Abstract
Objective:
Carotid artery restenosis can occur after both carotid artery stenting (CAS) and carotid endarterectomy (CEA). This systematic review and meta-analysis aim to determine which revascularization technique, CAS, or CEA, is superior for treating primary carotid restenosis, irrespective of the initial revascularization method used.
Design:
Systematic review and meta-analysis.
Methods:
MEDLINE, EMBASE, and Cochrane Central Register of Controlled Trials (CENTRALs) databases were searched for eligible studies on December 19th, 2023. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was followed. Primary endpoint was the occurrence of transient ischemic attack (TIA) or any stroke. Secondary endpoints were technical success, death within 30 days, myocardial infarction (MI), local complications, cerebral hyperperfusion syndrome (CHS), cranial nerve injury (CNI), dys-/arrythmia, secondary restenosis, repeat revascularization, and long-term survival. Results were adjusted for symptomatic status and primary treatment strategy.
Results:
Nineteen studies comprising 10,171 procedures in 10,041 patients were included. Baseline characteristics were comparable between groups. Main findings were (1) No difference in primary outcome; however, if adjusted for symptomatic status the rate of TIA/any stroke is higher (OR: 2.05, 95% CI: 1.29–3.27, p < 0.01) after CEA compared to CAS; (2) Significant higher rate of MI (OR: 1.85, 95% CI: 1.19–2.86, p < 0.01) after CEA; (3) Besides CNI, which appears to be commonly temporary and occurred only after CEA (7.56%, 95% CI: 4.21%–13.22%), no significant differences in other secondary endpoints were observed between groups. Long-term risk of secondary restenosis was similar between CEA compared to CAS (OR: 0.98, 95% CI: 0.39–2.49, p = 0.95); (4) Correction for the index procedure did not affect conclusions.
Conclusion:
Based on limited-quality studies, mostly retrospective and nonrandomized in design, both CAS and CEA represent feasible treatment approaches for patients with primary restenosis, with comparable primary outcome between the two groups. However, based on the obtained results, CAS appears to be preferable. Patients should be critically evaluated in a multidisciplinary team and further research is desirable.
Clinical Impact
This review expands on previous studies by incorporating a larger patient cohort and more recent literature while offering new insights into restenosis. Unlike earlier research, this study uniquely evaluates first repeat revascularization outcomes (CAS and CEA) independently of the initial procedure, suggesting that patient and plaque characteristics might be more influential than the primary technique. Sensitivity analysis confirmed this, as stratification by index procedure did not alter conclusions. Although lower TIA/stroke and mortality rates were observed in CAS-treated patients, these findings were not statistically significant in the overall group. These results may help guide clinical decision-making for optimal restenosis management.
Keywords
Clinical Relevance
With this systematic review and meta-analysis, the outcome of repeat revascularization for asymptomatic and symptomatic primary carotid restenosis after previous carotid artery stenting (CAS) or carotid endarterectomy (CEA) is investigated. Outcomes were evaluated irrespective of the initial revascularization method. Using this approach, insights in the risk of treating carotid restenosis should be gained and more easily translated to clinical practice. Primary outcome is the incidence of transient ischemic attack (TIA) or any stroke. Secondary outcomes are technical success, death within 30 days, myocardial infarction (MI), local complication, cerebral hyperperfusion syndrome (CHS), cranial nerve injury (CNI), dys-/arrythmia, secondary restenosis, repeat revascularization, and long-term survival. In clinical practice, the primary intervention for carotid stenosis is crucial when determining the optimal strategy for managing a primary restenosis. By correcting for the primary intervention in the outcomes, confounding effects of the primary intervention were considered.
The current guidelines from the Society for Vascular Surgery leave room for debate regarding the most appropriate treatment for primary restenosis. Their recommendations are partly based on a 2018 systematic review and meta-analysis of restenosis after CEA by Texakalidis et al., as well as cohort studies examining restenosis after CAS. This review incorporates more recent studies and includes a larger patient population, providing a more comprehensive evidence base that may enhance decision-making in clinical practice. Importantly, the results of this review demonstrate that the primary revascularization technique does not influence the outcome of the secondary revascularization technique.
Introduction
In accordance with guidelines issued by the Society for Vascular Surgery (SVS), symptomatic patients with carotid stenosis of ≥50% and selected asymptomatic patients with ≥70% stenosis are eligible candidates for surgical or endovascular treatment, combined with best medical treatment (BMT). Although CEA, representing surgical treatment, is the current gold standard, CAS, representing endovascular treatment, could be considered for selected patients. 1
One complication that can occur after carotid revascularization is restenosis. Criteria for diagnosing primary restenosis after CAS are not well-established hindering direct comparison with CEA. Moderate primary restenosis (≥50%) is significantly more common after CAS compared to CEA during a follow-up period of up to 5 years. However, there is no significant difference in the rate of severe primary restenosis (≥70%) after CAS compared to CEA. 2 Severe primary restenosis (>70%) is associated with an inherent risk of any ipsilateral stroke after CEA. No increased rate of ipsilateral stroke was found after CAS during a mean follow-up period of 50 months. 3
Both the SVS and European SVS (ESVS) guidelines recommend treating patients with recurrent TIA or stroke with ipsilateral 50% to 99% restenosis who were initially treated by CEA. CAS or redo-CEA is advised within 14 days of symptom onset. The choice of treatment should be discussed in a multidisciplinary team and align with patient’s, local surgeon’s and interventionist’s preference.1,4 Additionally, guidelines issued by the SVS recommend an endovascular treatment first, with drug-eluting techniques gaining interest, in patients with symptomatic primary restenosis after CAS. 1
Patients with asymptomatic primary restenosis would probably not benefit from revascularization since the incidence of ipsilateral stroke is low in this specific category of patients. Of all late (i.e., non-periprocedural) ipsilateral strokes during follow-up, 97% of the CAS and 85% of the CEA had no evidence of significant restenosis or occlusion. 3 Nevertheless, the latter patient category should be selected for repeat revascularization in a manner similar to that of primary asymptomatic patients, while aggressive BMT is advised for the former. 1
The most suitable treatment approach for managing primary carotid restenosis remains a highly debated topic with evidence-based recommendations lacking. Various different treatment options are applied, including CAS and CEA. This systematic review and meta-analysis aim to provide an overview of the current literature to evaluate the differences in terms of safety and outcome of secondary revascularization in patients with symptomatic and asymptomatic primary carotid artery restenosis after previous CAS or CEA.
Methods
A systematic review and meta-analysis of the literature were conducted following the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 5 The review was registered in the International Prospective Register of Systematic Reviews (PROSPERO) database (ID: CRD42024543598).
Eligible Criteria
Eligible studies where those reporting original data of patients with primary carotid artery restenosis after prior CAS or CEA (population) who underwent repeat endovascular revascularization (CAS or redo-CAS; intervention) and/or repeat open revascularization (CEA, or redo-CEA; control group) and reported on at least TIA/stroke within 30 days after the procedure (outcome). Eligible study designs included randomized controlled trials as well as prospective and retrospective cohort studies. Exclusion criteria were (1) duplicates; (2) absence of carotid artery disease; (3) no significant restenosis (<50%); (4) absence of repeat revascularization; (5) revascularization strategy other than CEA or CAS; (6) lack of subgroup analysis for each repeat revascularization technique used, resulting in pooled results; (7) no reported endpoints; (8) case-report or series with <10 cases; (9) language other than English, German, or Dutch; (10) conference abstract, opinion letter, review; (11) full-text not available.
Search Strategy
A systematic literature search of four electronic databases [MEDLINE, EMBASE, and Cochrane Central Register of Controlled Trials (CENTRALs); all inception to December 19, 2023] was conducted. The search strategy included medical subject heading terms and free-text words related to carotid artery restenosis and repeat revascularization (CAS or CEA) after prior CAS or CEA. Only studies published in 2008 or later were included since in 2008 the first SVS guideline regarding management of extracranial carotid disease was published. 6 The full search strategy can be found in Supplemental Table SI.
