Abstract
Objective:
Physician-modified endografts (PMEGs) are increasingly used for urgent or off-label repair of complex abdominal aortic aneurysms (AAAs) when custom-made devices are unavailable. A key technical step in PMEG preparation is reloading of the modified stent graft into its original delivery system. Aortic valve stent crimpers have recently been repurposed to assist in reloading, but direct comparisons are lacking with the traditional tourniquet technique. This study aims to compare the efficiency and reliability of tourniquet- and crimper-assisted reloading in a controlled benchtop setting.
Methods:
Eighteen reloading procedures were performed using a Medtronic Valiant 38×200 mm thoracic stent graft, with 9 per technique. Trained operators conducted the reloading under standardized conditions. The primary outcome was reloading time. The secondary endpoints included the infolding potential following deployment in simulated aortic models with oversizing conditions of 21%, 34%, and 42%. Postdeployment morphology was assessed by computed tomography (CT) imaging and reviewed by a blinded observer. Continuous data were analyzed using the Mann-Whitney U test and categorical data by Fisher’s exact test.
Results:
The crimper technique significantly reduced the reloading time compared with the tourniquet method (median 3.98 minutes, interquartile range [IQR=3.27–5.23] vs 11.48 minutes [IQR=8.82–17.35]; p=0.001). No infolding was observed in any of the 18 deployed grafts across all oversizing subgroups. Inadvertent proximal clasp release complicated the tourniquet technique (n=3), while this issue was absent with the crimper (n=0, p=0.206). Device integrity was preserved throughout, although the delivery system required replacement once after repeated reuse.
Conclusion:
This benchtop study demonstrates that the crimper technique provides a faster and more consistent alternative to the traditional tourniquet method for PMEG stent graft reloading. The use of an aortic valve crimper may enhance standardization and workflow efficiency, without infolding.
Clinical Impact
In this bench-top comparison of PMEG reloading techniques, a repurposed aortic valve stent crimper outperformed the traditional tourniquet method by reducing reloading time, without an increase in graft infolding. The improved efficiency associated with the crimper technique highlight its potential value as a standardized approach to PMEG preparation, particularly in time-critical scenarios.
Keywords
Article Highlights
Type of Research
Single-center benchtop experimental study
Key Findings
Reloading time was significantly shorter with the crimper technique than the tourniquet method (median 3.98 vs 11.48 minutes; p=0.001). No infolding was observed in 18 simulated deployments across oversizing levels. Proximal clasp release occurred in 3 tourniquet cases vs 0 crimper cases.
Take Home Message
Using an aortic valve crimper for PMEG reloading is faster than the tourniquet method, without increasing infolding risk, and may enhance workflow efficiency in complex endovascular aortic procedures.
Table of Content Summary
In this single-center benchtop study, the crimper technique significantly reduced PMEG reloading time compared with the traditional tourniquet method, no infolding was observed in any of the deployment attempts. The authors conclude that the crimper technique may provide a more efficient and standardized alternative for PMEG preparation.
Introduction
The treatment of complex abdominal aortic aneurysms (AAAs) has increasingly shifted toward endovascular repair in most high-volume centers.1–5 Current European Society for Vascular Surgery (ESVS) guidelines favor fenestrated or branched endovascular repair (F/B-EVAR) over open surgical repair in high-risk patients.2,3,6–8 In contrast to custom-made fenestrated/branched devices, which typically require several weeks for manufacturing and delivery, physician-modified endografts (PMEGs) offer a cost-effective and readily available alternative, enabling the urgent or semi-urgent repair of complex AAAs. 9 Thus, PMEGs have become an essential component of the armamentarium of vascular surgeons, particularly in urgent scenarios where custom devices are not available. While PMEGs are not recommended for routine clinical use, being restricted to clinical studies under current guidelines, emerging evidence challenges this position regarding the safety and comparability of midterm outcomes of complex AAAs treated with PMEGs.2,10–12 A recent Vascular Quality Initiative analysis concluded that outside of investigational device exemptions PMEGs represent the predominant endovascular management option for thoracoabdominal aortic aneurysms and complex AAAs in the United States underlining the importance of studies evaluating techniques related to PMEGs. 13
Following the initiation of the PMEG program in 2022 at the authors’ institution, a tertiary aortic referral center, several additions were made to the armamentarium, contributing to a more streamlined workflow for the endovascular management of complex AAAs.14–17 One key procedural step in the PMEG workflow is reloading of the modified stent graft into its original delivery system. Traditionally, this has been performed using the tourniquet technique, which involves the use of umbilical tape, vessel loops, and hemostats. 18 However, this method is time-consuming, highly operator-dependent, and may introduce variability into the procedure.
