Abstract
Purpose:
The objective of this systematic review was to report the cumulative incidence of endograft migration (EM), as well as the morbidity, reintervention rates, and mortality related to EM. This study aimed to provide evidence-based data on patient-relevant sequelae of EM after thoracic endovascular aortic repair (TEVAR) performed using contemporary aortic endografts.
Materials and Methods:
A systematic electronic search of literature in MEDLINE (via PubMed), Web of Science, and Cochrane Central Register of Controlled Trials was performed. The pooled synthesis of outcomes was performed using the inverse variance method.
Results:
Seven prospective non-randomized and 4 retrospective studies, including a total of 1783 patients presenting 70 EMs, were considered for the quantitative analysis. The pooled rate of EM was 4% (95% CI, 2%–7%; range, 0.2%–11%; I2=82%); pooled morbidity rate was 31% (95% CI, 12%–59%; range, 0%–100%; I2=64%) and pooled reintervention rate was 32% (95% CI, 15%–56%; range, 0%–100%; I2=55%). The pooled mortality rate due to EM was 5% (95% CI, 1%–21%; range, 0%–40%; I2=24%).
Conclusion:
For the first time, this meta-analysis provides pooled reference estimates of EM after TEVAR. Thus, the results hold the potential to further characterize EM after TEVAR. The clinical relevance of EM is underlined by its association with high rates of endoleak-related morbidity, reintervention, and mortality. Close standardized surveillance after TEVAR for early detection of EM and prophylaxis of its sequelae is essential.
Clinical Impact
The study underline the essentiality of the close standardized surveillance after TEVAR for early detection of endograft migration and prophylaxis of its sequelae. The study prove the clinical relevance of endograft migration, underlining its association with high rates of endoleak-related morbidity, reintervention and mortality. For the first time, this meta-analysis provides pooled reference estimates of endograft migration after TEVAR. Thus, the results hold the potential to further characterize endograft migration after TEVAR.
Introduction
More than 27 years after the first implant by MD Dake, 1 thoracic endovascular aortic repair (TEVAR) is the treatment of choice for a variety of thoracic aortic pathologies. 2 Late endograft migration (EM) after TEVAR refers to endograft-related complications, including endoleaks, aortic rupture, and reintervention.3–5 Early diagnosis of EM may prevent its clinical sequelae and improve rupture-free survival. A protocol of reporting standards for TEVAR including assessment of endograft location was published in 2010.3 However, literature and evidence on this clinically important topic remain limited.5,6
The majority of related studies failed to analyze EM per-standard and reported heterogeneous EM rates using diverse assessment methods and definitions. Furthermore, reported EM-related reintervention and morbidity/mortality rates vary widely between studies. Thus, the objective of this systematic review was to report the cumulative incidence of EM, as well as morbidity, reintervention rates, and mortality related to EM. This meta-analysis aimed to provide evidence-based data on patient-relevant sequelae of EM after TEVAR performed using contemporary aortic endografts.
Materials and Methods
Protocol and Registration
This systematic review was reported according to recommendations in the Preferred Reporting Terms for Systematic Review and Meta-analysis (PRISMA). 7 The review was registered in the International Prospective Register of Systematic Reviews (PROSPERO, No. CRD42020194309, registered 01/08/2020).
Eligibility Criteria
Prospective and retrospective studies that reported the results of TEVAR due to thoracic aortic aneurysm (TAA), dissection of the aortic arch and the descending thoracic aorta (TAD), penetrating aortic ulcer (PAU), intramural hematoma (IMH), traumatic aortic rupture (TAR), anastomotic aneurysm (AA), or aorto-oesophageal/aorto-bronchial fistula (ABF/AEF) and reported endograft migration in the follow-up (FU) were eligible. All included studies used a definition of EM according to the reporting standard, and assessment of endograft position was performed using contrast-enhanced computed tomography (CTA). 3 A minimum of 6 months of FU were deemed necessary for inclusion. The maximum FU-time was not restricted.
Studies that reported branched/chimney technique/fenestrated TEVAR or the results of solely open thoracic aortic repair were excluded. If the outcomes of TEVAR and open repair of thoracic aorta were reported in the trial and the outcomes for TEVAR branch were fully accessible individually, the TEVAR branch was included in the analysis.
Case reports and case series (<50 patients in study cohort) were excluded. Furthermore, animal studies, meeting abstracts, letters/comments/editorials, articles pre-dating 2010, and publications for which the full text was irretrievable were excluded.
Information Sources
A systematic electronic search of the literature in MEDLINE (via PubMed), Web of Science and Cochrane Central Register of Controlled Trials was performed. 8 Moreover, a hand search of the reference lists of relevant articles, as well as citing references in the Web of Science, was conducted.
Search Strategy
The search strategy was built using a combination of the following key search terms and synonyms as index or free text words and MeSH terms:
((((treatment [tiab] OR repair* [tiab] OR approach [tiab] OR therap* [tiab] OR management [tiab] OR surger* [tiab] OR stent* [tiab] OR graft* [tiab] OR stentgraft* [tiab] OR stent graft* [tiab] OR endostent* [tiab] OR endograft* [tiab] OR endoprosthe* [tiab]) OR (“Stents”[Mesh] OR “Blood Vessel Prosthesis” [Mesh] OR “Blood Vessel Prosthesis Implantation/adverse effects”[Mesh] OR “Prosthesis Failure”[Mesh] OR “Foreign-Body Migration/surgery “[Mesh])) AND (thorax* [tiab] OR thoraces [tiab] OR thoracic [tiab] OR thoracal [tiab] OR ascend*[tiab] OR descend*[tiab]) AND (endovascul*[tiab] OR aort*[tiab] OR “Endovascular Procedures”[Mesh] OR “Aorta, Thoracic “[Mesh])) OR (TEVAR [tiab] OR “thoracic aortic endovascular aneurysm repair” [tiab] OR “thoracic endovascular aortic repair” [tiab] OR “thoracic endovascular repair”[tiab])) AND (dislocat* [tiab] OR displace* [tiab] OR movement* [tiab] OR shift* [tiab] OR drift* [tiab] OR misplace*[tiab] OR dislodgement [tiab] OR dislodgment [tiab] OR migrat* [tiab] OR disconnect*[tiab] OR separation*[tiab] OR angulation*[tiab] OR malpositioning [tiab]) NOT (animals [mh] NOT humans [mh]). 8
To exclude studies investigating TEVAR performed with first-generation thoracic endografts, and studies published before the establishment of reporting standards, which defined a protocol of endograft position assessment, articles were searched starting with a publication date after January 1, 2010. The last search was performed on October 1, 2021.
