Abstract
Objective:
Fenestrated Endovascular Aortic Repair (F-EVAR) using Physician-Modified Endografts (PMEGs) has become an important option in the treatment of complex aortic aneurysms, particularly when custom-made devices are not available. However, difficult target vessel (TV) cannulations, notably with misalignment, often limit total transfemoral repair. To address this condition, we developed the Gap Steering (GPS) technique, which is a transfemoral-only adjunctive method using a steerable sheath to create a controlled gap between the stent graft and the aortic wall, enabling on-demand cannulation. In this study, we aimed to evaluate the technical feasibility and procedural features, outcomes, and risks associated with the GPS technique in difficult target vessel cannulation owing to fenestration misalignment.
Methods:
We retrospectively reviewed 42 patients (14 thoracoabdominal, 28 complex abdominal aortic aneurysms) undergoing F-EVAR using PMEGs with bridging stent placement between October 2020 and January 2025. Although not designed as a comparative study, patients were stratified by treatment period: Pre-GPS technique group (n = 10, TV = 30) and Post-GPS technique group (n = 32, TV = 114). The GPS technique was used as the primary bailout for difficult cannulations in the Post-GPS technique group. Among the 114 TVs, the GPS technique was selectively applied to 18 challenging vessels (15.8%).
Results:
Among 18 target vessels treated with the GPS technique, successful cannulation was achieved in 14 (77.8%), with no GPS technique–related complications. At the per patient level, the Post-GPS technique group showed significantly higher technical success (96.8% vs 60.0%, p=0.008) and a higher target vessel success rate (100% vs 80.0%, p=0.052). TV instability was significantly lower (3.1% vs 30.0%, p=0.03). At the per-vessel level, TV technical success was higher (100% vs 93.3%, p=0.042), with lower rates of TV occlusion (0.0% vs 6.7%, p=0.042) and TV instability (0.9% vs 13.3%, p=0.007). All GPS technique-treated vessels remained patent at the 14 month median follow-up.
Conclusion:
The GPS technique is a feasible adjunct for difficult cannulations in F-EVAR using PMEGs. It avoids additional devices and is particularly suited for total transfemoral repair using currently available steerable sheaths.
Clinical Impact
Difficult renovisceral target vessel cannulation remains a major limitation to achieving total transfemoral F-EVAR using physician-modified endografts (PMEGs). This challenge often necessitates upper-extremity access or complex adjunctive maneuvers, increasing procedural invasiveness and access-related risks. The Gap Steering (GPS) technique offers a practical adjunctive strategy using a commercially available steerable sheath to achieve controlled directional guidewire manipulation at the fenestration–aortic wall interface. It enables selective cannulation without additional access routes, larger delivery systems, or device modification. This reproducible and fully transfemoral approach has the potential to improve procedural efficiency and expand the applicability of PMEG-based F-EVAR in complex aortic repair.
Keywords
Introduction
Endovascular repair of thoracoabdominal aortic aneurysms (TAAAs) and complex abdominal aortic aneurysms (cAAAs) often requires advanced endovascular techniques owing to the involvement of the renovisceral branches. Fenestrated and branched endovascular aortic repair (F/B-EVAR) has emerged as a viable option, demonstrating high technical success rates, low early mortality, and favorable midterm outcomes.1–3 F/B-EVAR is now applied to both TAAAs and cAAAs and has become an essential strategy for the treatment of complex aortic pathology. In elective cases, custom-made devices (CMDs) are preferred; however, CMDs require prolonged manufacturing times and may not be available in all regions. 4 Physician-modified endografts (PMEGs) have steadily gained acceptance as a practical alternative, offering immediate availability and comparable durability.5–8 Although PMEGs have been increasingly used worldwide, they have not yet been formally incorporated into current guideline recommendations, and further validation along with cautious application remains necessary.
A successful fenestrated endovascular aortic repair (F-EVAR) procedure, using either PMEGs or CMDs, requires 2 critical elements: accurate alignment of fenestrations with the target vessels and controlled bridging stent delivery. Misalignment between fenestrations and target vessel ostia remains a significant technical challenge, often complicating subsequent cannulation and delivery procedures.9,10 Previous reports have quantified this challenge, with misalignment reported in approximately 40% of patients in the axial (horizontal) plane and 32.3% in the cranio-caudal (vertical) axis. 9 Once the main graft is fully deployed, it becomes difficult to correct any misalignment before bridging stent completion. Moreover, target vessel cannulation and bridging steps account for the majority of procedural complexity and radiation exposure. 11
Although various adjunctive techniques, such as balloon-assisted techniques, 12 preload systems, 13 and sequential deployment methods, 14 have been developed to facilitate difficult cannulations, these approaches often require additional access routes or increased device complexity. Given the increasing use of the total transfemoral approach to reduce upper-extremity complications, there is a growing need for simpler adjunctive techniques.15,16
To address difficult target vessel cannulation in cases of fenestration misalignment, we developed the Gap Steering (GPS) technique as a less complex, transfemoral-only adjunctive method. This technique was developed and evaluated in the context of F-EVAR using PMEGs, exclusively via the femoral access. It is theoretically applicable to target vessel cannulation during bridging stent delivery in F-EVAR using CMDs. In this study, we aimed to evaluate the technical feasibility and procedural features, outcomes, and risks associated with the GPS technique in difficult target vessel cannulation owing to fenestration misalignment.
Materials and Methods
Study Design and Patient Selection
This study was a retrospective, single-center investigation conducted at a tertiary aortic referral center. The subjects were selected from a total of 82 consecutive patients who underwent F-EVAR using PMEGs with bridging stent placement from October 2020 to January 2025.
Inclusion and Exclusion Criteria
Of the 82 consecutive patients, 22 with aortic dissection were excluded. We also excluded 10 patients who were treated before the introduction of steerable sheath techniques, as well as 7 patients in which a homemade steerable sheath was used, and 1 patient with a ruptured aneurysm. The remaining 42 patients were included in the present analysis.
Patient Stratification and Study Groups
To evaluate the impact of the GPS technique, the 42 included patients were stratified into 2 main study groups based on the institutional adoption timeline of the GPS technique as follows: Pre-GPS technique group and Post-GPS technique group.
Pre-GPS Technique Group (n = 10)
This group comprised the first 10 consecutive patients who underwent F-EVAR with a commercially available steerable sheath before the formal introduction and standardization of the GPS technique as a bailout option for challenging target vessel cannulation. In these initial cases, difficult cannulations were managed through a variety of laborious and technically demanding maneuvers, including microguidewire probing and balloon catheter-assisted redirection, as opposed to the structured approach of the GPS technique.
Post-GPS Technique Group (n = 32)
This group included patients treated after the institutional implementation of the GPS technique as the primary bailout strategy for challenging target vessel cannulations (from September 2023 onward). The specific procedural criteria for employing the GPS technique are described in the “Device Usage and Target Vessels” section.
Aneurysm Classification
Among the 42 included patients, 14 patients were classified as TAAAs and 28 as cAAAs. TAAAs were defined as aneurysms involving both the thoracic aorta and the abdominal aorta, including at least 1 visceral artery, based on the Crawford and modified Safi classifications. cAAAs were defined as infrarenal or juxtarenal aneurysms with short necks or involvement of visceral branches, but without thoracic aortic extension. 17
Device Usage and Target Vessels
In all the 42 patients, commercially available steerable sheaths were employed to facilitate bridging stent placement. A total of 144 target vessels were treated, including the celiac artery, superior mesenteric artery (SMA), and bilateral renal arteries.
The GPS technique was applied to patients where the initial target vessel cannulation, which used a commercially available steerable sheath, a 4 Fr angiographic catheter, and a 0.035 inch hydrophilic guidewire, was unsuccessful. In all cases, misalignment between the fenestration and the aortic wall was the underlying cause of cannulation difficulty. This anatomical mismatch led to 2 main scenarios as follows: (1) the guidewire could not be advanced into the aneurysmal sac; (2) the subsequent catheterization of the target vessel was hindered by an unfavorable takeoff angle or limited maneuverability even though the guidewire could successfully enter the sac. Under these circumstances, within the post-GPS technique group, the GPS technique was used as the primary bailout strategy to create a controlled space between the stent graft and the aortic wall, thereby facilitating redirection of the guidewire toward the target artery. Clinical data were obtained from electronic medical records reviewed using surgical records and procedural videos.