To streamline this critical step, an aortic valve stent crimper has been adopted as an alternative reloading tool, offering a more standardized and potentially faster technique. In this benchtop study, we aimed to quantitatively compare the reloading times and assess the infolding potential of the tourniquet versus crimper techniques under simulated conditions.
Materials and Methods
Study Design and Graft Selection
This benchtop experimental study was designed to compare 2 stent graft reloading techniques: the traditional tourniquet method and the novel application of an aortic valve stent crimper. Institutional Review Board approval was waved. All procedures were carried out with a Medtronic Valiant 38×200 mm thoracic stent graft (Medtronic, Minneapolis, Minnesota), a commonly used device in our clinical PMEG practice because of its availability and favorable mechanical properties.
The stent graft was fully unsheathed and then reloaded into the original delivery system using either the tourniquet or the crimper technique. Eighteen tests were conducted with 9 independent reloading attempts per technique (n=9 per group). All procedures were performed in a controlled benchtop environment.
Operators and Standardization Measures
All reloading procedures were performed by a vascular surgery resident (PGY5) with a radiology resident assistant (PGY5) after being trained by an expert in complex endovascular aortic aneurysm repair and having performed 10-10 reloading attempts per technique. Environmental factors, such as room temperature, surface setup, and lighting, were standardized throughout the study to minimize external influences.
Reloading Techniques
The tourniquet technique was performed using umbilical tapes, rubber tubes, a metal obturator, surgical vessel loops, and hemostats to gradually collapse the stent graft into the delivery sheath (Figure 1A). The technique was carried out using 2 sets of umbilical tapes, hemostats, and rubber tubes. The stent graft fabric was constricted manually by the assistant while an umbilical tape, secured by a rubber tube and hemostat were used to maintain graft contraction. As the stent graft was fed back to the delivery system, the umbilical tapes were moved further toward the end of the graft. A vessel loop secured the proximal corona of the stent graft. This technique relies heavily on tactile feedback and fine motor coordination, which may introduce variability between the operators. To maintain consistency throughout the same team performed each reloads.
