Abstract
In this case report, we describe the feasibility and technical considerations of an off-label use of the new Gore® Aortic Extender (W.L. Gore & Associates), which is part of the Gore® TAG® Thoracic Branch Endoprosthesis (W.L. Gore & Associates), to repair a huge false aneurysm originating from an ascending aortic prosthesis. The patient’s medical history reported an acute type A aortic dissection 10 years ago, for which he was treated with a Bentall procedure. This was complicated by obstruction of the left main coronary after it was reconstructed and thus a coronary artery bypass was also performed. During follow-up, a huge false aneurysm of 11 cm was found, caused by a dehiscent saphenous vein graft on the Bentall prosthesis. Since the patient was clearly not fit for redo surgery, we decided to treat this patient endovascularly with the Gore® Aortic Extender (W.L. Gore & Associates). This cuff has recently been introduced and is off-the-shelf available and seemed to fit perfectly in this patient.
Clinical impact
With the introduction of the new, Gore® TAG® Thoracic Branch Endoprosthesis (W.L. Gore & Associates), a very short endoprosthesis became available for proximal extension of the Thoracic Branch Endoprosthesis. This graft, the Gore® Aortic Extender (W.L. Gore & Associates) seems to come in favorable sizes for the ascending aorta. As long as dedicated endoprosthesis for the ascending aorta are not off-the-shelf availabe, off-label use of such endovascular devices is an useful option whenever open surgery seems too high risk.
Introduction
Endovascular treatment of thoracic aortic aneurysms is rising and is now standard of care in descending aortic pathologies. However, open surgery remains the golden standard for the aortic root, ascending aorta, and aortic arch. 1 Yet there are reports, that show the feasibility of endovascular treatment of the aortic arch with branched devices 2 and the treatment of the ascending aorta for acute type A aortic dissection.3,4 Due to the complexity of ascending and aortic arch endovascular procedures, there is a lack of approved off-the-shelf devices, requiring time-consuming custom-made stent-grafts. Although open surgery is the standard of care, some patients might benefit more from an endovascular treatment. In this report, we describe the off-label use of the recently introduced, off-the-shelf available Gore® Aortic Extender (W.L. Gore & Associates, Flagstaff, AZ, USA), which is part of the Gore® TAG® Thoracic Branch Endoprosthesis (TBE), to exclude a huge false aneurysm of 11 cm in the ascending aorta, caused by a dehiscent saphenous vein graft. Our patient provided informed consent for the report and the images.
Case Description
A 70-year-old male patient was referred to our hospital regarding a huge false aneurysm of 11 cm from the ascending aorta due to a dehiscent saphenous vein graft (Figure 1). Ten years earlier, the patient had undergone a subacute type A repair by the referring hospital. The repair consisted of a Bentall procedure with a Bio-Valsalva Biplex 23 (Vascutek Terumo, Renfrewshire, Scotland) with hemiarch replacement. The Bentall consisted of a 23 valve with a 24 mm Dacron prosthesis. The main stem was reconstructed with a short venous interposition graft. This graft failed the next day and therefore an additional coronary artery bypass graft (CABG) was performed: the left internal mammary artery (LIMA) was anastomosed on the left anterior descending and a saphenous vein graft was anastomosed from the 24 mm Dacron graft to the obtuse marginal and left posterolateral branch. Postoperative course was complicated by a stroke with left-sided paresis and a pacemaker implantation for total Atrioventricular block.