Study Selection and Data Extraction
Data were extracted from the trials that met the inclusion criteria using a standardized form. Two reviewers (D.S. and E.K.) independently extracted the data and any queries/discrepancies were resolved by a third reviewer (M.S.B.).
The following variables were gathered: author, full citation, study design (prospective or retrospective), demographic data (age, gender), clinical data (coronary artery disease [CAD], arterial hypertension [AH], congestive heart failure, diabetes mellitus [DM], prior vascular or aortic intervention, renal failure/dialysis and smoking, diagnosis, type of proximal landing zone, type of endograft), and follow-up (FU) settings. Other data included migration rate, time point of migration, migration site (proximal/distal landing zone or overlapping zone), migration-associated reintervention rate, and morbidity/mortality rates.
Outcomes and Definitions
The outcomes of the review were incidence of EM, EM-related reintervention rate, as well as EM-related morbidity and mortality.
EM was defined according to current reporting standards as an asymptomatic endograft displacement of >10 mm relative to a primary anatomic landmark or any displacement that led to symptoms or required therapy during follow-up. 3
EM-related morbidity was defined as aortic rupture, endoleaks (ELs) type I/III, or aneurysm sack expansion without visible EL due to EM.
EM-related reintervention was defined as endovascular or open reintervention performed due to EM-related morbidity.
EM-related mortality was defined as death due to EM-related morbidity.
The classification of ELs and proximal landing zones (PLZ) was performed according to the previously published literature.9,10
Mid-term FU was defined as between 1 and 5 years after TEVAR, and long-term FU thereafter. 3
Quality Assessment
The risk of bias was assessed in accordance with the Cochrane bias assessment tool for non-randomized intervention studies adapted for non-interventional studies (ROBINS-I tool). 11 Each of the following domains were evaluated: bias due to confounding, bias in selection of participants into the study, bias in classification of intervention, bias due to deviations from intended interventions, bias due to missing data, bias in measurement of outcomes, and bias in selection of the reported result. Each potential source of bias was graded as low risk, moderate risk, serious risk, critical risk, or no information/unclear. An overall judgment of the presence of bias in each study was subsequently performed.
Statistical Analysis