Study Selection
The title and abstracts of articles that met the search criteria were screened followed by assessing eligibility based on full text. Related reviews and selected studies were manually cross-referenced to identify any missing articles. The studies cover an inclusion period from 1985 to 2021.7,8 All steps of the study selection were performed by two independent reviewers. Adjudication was resolved by discussion with a third reviewer.
Data Extraction
Several predefined variables were extracted, including study characteristics (year of publication, study design, inclusion period), patient characteristics (age, sex, comorbidities, risk factors, presence of hostile neck), primary treatment strategy (CAS or CEA), indication for repeat revascularization (asymptomatic or symptomatic), interval until primary restenosis, and/or repeat revascularization, total number of repeat revascularization performed, follow-up time, and imaging modality used for follow-up.
The primary endpoint was any TIA/stroke within 30 days. Secondary endpoints were technical success, death within 30 days, MI, local complications (e.g., haemorrhage), CHS, CNI, dys-/arrythmia, secondary restenosis, second repeat revascularization, and long-term survival. Two independent reviewers performed data extraction. Consensus of correctness of data retrieval was obtained by discussion with a third reviewer.
Critical Appraisal
Methodological quality of the selected articles was assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for cohort studies or randomized controlled trials. 9 The tool for cohort studies consists of 11 questions regarding study design. Each question can be answered with “yes,” “no,” “unclear,” or “not applicable.” The answer “yes” indicates greater quality. This checklist addresses selection bias, validity, and reliability of methodologies used to evaluate exposure and outcomes, confounding, study duration, reverse causality, suitability of statistical analysis, and correction for significant confounders. The tool for randomized controlled trials consists of 13 questions and addresses internal validity, bias related to participant retention, and statistical conclusion validity. These tools allow users to include or exclude articles depending on their overall quality. A study was excluded from the analysis if it had five or more “no” or “unclear” quality categories. Two reviewers performed quality assessment. Consensus of correctness of quality assessment was obtained by discussion with a third reviewer.
Statistical Analysis
Study and baseline characteristics were displayed as mean [± standard deviation (SD)] for normally distributed data, as median (25th to 75th percentile) for nonparametric data, and as frequencies [percentages (%)] for a number of observations.
The effect of estimates of the primary and secondary outcomes were presented per intervention type (i.e., CAS and CEA) as proportions with corresponding 95% confidence interval (95% CI) and were compared using the Chi-squared test. In addition, (adjusted) odds ratios with corresponding 95% CI were calculated. A summary effect estimate was calculated if ≥2 studies could be pooled. If no summary effect estimate could be calculated, a qualitative synthesis was performed. Statistical heterogeneity was regarded substantial if I2 > 50%. Publication bias was assessed through funnel plots for the primary outcome.
A priori defined meta-regression analysis was conducted to assess whether preoperative symptomatic status confounded the effect of the intervention type on the primary and secondary outcomes. In addition, sensitivity analyses were performed to assess whether the index procedures (i.e., primary CAS or CEA) altered the conclusion of the outcomes of the procedure (i.e., CAS or CEA) after restenosis. These sensitivity analyses included meta-analysis of primary and secondary outcomes per index procedure as well as performing the meta-regression analysis stratified by index procedure. All analyses, including the meta-regression analysis, were performed in R (R Core Team, 2024, version 4.3.3, meta-package) using a random effects model with the DerSiminonian–Laird estimator. p < 0.05 was considered statistically significant in all analyses.
Results
A total of 1335 studies were identified. After exclusion of 363 duplicate articles, 972 remained for screening by title and abstract. Forty-six studies underwent full-text assessment for eligibility, resulting in 18 studies being included in this review.7,8,10–25 Five articles were excluded due to potential overlapping data.26–30 Additionally, one study was identified and included after cross-referencing. 31 A detailed flowchart of the selection process, including reasons for exclusion, is presented in Figure 1. No studies were excluded after critical appraisal (Supplemental file II).
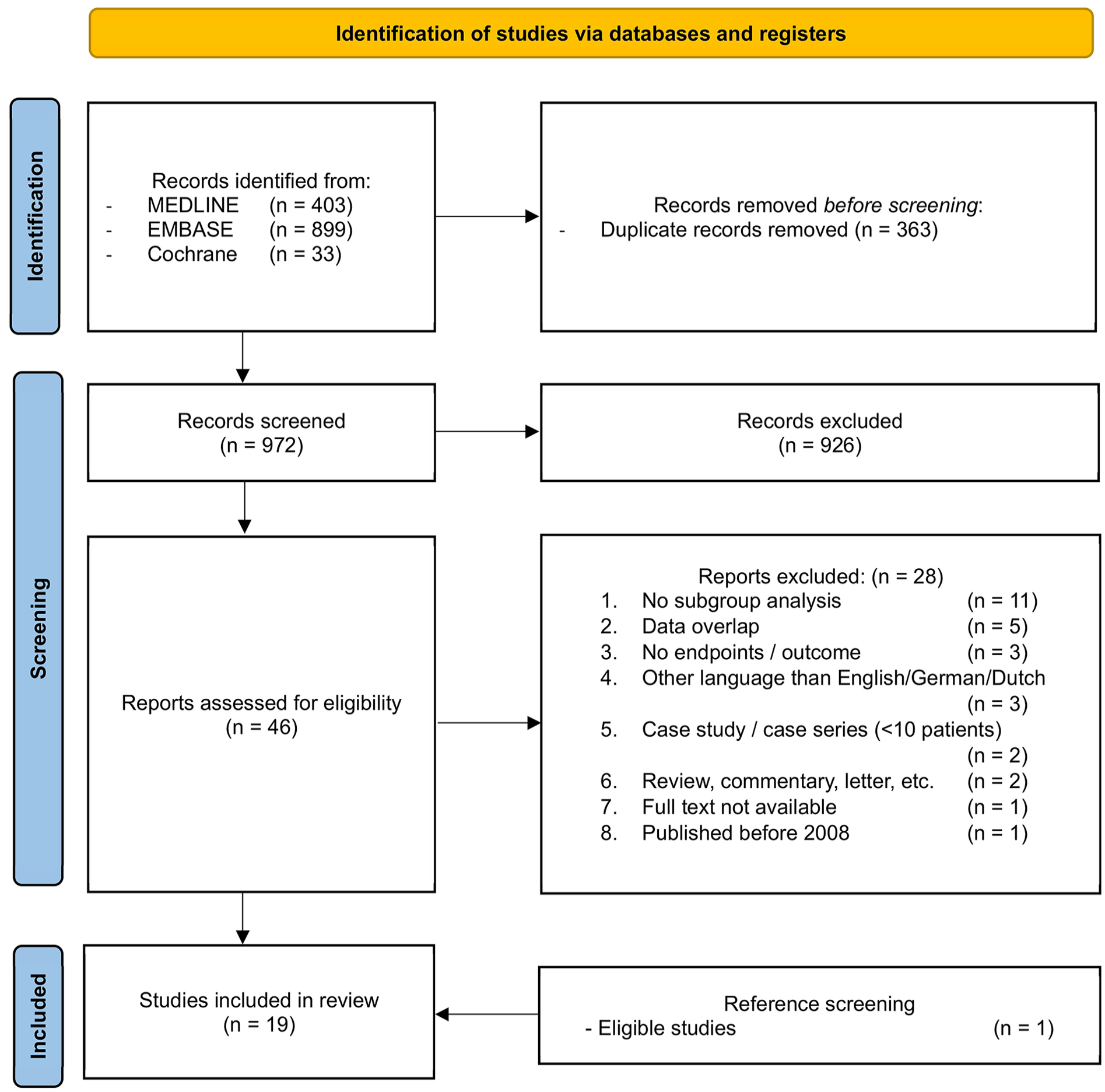
PRISMA flow chart of systematic literature search for studies evaluating repeat revascularization in patients with recurrent carotid stenosis.