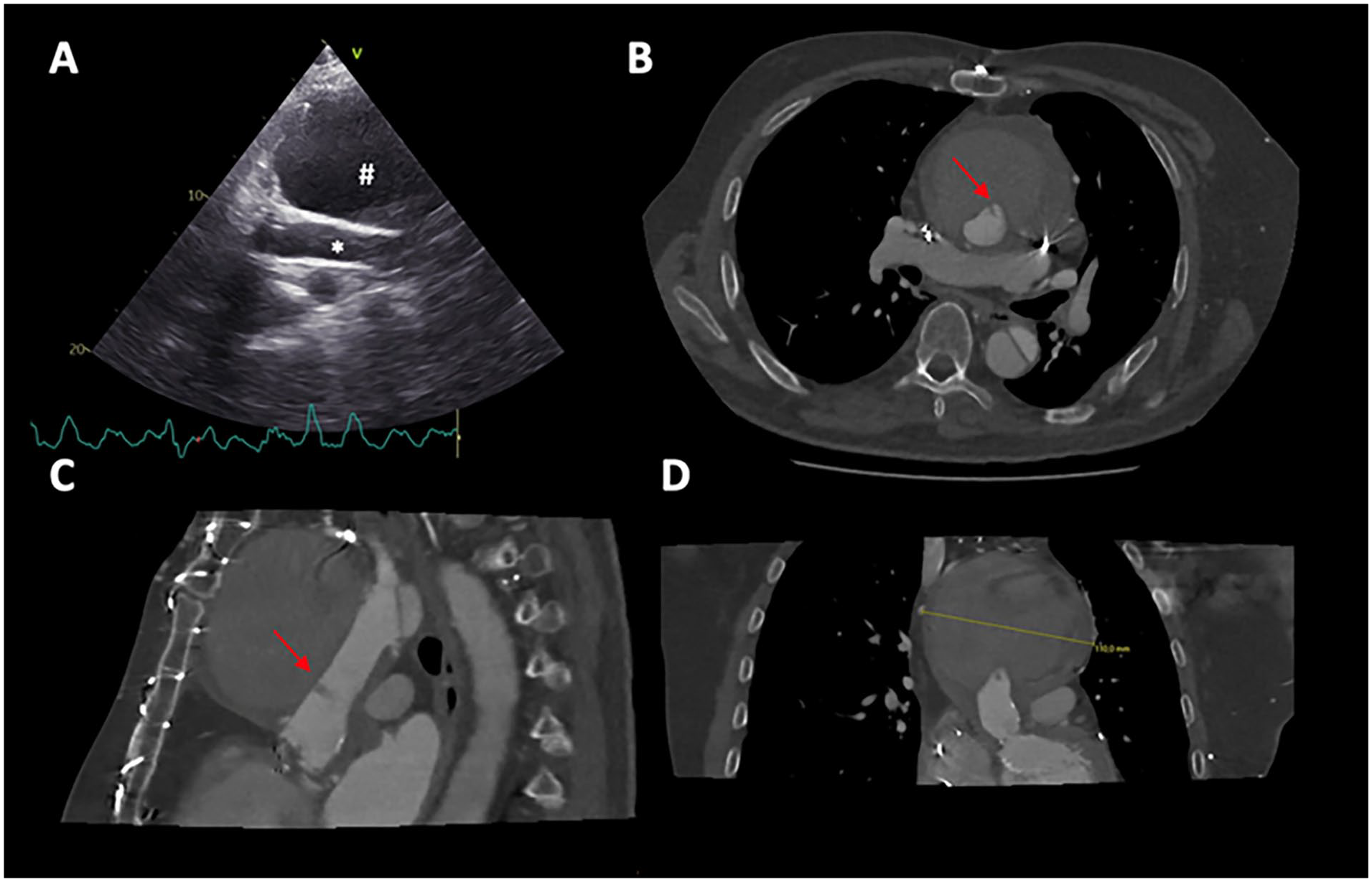
(A) Echocardiography showing a large mass (#) on the ascending prosthesis (*). (B) Axial view showing a big mass with blood leaking (red arrow) toward the ascending prosthesis lumen in diastole. (C) Sagittal view. (D) Frontal view, maximum diameter 11 cm.
In the last 10 years the follow-up was minimal. Now, a routine echocardiogram showed a mass around the aortic graft of the Bentall (Figure 1A). A computed tomography (CT) scan showed a huge false aneurysm of 11 cm (Figure 1B–D), and the patient was referred to our hospital.
An angiogram showed a dehiscent saphenous vein graft (Figure 2). Furthermore, a remnant of the left main stem reconstruction was seen in the root. The risk of redo surgery was deemed too high, because of his co-morbidities and functional performance. Therefore, we explored an endovascular option. The LIMA to the left anterior descending was patent on CT scan and provided the whole left side of the heart with blood. The dehiscent saphenous vein graft was already occluded on CT scan and full coverage of the saphenous vein was feasible. A custom-made device was considered, but is time-consuming. With the recent introduction of the Gore® TAG® TBE, a short extension cuff came available. We felt that this cuff, Gore® Aortic Extender (W.L. Gore & Associates), would be the ideal prosthesis since it is off-the-shelf available and has the mechanical properties of the Gore® cTAG® which we are familiar with. The aortic valve prosthesis was sclerotic on echocardiography but did not require a re-intervention.