For all outcomes, a random-effects model was applied to account for methodological and clinical differences. Statistical heterogeneity among the effect estimates of the included trials was evaluated using the I2 statistic. In the analysis, an I2 less than 25% was considered to indicate low heterogeneity and an I2 > 75% to indicate high heterogeneity. 12
Pooled rates and means with 95% CI for the above-mentioned outcomes were calculated using the inverse variance method. Primary statistical analysis and meta-analysis was performed using program R (R Foundation for Statistical Computing, Vienna, Austria). Forest plots were used for graphical presentation of overall effect estimates.
Results
Study Selection
A total of 439 articles were screened for eligibility (Figure 1). After removing duplicates and screening abstracts, the evaluation of 48 full-text articles was performed. Fourteen studies were excluded from further analysis because they failed to define EM and 11 due to duplication; 6 studies reported non-interpretable data, 2 studies reported a short FU (<6 months), and 4 studies did not present EM as an endpoint (Figure 1). If duplication of results occurred, the study that reported a longer FU was included. Two trials did not indicate the definition of EM, but this was clarified through communication with the corresponding author.13,14 One trial reported migration as an endpoint, failing to describe EM-related clinical sequelae. This lack of information was resolved through communication with the corresponding author. 15 Eleven studies with a total of 1783 patients presenting 70 EMs were included in the qualitative and quantitative analysis.
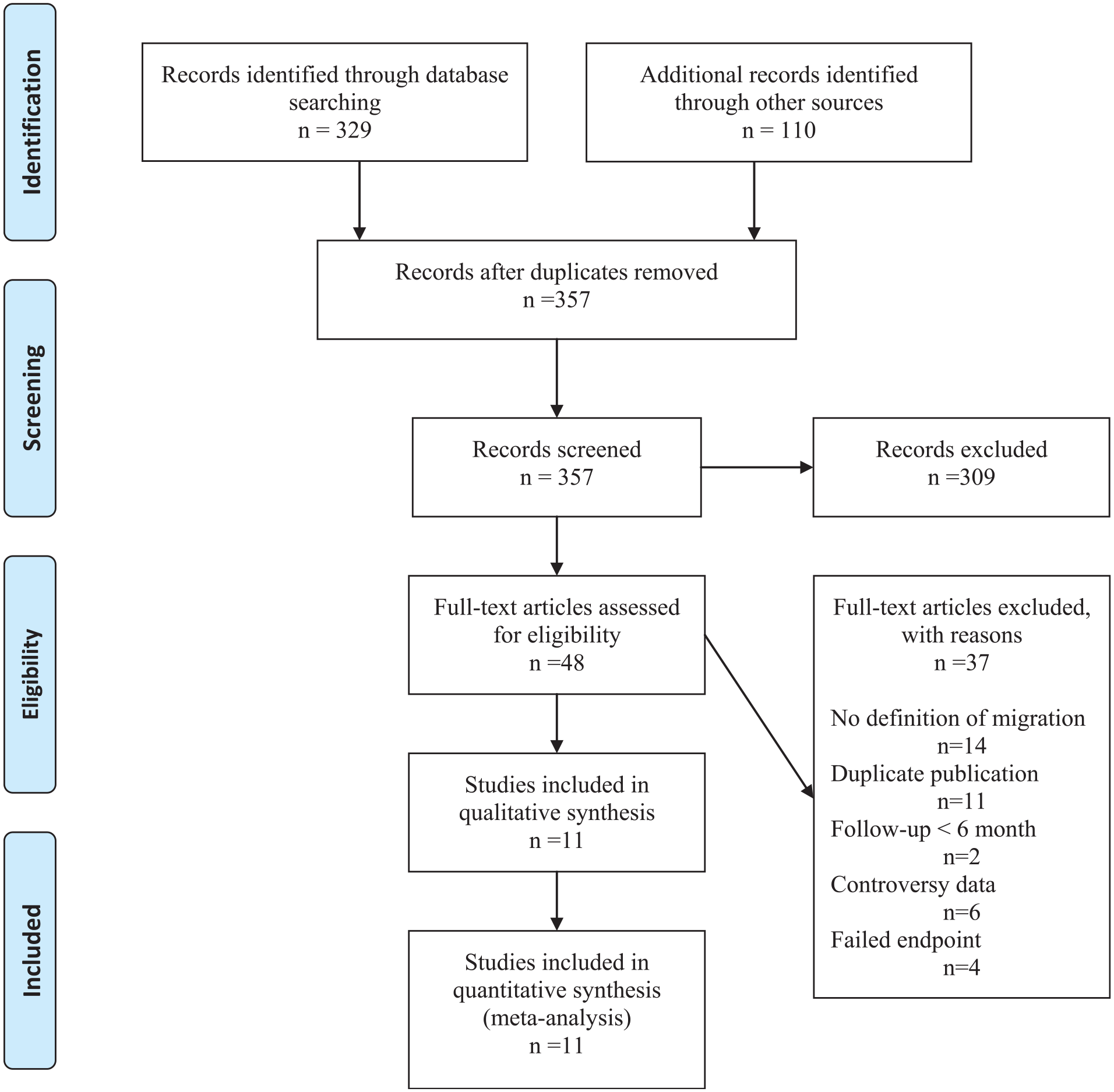
PRISMA 2009 flow diagram.a
Study Characteristics
No randomized controlled trials reporting EM after TEVAR were identified. Predominantly prospective, non-randomized studies were included in the study (63.6%, 7/11); 36.4% (4/11) of the included trials were of a retrospective nature. The pooled analysis included 1783 patients (Table 1). Male gender was predominant (range, 59%–77%), as well as age over 50 years (range, 53.3±20.6–72.2±9.4 years) and the profile of risk factors included AH (range, 41%–98.5%), congestive heart failure (range, 2.4%–27.9%), CAD (range, 7.9%–47%), DM (range, 6%–21%), renal failure/dialysis (range, 0.9%–17.5%), and smoking (range, 21.5%–100%) (Table 1). The indication for TEVAR was TAA, AD, or PAU (11/11, 6/11, and 6/11 of trials, respectively) (Table 2).
Study Characteristics and Demographics.
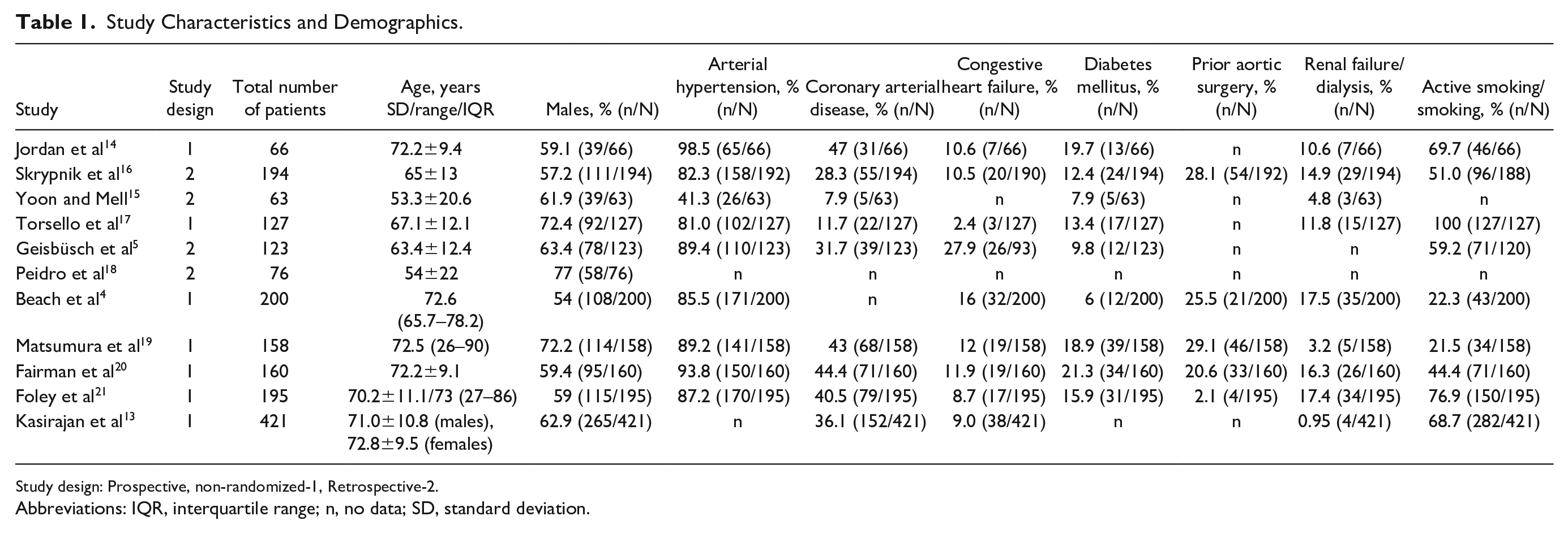
Study design: Prospective, non-randomized-1, Retrospective-2.
Abbreviations: IQR, interquartile range; n, no data; SD, standard deviation.
Indication for Thoracic Endovascular Aortic Repair (TEVAR), Operative Settings, and Length of FU.