Study Characteristics
Since 2008 one blind assessed clinical randomized trial 8 and 18 retrospective cohort studies7,10–25,31 have been published. A total of 10,041 patients underwent 10,171 procedures. Four studies used the same database of the Vascular Quality Initiative, but inclusion periods did not overlap.12,13,16,18 The patient population of four studies did not completely match the in- and exclusion criteria; therefore, only partial data could be retrieved (Tables 1–4).8,11,21,22
Summary of Study Characteristics of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
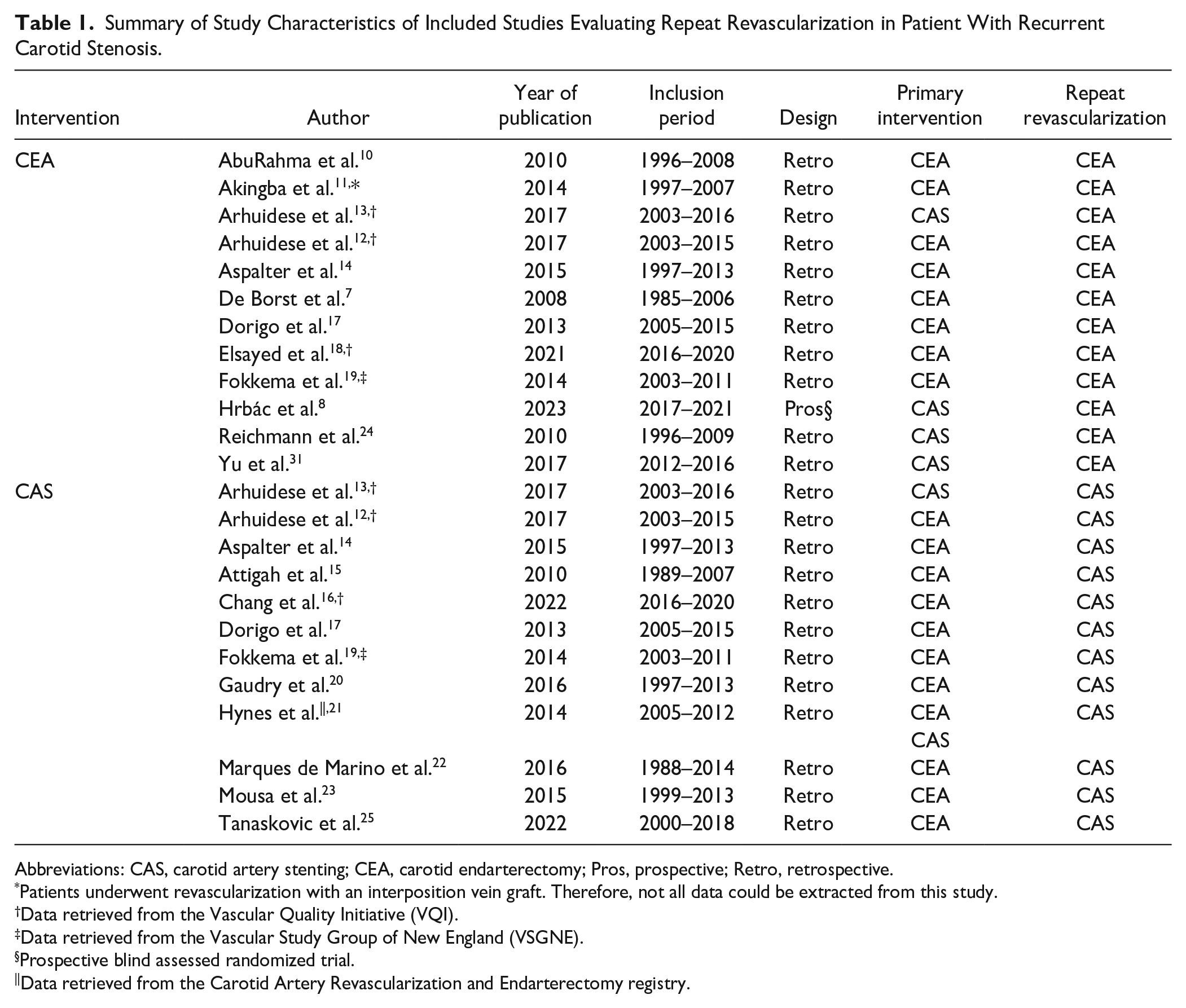
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; Pros, prospective; Retro, retrospective.
Patients underwent revascularization with an interposition vein graft. Therefore, not all data could be extracted from this study.
Data retrieved from the Vascular Quality Initiative (VQI).
Data retrieved from the Vascular Study Group of New England (VSGNE).
Prospective blind assessed randomized trial.
Data retrieved from the Carotid Artery Revascularization and Endarterectomy registry.
Summary of Patient Characteristics of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
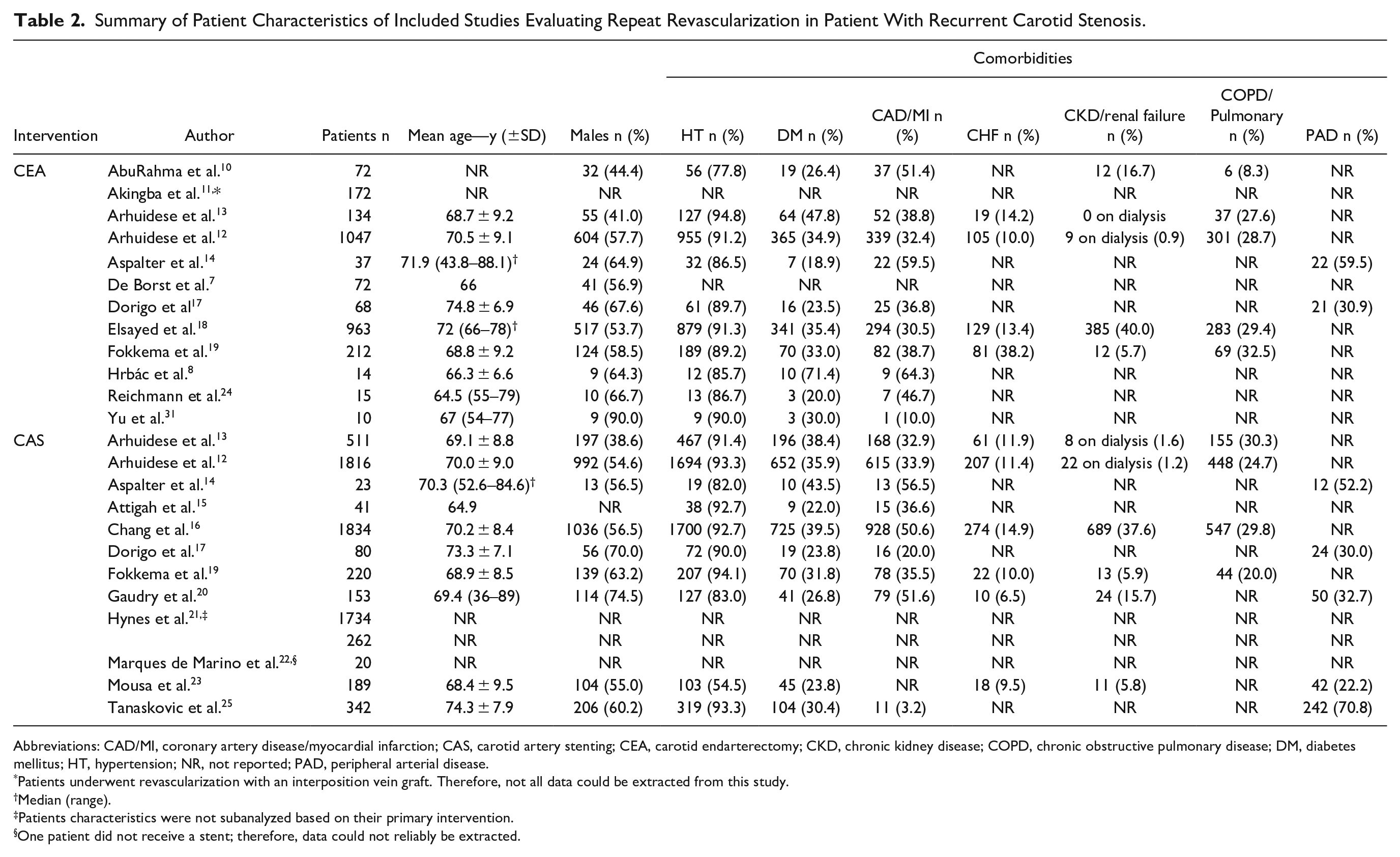
Abbreviations: CAD/MI, coronary artery disease/myocardial infarction; CAS, carotid artery stenting; CEA, carotid endarterectomy; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; HT, hypertension; NR, not reported; PAD, peripheral arterial disease.