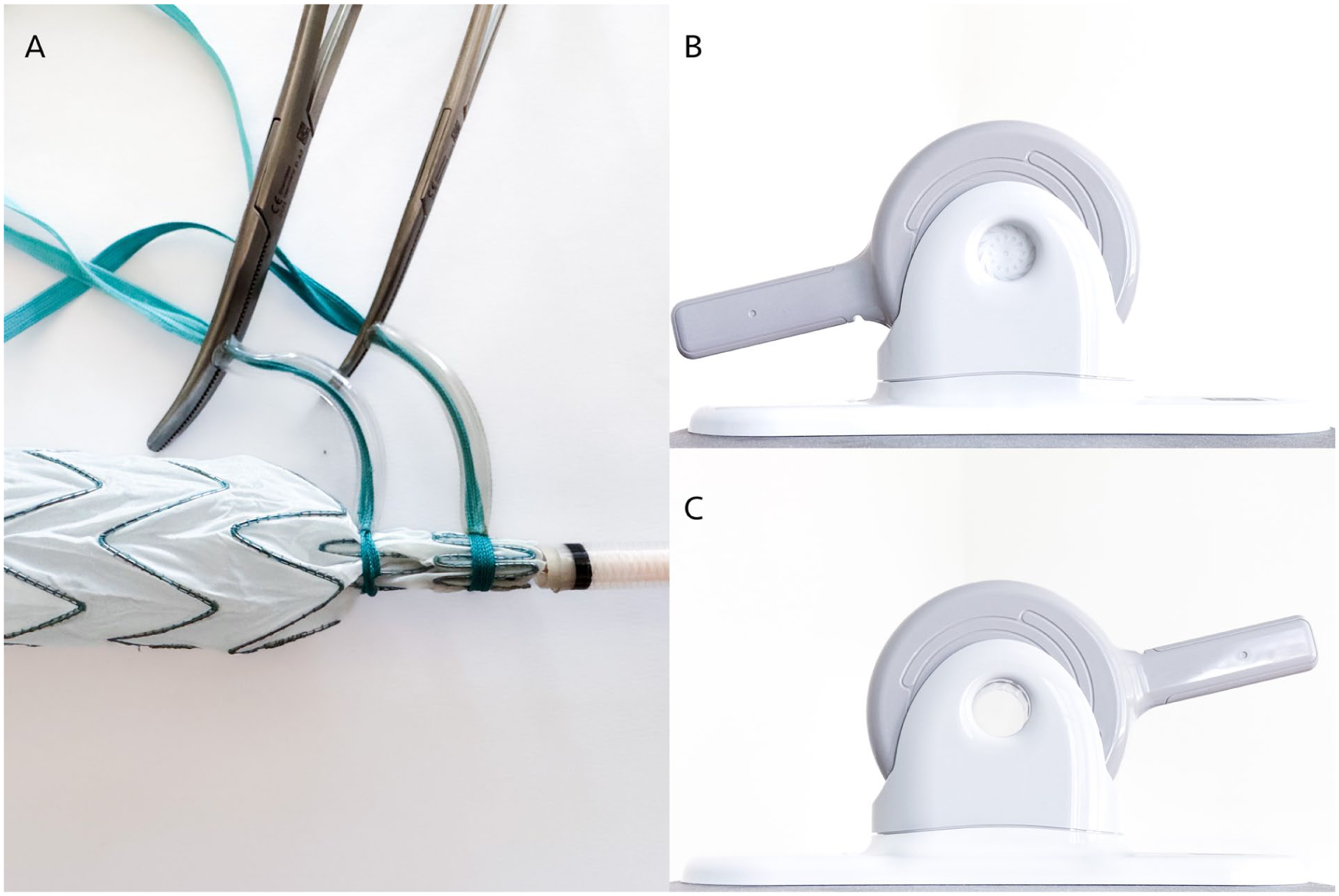
Illustration of the technique used for the tourniquet (A), and the Edwards crimper in its closed (B) and open (C) positions, as applied in the crimper technique.
The crimper technique utilized a commercially available aortic valve stent crimper (Edwards Lifesciences, Santa Ana, California) typically used in transcatheter aortic valve implantation (TAVI) procedures. The device was repurposed to gradually reduce the diameter of the stent graft before it was reintroduced into the original sheath (Figure 1B–C).
Timing and Outcome Measurements
The primary endpoint was reloading time, defined as the time from the beginning of the reloading process—the fully deployed stent graft was ready to be reloaded—until complete reinsertion into the delivery sheath. Timing was recorded by an independent observer using a digital stopwatch.