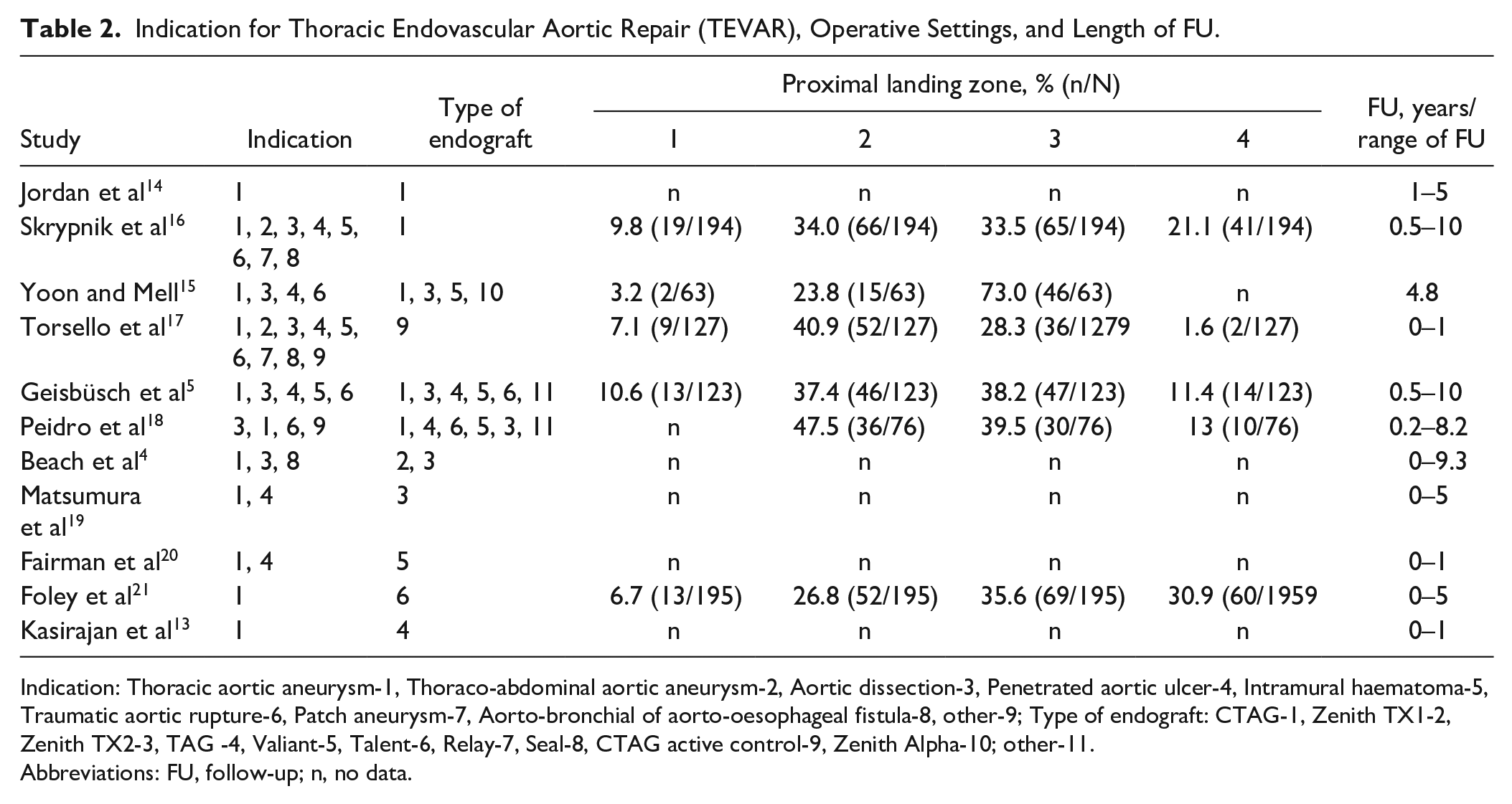
Indication: Thoracic aortic aneurysm-1, Thoraco-abdominal aortic aneurysm-2, Aortic dissection-3, Penetrated aortic ulcer-4, Intramural haematoma-5, Traumatic aortic rupture-6, Patch aneurysm-7, Aorto-bronchial of aorto-oesophageal fistula-8, other-9; Type of endograft: CTAG-1, Zenith TX1-2, Zenith TX2-3, TAG -4, Valiant-5, Talent-6, Relay-7, Seal-8, CTAG active control-9, Zenith Alpha-10; other-11.
Abbreviations: FU, follow-up; n, no data.
The PLZ was predominantly in zones 2–3 and FU was up to 10 years after TEVAR (Table 2).
One type of endograft was used for TEVAR in 63.7% (7/11) of studies, while various types of endografts were used for TEVAR in the remaining 36.4% (4/11). All types of endografts are listed in Table 2. Long-term outcomes were reported in 63.7% (7/11) of studies, while the rest showed outcomes within the mid-term FU (Table 2).
The studies reported migration rates ranging between 0.2% and 11%. The time point of migration was randomly distributed between 8 and 64 months in all landing zones (Table 3). All trials reported close surveillance during FU after TEVAR with CTA after the intervention, predominantly at 6 or 12 months and annually thereafter.
Migration of Endografts.
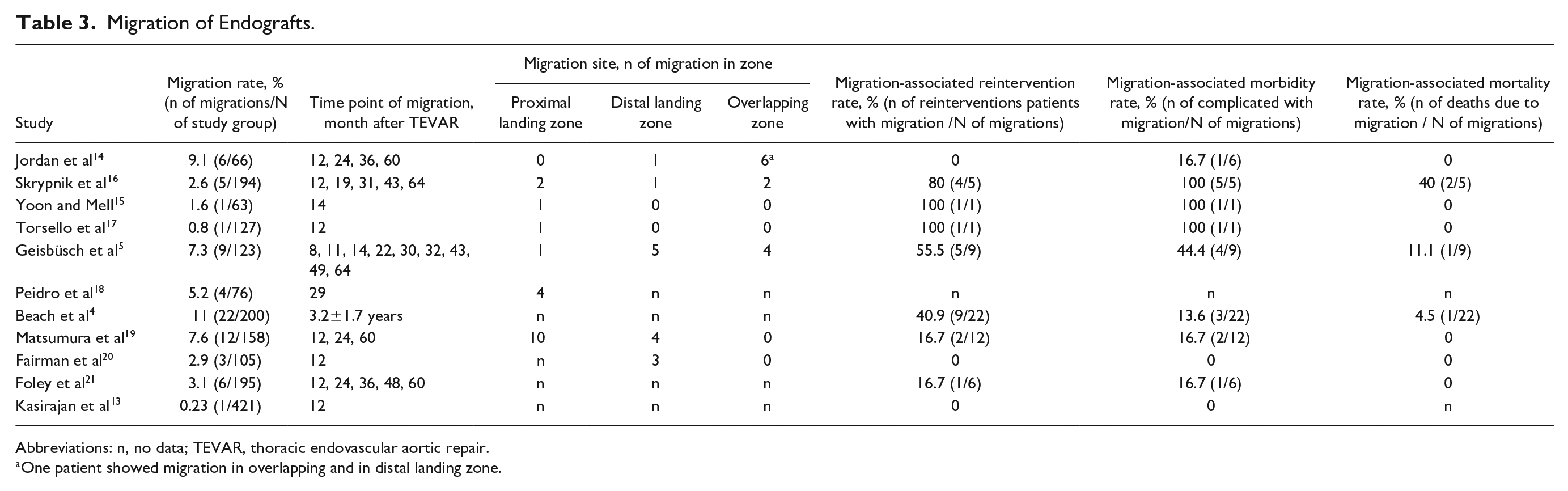
Abbreviations: n, no data; TEVAR, thoracic endovascular aortic repair.