Procedure Details
Figure 1A–F illustrates the procedural steps of the GPS technique. The procedure was performed in a hybrid operating room under general anesthesia using bilateral femoral artery cut-downs. Surgical cut-down was performed to secure swift additional perfusion if necessary. Bridging stent graft placement was managed by a single interventional radiologist who specializes in vascular interventional radiology. All other procedural steps were performed by a cardiovascular surgeon experienced in aortic stent grafting. A total transfemoral approach was used in all cases except 1, in which an open iliac artery approach was performed.
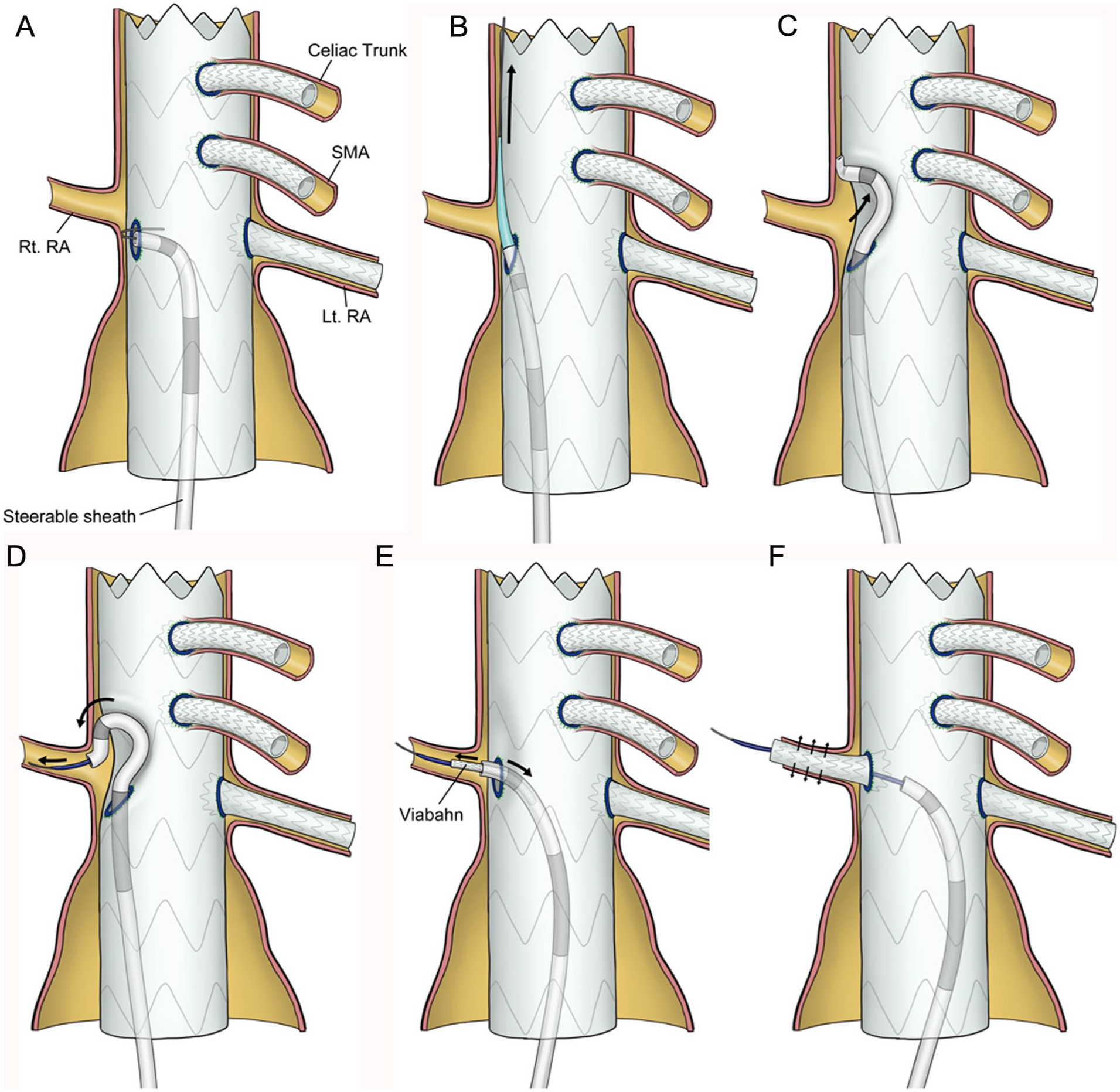
Schematic Diagram of Gap Steering (GPS) Technique. (A) The fenestration is misaligned, causing the guidewire to come in contact with the aortic wall and preventing further advancement. (B) The steerable sheath is inserted into the gap outside the stent graft using the stiff guidewire and inner dilator. (C) Steering in the gap, the steerable sheath is maneuvered to create an additional space. (D) The guidewire is advanced into the target vessel. (E) A bridging stent is inserted. (F) The bridging stent is deployed. Lt, left; RA, renal artery; Rt, right; SMA, superior mesenteric artery.
The commercially available steerable sheath used was Agilis NxT S (Abbott Laboratories, USA; length, 60 cm). The fenestrated stent graft used was the Zenith Alpha Thoracic (Cook Medical, USA). For bridging stents, the self-expanding GORE VIABAHN Endoprosthesis (W. L. Gore & Associates, Flagstaff, AZ, USA) was primarily used, whereas the balloon-expandable GORE VIABAHN VBX Balloon Expandable Endoprosthesis (W. L. Gore & Associates, Flagstaff, AZ, USA) was applied selectively. In the initial phase of this study, VBX stents were the main devices used; however, following an incident of branch perfusion compromise owing to VBX deformation, our institutional strategy was revised to favor Viabahn as the primary choice. VBX devices were thereafter used only when a greater radial force was required at the fenestration site, particularly in cases of suspected type IIIc endoleak. The femoral artery sheaths were 20 to 22 Fr for the main stent graft side and 12 to 14 Fr DrySeal Flex Introducer Sheath (W. L. Gore & Associates, Flagstaff, AZ, USA) for the contralateral side.
The position of the fenestrated stent graft was planned using three-dimensional computed tomography angiography (3D CTA) with 1 mm slice data and the 3mensio workstation (Pie Medical Imaging, the Netherlands), with fine adjustments to the fenestration site made intraoperatively. Upper-extremity sheath insertion was avoided except in cases requiring a pull-through wire between the brachial and femoral arteries. Lower extremity perfusion was supplemented as necessary. From the contralateral femoral artery sheath, a 5 Fr hook-shaped angiographic catheter (Type SU; Hanako Medical, Tokyo, Japan; length, 80 cm) and a 0.035 inch guidewire (Radifocus Guidewire M; Terumo Corporation, Tokyo, Japan; length, 260 cm) were advanced to the SMA, whereas the other branches were not presecured. The main stent graft was deployed cautiously using fusion images created from 3D CTA and fluoroscopic findings. Intraoperative cone beam computed tomography (CBCT) was not used. After aortography, bridging stents were inserted sequentially into each fenestration. The steerable sheath was first used to engage the fenestration, visualize its relationship with the target vessel, and attempt guidewire advancement using a 4 Fr angiographic catheter (TEMPO Angiographic Catheter, Cordis, FL, USA; length, 125 cm) and a 0.035 inch guidewire. A 0.014 inch microguidewire (Gradius MG, Asahi Intec, Aichi, Japan; length 300 cm) and a 1.6 Fr microcatheter (Prominent BTA, Tokai Medical Products, Aichi, Japan; length 150 cm) were used as needed.
If target vessel cannulation proved difficult owing to misalignment between the fenestration and the target vessel ostium, the GPS technique was used. The timing of its adoption was determined by the operator based on the fenestration–target vessel relationship and elapsed time. Difficult situations usually arose when the stent graft and branch vessels were misaligned, resulting in malpositioned fenestrations (Figure 1A). When the fenestration was flush with the aortic wall, making direct selection of the target vessel ostium impossible, the same microguidewire was used to carefully probe the gap between the fenestration and the aortic wall. The microguidewire and microcatheter were then advanced cranially outside the stent graft, followed by an angiographic catheter over the same microguidewire and microcatheter. The guidewire was then switched to a 0.035 inch guidewire stiff type (Radifocus Guidewire M stiff type, Terumo Corporation, Japan; length, 260 cm), and the steerable sheath was advanced outside the stent graft using its inner dilator (Figure 1B). The fluoroscopic angle was carefully selected to avoid interference with already deployed bridging stents during manipulation. The steerable sheath tip was angulated in the gap, directed toward the origin of the target vessel, and retracted while rotating toward the branch (Figure 1C). After the guidewire successfully cannulated the target vessel (Figure 1D), the bridging stent was delivered. Guiding catheters were not used. Once the bridging stent was delivered, the curved portion of the steerable sheath within the gap was retracted into the stent graft and gradually unsheathed (Figure 1E). The bridging stent was then deployed sequentially (Figure 1F). After complete deployment, the fenestration was dilated with either the Metacross RX PTA Balloon Dilatation Catheter (Terumo Corporation, Japan) or the Senri PTA Balloon Dilatation Catheter (Terumo Corporation, Japan), and the result was confirmed by angiography. Following the procedural description, an illustrative case was provided to demonstrate the clinical application of the GPS technique. A representative clinical application is illustrated in Figure 2. The fluoroscopic images demonstrate the sequential steps, including steerable sheath angulation within the gap (Figure 2A), target vessel cannulation (Figure 2B), bridging stent delivery (Figure 2C), gradual straightening and retraction of the angulated steerable sheath into the stent graft (Figure 2D), and full deployment (Figure 2E). Preoperative and postoperative three-dimensional computed tomography (3D CT) scans (Figure 2F and G, respectively) confirmed successful aneurysm exclusion and preservation of the renovisceral branches.