Patients underwent revascularization with an interposition vein graft. Therefore, not all data could be extracted from this study.
Median (range).
Patients characteristics were not subanalyzed based on their primary intervention.
One patient did not receive a stent; therefore, data could not reliably be extracted.
Summary of Risk Factors of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
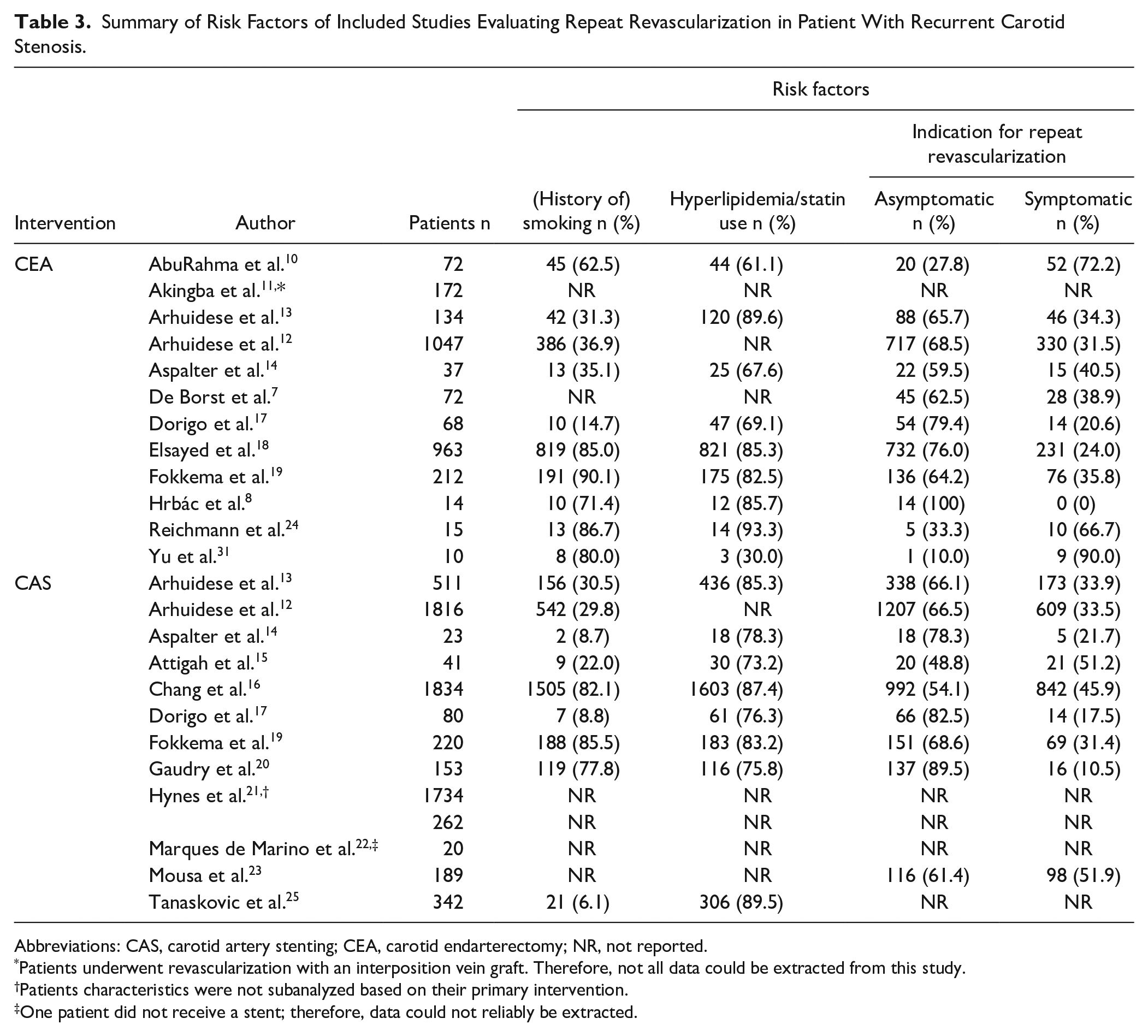
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; NR, not reported.
Patients underwent revascularization with an interposition vein graft. Therefore, not all data could be extracted from this study.
Patients characteristics were not subanalyzed based on their primary intervention.
One patient did not receive a stent; therefore, data could not reliably be extracted.
Summary of Primary Outcome of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
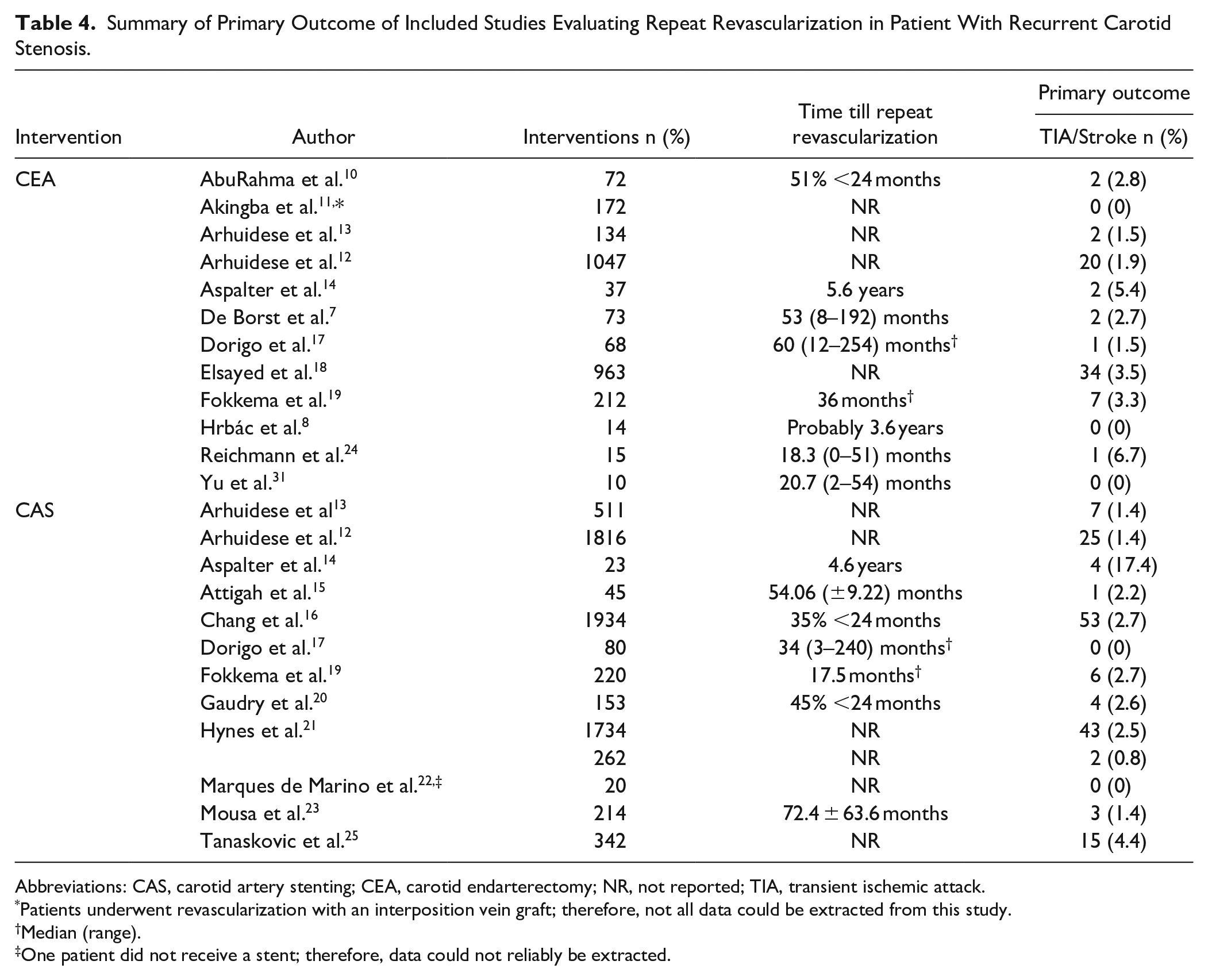
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; NR, not reported; TIA, transient ischemic attack.