One patient showed migration in overlapping and in distal landing zone.
EM-related morbidity was reported in 8 trials.4,5,14–17,19,21 Two trials reported no EM-related morbidity and 1 trial failed to report the outcomes in this term (Table 3).
Three trials reported no reinterventions during mid-term FU.13,14,20 The trials reporting on long-term FU showed that reintervention rates due to EM ranged between 0% and 100%.4,5,15–17,19,21 The one trial that formally reported late mid-term results with a maximal FU of 1750 days reported 100% (1/1) reintervention in the migration group. 15
The EM-related reinterventions that were performed were predominantly endovascular (82.6%; 19/23): 6 trials reported endolining or proximal/distal extension;4,5,16,17,19,21 2 trials reported FEVAR.4,5 Overall, 4 open conversions in 4 trials were reported.4,5,15,16
The EM-related mortality in the migration group ranged from 0% to 40% (Table 3).
Four trials reported the cause of EM. Skrypnik et al 16 reported EL type II and progression of aortic degeneration as a leading cause of EM. Beach et al 4 reported progression of aortic degeneration and aneurysm sack growth as a leading cause of EM. EM due to unfavorable oversizing and funnel-shaped landing zones was reported by Matsumura et al. 19 Only 1 trial reported statistical grounding as a risk factor of EM (aortic elongation [p=0.003] and TAA [p=0.01]), thus quantitative analysis of the risk factors could not be performed. 5
Risk-of-Bias Assessment
The ROBINS-I tool assessment adapted for non-interventional studies revealed that all trials included in quantitative analyses showed a low risk of bias of confounding, selection of participants, and classification of intervention (Table 4). Moreover, the risk of bias due to deviations from intended interventions, bias in measurement of outcomes, and bias in selection of reported results was also low.
Risk-of-Bias Assessment.
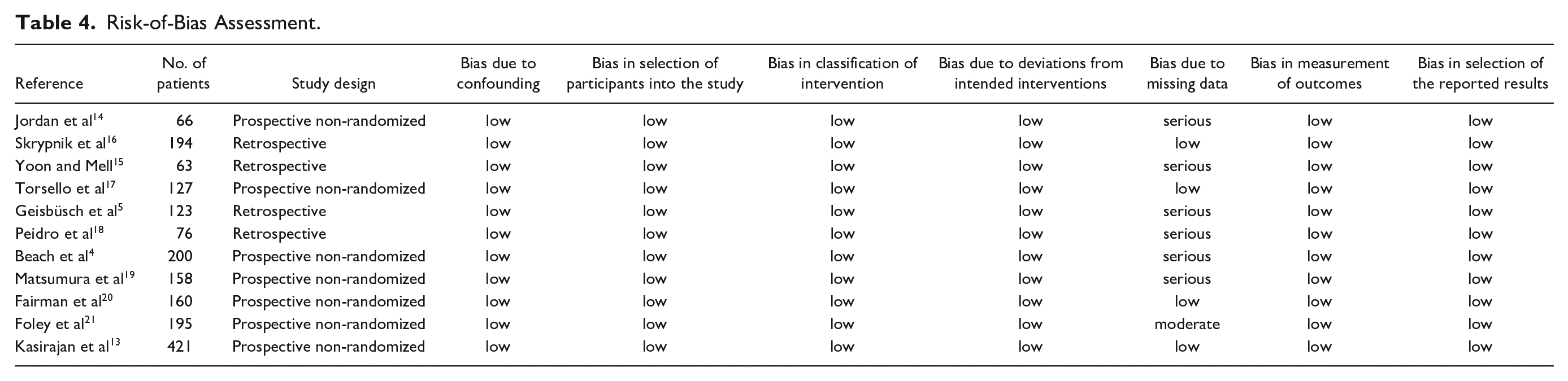
However, bias due to missing data was serious in 54.5% (6/11) of trials, as only a minority of patients achieved maximal FU in the trial (Table 4). In 1 trial, where >50% of patients achieved maximal FU, bias due to missing data was moderate (Table 4). Accordingly, the overall bias of trials included in the analysis was predominantly serious (54.5%, 6/11). Only 36.4% (4/11) of trials presented a low risk of overall bias (Table 4).
Incidence of EM
Eleven trials reported migration rates. The pooled migration rate was 4% (95% CI, 2%–7%; range, 0.2%–11%; I2=82%; Figure 2). The heterogeneity, however, was significant (I2=82, p<0.01), likely due to an inconsistent protocol for assessment of endograft position. Only Geisbüsch et al 5 reported the per-standard assessment using primary and secondary analysis and dual reference point assessment for the proximal and distal ends of the endograft.
