Abstract
Introduction:
This is a descriptive study analyzing all surgically treated complex aneurysm patients in a tertiary referral center, during a 15-year time period in which complex endovascular aortic repair (complex EVAR) was introduced alongside open surgical repair (OSR). The goal was to present the real-world clinical profiles and surgical outcomes of complex (thoraco-)abdominal aneurysm patients, based on the current decision-making process.
Methods:
Patients undergoing OSR and complex EVAR between January 2008 and August 2023 were retrospectively included. The reasoning behind each treatment decision was analyzed. Both groups were described in terms of baseline characteristics and outcomes. A subanalysis was made for the frailest complex EVAR patients, considered unfit for OSR.
Results:
As of 2008, 112 patients underwent OSR, whereas 119 patients underwent complex EVAR since 2013. When both modalities were considered opportune, the patient’s preference toward complex EVAR was often decisive in shared decision-making. Based on the current selection process, 47 patients were considered too frail for OSR. Complex EVAR patients had a higher Charlson Comorbidity Index (3.0 [IQR: 2.0,4.0] vs 4.0 [3.0, 5.0], p=0.002). More complications occurred after OSR (31.3% vs 18.5%, p=0.027), while reinterventions were more common after complex EVAR (20.2% vs 8.0%, p=0.008). The 47 frailest complex EVAR patients had a lower median survival time and high 1-year mortality of 23.8%.
Conclusion:
Although less invasive compared to OSR regarding complications, complex EVAR still comes with considerable risks. For some frailest patients, complex EVAR might be a bridge too far. Specific insights in patient selection are warranted.
Clinical Impact
With the introduction of complex EVAR, more complex aneurysms were treated, in an overall frailer patient cohort. The frailest complex EVAR patients had a high 1-year mortality and a significantly lower mean survival compared to fit complex EVAR patients and patients undergoing OSR. The current selection process seems effective in recognizing the frailest patients and assigning them to complex EVAR, as guidelines propose. However, it might not yet be able to select patients that would benefit more from conservative non-surgical management. Specific insights in patient selection are warranted.
Keywords
Introduction
Aortic aneurysms that comprise segments of the aorta including visceral side branches are classified as complex. Such aneurysms can be treated by open surgical repair (OSR) or by endovascular aortic repair (complex EVAR), using fenestrated (FEVAR) or branched (BEVAR) stents. Evidence suggests that complex EVAR provides favorable perioperative outcomes compared to OSR. These advantages are counterbalanced by a higher number of reinterventions and the need for life long surveillance. Moreover, it appears that the short-term survival benefit is not maintained during longer-term follow-up.1–4 Guidelines have therefore been somewhat cautious in their advice on the implementation of complex EVAR, mainly recommending it for patients considered frail or unfit for OSR.5–9
In practice, complex EVAR has become more and more conventional, extending beyond a treatment modality only for the frailest patients. Implementation of this technique is limited by the need for advanced materials, patient exposure, and related costs.10,11 In addition, the introduction of new techniques necessitates adequate patient selection and information provision. For future planning and policy making, it is essential to understand how the introduction of complex EVAR has shaped real-world treatment patterns and outcomes.
This descriptive study provides a detailed overview of how complex (thoraco-)abdominal aneurysm patients were selected and treated in a tertiary referral center for aortic pathology in the Netherlands, in a 15-year time period during which complex EVAR was introduced. The goal was to analyze the clinical profiles and surgical outcomes of complex aneurysm patients, based on the current decision-making process. A subanalysis was made of patients deemed unfit for OSR; the initial target group, intended to benefit the most from complex EVAR. We aspire to contribute real-world data to the ongoing research on complex EVAR, to better understand the changing landscape of complex aneurysm repair.
Materials and Methods
Study Design
The current study is a descriptive single-center retrospective cohort study, within a tertiary referral center for aortic pathology in the Netherlands; Leiden University Medical Center (LUMC). This study was not designed to evaluate treatment efficacy, but to provide a detailed overview of how complex (thoraco-)abdominal aneurysm patients were selected and treated in current practice. As is required by Dutch law, approval was issued by the LUMC Medical Ethics Committee (METC, registration number 2024-010). Due to the merely retrospective character of our study, the anonymized use of data, and the fact that there was no interference in the standard care pathway, explicit patient consent was not obtained. All data was subtracted from patient’s medical records and archived in an online secured computerized database (Castor). 12
Decision-Making
In this study, a complex aneurysm was defined as a (thoraco-)abdominal aneurysm that included or was narrowly adjacent to segments of the aorta with visceral side branches, to which perfusion needed to be maintained when surgery was performed. Prior to the introduction of complex EVAR, open repair was the sole treatment option for complex aneurysms. Complex EVAR was introduced in the LUMC in July 2013, when an endovascular treatment team (ETT) was formed, consisting of vascular surgeons, cardiothoracic surgeons, interventional radiologists, anesthesiologists, dedicated operating room (OR) staff, and radiology technicians. The main stent graft manufacturer was COOK Medical, using the Zenith.
When a complex aneurysm is discovered, imaging follow-up is initiated until the treatment threshold of 6 cm is reached, and a treatment decision has to be made. Patients are usually referred to an anesthesiologist, geriatric specialist, dietician, and physiotherapist, in order to examine the patient’s physical and cognitive condition, all using their own indicators, such as cardiopulmonary fitness tests, American Society of Anesthesiologists score, hand grip strength, Mini Nutritional Assessment, and (Instrumental) Activities of Daily Living scores. Other specialists can be consulted if comorbidities require so, for example, a pulmonologist when concerns arise regarding pulmonary function. Complex EVAR, OSR, and conservative non-surgical management are discussed whithin the multidisciplinary treatment team. The ETT forms an advice in a multidisciplinary meeting, taking into account preoperative screening, the aneurysm’s anatomy, and the patient’s preferences. This advice is discussed with the patient and, preferably, its relatives. Usually, there is a reflection period of at least 2 weeks before the final treatment decision has to be made.
After complex EVAR, patients are seen at 6 weeks, 6 months, and 12 months post-surgery and yearly after that for lifelong follow-up. Additional appointments are made if deemed necessary. Computed tomography angiography and Duplex ultrasonography are used for detection of complications such as stent migration, aneurysm sac enlargement, and endoleaks. After OSR, patients are seen at 6 weeks, 6 months, and after 1, 3, and 5 years. Duplex ultrasonography is usually applied within year 1 and after 3 to 5 years.
Patient Inclusion
All patients electively treated for a complex aneurysm between January 2008 and August 2023 were included. January 2008 was chosen as the starting point of inclusion, because by that time rigorous registration had started within the currently still accessible electronic patient file software (ChipSoft HiX). Inclusion ended by August 2023, in order to reach a minimum follow-up period of 6 months when retrospective data collection started in January 2024. This means we were able to include patients treated during 5 years before complex EVAR was introduced (2008–2012) and during approximately 10 years in which both treatment modalities were available (July 2013–August 2023). As of 2021, patients with a thoraco-abdominal aneurysm undergoing OSR were treated in collaboration with a surgical team of the Amsterdam University Medical Center (AUMC).