Patients underwent revascularization with an interposition vein graft; therefore, not all data could be extracted from this study.
Median (range).
One patient did not receive a stent; therefore, data could not reliably be extracted.
Seven CEA studies8,10,12,13,17–19 and four CAS studies stratified outcomes by preoperative symptomatic status.12,13,16,19 Technical success was only applicable after CAS, which was reported in all studies regarding CAS, expect two studies.21,25
Follow-up time varied between 1.0 to 5.6 years after CAS and 1.0 to 6.2 years after CEA.8,13,14 Main modality for follow-up imaging was duplex ultrasound. Other modalities used were computed tomography angiography, magnetic resonance angiography, and catheter angiography. Lost to follow-up was poorly reported (Table 5).
Summary of Follow Up of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
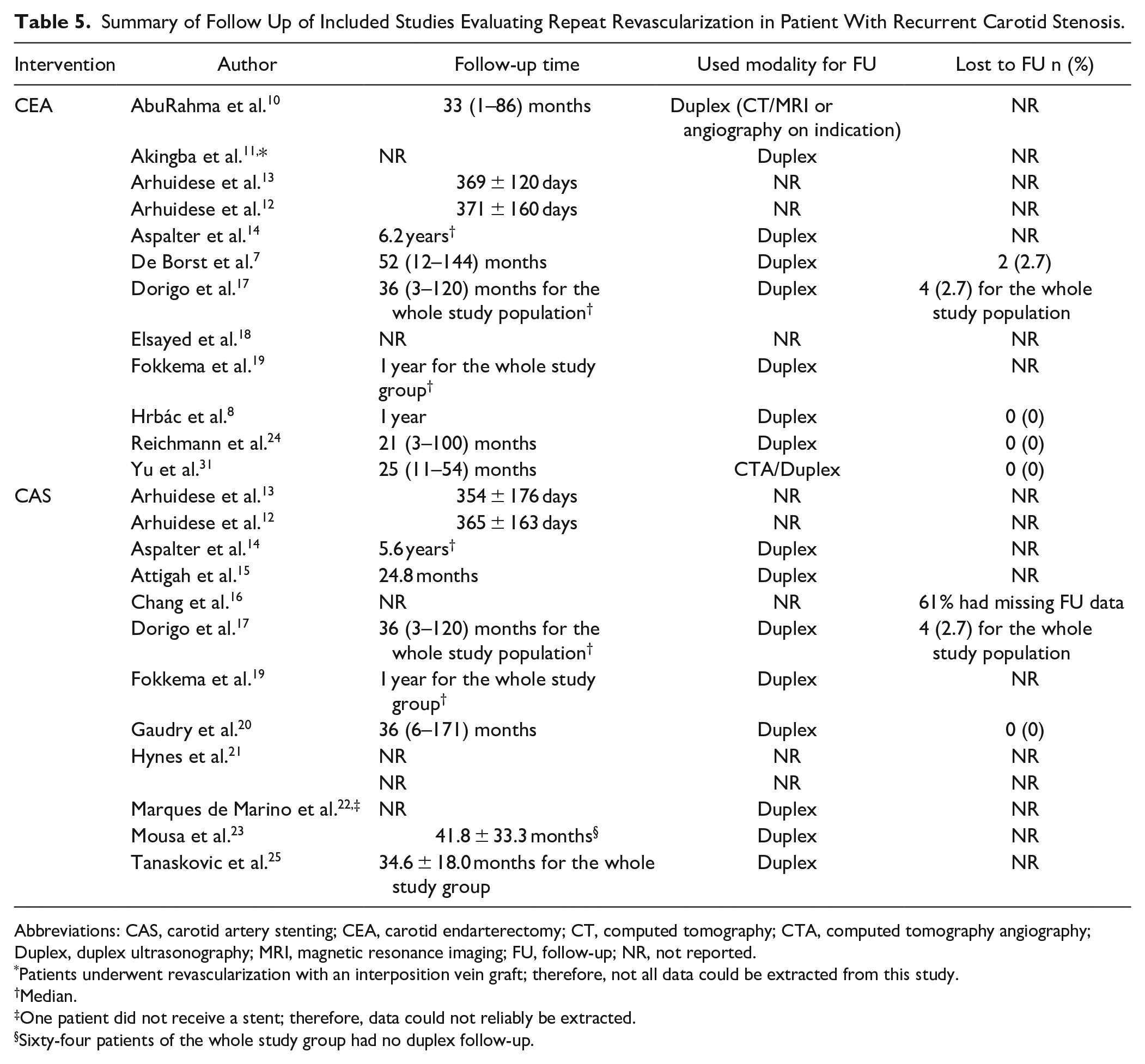
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; CT, computed tomography; CTA, computed tomography angiography; Duplex, duplex ultrasonography; MRI, magnetic resonance imaging; FU, follow-up; NR, not reported.
Patients underwent revascularization with an interposition vein graft; therefore, not all data could be extracted from this study.
Median.
One patient did not receive a stent; therefore, data could not reliably be extracted.
Sixty-four patients of the whole study group had no duplex follow-up.
Patient Characteristics
Out of 10,041 patients, 7225 underwent CAS and 2816 CEA as the repeat revascularization technique. Fifteen studies reported the sex distribution of the included patients (55.3% vs 55.6% male in CAS and CEA groups, respectively).7,8,10,12–14,16–20,23–25,31 Fifteen studies reported preoperative symptomatic status of the included patients (62.6% vs 69.4% asymptomatic; 37.9% vs 30.7% symptomatic in CAS and CEA groups, respectively).7,8,10,12–20,23,24,31 Baseline characteristics and risk factors were comparable in both (Tables 2 and 3).