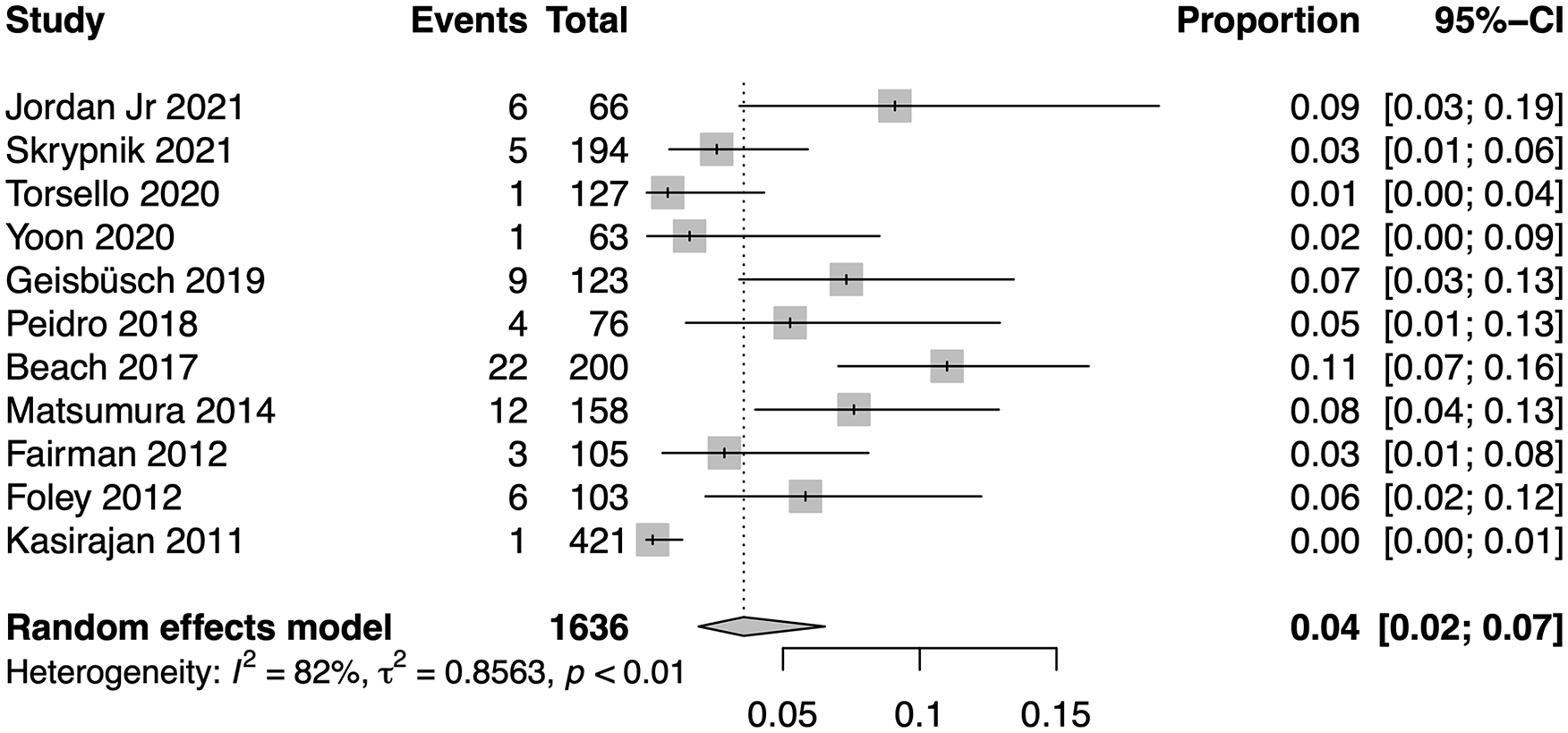
Pooled migration rate. CI, confidence interval.
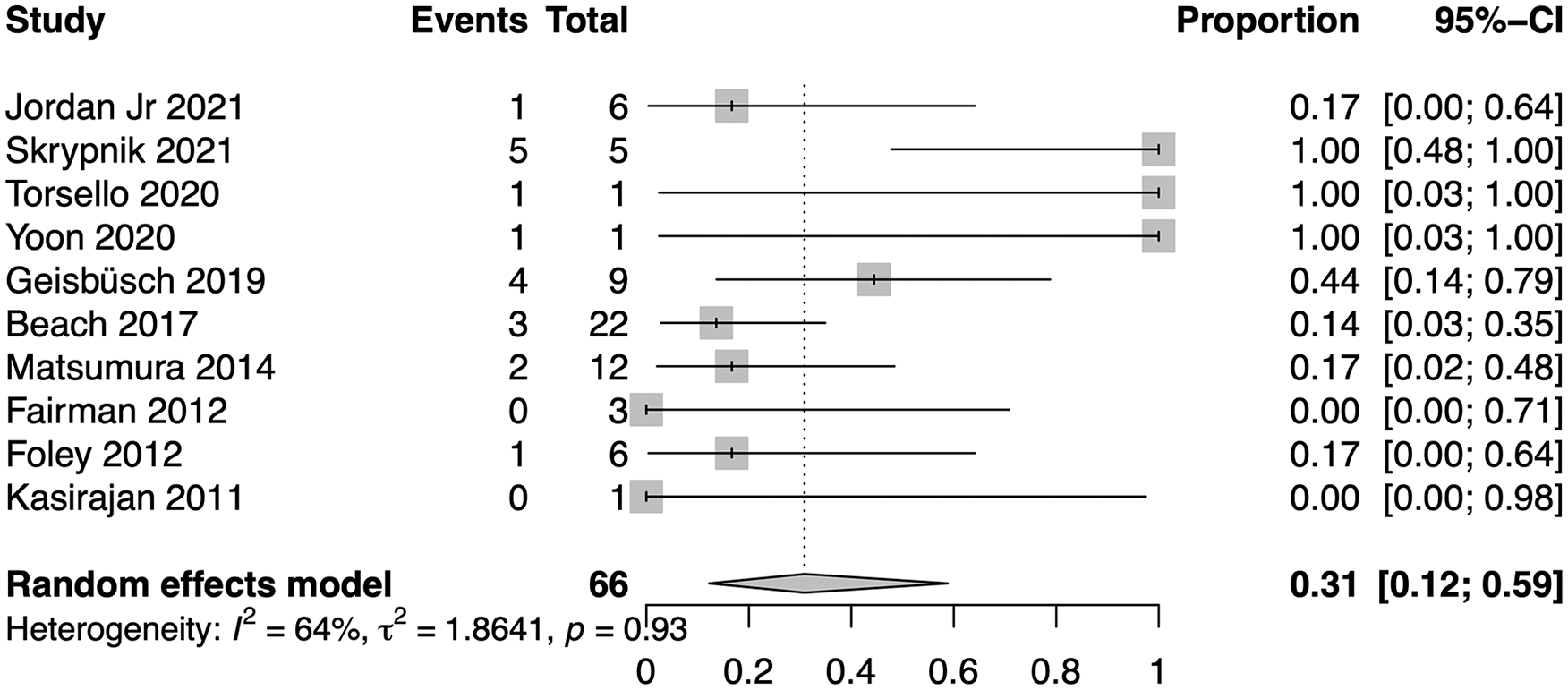
Pooled morbidity rate. CI, confidence interval.