The endovascular cohort consisted of all patients that underwent fenestrated EVAR (FEVAR), branched EVAR (BEVAR), or a combination (F/B-EVAR). The open repair group included patients that underwent a Crawford procedure for thoraco-abdominal aneurysms and patients that underwent open reconstruction for suprarenal, pararenal, or juxtarenal aneurysms. Juxtarenal aneurysms were considered complex if they were unsuitable for regular EVAR (within instructions for use) due to hostile neck anatomy, based on a combination of criteria: aortic length (<10 mm), excessive aortic neck angulation, aortic neck diameter (>30 mm), conical neck, and presence of circumferential calcification or thrombus. Reasons for exclusion were: (semi-)emergent repair of ruptured and symptomatic complex aortic aneurysms, explantation of an infected or otherwise failing EVAR-stent with conversion to open repair. In addition, patients who underwent both complex EVAR and open repair were excluded. Arch-EVAR patients were excluded, due to small patient numbers and to comply with inclusion criteria of previous research, as including these cases would impede meaningful comparison.
Outcomes of Interest
The nature of this study was descriptive, aiming to analyze changes in the number of patients being treated, differences in patient characteristics between OSR and complex EVAR, and the reason for choosing either treatment modality. For these primary outcomes, the following baseline and surgical characteristics were gathered: age at the time of surgery, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) score, comorbidities, type and size of the aneurysm, treatment modality, and the documented reasoning behind the treatment decision between open and endovascular repair as of July 2013. Baseline characteristics were used to determine the Charlson Comorbidity Index, in order to measure and compare the burden of comorbid disease, ranging from 0 to 37. The modified 5-Item Frailty Index (mFI-5) was calculated as an indicator of the patient’s frailty, ranging from 0 to 5.13,14
The secondary aim was to analyze the postoperative outcomes, resulting from the current selection process. Outcomes of interest were: technical or modified technical success, Major Adverse Events (MAE), length of hospital stay, reinterventions, 30-day mortality, 1-year mortality, and all-cause mortality during follow-up. The definition of modified technical success was based on reporting standards for complex EVAR and adapted to apply to OSR as well; a procedure needing an alteration of the preoperative plan, but resulting in the successful employment of a stent or implantation of a graft. 15 A subgroup analysis was made regarding complex EVAR patients deemed unfit for OSR by the ETT, due to comorbidities, physical condition, and/or age. The goal was to examine the baseline characteristics and outcomes of these frailest patients, compared to patients eligible for both treatment modalities.
Statistics
Categorical patient characteristics were expressed by number of patients and percentages. Continuous variables were tested for normal distribution using histograms, assessing skewness. Normally distributed data was expressed as mean with the standard deviation (SD). Median with the interquartile range (Q1–Q3) was used in case of skewed distribution. The independent t-test (2 groups) or 1-way ANOVA test (3 groups) was used to compare continuous normally distributed data, Chi-square test for categorical data, and the Mann-Whitney U test (2 groups) and Kruskal-Wallis (3 groups) for skewed data. When less than 5 events occurred, the Fisher’s exact test was used instead of chi-square. Post-hoc testing was performed using Dunn’s test after Kruskal-Wallis analysis. All-cause mortality was tested by Kaplan-Meier with log-rank testing. Data was terminated when less than 10 patients were at risk in any of the groups and/or when the standard error reached below 10%. The study design was descriptive, aiming to present real-world data based on clinical practice and the current decision-making process. Therefore, propensity score methods to artificially balance the (sub)groups, were not applied, in order to prevent the loss of selection effects. P-values are presented to indicate observed trends in both treatment groups, in the context of their known baseline characteristics. They do not aim to indicate treatment efficacy or superiority. All analyses were made using IBM SPSS Statistics version 27.
Results
Patient Numbers
A total of 231 elective complex aneurysm patients were included (Figure 1). From 2008 until August 2023, 112 patients underwent open complex aneurysm repair, whereas 119 patients underwent complex EVAR between July 2013 and August 2023. Figure 2 depicts a distribution of the number of treated patients over the years, showing an increase in the total number of complex aortic aneurysms being treated and a rise in endovascular procedures. By 2016, complex EVAR exceeded open repair as the preferred treatment modality, except for the year 2017.
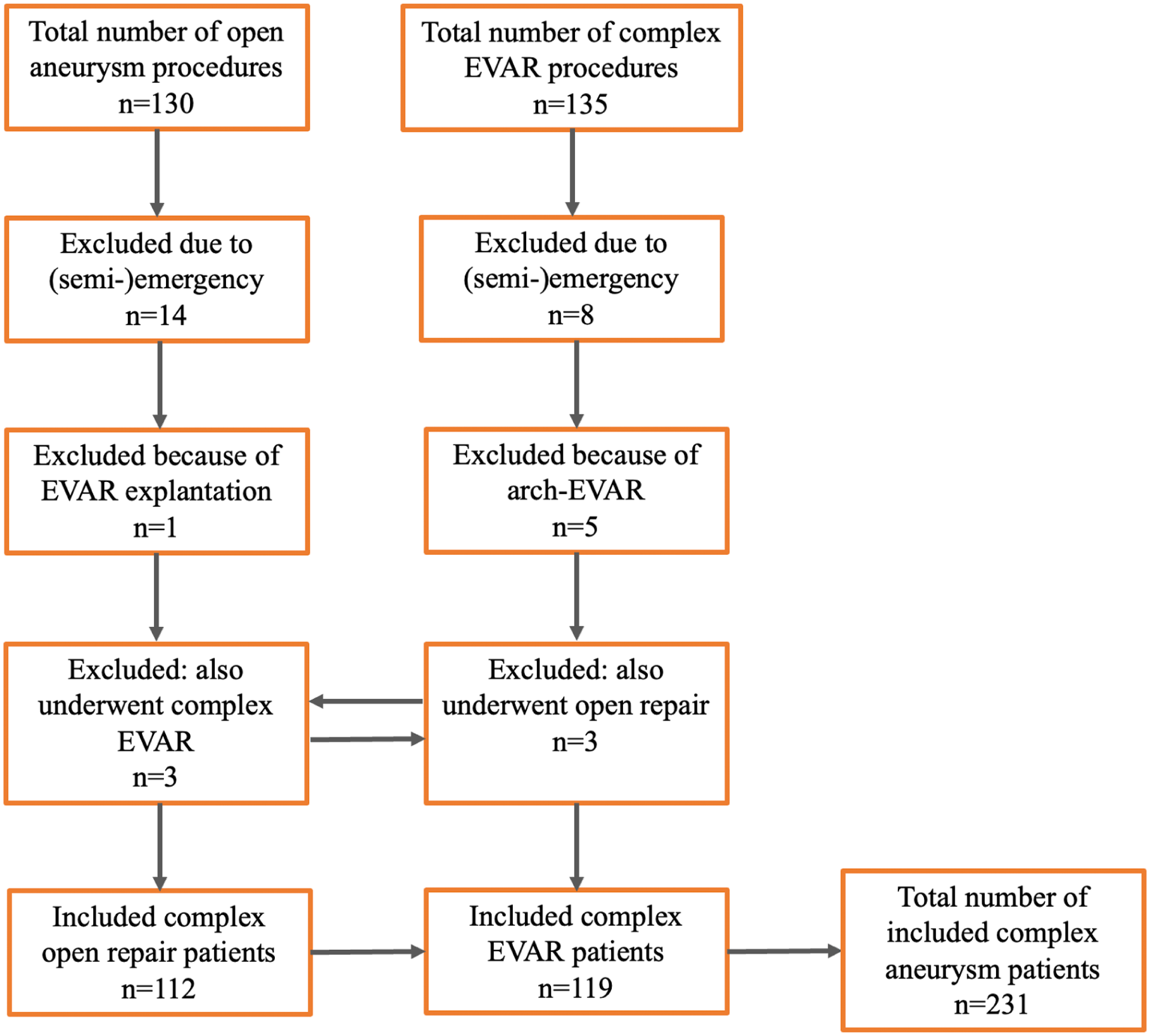
Inclusion flowchart. EVAR, endovascular aortic repair.