The secondary outcomes included the infolding potential upon simulated deployment the influence of varying degrees of oversizing, and loss of device integrity (defined as damage to the stent graft fabric- presence of fabric tear, stent fracture)
Simulated Deployment and Oversizing Conditions
To mimic in vivo circumstances, each reloaded stent graft was deployed in rigid transparent plastic tubes to simulate the aorta. (Figure 2A) The oversizing conditions of 42%, 34%, and 21% were tested. Each oversizing group included 3 grafts per reloading technique (3 crimpers and 3 tourniquets per oversizing subgroup). Deployment was performed according to a standard protocol. Efforts were made to avoid opening the proximal clasp to leave the device prepared for the next reloading session.
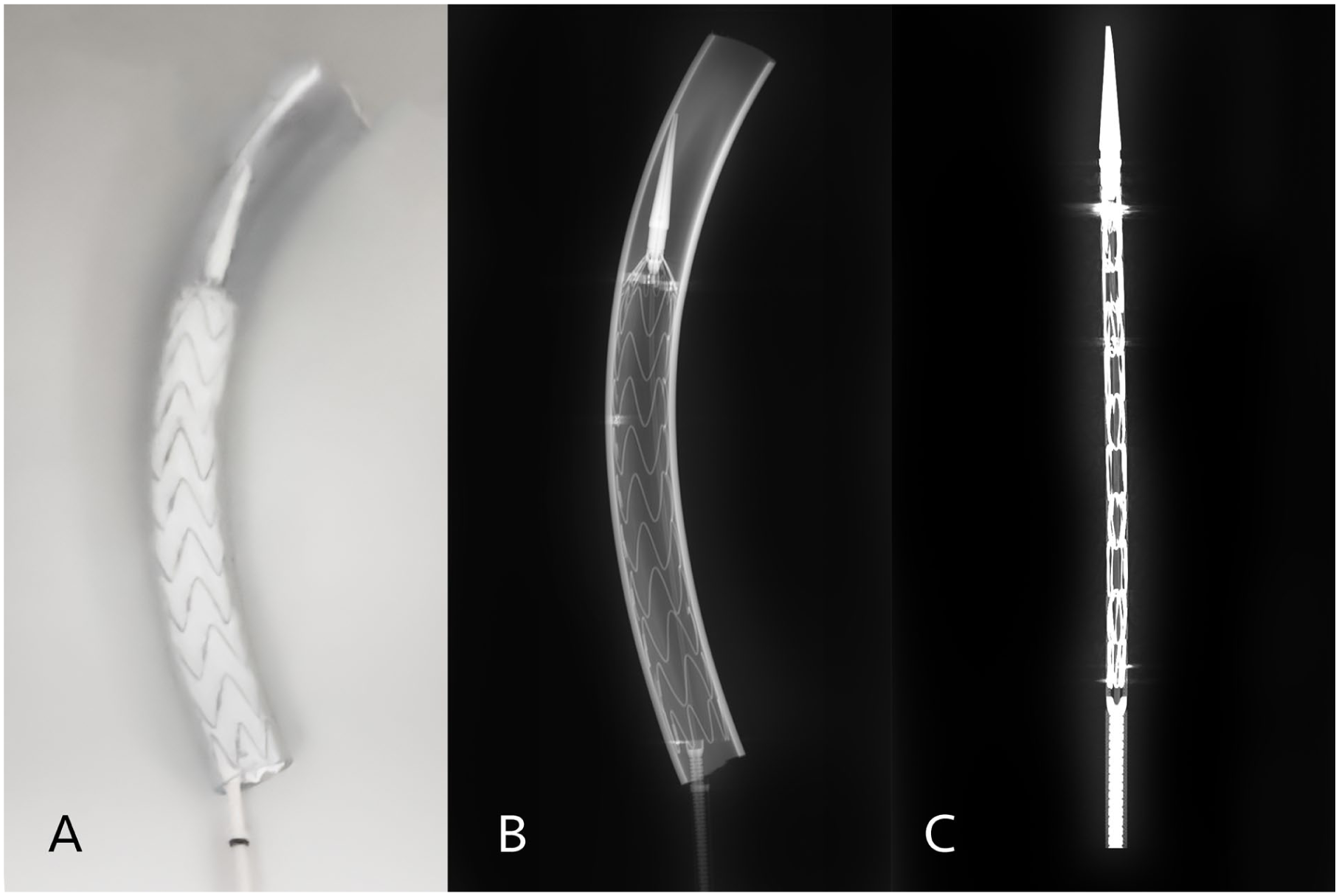
The reloaded graft was deployed into a plastic tube (A) and a CT scan was performed predeployment (C) and postdeployment (B).