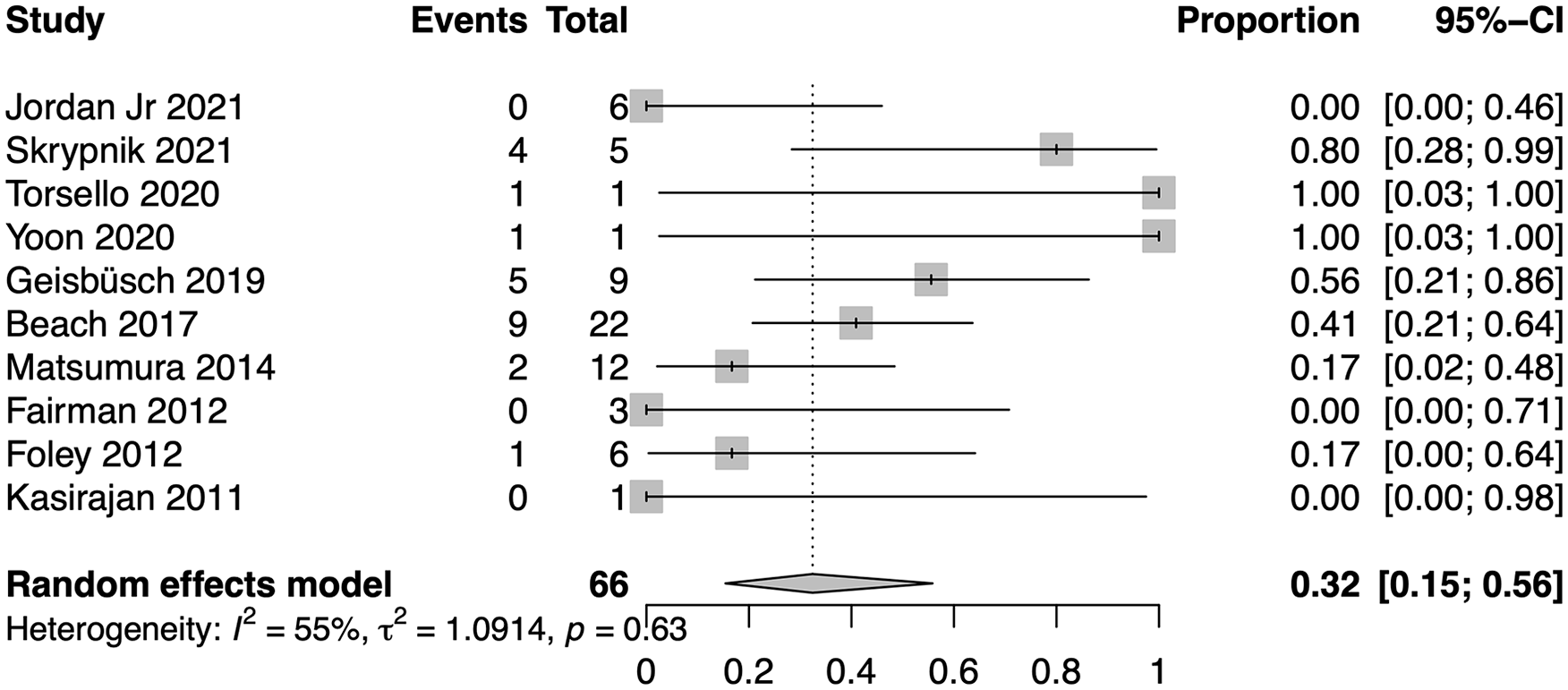
Pooled reintervention rate. CI, confidence interval.
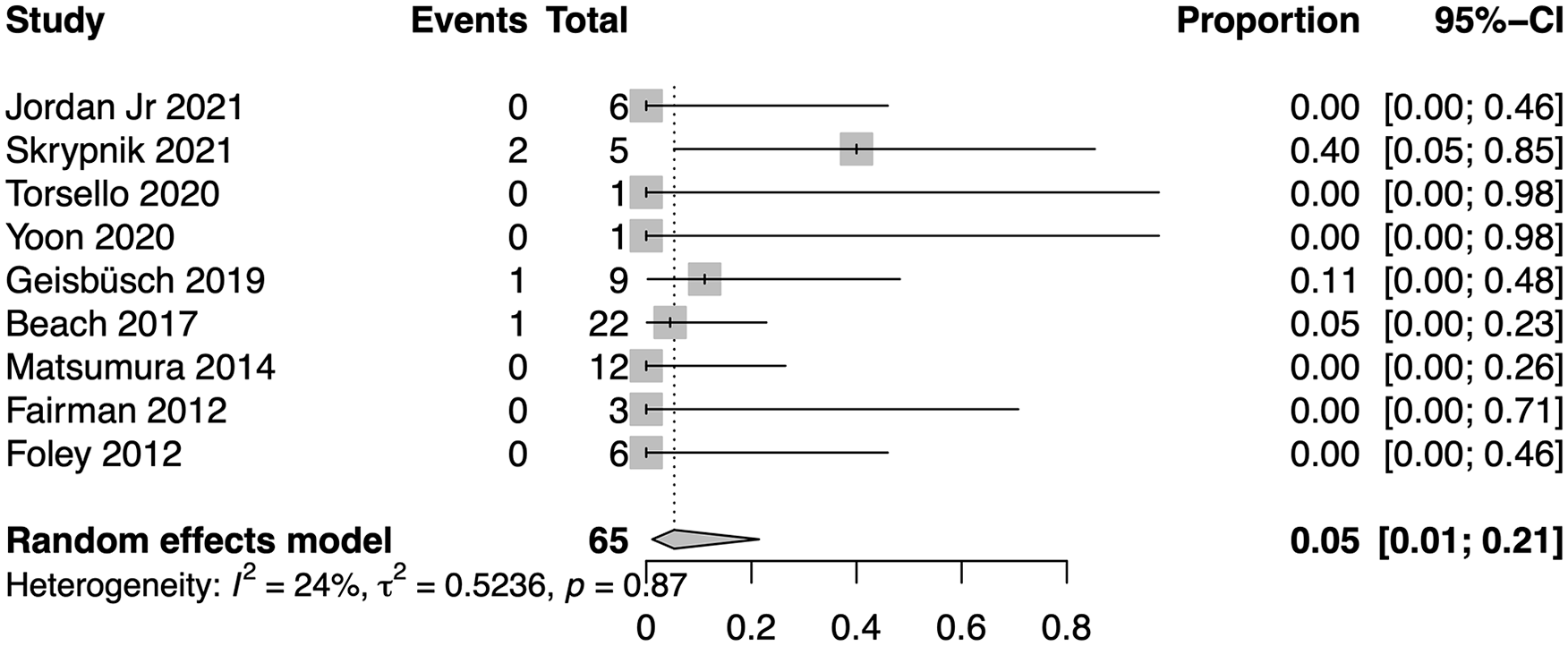
Pooled mortality rate. CI, confidence interval.