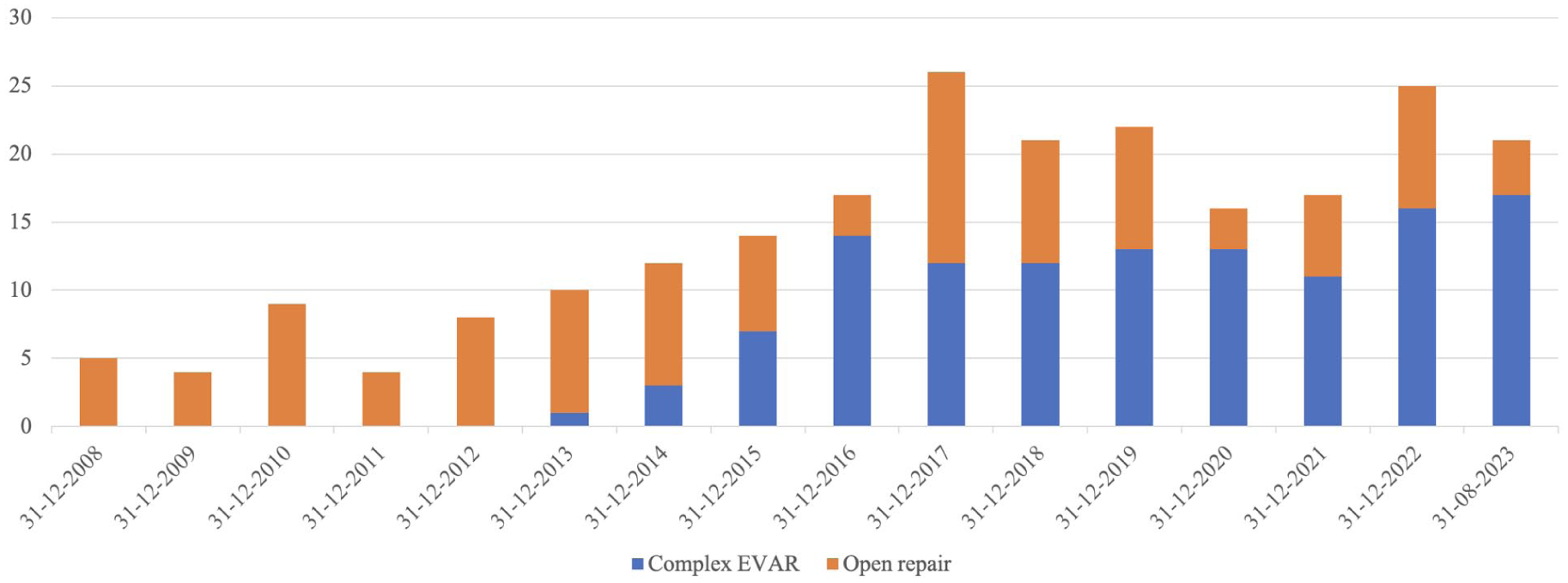
Number of complex aneurysm patients treated in the Leiden University Medical Center from 2008 until August 2023. EVAR, endovascular aortic repair.
Reasoning Behind Treatment Decisions
Table 1 shows the reasoning for choosing between OSR and complex EVAR. Figure 3 depicts a flow-chart. This analysis included patients treated as of 2013 (n=201), when both complex EVAR and OSR were practiced in the LUMC. It indicates the number of patients for whom each consideration was relevant. For some patients, multiple reasons were registered.
Main Reasoning Behind the Treatment Decision Between Open Repair (n=82) and Complex EVAR (n=119) in the Era That Both Procedures Were Being Performed in the LUMC (2013–2023).

For some patients, multiple considerations were decisive.
Abbreviations: complex EVAR: complex endovascular aortic repair; ETT: endovascular treatment team; LUMC: Leiden University Medical Center; OSR: open surgical repair.
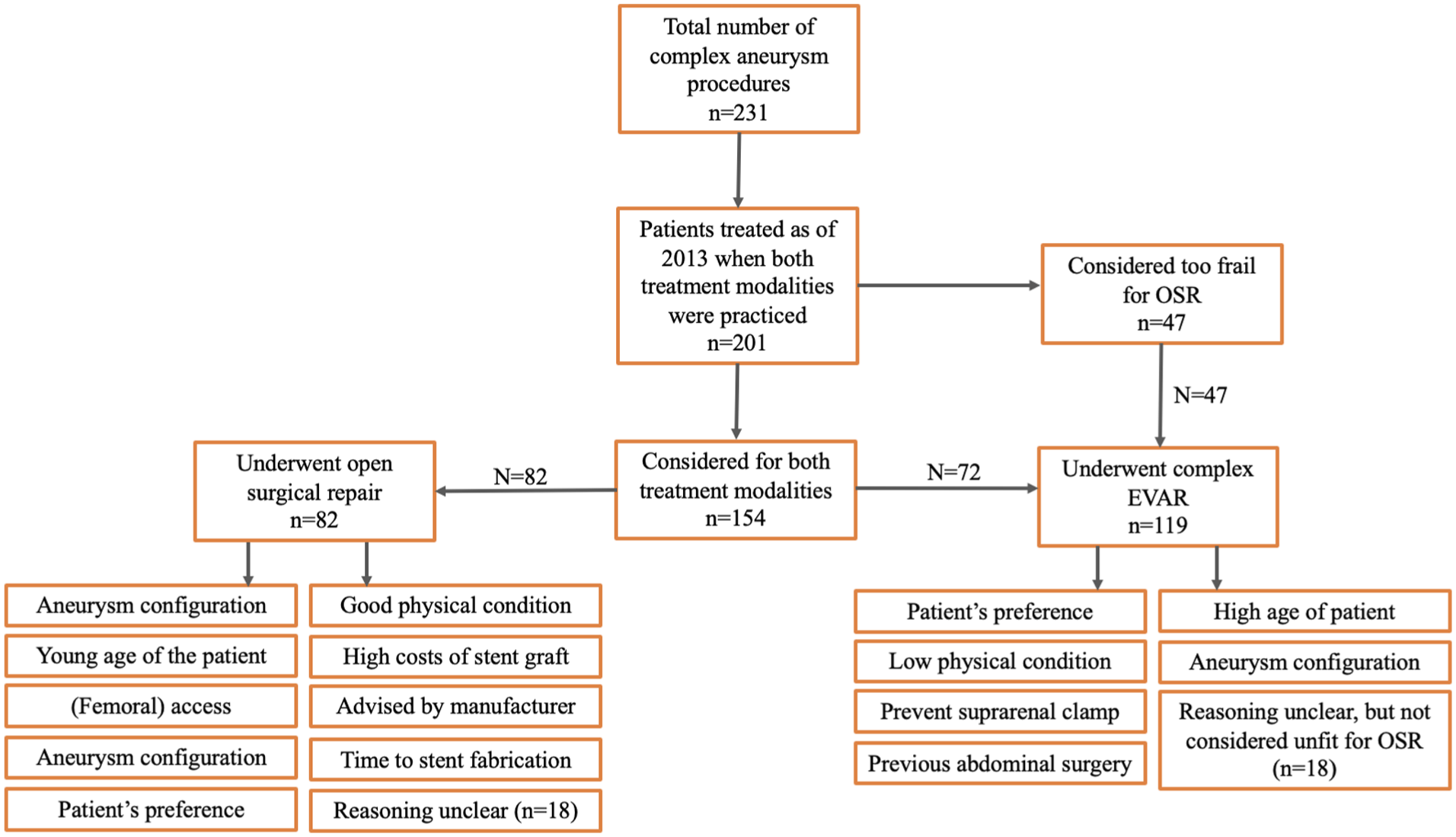
Flowchart of decision-making when both open surgical repair (OSR) and complex endovascular aortic repair (complex EVAR) were practiced.