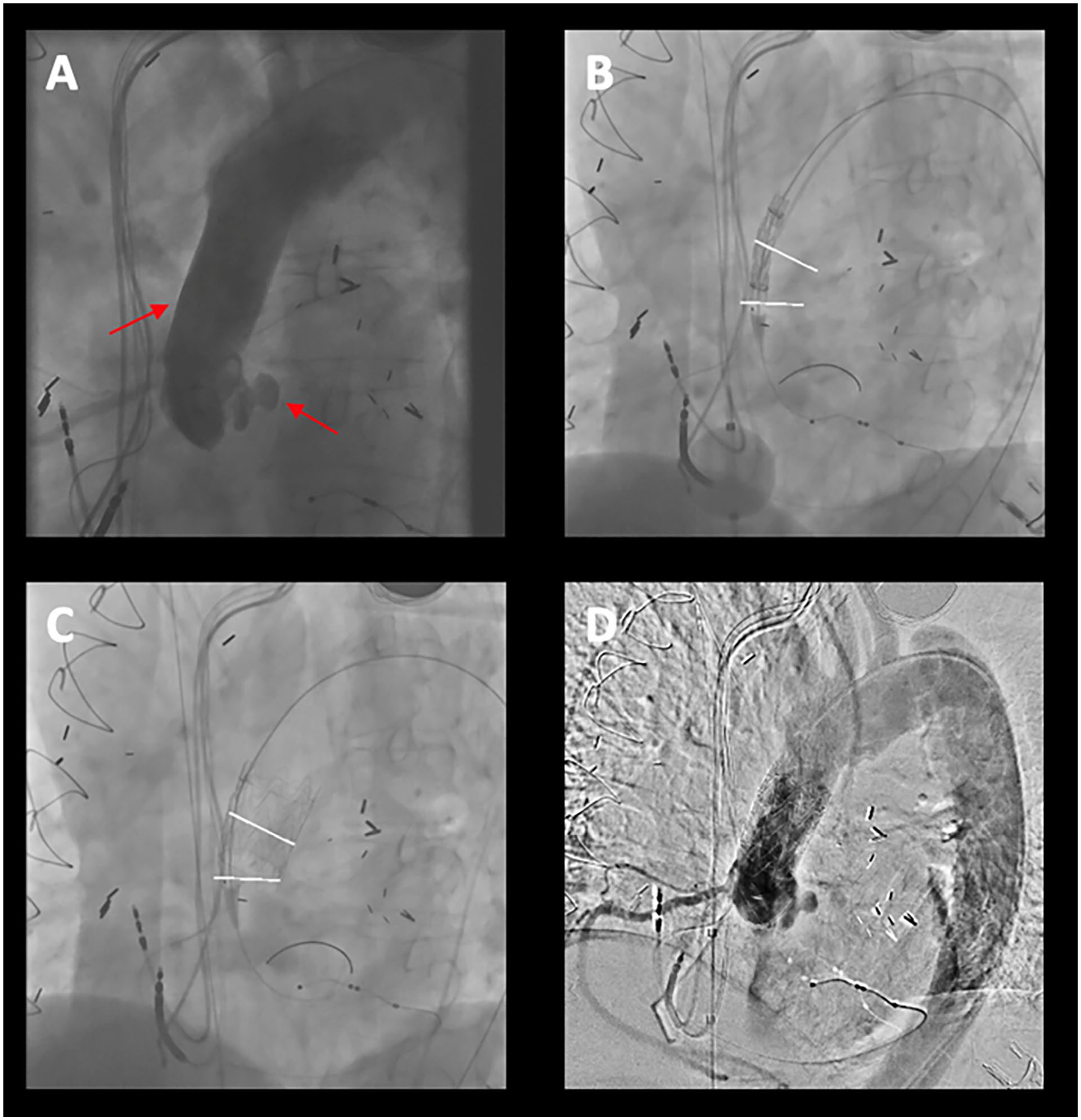
(A) Aortic angiogram, showing the dehiscent anastomosis with leakage and remnant of the reconstructed main stem. (B) Positioning of the stent in the prosthesis, upper marker is the point of leakage, lower marker is the origin of the right coronary artery. (C) Result after deployment of the stent. (D) Control angiogram showed no leakage anymore.