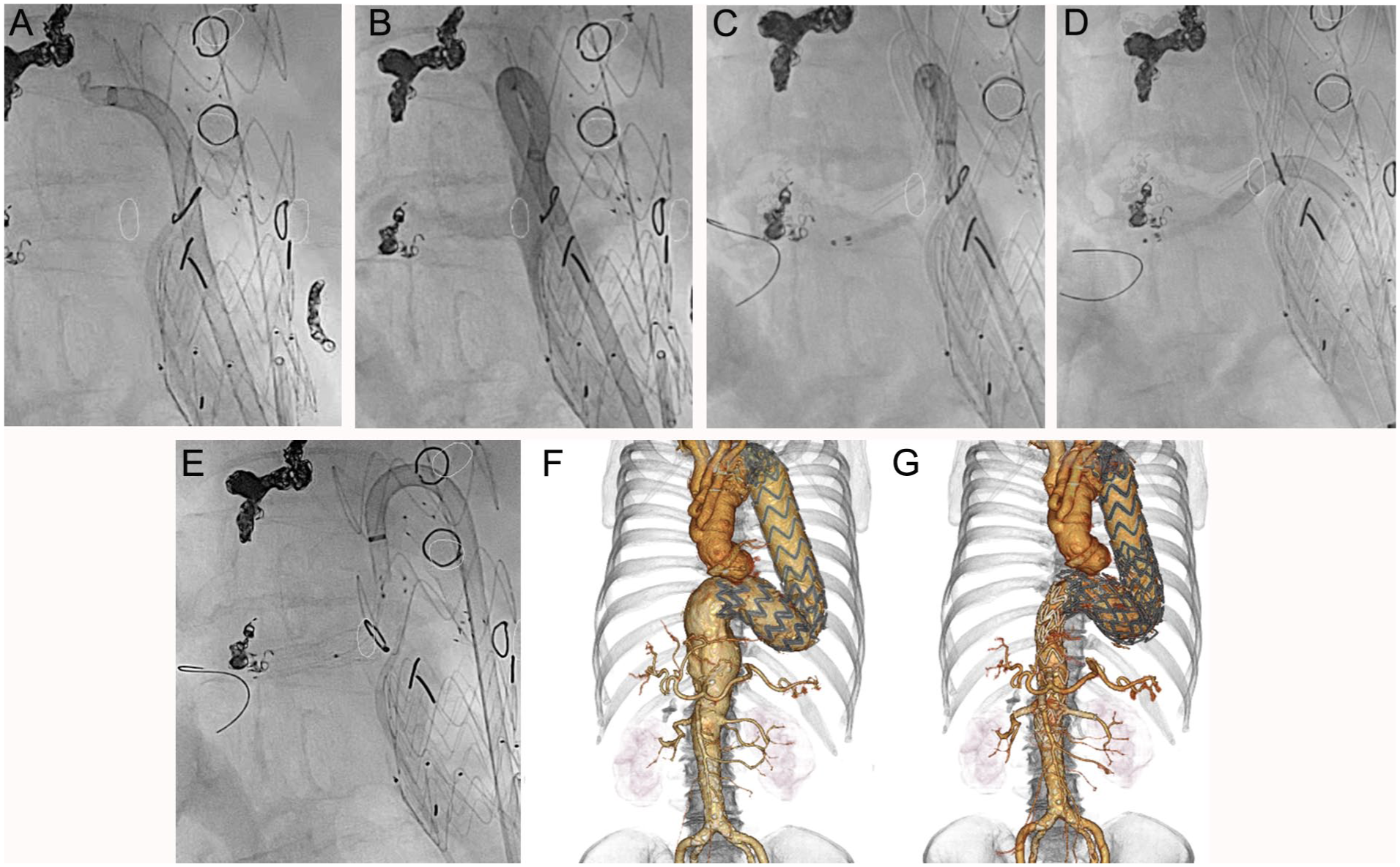
A case using gap steering technique. *The white circles in the figure indicate the positions of the target vessel ostium, which were determined from the fusion images and displayed on the fluoroscopic screen. (A) Steering is achieved by angulating the steerable sheath within the narrow aortic space beyond the fenestration to create an additional space. (B) After confirming the contrast flow and branch anatomy through the steerable sheath, cannulation is performed. (C) A bridging stent is placed over the guidewire. (D) The angulated steerable sheath is gradually straightened and retracted into the stent graft. (E) The bridging stent is fully deployed after withdrawal of the steerable sheath. (F) The preoperative 3D CT showed that the thoracoabdominal aneurysm extended to the renovisceral branches. (G) The postoperative 3D CT scan demonstrated that the fenestrated and bridging stent graft successfully covered the aneurysm while preserving the visceral branches of the aorta. 3D CT, three-dimensional computed tomography.
Endpoints
The primary endpoints were technical success and target vessel technical success, both defined according to the Society for Vascular Surgery (SVS) reporting standards. Technical success was defined as meeting all of the following criteria: (1) successful deployment of the fenestrated stent graft at the intended position; (2) successful cannulation and bridging of all target vessels; (3) absence of type I or III endoleaks on completion angiography and on follow-up imaging within 30 days; and (4) no conversion to open surgery. Target vessel technical success was defined by successful catheterization and stent placement in all intended target vessels. 17
The secondary endpoints were categorized and assessed in alignment with the SVS reporting standards for endovascular aortic repair. 17
Intraoperative parameters included total operating time, fluoroscopy time, contrast volume, target vessel bridging time, and the need for intraoperative blood transfusion.
Intraoperative adverse events included target vessel injury and access-related complications.
Early clinical outcomes included 30 day mortality, major adverse events (MAEs), stroke, spinal cord ischemia, myocardial infarction, new-onset dialysis, ICU length of stay, and hospital length of stay.
Device-related events included type I or III endoleak during follow-up, endograft migration >10 mm, target vessel instability (defined as bridging stent migration, stenosis, or kinking), and target vessel patency at 30 days.
Reintervention was evaluated as freedom from reintervention at 30 days.
In addition to evaluating the primary endpoints, branch-specific metrics were also assessed to provide a more comprehensive procedural analysis. These included GPS technique–related complications, total target vessel bridging success rate, and target vessel bridging time. Branch-related complications such as dissection, occlusion, endoleak, and stent dislodgement were documented. Intraoperative and postoperative branch–related reinterventions were also evaluated. Patency was assessed based on imaging obtained at 30 days after the procedure.
There was no directly comparable technique in this study, as the GPS technique was uniquely applied as a salvage method when initial target vessel cannulation using a steerable sheath and a 4 Fr angiographic catheter with a 0.035 inch hydrophilic guidewire proved unsuccessful. Accordingly, the technical success of the GPS technique was defined as successful bridging of misaligned target vessels without the need for access conversion or additional bailout maneuvers.
Statistical Analysis
Data were analyzed using SPSS version 30 (IBM Corp., Armonk, NY, USA). Patients were compared between the pre-GPS technique group and the post-GPS technique group. Comparisons were also conducted on a per-target vessel basis. Additionally, the target vessels in which the GPS technique was successful were compared with the target vessels in which the GPS technique was not successful. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed using the Mann–Whitney U test. A p-value of < 0.05 was considered to indicate a statistically significant difference.