Imaging and Infolding Assessment
All stent grafts were scanned using a Siemens SOMATOM Force CT scanner (Siemens Healthineers, Erlangen, Germany) with a 0.5 mm isotropic resolution. Images were reconstructed using multiplanar reformation (MPR) on the Syngo.via workstation (Siemens Healthineers) to assess stent morphology. (Figure 2B–C)
Infolding was defined as duplication of the stent graft fabric extending over the center point of the stent graft. Center point was defined as the intersection of the major and minor axes. The assessment was performed by a vascular radiologist blinded to the reloading technique. Partial folding, fabric wrinkles, or minor asymmetries were recorded, but not classified as infolding unless the predefined threshold was met.
Statistical Analysis
Statistical analysis was performed using SPSS version 28.0 (IBM Corp., Armonk, New York). Continuous variables are expressed as medians with interquartile ranges (IQRs). Group comparisons for reloading times were conducted using the non-parametric Mann-Whitney U test because of the small sample size. Categorical data were assessed using Fisher’s exact test. Statistical significance was set at p<0.05.
Results
A total of 18 stent graft reloading procedures were completed—9 using the tourniquet technique and 9 using the crimper technique. All reloading attempts were successful with no device damage.
Reloading Time
The crimper technique demonstrated a significantly shorter reloading time than that of the traditional tourniquet method. The median reloading time with the crimper technique was 3.98 minutes (IQR=3.27–5.23), while the tourniquet technique required a median of 11.48 minutes (IQR=8.82–17.35) (p=0.001, Mann-Whitney U test). The crimper group also showed less variability in reloading duration, suggesting more consistency across operators.
The tourniquet group saw unwanted proximal clasps and proximal corona openings during 3 of the reloading attempts, which were dealt with by the reloading team. In contrast, the crimper technique showed no tendency toward this potentially workflow-disrupting complication.
A violin plot comparing reloading times between techniques is provided in Figure 3.
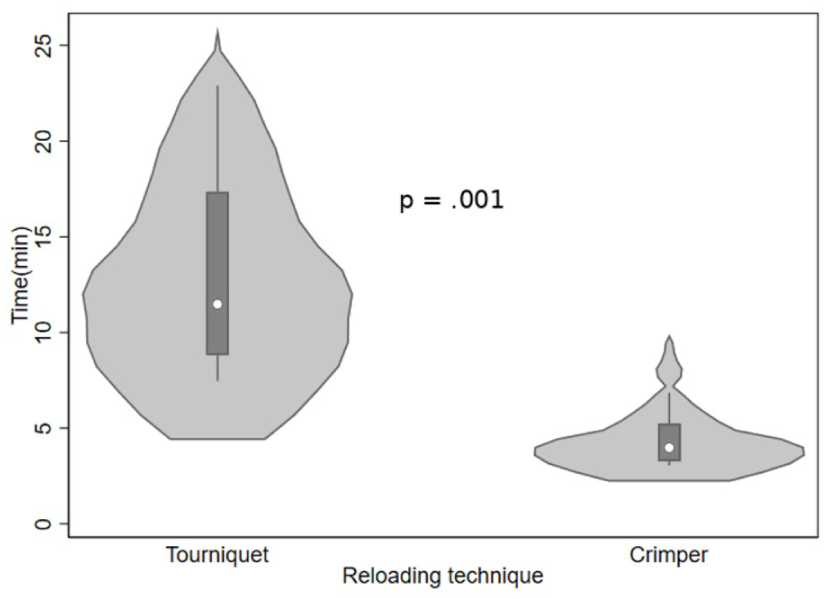
Violin plot representing the significantly faster reloading time with the crimper technique.