Sizing
CT measurements were made with the Endosize Software (Therenva, Rennes, France). The length of the outer curvature of the ascending prosthesis from the sinotubular junction until the distal anastomosis was 43 mm. The diameter of the graft of the Bentall was oval-shaped with a perimeter-derived diameter of 24 mm (Figure 3), which was consistent with the implanted surgical prosthesis (24 mm). We chose a 31 mm device diameter which has an intended aortic diameter of 24 to 29 mm according to the instruction for use of Gore (see Figure 4A). The Gore® Aortic Extender (W.L. Gore & Associates; which is an extender of the TBE graft) is a straight graft (31 mm) with an overall length of 4 cm of which 32 mm is covered (Figure 4A).
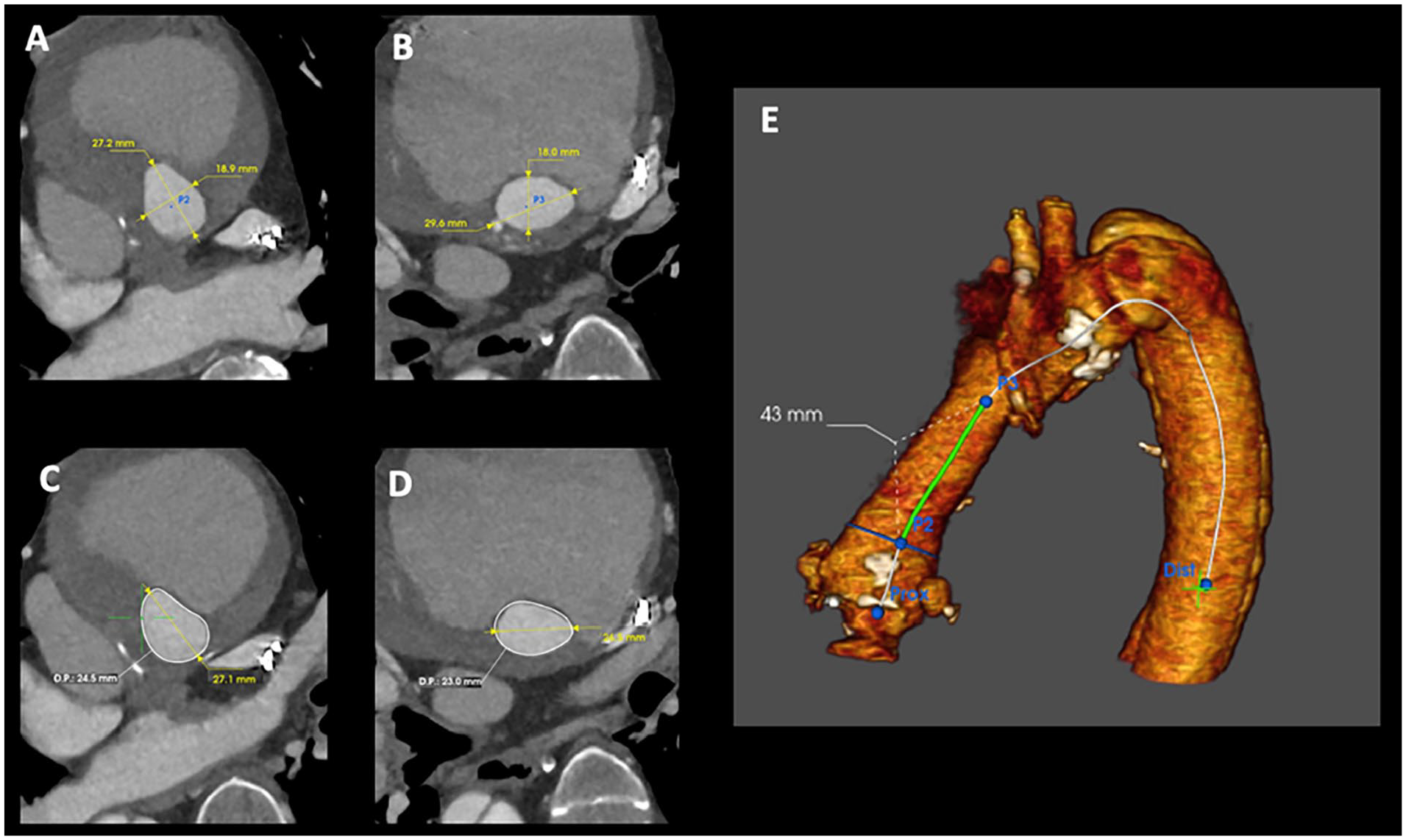
(A) CT measurement of the ascending prosthesis at the level of P2, 27.2 × 18.9 mm. (B) CT measurement of the ascending prosthesis at the level of P3, 29.6 × 18.0 mm. (C) Perimeter at level P2, 24.6 mm. (D) Perimeter at level P3, 23.0 mm. (E) 3D reconstruction of the aorta and locations of P2 and P3 measurements.