Results
Patient Characteristics
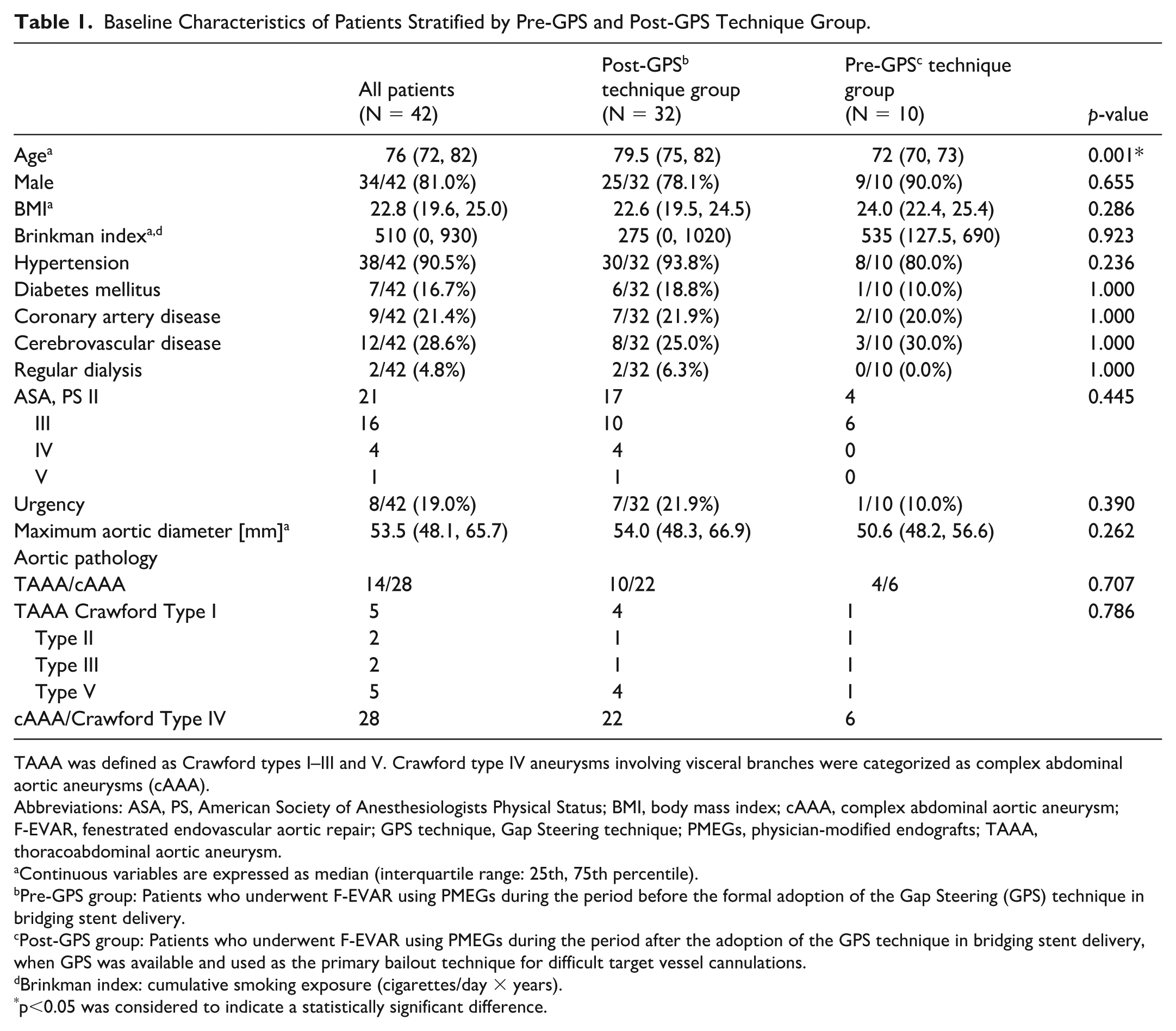
A summary of the baseline characteristics of all 42 patients, stratified by Pre-GPS and Post-GPS technique groups, is presented in Table 1. The cohort was stratified based on whether the GPS technique had been adopted at the time of treatment as follows: the Post-GPS technique group (n = 32), in which the GPS technique was available and selectively used as a bailout method for difficult target vessel cannulation; the Pre-GPS technique group (n = 10), in which PMEG was used before the formal adoption of the GPS technique.
Baseline Characteristics of Patients Stratified by Pre-GPS and Post-GPS Technique Group.
TAAA was defined as Crawford types I–III and V. Crawford type IV aneurysms involving visceral branches were categorized as complex abdominal aortic aneurysms (cAAA).
Abbreviations: ASA, PS, American Society of Anesthesiologists Physical Status; BMI, body mass index; cAAA, complex abdominal aortic aneurysm; F-EVAR, fenestrated endovascular aortic repair; GPS technique, Gap Steering technique; PMEGs, physician-modified endografts; TAAA, thoracoabdominal aortic aneurysm.
Continuous variables are expressed as median (interquartile range: 25th, 75th percentile).
Pre-GPS group: Patients who underwent F-EVAR using PMEGs during the period before the formal adoption of the Gap Steering (GPS) technique in bridging stent delivery.
Post-GPS group: Patients who underwent F-EVAR using PMEGs during the period after the adoption of the GPS technique in bridging stent delivery, when GPS was available and used as the primary bailout technique for difficult target vessel cannulations.
Brinkman index: cumulative smoking exposure (cigarettes/day × years).
p<0.05 was considered to indicate a statistically significant difference.
Regarding patient demographics, the median age was significantly higher in the Post-GPS technique group than in the Pre-GPS technique group (79.5 vs 72 years, p=0.001). Other baseline characteristics, including male percentage, body mass index (BMI), Brinkman index, prevalence of major comorbidities (hypertension, diabetes mellitus, coronary artery disease, cerebrovascular disease, regular dialysis), American Society of Anesthesiologists Physical Status classification (ASA-PS) classifications, procedural urgency, and median maximum aortic diameter, were comparable between the 2 groups.
The distribution of aortic pathology (TAAA vs cAAA) was not significantly different between the Post-GPS technique group (TAAA 10, cAAA 22) and the Pre-GPS technique group (TAAA 4, cAAA 6). Within the TAAA subgroup, the distribution across Crawford types was comparable between the 2 groups.
Primary Endpoints (Per Patient Analysis)
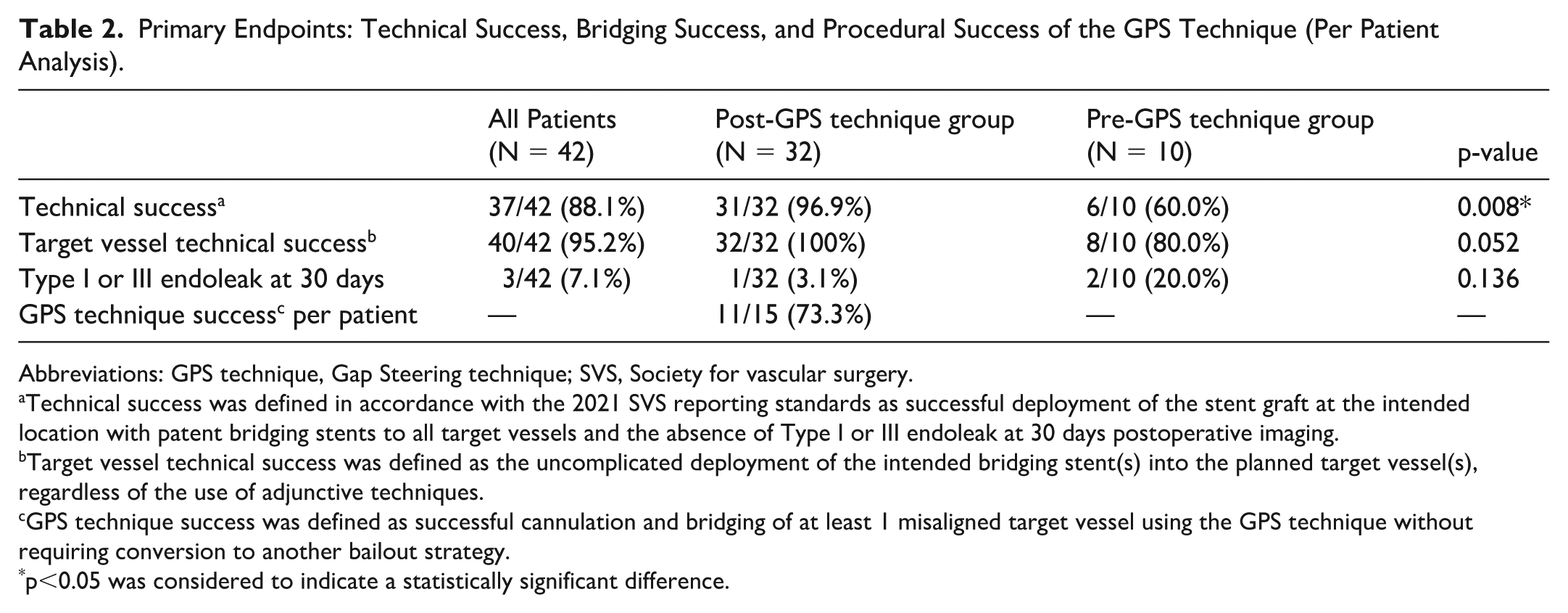
The results for the primary endpoints are summarized in Table 2. The overall technical success rate for the entire cohort was 88.1% (37/42). Technical success, which is defined according to the 2021 SVS reporting standards, was significantly higher in the Post-GPS technique group (96.9%, 31/32) than in the Pre-GPS technique group (60.0%, 6/10; p=0.008). The target vessel technical success rate was high overall (95.2%, 40/42). Although numerically higher in the Post-GPS technique group (100%, 32/32) than in the Pre-GPS technique group (80.0%, 8/10), this difference was not significant (p=0.052). The incidence of type I or III endoleak at completion or on 30 day CTA was comparable between the 2 groups. Among patients in whom the GPS technique was attempted (n = 15), procedural success was achieved in 73.3% (11/15).