The main reason for the treatment team to prefer open repair (n=82) in this era was based on the aneurysm’s configuration (n=27, 32.9%), for example, due to doubts regarding stent sealing or the need for extended aortic coverage. In addition, factors complicating femoral access (n=9) and the etiology of the aneurysm (connective tissue disease, infectious) could favor open repair (n=9). Young age (n=12, ranging from 43 to 77 years old) and good physical condition (n=5) were patient-specific characteristics leading to a preference for OSR. In 8 cases, the patient’s own preference toward OSR was one of the decisive reasons in shared decision-making.
In the same era, 119 patients underwent complex EVAR. In 47 cases (39.5%), the ETT decided that the patient was too frail for OSR, based on physical condition, preoperative screening, and/or age. When both modalities were considered possible, the patient’s preference toward endovascular repair was often decisive in shared decision-making (n=49, 41.2%). Other considerations for choosing complex EVAR over OSR were: wanting to prevent the need for suprarenal clamping (n=4), adhesions after previous abdominal surgery (n=3), and the aneurysm’s configuration being regarded as less compatible with OSR (n=2). In 18 patients, the reason for choosing complex EVAR did not became clear from their medical record, although they did not seem to be considered unfit for OSR.
Patient Characteristics
Table 2 includes the patient characteristics, comparing patients treated by OSR versus complex EVAR. Following the current patient selection process, patients selected for complex EVAR turn out to be more often male (69.6% vs 85.7%, p=0.003), older (67.9 [SD: 9.6] vs 74.1 [SD: 6.5] years, p<0.001), with a higher median Charlson Comorbidity Index (3.0 [IQR: 2.0, 4.0] vs 4.0 [IQR: 3.0, 5.0], p=0.002), and more often a Modified 5-Item Frailty scale of 2 or higher (16.1% vs 26.9%, p=0.046). This is indicative of a heavier burden of comorbid disease and more frail patients among the complex EVAR group.
Baseline Characteristics of Complex Aneurysm Patients, Treated by Complex EVAR Versus Open Surgical Repair.
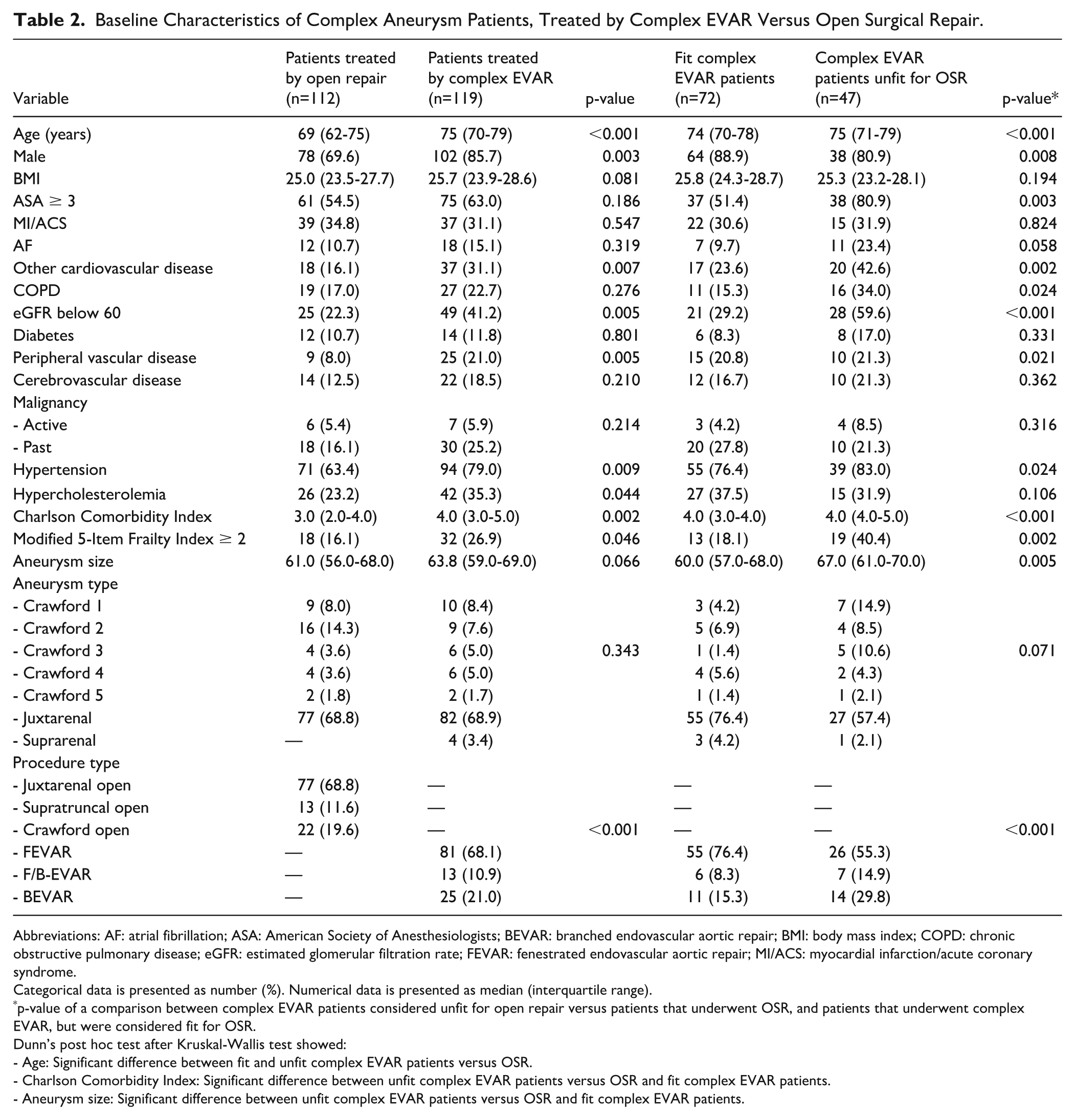
Abbreviations: AF: atrial fibrillation; ASA: American Society of Anesthesiologists; BEVAR: branched endovascular aortic repair; BMI: body mass index; COPD: chronic obstructive pulmonary disease; eGFR: estimated glomerular filtration rate; FEVAR: fenestrated endovascular aortic repair; MI/ACS: myocardial infarction/acute coronary syndrome.