(A) Stent-graft (size 31) with measurements. (B) Delivery device with sheath.
Operative Procedure
Intra-operative angiograms are shown in Figure 2. Ultrasound-guided access was obtained percutaneously in the right femoral artery and preclosure devices were deployed (Perclose ProGlide system; Abbott Vascular, Redwood City, CA, USA). A stiff wire (Lunderquist; Cook Medical, Bloomington, IN, USA) was positioned in the left ventricle. A standard pigtail angiography catheter was used to locate the dehiscent saphenous vein graft and 2 markers were placed on the screen: 1 above the right coronary artery and 1 on the defect. The stent-graft was then introduced and positioned in the ascending aorta, above the right coronary artery and covering the dehiscent vein-graft in the middle. Lowering of the cardiac output was obtained by occlusion of the inferior caval vein by using a Reliant® balloon (Medtronic, Dublin, Ireland), followed by deployment of the stent-graft. Because the deployment sleeve is attached to the catheter itself and is trapped between the stent-graft and the aorta, the nose cone was not entirely free to move (Figure 4B). When retrieving the device, the nose cone would be caught on the prosthesis. We solved this by rotating the catheter 90° back and forth. This reduced tension on the sleeve and, therefore, the nose cone was free to move for retrieval. Control angiogram showed no leakage.
Postoperative Course
Postoperative course was uneventful. Patient was extubated in the operating room and discharged home 2 days postoperative. Follow-up CT scan on day 1 showed excellent position of the stent-graft without leakage (Figure 5). CT scan was repeated at 3 months follow-up and showed a completely thrombosed false aneurysm.
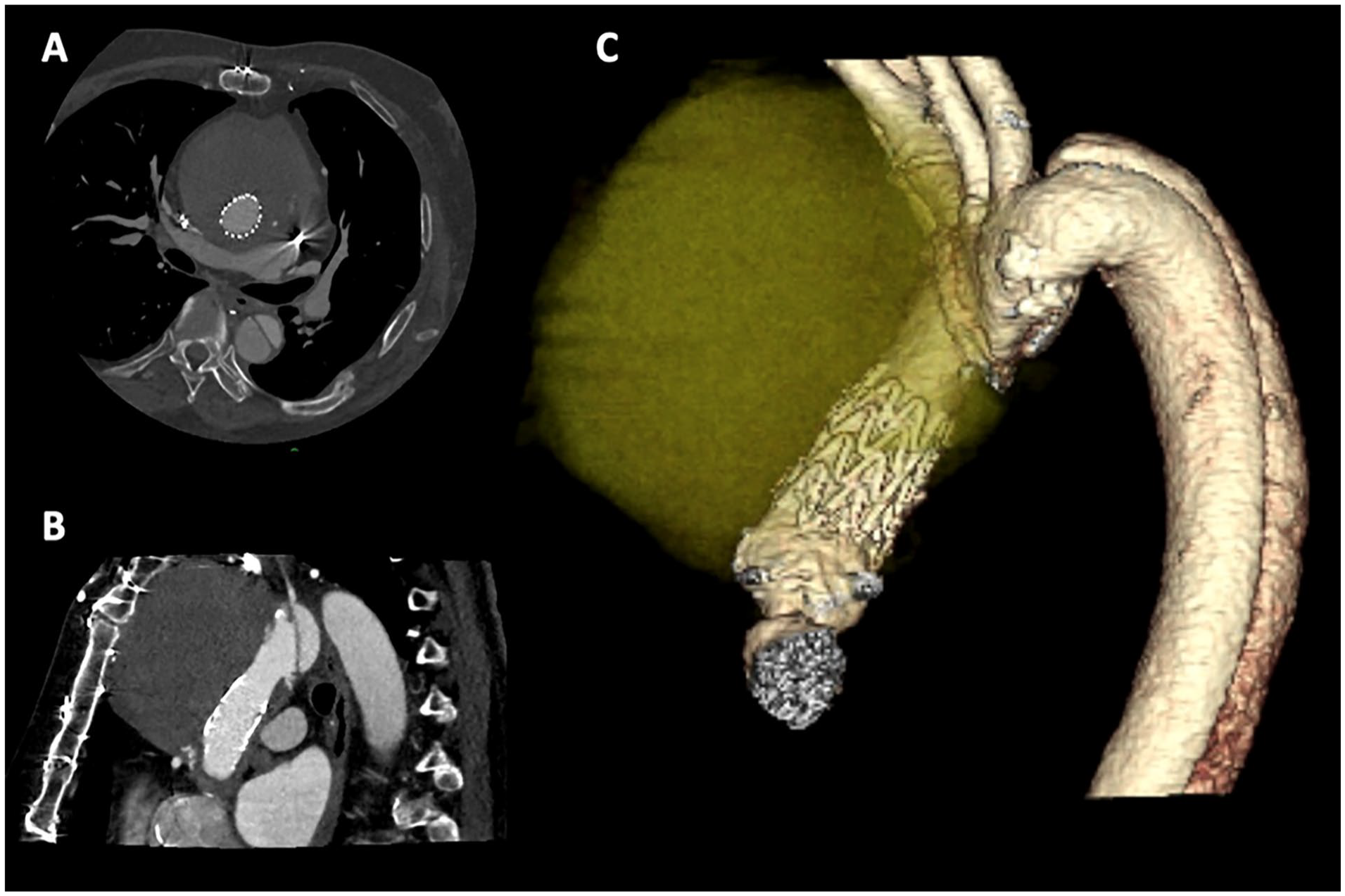
(A) Postoperative day 1 CT scan, axial view. Follow up at 3 months showed similar finding. (B) Sagittal view. (C) 3D reconstruction.
Discussion
We present the off-label use of the Gore® Aortic Extender to treat a huge false ascending aortic aneurysm of 11 cm due to a dehiscent saphenous vein-graft.