Primary and Secondary Outcomes
Outcomes Within 30 Days
The primary outcome TIA/stroke occurred in 2.54% (95% CI: 1.98–3.27) of cases after repeat revascularization. TIA/stroke following CAS occurred in 2.42% of cases (95% CI: 1.61–3.64) and following CEA in 2.75% cases (95% CI: 2.02–3.75%; OR: 1.48; 95% CI: 0.93–2.36; p = 0.10) (Figure 2, Tables 4 and 6). In the included trial, none of the 14 patients experienced a TIA or stroke following CEA with stent retrieval. 8
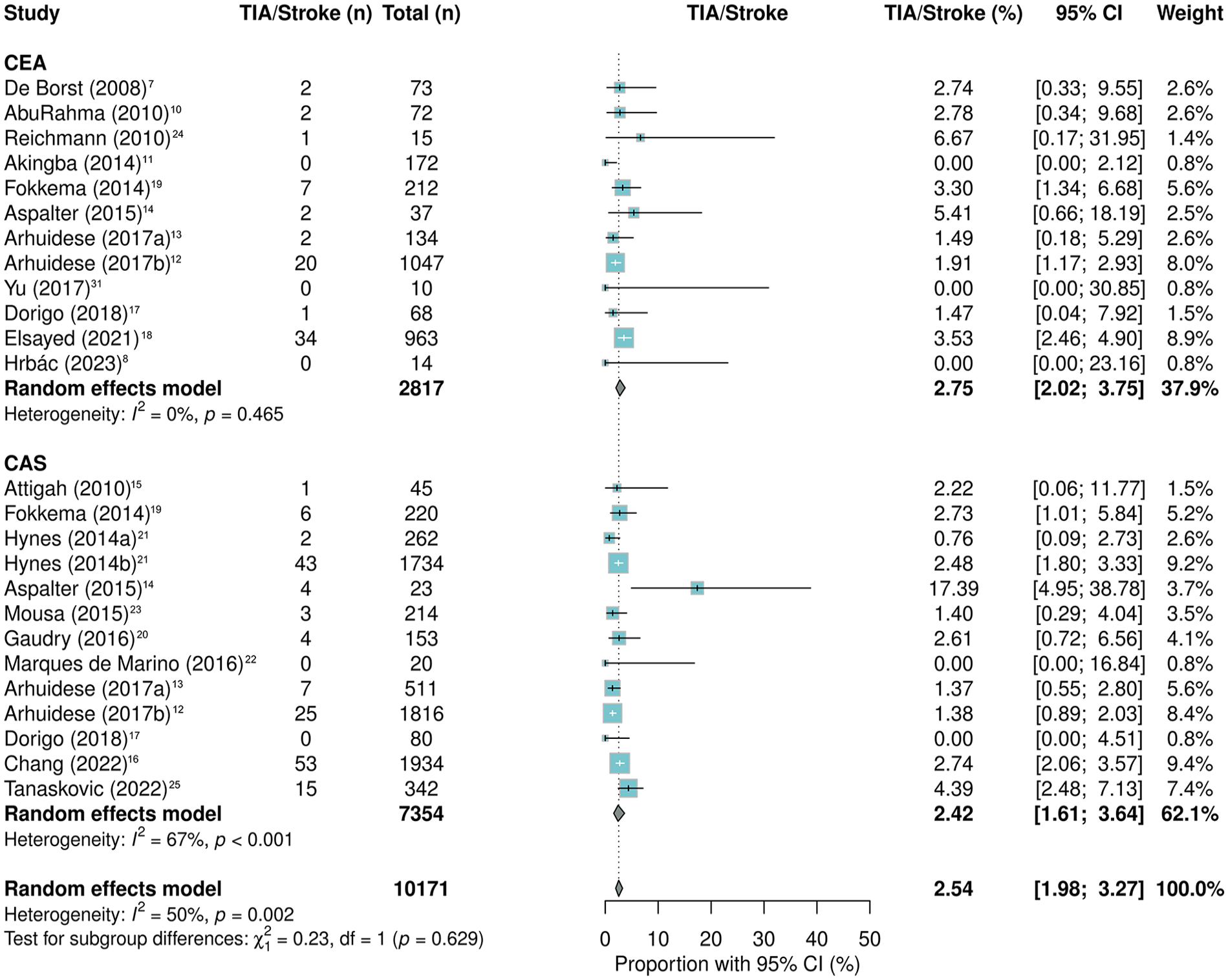
Forrest plot for any TIA/stroke after CEA and CAS in patients with restenosis after prior carotid intervention.
After CAS, 0.62% (95% CI: 0.45–0.84) died versus 1.19% (95% CI: 0.62–2.28; OR: 2.14; 95% CI: 0.96–4.75; p = 0.06) after CEA. MI following CAS occurred in 0.78% of cases (95% CI: 0.54–1.12) and in 1.61% after CEA (95% CI: 1.19–2.18; OR: 1.85; 95% CI: 1.19–2.86; p < 0.01) (Figures 3 and 4, Tables 6 and 7). The funnel plot did not show signs of publication bias (Supplemental Figures SI–II).
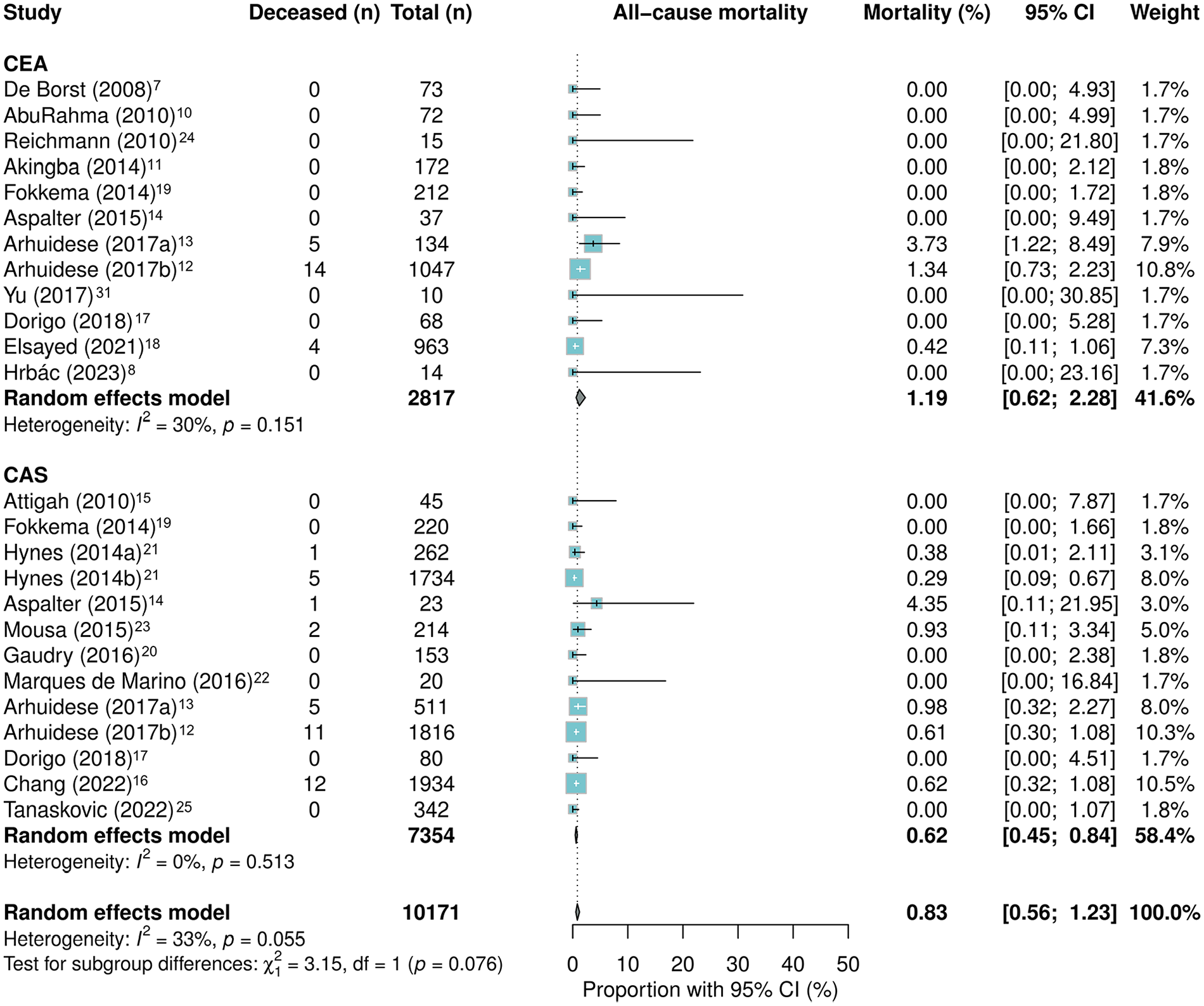
Forrest plot for all-cause mortality after CEA and CAS in patients with restenosis after prior carotid intervention.