Categorical data is presented as number (%). Numerical data is presented as median (interquartile range).
p-value of a comparison between complex EVAR patients considered unfit for open repair versus patients that underwent OSR, and patients that underwent complex EVAR, but were considered fit for OSR.
Dunn’s post hoc test after Kruskal-Wallis test showed:
- Age: Significant difference between fit and unfit complex EVAR patients versus OSR.
- Charlson Comorbidity Index: Significant difference between unfit complex EVAR patients versus OSR and fit complex EVAR patients.
- Aneurysm size: Significant difference between unfit complex EVAR patients versus OSR and fit complex EVAR patients.
A subanalysis (Table 2) was made comparing complex EVAR patients considered unfit for OSR (n=47), with patients that were considered fit for both modalities. Patients considered unfit for OSR had a higher Charlson Comorbidity Index compared to fit complex EVAR and OSR patients, while there was no statistically significance in this score between fit complex EVAR versus OSR patients (4.0 [IQR: 4.0, 5.0] vs 4.0 [IQR: 3.0, 4.0] and 3.0 [IQR: 2.0, 4.0] respectively). In addition, the frailest complex EVAR patients more often had an ASA score of 3 or higher and a mFI-5 of 2 or above, while these numbers were not statistically different between fit complex EVAR patients and patients that underwent OSR. In addition, the frailest complex EVAR patients were more often diagnosed with an eGFR below 60 mL/min and COPD.
Perioperative Outcomes and Reinterventions During Follow-up
The (modified) technical success rate in the OSR group was 100%, with 11 cases (9.8%) needing a perioperative alteration from the preoperative procedure plan, such as a splenectomy for capsule tear or a bypass for encountered artery occlusion. In the complex EVAR group, the (modified) technical success rate was 96.6% (p=0.125), with 13 patients (10.9%) needing a perioperative alteration in order to reach technical success, such as an unplanned iliac extension due to type 1B endoleak or intentionally leaving a branch open due to stent rotation. Table 3 shows the Major Adverse Events (MAE). In total 35 (31.3%) patients experienced 58 MAE in the open repair group versus 22 (18.5%) patients with a total of 31 MAE in the complex EVAR group (p=0.027). In the open repair group, a significantly higher number of patients experienced respiratory failure (15.2% vs 4.2%, p=0.006), as well as renal function decline (16.1% vs 7.6%, p=0.047).
The Number of Major Adverse Events (MAE) Compared Between Patients Treated by Open Surgical Repair and Complex Endovascular Aortic Repair (Complex EVAR).
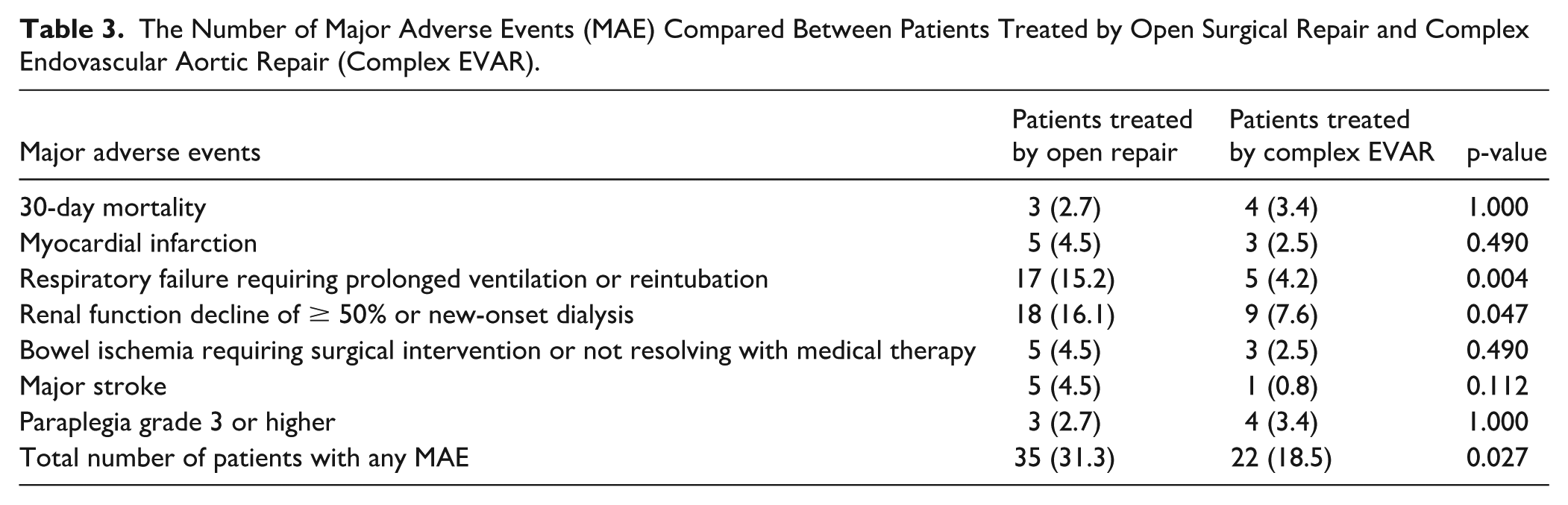
Thirty-day mortality was comparable: 2.7% (n=3) in the open repair group and 3.4% (n=4) in the complex EVAR group (p=1.000). The causes of these deaths are indicated in Appendix A. Median length of hospital stay in the OSR group was 11.0 (IQR: 8, 20) days versus 6.0 (IQR: 3, 11) days after complex EVAR (p<0.001).
During follow-up, 17 complex EVAR patients (14.3%) needed a reintervention for one or more endoleaks (8 type 1 endoleaks, 6 type 3 endoleaks, and 6 other endoleaks). In addition, there were 9 patients (7.6%) in this group that had to undergo a reintervention for other aneurysm-related complications. In total, 24 complex EVAR patients (20.2%) needed any aneurysm or procedure-related reintervention during follow-up versus 8 patients (7.1%) in the OSR group (p=0.004). The types of reinterventions are included in Appendix B.
