often used for such purpose (22–24F for the Valiant [Medtronic, Minneapolis, MN, USA]), while sharing the same design benefits of those implants. A low-profile delivery system increases the applicability of this technique in cases with difficult access anatomies. Still, access complications (n=4) contributed to the 80% of the in-hospital adverse events (n=5) in this initial cohort, as other major complications were absent.
The active fixation of the semi-deployed device within the delivery system is a unique feature of the Treo device that increases torque- and pull-control during deployment—a minor addition that is paramount in the longitudinal and circumferential precision of the deployment. This feature is often augmented by additional modifications of the delivery system on other platforms.
Despite the potential advantages of PMEGs, several drawbacks and uncertainties remain. The most important limitation is the lack of long-term durability data compared with CMDs. While early reports, including the present study, indicate promising technical and clinical outcomes, concerns persist regarding the structural integrity of modified fenestrations, variability in reinforcement techniques, and the absence of standardized manufacturing and quality control processes. These factors may contribute to heterogeneity in outcomes and limit the generalizability of the current findings.
Furthermore, the PMEG technique is technically demanding and highly operator-dependent. Differences in device platforms, back-table modification methods, and intraoperative handling may influence outcomes, especially in less experienced centers. In addition, potential regulatory and medicolegal issues remain unresolved, as these devices do not undergo the same rigorous approval pathways as CMDs. Finally, economic considerations may vary substantially between health care systems, and the financial implications of adopting PMEGs on a broader scale remain uncertain.
Limitations of Study
Some inherent disadvantages due to the retrospective nature of this study have to be acknowledged. The retrospective data collection increases the risk of reporting and recall bias, as well as of missing some data. This is a rather small patient cohort, particularly when compared with studies from the overseas, where the limited availability of CMDs paved the way for the general acceptance and more widespread use of the PMEG technique. The low sample count precluded analyses of statistical correlations.
Conclusion
This international multicenter observational study summarizes the initial European experience of the PMEG technique for elective CAAAs using an abdominal bifurcated endograft. The approach was associated with a high-technical-success rate, and major clinical complications were uncommon. However, given the small cohort size and limited follow-up, no firm conclusions can be drawn regarding safety or efficacy. Long-term data from larger, prospective studies are required before the role of the PMEG technique in the elective treatment of complex AAAs can be established.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Study protocol was approved by the local ethics committee (129/2024).
Informed Consent
Informed consent to participate has been waived by the relevant Ethics Committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Csaba Csobay-Novák was supported by a grant from the National Research, Development and Innovation Office (NKFIH) of Hungary (STARTING 152421) and by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.C.N. is a trainer, speaker, and proctor for Cook Medical, Gore Medical and Medtronic. V.M. is a consultant and proctor for Terumo Aortic.
Data Availability Statement
The data supporting the findings of this study are available from the authors upon reasonable request.