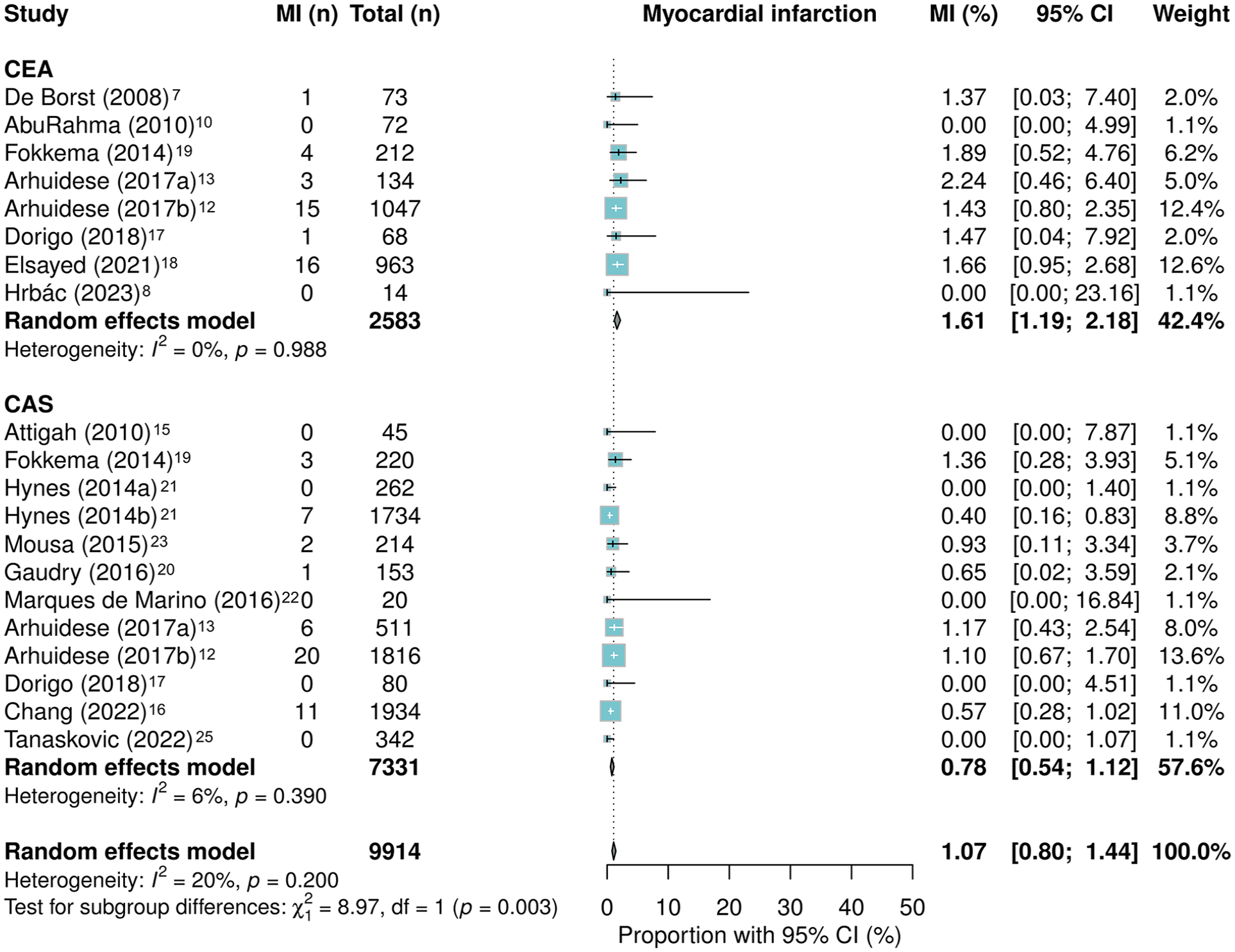
Forrest plot for myocardial infarction after CEA and CAS in patients with restenosis after prior carotid intervention.
Summary of Outcome of CEA Compared to CAS After Prior Carotid Intervention for Carotid Stenosis.
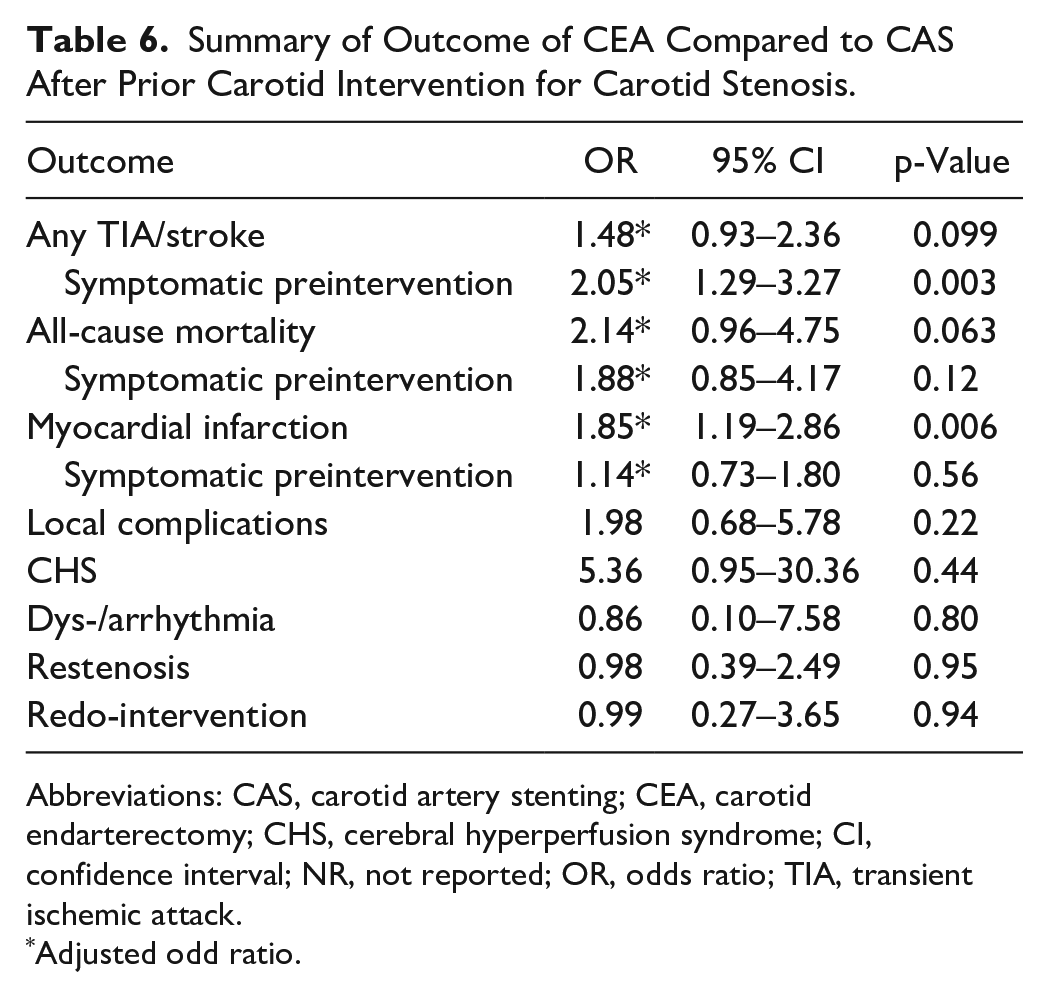
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; CHS, cerebral hyperperfusion syndrome; CI, confidence interval; NR, not reported; OR, odds ratio; TIA, transient ischemic attack.