One-Year and All-Cause Mortality
There was no statistically significant difference in 1-year mortality between all complex EVAR versus OSR patients (15.8% vs 8.9% respectively, p=0.142). However, there was a significant difference when analyzing the 1-year mortality between the frailest complex EVAR patients (23.8%), compared to fit complex EVAR patients (9.4%), and OSR (8.9%), p=0.047. The overall survival curve (Figure 4) showed a trend toward lower estimated mean survival time for complex EVAR patients (57.7 months, CI: 51.3–64.1), compared with patients treated by open repair (66.7 months, CI: 61.4–72.1), p=0.048. Data was censored after 82 months, when less than 10 patients remained at risk in the complex EVAR group. A subanalysis showed that the frailest complex EVAR patients had a significantly lower mean survival time (33.0 months, CI: 27.5–38.6), compared to fit complex EVAR patients (43.3 months, CI: 40.2–46.5) and patients that underwent OSR (41.8 months, CI: 39.0–44.5), with p<0.001 (Figure 5). The apparent lower survival in the OSR group, compared to the analysis above, is not due to worse survival, but is caused by the fact that data was truncated earlier in this analysis, at 48 months, when less than 10 frail complex EVAR patients remained at risk.
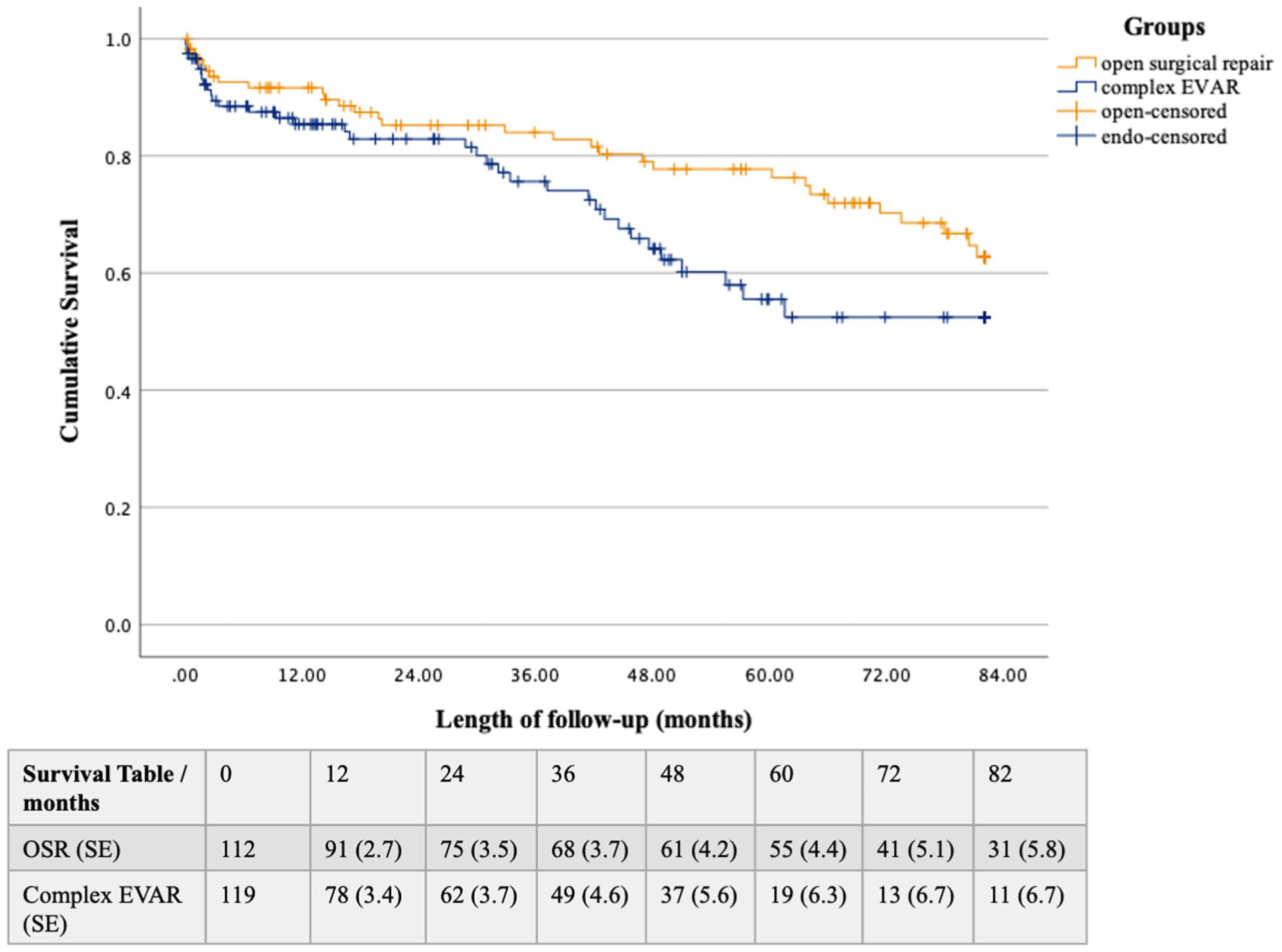
Kaplan-Meier survival curve of patients treated by complex endovascular aortic repair (complex EVAR) versus open surgical repair (OSR). Standard error (SE) is provided in %. Data is censored beyond 82.2 months, when less than 10 patients remained in the complex EVAR group.
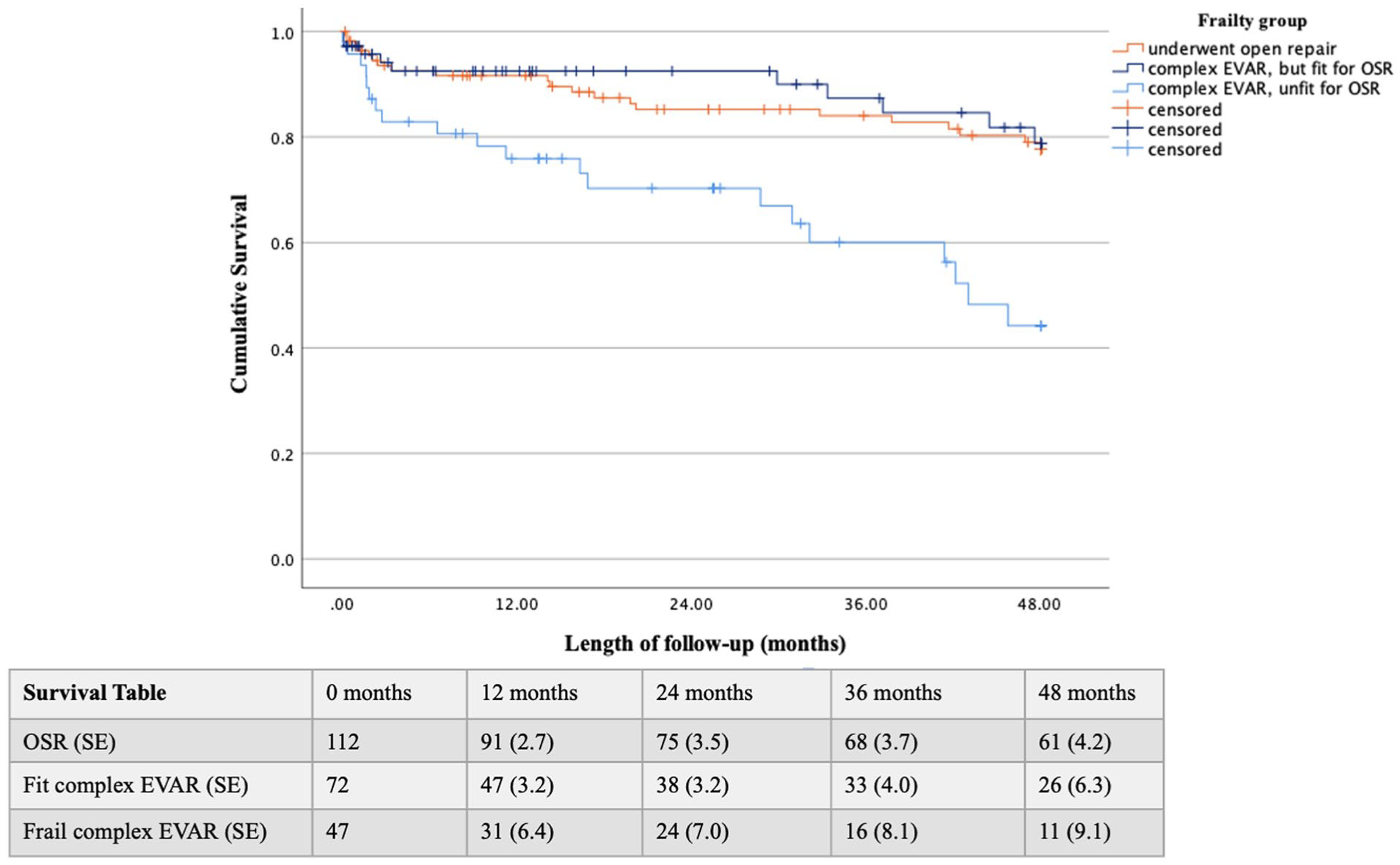
Kaplan-Meier survival curve with a sub analysis of frail complex endovascular aortic repair (complex EVAR) patients. Standard error (SE) is provided in %. Data is censored beyond 48.1 months, when less than 10 patients remained in the frail complex EVAR group.