Primary Endpoints: Technical Success, Bridging Success, and Procedural Success of the GPS Technique (Per Patient Analysis).
Abbreviations: GPS technique, Gap Steering technique; SVS, Society for vascular surgery.
Technical success was defined in accordance with the 2021 SVS reporting standards as successful deployment of the stent graft at the intended location with patent bridging stents to all target vessels and the absence of Type I or III endoleak at 30 days postoperative imaging.
Target vessel technical success was defined as the uncomplicated deployment of the intended bridging stent(s) into the planned target vessel(s), regardless of the use of adjunctive techniques.
GPS technique success was defined as successful cannulation and bridging of at least 1 misaligned target vessel using the GPS technique without requiring conversion to another bailout strategy.
p<0.05 was considered to indicate a statistically significant difference.
Secondary Endpoints (Per Patient Analysis)
The secondary endpoints, including intraoperative parameters, early clinical outcomes, and device-related events, are summarized in Table 3.
Secondary Endpoints: Intraoperative Metrics, Early Clinical Outcomes, and Device-Related Events (Per Patient Analysis).
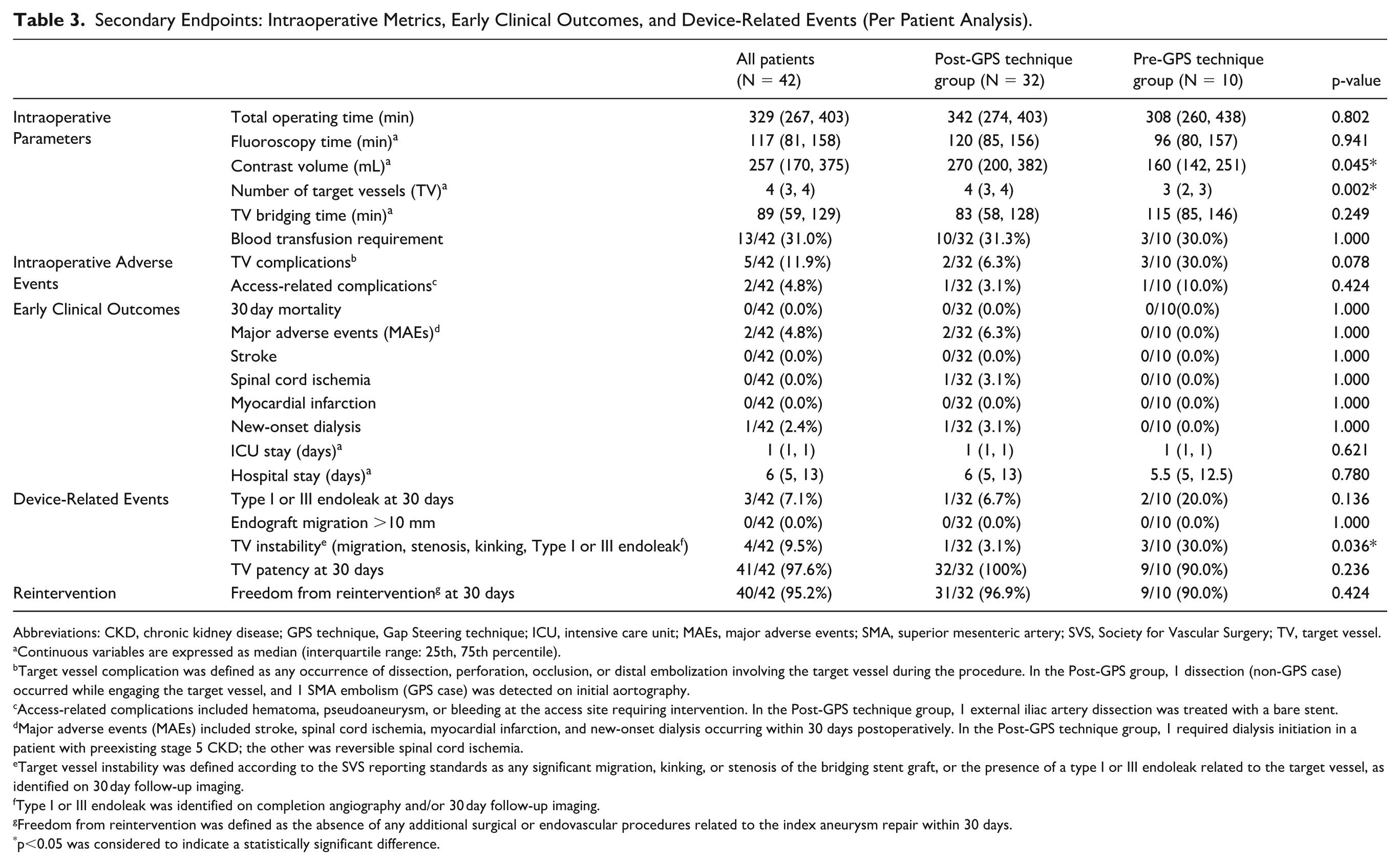
Abbreviations: CKD, chronic kidney disease; GPS technique, Gap Steering technique; ICU, intensive care unit; MAEs, major adverse events; SMA, superior mesenteric artery; SVS, Society for Vascular Surgery; TV, target vessel.
Continuous variables are expressed as median (interquartile range: 25th, 75th percentile).
Target vessel complication was defined as any occurrence of dissection, perforation, occlusion, or distal embolization involving the target vessel during the procedure. In the Post-GPS group, 1 dissection (non-GPS case) occurred while engaging the target vessel, and 1 SMA embolism (GPS case) was detected on initial aortography.
Access-related complications included hematoma, pseudoaneurysm, or bleeding at the access site requiring intervention. In the Post-GPS technique group, 1 external iliac artery dissection was treated with a bare stent.
Major adverse events (MAEs) included stroke, spinal cord ischemia, myocardial infarction, and new-onset dialysis occurring within 30 days postoperatively. In the Post-GPS technique group, 1 required dialysis initiation in a patient with preexisting stage 5 CKD; the other was reversible spinal cord ischemia.
Target vessel instability was defined according to the SVS reporting standards as any significant migration, kinking, or stenosis of the bridging stent graft, or the presence of a type I or III endoleak related to the target vessel, as identified on 30 day follow-up imaging.
Type I or III endoleak was identified on completion angiography and/or 30 day follow-up imaging.
Freedom from reintervention was defined as the absence of any additional surgical or endovascular procedures related to the index aneurysm repair within 30 days.
p<0.05 was considered to indicate a statistically significant difference.
For the intraoperative parameters, the total operating time, fluoroscopy time, and target vessel bridging time were comparable between the 2 groups. However, the contrast volume was significantly higher in the Post-GPS technique group (median 270 vs 160 mL, p=0.045). The median number of target vessels bridged was also significantly greater in the Post-GPS technique group than in the Pre-GPS technique group (4 vs 3, p=0.002).
Adverse Events and Device-Related Outcomes
Intraoperative adverse events, early clinical outcomes (including 30 day mortality and major adverse events), and freedom from reintervention at 30 days were comparable between the 2 groups. Type I or III endoleak and endograft migration were also comparable. Notably, target vessel instability was significantly lower in the Post-GPS technique group (3.1%, 1/32) than in the Pre-GPS technique group (30.0%, 3/10; p=0.036). The overall target vessel patency at 30 days remained high and comparable.