Infolding Assessment
All 18 deployments were successfully performed in plastic aortic simulators under their respective oversizing conditions (21%, 34%, and 42%). Postdeployment computed tomography (CT) imaging revealed no instances of infolding in any of the stent grafts, regardless of the reloading technique or the oversizing level (Figure 4A–F). Minor irregularities in the stent fabric were observed in both groups but did not meet the predefined criteria for infolding and were considered insignificant (Figure 4F—pink dotted oval line, pink straight line).
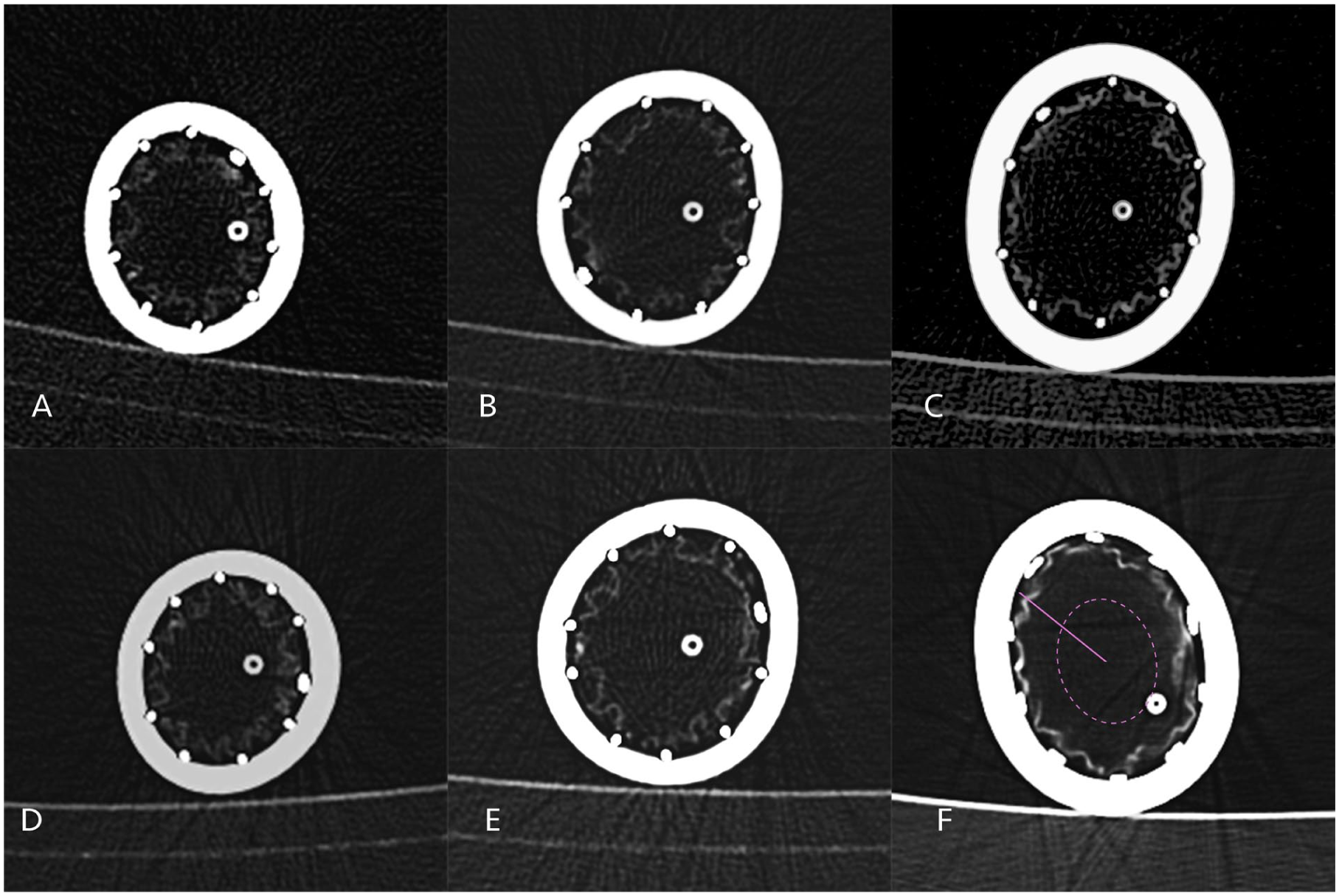
Computed tomographic scans were performed on the different diameter plastic tubes with the deployed stent graft which were previously reloaded either using the tourniquet (A–C) or crimper (D–F) technique. Infolding was defined as duplication of the stent graft fabric extending over the midpoint (dotted pink line) of the stent graft radius (straight pink line).