Discussion
An analysis of 15 years of complex aneurysm management in our tertiary referral center showed a steady increase in the number of complex aneurysm procedures being performed, with a temporary decrease during the Covid pandemic (2020–2021). Complex EVAR became the preferred treatment method soon after its introduction, which should be anticipated when deciding to introduce this technique. Over one third of complex EVAR patients was deemed too frail for open repair by the ETT. These patients would likely not have been treated in the pre-EVAR era. When both modalities were considered opportune, the patient’s preference toward complex EVAR was often decisive in shared decision-making.
Our decision-making process is currently based on the professional opinion and expertise of the multidisciplinary treatment team, including patient preferences in shared decision-making. Preoperative screening is often performed, using several indicators of a patient’s physical and cognitive condition. This practice-based approach resulted in the fact that complex EVAR patients were more likely to be frail, with a higher burden of comorbid disease. These differences were mainly caused by the frailest complex EVAR patients, considered unfit for OSR.
The reason for complex EVAR being the preferred treatment option for frail patients is the fact that studies have shown a lower perioperative risk of morbidity compared to OSR and a trend toward lower perioperative mortality.1,2,4 In addition, studies have shown that complex EVAR can safely be performed in carefully selected octogenarians. 16 In our real-world cohort, there was indeed a lower number of MAE after complex EVAR, while 30-day mortality was comparable. The frailest complex EVAR patients, considered unfit for OSR, had a high 1-year mortality (23.8%) and an estimated median survival time of about 33 months. A notable proportion of complex EVAR patients had to undergo reinterventions during follow-up. However, it is possible that due to the extensive surveillance after complex EVAR more minor complications are detected and treated, in addition to the fact that reinterventions after complex EVAR are usually less intensive compared to reinterventions after OSR. In addition, center-specific practices can be of importance when interpreting MAE results. In our hospital, for example, epidural anesthetics are usually applied and early extubation is pursued. In open repair, renal preservation techniques are usually not applied. However, the application of such techniques can vary over time. Given the relatively small patient cohort, meaningful trend analyses are currently not feasible.
Although it is not clear whether these survival differences are entirely attributable to frailty, as the difference in endovascular procedures type (BEVAR vs FEVAR) could for example also play a role, it is debatable whether all these frailest patients ultimately benefit from undergoing surgery. Especially when considering prolonged recovery and possible decline in physical functioning. The current selection process seems effective in recognizing the frailest patients and assigning them to complex EVAR, as guidelines propose. However, it might not yet be able to select those who would benefit more from conservative non-surgical management.
Being able to predict which patients are most at risk for an unfavorable outcome after surgery, would contribute to patient information and selection. 17 In our cohort, unfit patients who experienced more unfavorable outcomes, were more likely to have cardiovascular disease, COPD, and renal function insufficiency. Efforts are being taken to investigate such potential predicting factors, not just regarding comorbidities, but including a patient’s physical and daily functioning, nutritional status, and quality of life as well. Some indicators might be modifiable, like malnutrition and low muscle mass and could be optimized by prehabilitation.18,19 In addition, survival time does not provide information on quality of life after surgery. Patients decide to get treated with expectations and hopes regarding their life after surgery, with a certain level of functional performance in mind that they wish to uphold. 20 Being able to provide more information on this aspect of postoperative outcomes, by performing more research into functional performance after complex aneurysm repair, would further aid in preoperative counseling. 21
For some patients, complex EVAR is a bridge too far. Their life expectancy is limited more or equally by their level of comorbid disease, then by the risk of aneurysm rupture. This is supported in guidelines stating that elective aneurysm repair is not suitable for patients with a limited life expectancy of 2 to 3 years. 6 Previous research has shown, however, that conservative management is often not regarded as a realistic treatment option by patients. 20 We hypothesize that this is due to the fear of aneurysm rupture; the feeling of “living with a time bomb”. In this perspective, patients might overestimate the chance of aneurysm rupture, while underestimating their frailty and surgical risks. Treating the patient’s fear should not be the main reason for complex aneurysm surgery. The reality of this fear, compared to the risks of surgery, should be addressed during preoperative counseling. Professional psychological counseling could be offered, addressing the concerns that might arise when a conservative treatment decision is made.
Limitations
Inclusion was limited to the number of patients treated in the center of interest, which resulted in a relatively small patients cohort compared to multicenter studies or studies within high-volume centers. This impeded meaningful subgroup analyses regarding the different surgical subtypes, which were therefore not performed. The external validity of the results is confined by restricted generalizability. However, this approach fitted our goal of reporting on the patient population and outcomes within this specific center.
This study was constructed retrospectively. Analyses were therefore limited by the data available in patients’ medical records. Gathering data prospectively might lead to more data being available and more thorough research, especially regarding the analysis into the reasoning behind the treatment decisions. In 18 of the complex EVAR (15%) and 18 of the OSR patients (22%), the reason for choosing their treatment modality did not become clear. However, for these complex EVAR patients, OSR was considered as well, leading to the conclusion that they were not considered frail. In addition, complex aneurysm patients that ended up not being treated were not systematically registered and could therefore not be included in our study. Including these patients could shine a broader light on the decision-making process, baseline characteristics, and outcomes of all complex aneurysm patients presented in our hospital.