Target Vessel Outcomes (Per-Vessel Analysis)
The per-vessel outcomes of the 144 target vessels are shown in Table 4. The use of the VBX stent was significantly higher in the Pre-GPS technique group than in the Post-GPS technique group, reflecting differing institutional strategies (p<0.001). The GPS technique was employed in 18 of the 114 target vessels (15.8%) in the Post-GPS technique group, with no technique-related complications observed. The target vessel technical success rate per vessel was significantly higher in the Post-GPS technique group (114/114; 100%) than in the Pre-GPS technique group (28/30; 93.3%; p=0.042).
Variables and Results of Bridging Stent Delivery, and Target Vessel Specific Data (Per-Vessel Analysis).
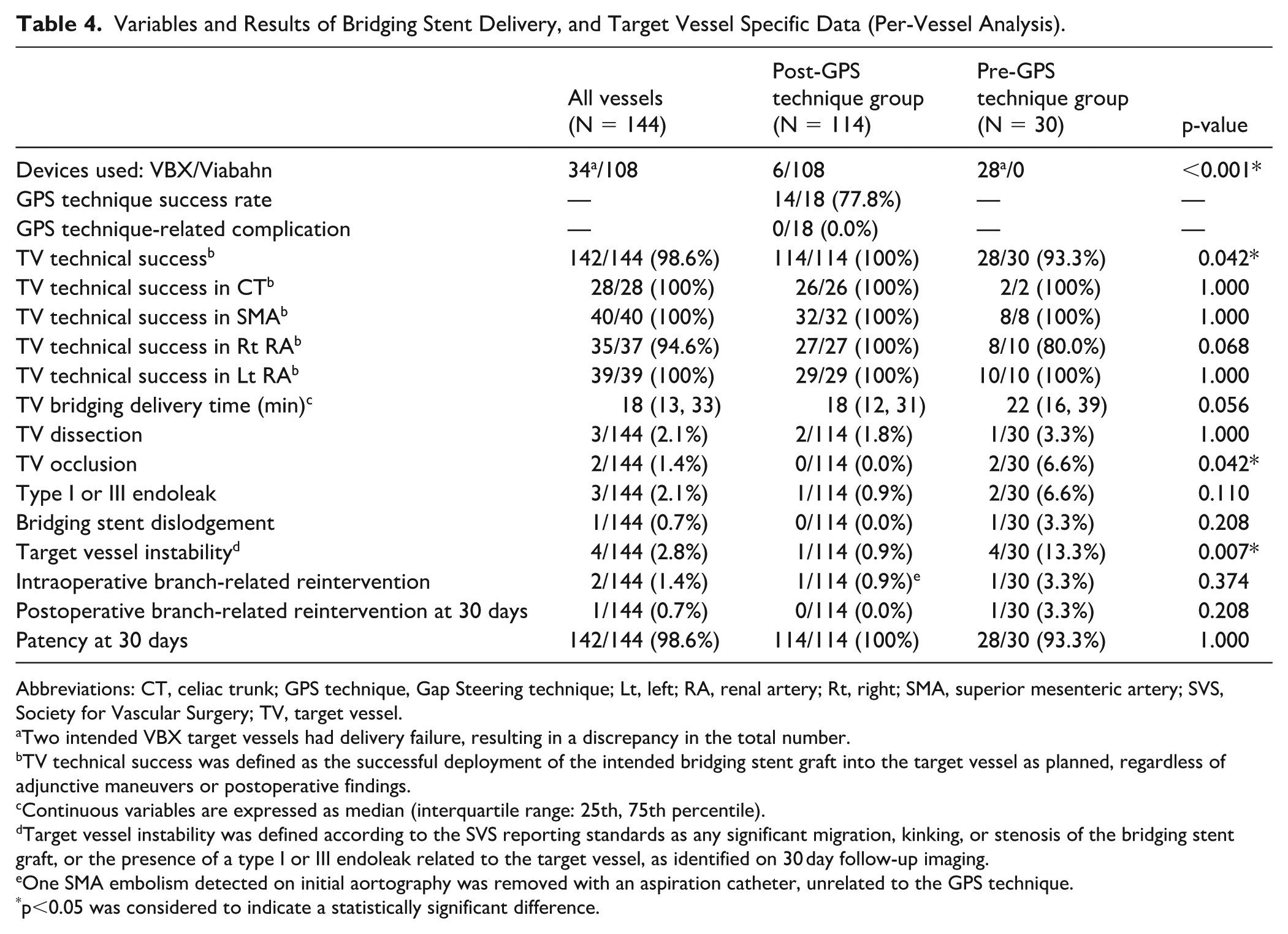
Abbreviations: CT, celiac trunk; GPS technique, Gap Steering technique; Lt, left; RA, renal artery; Rt, right; SMA, superior mesenteric artery; SVS, Society for Vascular Surgery; TV, target vessel.
Two intended VBX target vessels had delivery failure, resulting in a discrepancy in the total number.
TV technical success was defined as the successful deployment of the intended bridging stent graft into the target vessel as planned, regardless of adjunctive maneuvers or postoperative findings.
Continuous variables are expressed as median (interquartile range: 25th, 75th percentile).
Target vessel instability was defined according to the SVS reporting standards as any significant migration, kinking, or stenosis of the bridging stent graft, or the presence of a type I or III endoleak related to the target vessel, as identified on 30 day follow-up imaging.
One SMA embolism detected on initial aortography was removed with an aspiration catheter, unrelated to the GPS technique.
p<0.05 was considered to indicate a statistically significant difference.
The target vessel technical success rates for the celiac trunk (CT), superior mesenteric artery (SMA), and left renal artery (Lt RA) were uniformly 100% in both the Pre- and Post-GPS technique groups. For the right renal artery (Rt RA), intraoperative bridging success was slightly lower in the Pre-GPS technique group (8/10; 80.0%) than in the Post-GPS technique group (27/27; 100%), although the difference was not significant (p=0.068). The median target vessel bridging delivery time was comparable. Regarding per-vessel complications, the rates of target vessel dissection, type I or III endoleak, bridging stent dislodgement, and reintervention were not significantly different between the 2 groups. Importantly, target vessel occlusion occurred in 0/114 vessels (0.0%) in the Post-GPS technique group compared with 2/30 (6.7%) in the Pre-GPS technique group (p=0.042). Target vessel instability was also significantly lower in the Post-GPS technique group (1/114; 0.88%) than in the Pre-GPS technique group (4/30; 13.3%; p=0.007). Patency at 30 days remained high and comparable in both groups.
Procedural Performance of the GPS Technique
The GPS technique was attempted in 18 of the 114 target vessels (15.8%) within the Post-GPS technique group. As detailed in Table 5, the GPS technique achieved an overall success rate of 77.8% (14/18). Successful cannulation was achieved in all the ventral branches: CT (3/3) and SMA (2/2). All 4 failures occurred in the lateral branches—Rt RA (8/9) and Lt RA (1/4), where the values represent successful cannulations per attempted cases. Procedural timing metrics are also summarized therein. Although the time required for branch engagement, guidewire cannulation, and initiation of the GPS technique was similar between successful and failed cases, the duration of the GPS technique was significantly longer in successful cases (median 20 min) than in failed cases (median 5 min; p=0.004). This likely reflects the procedural sequence, as successful GPS cases progressed through stent delivery and balloon expansion, whereas failed GPS attempts were often abandoned early owing to inability to access the gap or advance the steerable sheath safely. Importantly, no GPS technique-related complications, such as target vessel dissection, occlusion, or bridging stent dislodgement, occurred. Although 1 type I or III endoleak and 1 reintervention were recorded among the GPS-attempted vessels, neither was directly attributed to the GPS technique, and patency was maintained in all 18 vessels at follow-up.
Variables and Results of the GPS Technique Branch Specific Data (Per-Vessel Analysis).