Subgroup Analysis by Oversizing
No statistically significant difference in post-deployment morphology was observed between the 3 oversizing subgroups for either reloading technique. The deployment across all oversizing levels was uniform, and the reloaded stent grafts expanded symmetrically within the rigid-tube models.
Device Integrity
The stent graft used for reloading has become increasingly difficult to resheath, probably because of fabric fatigue. To obtain comparable results, the operators alternated between reloading techniques. We experienced failure of the delivery system after 12 re-sheathing and deployment attempts, necessitating the use of another delivery system with the same stent graft. Inadvertent proximal clasp release complicated the tourniquet technique (n=3), while this issue was absent with the crimper (n=0, p=0.206).
Discussion
This benchtop study demonstrates that the use of a repurposed aortic valve stent crimper significantly reduces reloading time compared with the traditional tourniquet method during PMEG preparation. The crimper method showed superior efficiency, suggesting that it could contribute to a more standardized and streamlined workflow for complex endovascular aneurysm repairs.
The importance of stent graft reloading as a procedural step is often underemphasized in the broader context of PMEG preparation. Reloading is not only technically challenging but also time sensitive, especially in urgent or emergent cases. Delays in graft preparation can increase the overall operative duration and the risk of complications. By achieving faster reloading times without compromising device integrity or increasing the risk of infolding, the crimper method may help mitigate these risks and improve procedural efficiency.
The aortic valve crimper allowed uniform radial compression of the stent graft and required minimal manual manipulation, thereby reducing operator dependence. Inadvertent opening of the proximal clasp during device preparation and reloading represents an uncomfortable occurrence, which was not observed with the crimper technique. On the contrary, with the tourniquet technique proximal clasp opening occurred 3 times, which might hinder the entire PMEG workflow and could cause potential delays in patient management. As it requires expert technique to reload the proximal clasp, such an inadvertent release can easily become a major obstacle during preparation.
The tip capture mechanism of the Medtronic Valiant Captivia Freeflo stent graft (Medtronic, Minneapolis, Minnesota) includes a proximal clasp mounted on a relatively short thread which ensures capture of the proximal bare metal stent. Care must be taken not to disengage the proximal clasp by rotating the stent graft counterclockwise during reloading. The Valiant stent graft is fixed to the delivery system solely by the tip capture mechanism and the lack of distal attachment makes the device vulnerable to inadvertent deployment which is something the operator performing the reloading needs to be aware of. Future design improvements might be able to resolve this issue.
Importantly, no modification of the crimper was performed, underscoring the potential of repurposing existing tools to enhance the workflow without added cost or complexity.
Despite the widespread use of the tourniquet technique in clinical practice, it remains highly operator-dependent and time-intensive. Originally described by Uflacker et al 18 using the Cook Zenith platform, the tourniquet method requires fine motor control and constant visual monitoring, and is inherently variable. In contrast, the aortic valve crimper approach provides more consistent compression and could shorten the learning curve for endovascular specialists early in their PMEG adoption.