EM-Related Morbidity
The morbidity due to EM was reported in 10 trials.4,5,13–17,19–21 The pooled EM-related morbidity rate was 31% (95% CI, 12%–59%; range, 0%–100%; I2=64%; Figure 3), showing moderate heterogeneity of data (I2=64, p=0.93).
EM-Related Reintervention
Ten trials reported on reintervention due to EM.4,5,13–17,19–21 The pooled reintervention rate due to EM was 32% (95% CI, 15%–56%; range, 0%–100%; I2=55%; Figure 4). Similarly, to the pooled EM-related morbidity rate, the data showed moderate heterogeneity (I2=55%, p=0.63).
EM-Related Mortality
Nine trials reported mortality rates due to EM with a pooled mortality rate of 5% (95% CI, 1%–21%; range, 0%–40%; I2=24%; Figure 5).4,5,14–17,19–21 Cumulative data showed low heterogeneity (I2=24%, p=0.87).
Discussion
In this systematic review and meta-analysis, pooled estimates of late endograft migration after TEVAR are available for the first time in a group of 1783 patients presenting 70 EMs.
The pooled migration rates were shown to be 4% (95% CI, 2%–7%). The earliest studies reported EM rates up to 30%. However, these rates were observed with first-generation devices, different methods of assessment of endograft position, and diverse definitions of EM.6,22,23 After publishing the definition of EM in 2010, trials using contemporary endografts reported improved TEVAR performance with EM rates up to 8%.5,19,24
However, very few trials used the standard-based protocol of assessment of endograft position, which includes primary analysis of centerline distances between tips and covered parts of the endograft relative to large aortic branches and secondary analysis relative to anatomical landmarks (eg, calc, surgical clips, etc), when endograft movement >10 mm was suspected. 3 Among the trials included in the current meta-analysis, only Geisbüsch et al 5 reported standard-based assessment using primary and secondary analysis resulting in 7.3% of migrations. Other trials failed to report a standardized protocol for EM diagnosis and showed a predominantly lower incidence of EM of between 0.8% and 5.2%.14,15,17,18,20 The observed high heterogeneity of pooled data (I2=85.6, p<0.01) was thus likely due to the lack of standard-based assessment of endograft position in all trials included in the current study. Accordingly, despite the fact that the current analysis showed very low pooled EM rates, if the standard-based assessment of endograft position was performed more often, higher EM rates might be expected.
The EM and EM-related mortality are associated with ELs.5,15–17 Previously published studies reported substantial EM-related morbidity in the EM group, which was predominantly due to ELs.14,16–18,25 The rate of EM-related morbidity in the EM group ranged from 0% to 100%, reflecting the rarity of EM in study cohorts and the relatively small size of cohorts. The current study reported 31% of pooled EM-related morbidity rates in the EM group (95% CI, 12%–59%; range, 0%–100%; I2=64, p=0.93) in a large cohort, confirming the leading role of ELs in the occurrence of morbidity due to EM.
Although only a few trials focused on risk factors for EM, some anatomical assumptions could be made. Altnji et al 26 used a numeric simulation model and found that a length of the proximal landing zone <15 mm was a risk factor for EM. This was in line with the clinical study of Yoon and Mell, 15 who reported higher rates of EM (2.2%) in the group with a proximal landing length <20 mm compared with >20 mm (0%).
By using mathematical modeling, Liffman et al 27 showed that the risk of EM potentially increases after TEVAR for TAA if the overlapping zone is free of thrombotic material while pressure on the endograft lumen and aneurysm sack are comparable and lateral pulsatile friction increases. These results were consistent with clinical results published by Geisbüsch et al, 5 who identified TAA as a risk factor of EM (p=0.01).
Matsumura et al 19 reported that the quality of PLZ and undersizing were risk factors of migration. Chen et al 28 reported high tortuosity as a risk factor for both EL and EM.
In the current study, pooled analysis of risk factors for EM was impossible due to limitations. However, based on the limited data available, short, reversed funnel PLZ, aortic kinking, and thrombus-free large TAA require attention in future EM analyses.
The current literature reported a wide range of reintervention rates (7%–25%) after TEVAR predominantly due to ELs.29–31 This meta-analysis showed that, despite the rarity of EM, the pooled EM-related reintervention rate (32%; 95% CI, 15%–56%; range, 0%–100%; I2=55%, p=0.63) is higher than the general reintervention rate after TEVAR. Accordingly, based on current data, EM is considered an endograft-related complication that is highly likely to require reintervention.
Endograft-related complications are rare but associated with substantial mortality. For example, endograft infection after TEVAR showed an incidence <2% with a mortality rate beyond 50%. 32 Retrograde type A aortic dissection has an incidence of 2.5% associated with a 37% mortality rate. 33 The current meta-analysis showed that, compared with other device-related complications, EM is associated with reduced mortality (5%, 95% CI, 1%–21%), likely due to slow progression, close CTA-FU after TEVAR, and predominantly endovascular treatment of EM and its complications.
The herein presented systematic review and meta-analysis has several limitations. The analysis was limited by the heterogeneous designs (ie protocol, EM assessment) of the included studies as highlighted above. The failed long-term FU data in all studies included in the analysis made the relationship between EM and length of FU inconclusive. Moreover, a pooled analysis of anatomical risk factors was impossible due to missing data.
Despite these limitations, the current meta-analysis for the first time provides reference estimates of EM after TEVAR for further studies, highlighting the need for standard-based assessment of endograft position for accurate reporting of TEVAR outcome, early detection of EM, and prophylaxis of its sequelae.
Conclusions
For the first time, this meta-analysis provides pooled reference estimates of EM after TEVAR. Thus, the results hold the potential to further characterize EM after TEVAR. The clinical relevance of EM is underlined by its association with high rates of endoleak-related morbidity, reintervention, and mortality. Close standardized surveillance after TEVAR for early detection of EM and prophylaxis of its sequelae is essential.
Footnotes
Authors’ Note
This study was presented on CX Aortic Vienna, October 5–7, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