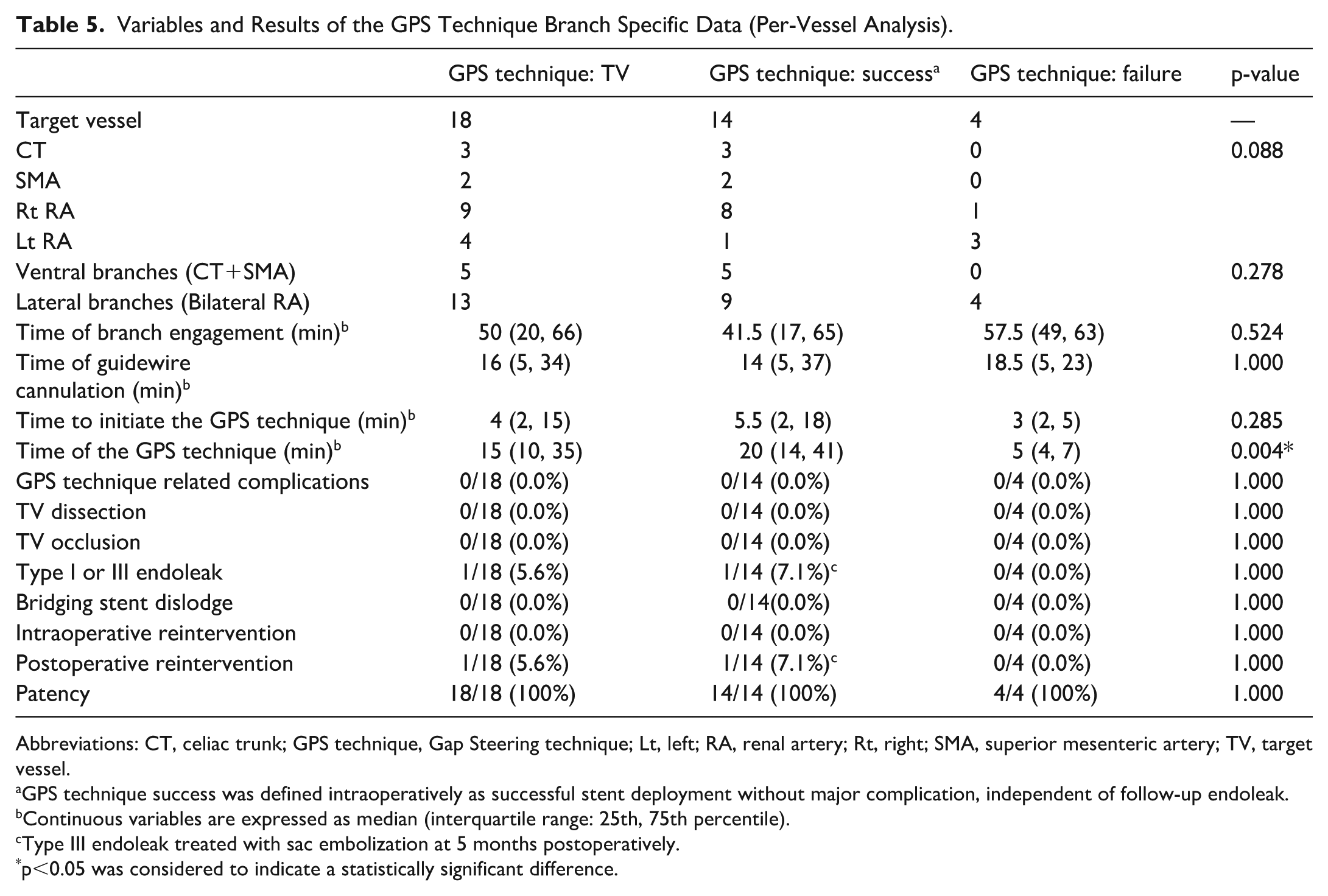
Abbreviations: CT, celiac trunk; GPS technique, Gap Steering technique; Lt, left; RA, renal artery; Rt, right; SMA, superior mesenteric artery; TV, target vessel.
GPS technique success was defined intraoperatively as successful stent deployment without major complication, independent of follow-up endoleak.
Continuous variables are expressed as median (interquartile range: 25th, 75th percentile).
Type III endoleak treated with sac embolization at 5 months postoperatively.
p<0.05 was considered to indicate a statistically significant difference.
Outcomes in GPS Technique Failures
Detailed outcomes for the 4 target vessels where the GPS technique failed are presented in Table 6. All failures occurred in renal arteries (1 right, 3 left). The possible causes for the failure included a near-perpendicular sac entry angle to the aortic wall, passage restriction owing to a preexisting suprarenal bare stent, and aortic sac access limitation owing to aortic curvature. Despite the initial GPS failure, all 4 target vessels were ultimately successfully cannulated and bridged. Subsequent management of these failures involved various adjunctive maneuvers such as looped microguidewire trajectory with balloon anchoring, or microguidewire-assisted selective cannulation with prolonged effort. Importantly, no postoperative reinterventions or endoleaks were observed in these vessels, and patency was maintained in all 4 vessels at follow-up. This demonstrates that although the GPS technique may not be universally successful, it does not preclude successful bridging via alternative means, and it does not compromise the ultimate outcome.
Branches With the GPS Technique Failure and Related Factors (Per-Vessel Analysis).
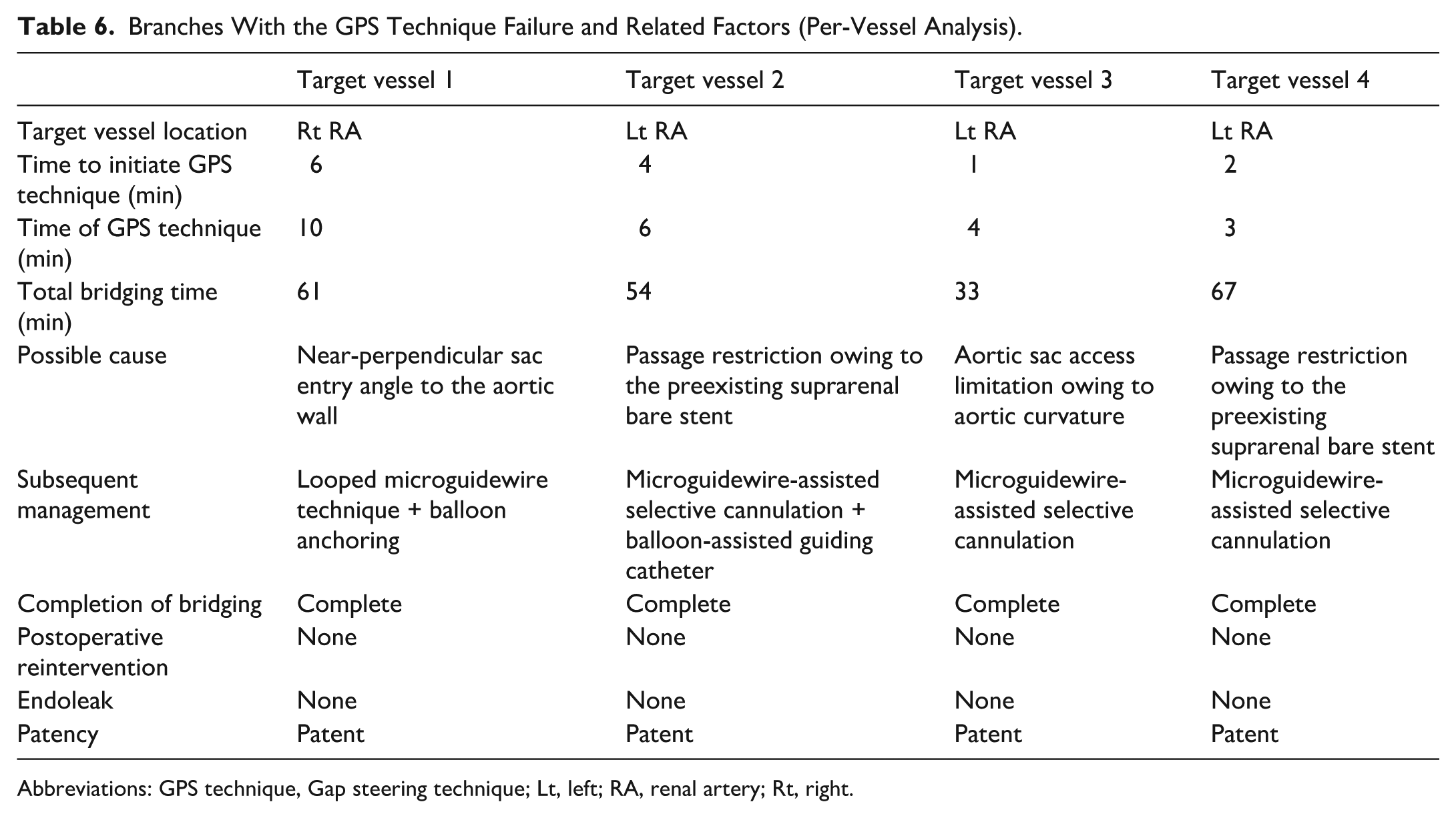
Abbreviations: GPS technique, Gap steering technique; Lt, left; RA, renal artery; Rt, right.