Literature on reloading techniques in PMEG remains limited. The recently described PERTINI technique, developed specifically for the Gore Excluder C3 system, is another example of center-specific innovation that addresses similar procedural challenges. 19 However, the diversity of graft platforms, fenestration strategies, and institutional protocols makes standardization difficult. Our findings support the use of a more reproducible and efficient reloading strategy that can be easily integrated into existing workflows, especially for centers that use the Medtronic Valiant platform.
We chose a benchtop model for this comparative study to eliminate confounding variables inherent in clinical cases, such as varied aneurysm anatomy, target vessel orientation, and procedural urgency. In addition, the simulated deployment into rigid tubes at multiple oversizing levels allowed for a controlled assessment of the risk of infolding. Despite the variations in oversizing (21%, 34%, and 42%, respectively), no infolding was observed in either group.
Since the aortic valve crimper is only available as part of a balloon-expandable TAVI delivery kit, its use in the PMEG workflow requires resterilization. Institutional experience indicates that the device tolerates repeated low-temperature hydrogen peroxide sterilization cycles without loss of function.
Limitations
The primary limitation of this study is its benchtop design, which may not fully replicate intraoperative conditions, including hemodynamic stress, graft rotation, fenestration reinforcement, or handling-related factors. While the operator had partaken in a preliminary learning phase, the tourniquet technique probably requires a higher volume to achieve expert level proficiency, which may explain the time difference between the 2 techniques. Nevertheless, the crimper technique may contribute to a better streamlined reloading workflow given its ease of use. It is important to note, that fenestration reloading was not assessed in this benchtop study. Current study assessed a single type of stent graft at a given diameter, which might not be representative of the various graft types used across the international PMEG landscape, nevertheless the crimper technique seems promising to deliver regardless of stent graft type. Although there is a trend toward “wet reloading,” the authors—according to their clinical practice—utilized dry reloading, which should not hinder results since the approach was adopted for both reloading arms. 20 In addition, although adequate for a controlled bench study, the sample size may not detect rare occurrences of infolding or device failure. Finally, the occurrence of inadvertent clasp release might show different frequency based on the varying techniques used across centers for securing the corona of the devices. Future studies, ideally prospective and clinical in nature, are needed to validate these findings in vivo and assess the broader applicability of the crimper technique across other graft platforms. In summary, this study supports the aortic valve stent crimper as a more efficient alternative to the traditional tourniquet method for stent graft reloading during PMEG. This method allows for faster reloading with a lower risk of inadvertent proximal clasp release. Following clinical validation, the technique’s integration into standardized PMEG workflows may enhance procedural efficiency, reduce operator variability, and improve the outcomes of complex endovascular aortic repair.
Footnotes
Ethical Considerations
This study was conducted entirely in a laboratory setting using commercially available medical devices. No human participants or animal subjects were involved. Therefore, ethical approval was not required under the guidelines of the institutional or national research ethics committee.
Author Contribution
J.C., A.H., and A.B. contributed to the design and implementation of the research project; J.C., P.O., and C.C.N. to the analysis of the results; and P.O., J.C., A.H., A.B., and C.C.N. to the writing of the manuscript. C.C.N. conceived the initial concept and supervised the project.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Csaba Csobay-Novák was supported by a grant from the National Research, Development and Innovation Office (NKFIH) of Hungary (STARTING 152421) and by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences. Judit Csőre was Supported by the 2025-2.1.1-EKÖP-2025-00014 University Research Scholarship Programme of the Ministry for Culture and Innovation from the source of the National Research, Development and Innovation Fund.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.C.N. is trainer, proctor, and speaker for Medtronic, Cook Medical, Gore Medical, and Terumo Aortic.