The off-label use of an extender for the ascending aorta has been described previously.5–7 However, the extender in these reports is for an abdominal endoprosthesis. There are important differences compared to the extender of the new introduced single-branched conformable stent-graft from Gore®: (1) The delivery system for the new extender is much longer (105 cm) than the abdominal extender (65 cm). Therefore, to position the stent-graft in the Bentall, access for the abdominal device is only possible through the carotid or axillary artery, while the extender of the new TBE can be introduced through the femoral artery. (2) The abdominal extender is deployed from distal to proximal, while the TBE extender is deployed from the middle toward proximal and distal. The TBE extender is therefore less at risk to migrate during deployment. (3) The abdominal extender is a full covered stent, while the TBE extender has a small uncovered proximal and distal section. (4) The TBE graft is designed for the thoracic aorta with more suitable sizes for the thoracic aorta. In our case, only 1 stent was needed to exclude the false aneurysm, while multiple stents were needed with the abdominal excluder. For these reasons, the new extender seems more suitable for our case.
A too long nose cone may damage the biological valve. In contrast to many other devices, the nose cone of the Gore® devices are particularly short. Furthermore, the outer sheath of the stent is attached to the delivery device (Figure 4B), which increases the risk of stent migration when retrieving the whole system, which could hamper the proximal sealing.
These are important considerations to keep in mind, as this device is not primarily designed to be implanted in the ascending aorta. Off-label use of these devices is currently a useful option to treat ascending aorta pathology, until dedicated devices for the ascending aorta are approved. 8
Conclusion
In select cases, the Gore® Aortic Extender (W.L. Gore & Associates) can be used off-label in the ascending aorta.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