The study design was descriptive, aiming to present real-world data based on clinical practice and the current decision-making process. Propensity score methods to artificially balance (sub)groups were not applied.22,23 Confounders are therefore inherently present. However, this reflects the complexity of clinical decision-making in a real-world setting. Instead of eliminating these factors, we chose to present the variation that occurred based on our routine clinical practice. The indicators of frailty (modified 5-Item Frailty Index and Charlson Comorbidity Score) have not yet been verified as indicators of frailty for complex aneurysm patients specifically, but have been for a number of other patient cohorts.24,25
Conclusions
An analysis of 15 years of complex aneurysm management showed that complex EVAR became the preferred treatment method soon after its introduction. The current selection process seems effective in recognizing the frailest patients and assigning them to complex EVAR, as guidelines propose. Although less invasive compared to OSR when focusing on major complications, complex EVAR still comes with considerable perioperative risks, mortality, and reintervention rates. For some frailest patients, complex EVAR might be a bridge too far. Specific insights in patient selection are warranted, identifying potential indicators of unfavorable outcomes or targets for preoperative optimization. In addition, we propose to include postoperative functioning into analyses regarding surgical outcomes.
Footnotes
Appendix
Type of Aneurysm or Procedure-Related Reinterventions During Follow-up After Open Surgical Repair and Complex Endovascular Aortic Repair (EVAR), With the Time After Surgery Indicated.
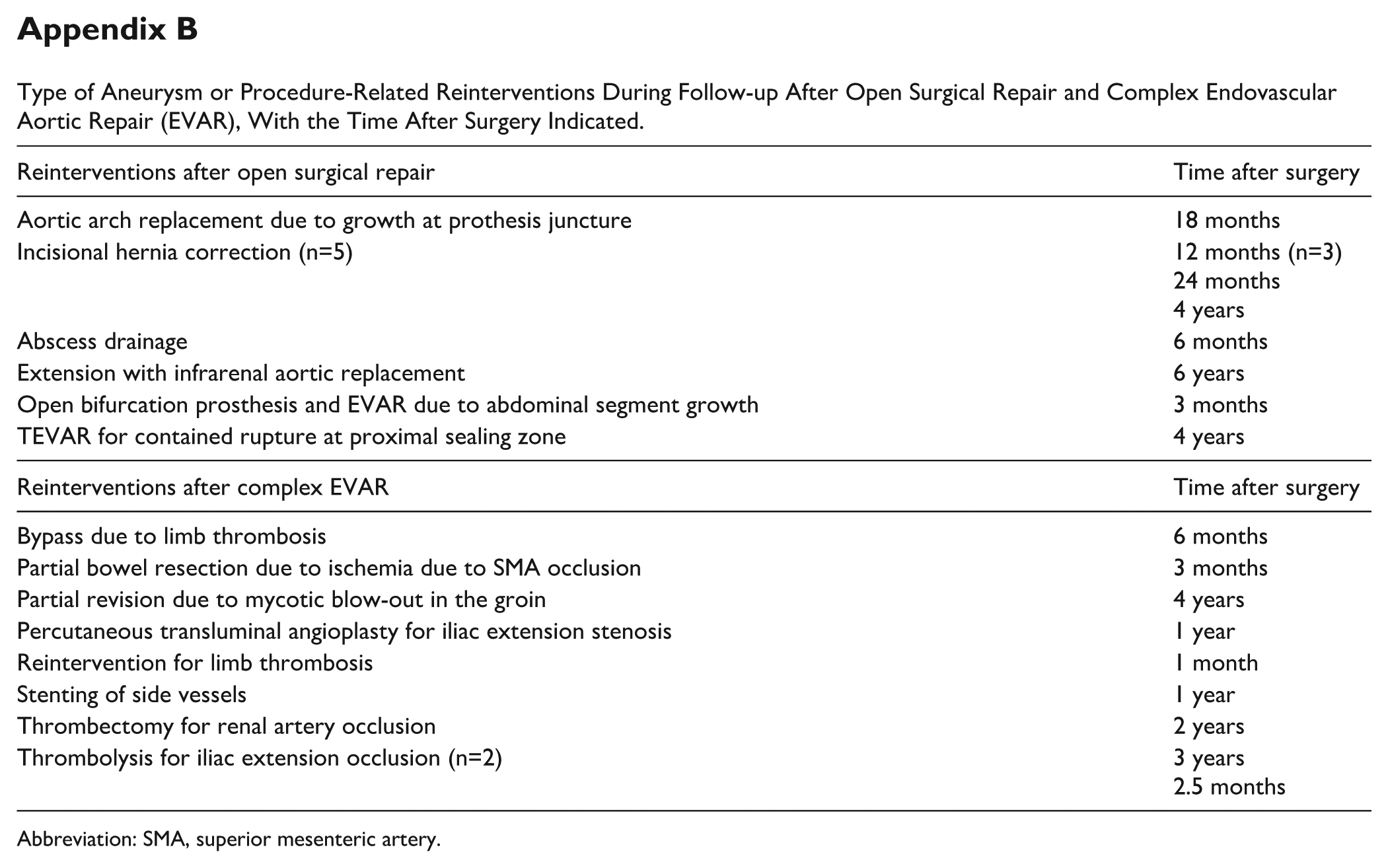
| Reinterventions after open surgical repair | Time after surgery |
|---|---|
| Aortic arch replacement due to growth at prothesis juncture | 18 months |
| Incisional hernia correction (n=5) | 12 months (n=3) 24 months 4 years |
| Abscess drainage | 6 months |
| Extension with infrarenal aortic replacement | 6 years |
| Open bifurcation prosthesis and EVAR due to abdominal segment growth | 3 months |
| TEVAR for contained rupture at proximal sealing zone | 4 years |
| Reinterventions after complex EVAR | Time after surgery |
| Bypass due to limb thrombosis | 6 months |
| Partial bowel resection due to ischemia due to SMA occlusion | 3 months |
| Partial revision due to mycotic blow-out in the groin | 4 years |
| Percutaneous transluminal angioplasty for iliac extension stenosis | 1 year |
| Reintervention for limb thrombosis | 1 month |
| Stenting of side vessels | 1 year |
| Thrombectomy for renal artery occlusion | 2 years |
| Thrombolysis for iliac extension occlusion (n=2) | 3 years 2.5 months |
Abbreviation: SMA, superior mesenteric artery.
Acknowledgements
None.
Author Contributions
Data was gathered and analyzed by authors Warmerdam, Van der Vorst, and Van Schaik. Together with author Hamming, they wrote a first draft of the article. This was then critically analyzed and rewritten using extensive feedback from authors Hjortnaes, Driessen, Eefting, Klautz, Van Rijswijk, and Van der Meer.
Ethical Committee/Ethical Statement/Informed Consent
As is required by Dutch law under the Medical Research Involving Human Subjects Act (WMO), a “non-WMO declaration” was issued by the LUMC Medical Ethics Committee (METC, registration number 2024-010). Due to the merely retrospective character of our study, the anonymized use of data, and the fact that there was no interference in the standard care pathway, explicit patient consent was not obtained, in accordance with Dutch law.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
To protect the patients’ privacy as much as possible, we choose not to share data at this point. Even when anonymized, there is a risk of identification, especially given the relatively small sample size. In addition, explicit patient consent for data sharing was not obtained.
Trial Registration Number
Not applicable.
Grant Number
None.