Discussion
We comprehensively evaluated the technical feasibility and procedural features, outcomes, and risks associated with the GPS technique in difficult target vessel cannulation owing to fenestration misalignment. The GPS technique is a novel transfemoral-only adjunctive method aimed at challenging renovisceral target vessel cannulation during F-EVAR using PMEGs. We selectively applied the GPS technique to 18 out of 144 target vessels across the 42 patients when guidewire advancement using the initial approach with an angiographic catheter and steerable sheath was unsuccessful. The GPS technique achieved technical success in 77.8% (14/18) of the attempted vessels without any direct procedure-related complications. Crucially, all intended bridging stents were ultimately placed successfully in the target vessels where the GPS technique was attempted. The absence of a significant difference in target vessel bridging time per patient may have been influenced by the significantly higher number of bridged vessels in the post-GPS technique group. In the per-vessel analysis, although not reaching statistical significance (p=0.056), there was a trend toward reduced bridging time. However, potential confounding factors such as the operator’s learning curve cannot be entirely excluded. Importantly, the application of the GPS technique did not result in higher rates of early complications, endoleak, or reintervention compared with cases where the GPS technique was not used. These findings underscore the safety and effectiveness of the GPS technique as an adjunctive strategy for intricate cannulation scenarios in F-EVAR.
Although F-EVAR using PMEGs has demonstrated high technical success rates, typically exceeding 90% in existing literature,5–8 a small but persistent percentage of cases still encounter significant challenges during bridging stent delivery. To facilitate bridging stent delivery, one possible strategy is to deliberately increase the distance between the fenestration and the target vessel ostium during preoperative planning. However, this approach must be carefully balanced, as previous reports have shown that a gap of >5 mm is associated with a higher risk of target vessel instability. 18 Consequently, current best practices emphasize minimizing the fenestration-to-ostium distance as much as possible. This anatomical proximity, although beneficial for long-term stability, inherently increases the technical difficulty when fenestrations are even slightly misaligned, creating a need for effective bailout strategies. Although not specifically targeting F-EVAR using PMEGs, a range of adjunctive techniques has been described to overcome this difficulty. Examples include balloon-assisted maneuvers,12,19,20 the use of preloaded systems, 13 sequential deployment strategies such as the sequential catheterization amid progressive endograft deployment (SCAPED) technique, 14 diameter-reducing techniques, 21 in situ fenestration,22–24 and techniques employing snare catheters.25–27 However, many of these approaches require additional access routes, such as upper-extremity access, which may increase the risk of complications such as stroke or upper-extremity ischemia, or involve more complex device configurations and extensive preprocedural planning. In contrast to adjuncts that require additional access and greater device complexity, steerable-sheath–based transfemoral approaches demonstrate high success rates28,29 while avoiding the known risks of upper-extremity access,15,16 supporting our continued preference for this strategy.
Our study uniquely presents the GPS technique as a practical adjunctive bailout option for managing misaligned fenestrations, with a focus on its selective, on-demand use and its potential to facilitate a total transfemoral approach in difficult scenarios. Unlike many published adjunctive techniques that primarily describe methods to achieve cannulation, this study quantifies the specific success rate (77.8%) of a particular bailout maneuver for difficult fenestrated cannulations, which is a valuable and core contribution of our study to the literature.
The GPS technique, leveraging a commercially available steerable sheath, offers distinct advantages. It provides real-time, multidirectional control (cranio-caudal, axial plane angulation, and tip deflection) through a single femoral access point, eliminating the need to reorient the main stent graft or utilize additional large-bore sheaths. This streamlined approach not only minimizes procedural complexity and potential access-related complications but also aligns with the growing trend toward fully transfemoral F/B-EVAR workflows. 15
Anatomical Considerations of the GPS Technique
Despite its beneficial uses, the effectiveness of the GPS technique is not universal. In our series, of the 18 attempted vessels (22.2%), 4 vessels predominantly involving the renal arteries (3 left, 1 right) showed failures. These failures were primarily attributed to challenging anatomical factors such as severe aortic angulation, interference from preexisting suprarenal bridging stents, or unfavorable fenestration-to-ostium trajectories. Such conditions likely limited the ability of the steerable sheath to create and maintain an adequate space or angle for guidewire advancement.
It is worth noting that in cases of the GPS technique failure, the attempt was often discontinued early (median 5 min). This prompt discontinuation allowed for the timely adoption of alternative adjunctive maneuvers, such as looped microguidewire techniques or balloon anchoring, which ultimately ensured successful bridging stent placement in all 4 failed cases without additional complications or reinterventions. This emphasizes the importance of operator judgment and adaptability when encountering resistance with the GPS technique. Preoperative imaging assessment remains crucial for identifying anatomical features that may limit the efficacy of the GPS technique and for planning alternative strategies. Future refinements, such as maintaining a stiff support guidewire (eg, Lunderquist Extra-Stiff Wire Guide [Cook Medical, USA]) parallel to the steerable sheath as it exits the fenestration, could potentially improve procedural stability by preventing the sheath from becoming excessively perpendicular to the aortic wall.
Safety Profile and Potential Pitfalls
The GPS technique demonstrated a favorable safety profile in our cohort, with no directly attributable intraoperative complications or increased rates of early adverse events, endoleaks, or reinterventions. However, caution is essential during steerable sheath manipulation. Although no bridging stent dislodgement occurred in this specific series, our prior institutional experience includes 1 case out of 6 branches (a renal artery) in a dissecting aneurysm where a previously deployed stent was inadvertently displaced during the application of the GPS technique, resulting in a type IIIc endoleak requiring further intervention. This underscores the critical importance of avoiding contact between the steerable sheath and already deployed stents.
Furthermore, there is a theoretical risk of target vessel embolization because of the potential intimal injury or disruption of the aortic plaque during retrograde sheath advancement. In our study, one instance of SMA embolism was observed before main body deployment. This event occurred during initial catheter manipulation and large-bore sheath placement, rather than during steerable sheath navigation for the GPS technique. Pathological examination of the retrieved embolic material revealed detached intima rather than thrombus, suggesting an aortic wall injury as the underlying cause. This finding reinforces the importance of careful manipulation to minimize iatrogenic aortic wall injury during complex endovascular procedures. Although the soft, atraumatic tip of modern steerable sheaths is designed to mitigate such risks, meticulous device manipulation and careful fluoroscopic guidance are paramount to ensure the safe and effective application of the GPS technique. The observation of a significantly higher contrast volume in the GPS group (median 350 vs 220 mL) likely reflects the increased complexity and prolonged manipulation time associated with these challenging cannulations, necessitating careful postprocedural renal function monitoring.
Collectively, these considerations highlight that although the GPS technique can be safely integrated into F-EVAR workflows, its application requires thoughtful case selection, meticulous execution, and heightened intra- and postprocedural vigilance.
Limitations
This study has several important limitations. First, it was a retrospective, single-center analysis with a relatively small sample size, particularly in the Pre-GPS technique group, which may limit the generalizability of our findings. Second, the decision to apply the GPS technique was based on subjective operator judgment at the time of the procedure, introducing potential selection bias. In this study, we were not able to include a direct comparator group employing alternative bailout techniques, such as the SCAPED technique, balloon-assisted tracking, or preloaded systems, which prevents head-to-head evaluation of efficacy. Finally, anatomical complexity was not uniformly stratified or quantified across groups, which may have affected procedural difficulty and outcomes. Future prospective, multicenter studies with standardized anatomical grading and predefined technical protocols are needed to validate the broader applicability and efficacy of the GPS technique.
Conclusion
The GPS technique is a transfemoral-only, adjunctive bailout strategy developed to overcome target vessel cannulation difficulties arising from misalignment between the fenestrations and the target vessel ostia in F-EVAR using PMEGs. Applied selectively when initial cannulation using an angiographic catheter and a 0.035 inch guidewire failed, the GPS technique achieved a procedural success rate of 77.8% (14/18) without any technique-related complications. Its on-demand use enabled completion of bridging entirely via the femoral route, avoiding the need for additional access or device escalation. Although not universally applicable, the GPS technique expands the procedural options available for managing misaligned fenestrations in complex endovascular aortic repair. Future studies should aim to identify anatomical predictors of success, refine technical steps to enhance stability in unfavorable orientations, and evaluate its integration into broader F/B-EVAR workflows across diverse clinical settings.
Footnotes
Acknowledgements
We thank Dr. Edward Barroga (https://orcid.org/0000-0002-8920-2607), Medical and Nursing Science Editor, Professor of Medical English Education Center and former Professor of Medical Education at Showa Medical University School of Medicine, Japan, and former Professor and currently Adjunct Faculty of Graduate Academic Writing at St. Luke’s International University, Japan for editing the manuscript.
Ethical Considerations
This study was conducted as part of a clinical investigation approved by the local ethics committee (approval no. 2022-34).
Consent to Participate
All patients provided informed consent.
Consent for Publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