Adjusted odd ratio.
In prior symptomatic patients, TIA/stroke occurred in at least 54 (3.2%) patients after CAS and 28 (3.7%) after CEA. In these groups, at least 17 (1.0%) and 8 (1.1%) patients died, respectively. MI occurred in at least 17 (1.0%) and 12 (1.6%) patients after CAS and CEA, respectively. The meta-analysis demonstrated that preoperative symptomatic patients carry a significantly higher TIA/stroke risk after CEA as secondary revascularization technique compared to CAS, irrespective from the initial revascularization technique (combining initial revascularization techniques: OR: 2.05; 95% CI: 1.29–3.27, p < 0.01; after initial CEA: OR: 1.85; 95% CI: 1.09–3.15, p = 0.02; after initial CAS: OR: 6.61; 95% CI: 1.36–32.1, p = 0.02) (Table 6 and Supplemental Table SII). The effect of the intervention on MI was not confounded by preoperative symptomatic status (Table 6).
Other short-term secondary outcomes did not differ significantly (Table 7, Supplemental Figures SIII–VII).
Summary of Short Term (≤30 days) Secondary Outcome of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
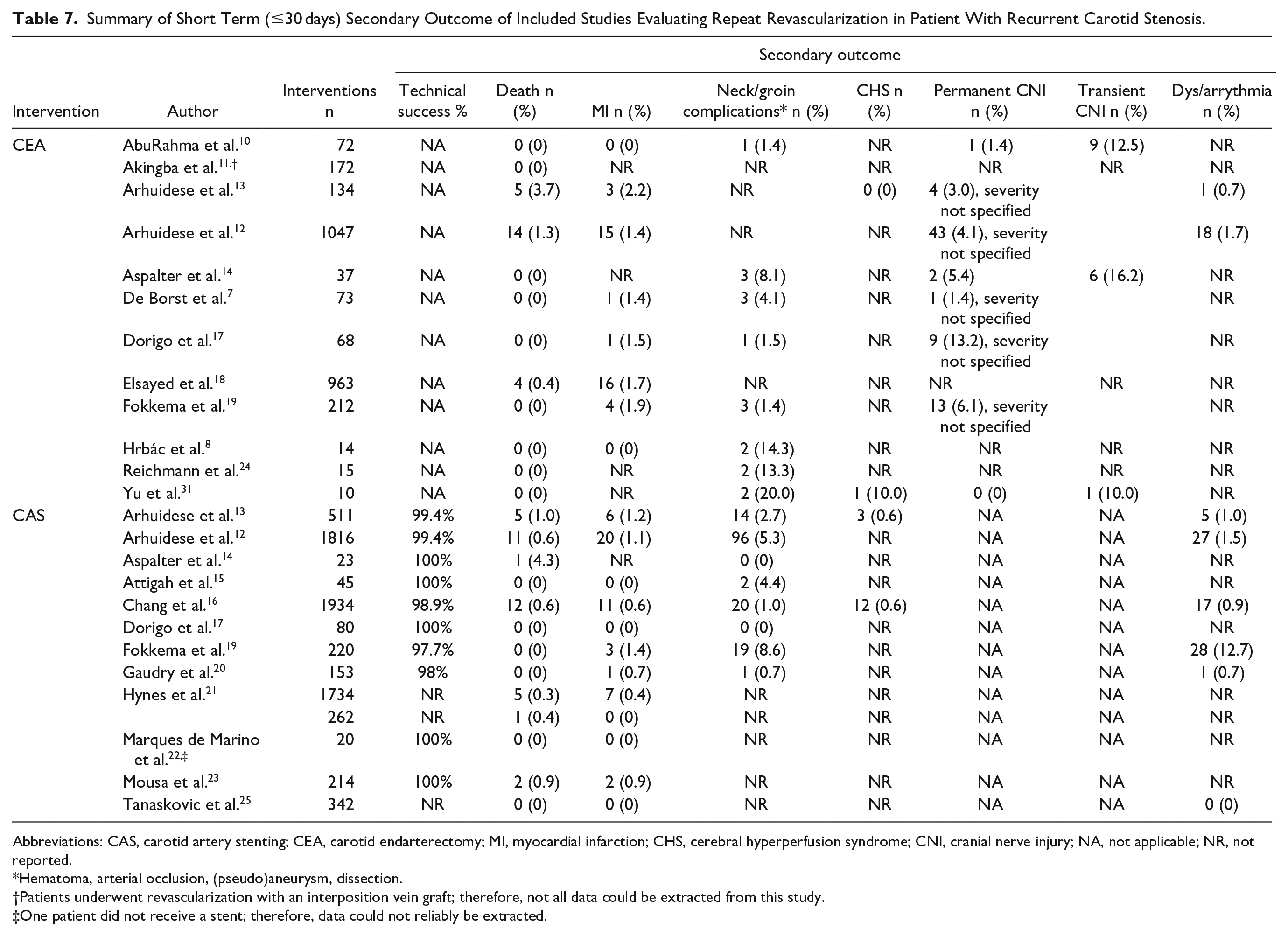
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; MI, myocardial infarction; CHS, cerebral hyperperfusion syndrome; CNI, cranial nerve injury; NA, not applicable; NR, not reported.
Hematoma, arterial occlusion, (pseudo)aneurysm, dissection.
Patients underwent revascularization with an interposition vein graft; therefore, not all data could be extracted from this study.
One patient did not receive a stent; therefore, data could not reliably be extracted.
Sensitivity Analysis
After stratification for the index procedure (CAS or CEA), the primary outcome (TIA/stroke) showed no significant difference between groups. CEA after CAS is associated with higher odds of all-cause mortality (OR: 3.96, 95% CI: 1.07–14.7, p = 0.04) and local complications (OR: 6.54, 95% CI: 2.36–18.18, p < 0.001) compared to CAS after CAS. However, the wide confidence intervals should be noted. Other outcomes did not differ between groups (Supplemental Figures SIX–SXVI and Table SII).
Long-Term Outcomes
Rate of restenosis was reported by 11 studies.7,8,10,11,15,17,19,20,23,25,31 Restenosis during a follow-up period of 1 to >4 years after CAS was 6.47% (95% CI: 3.26–12.43) and 6.65% (3.75–11.54) after CEA. No statistical difference was observed (p = 0.94). Several studies computed a Kaplan–Meier estimate for freedom of restenosis, freedom of further repeat revascularization and survival. Freedom of restenosis varied between 82% and 99% after 1 year and between 78% and 94% after 5 years.7,17,20,28 Freedom of repeat revascularization varied between 82% and 100% after 1 year and between 75% and 96% after 5 years.7,14,15,17,28 Correction for the index procedure did not influence the outcome (Table 8 and Supplemental Figures SVIII–IX and SXV-XVI).
Summary of Long-Term Secondary Outcome of Included Studies Evaluating Repeat Revascularization in Patient With Recurrent Carotid Stenosis.
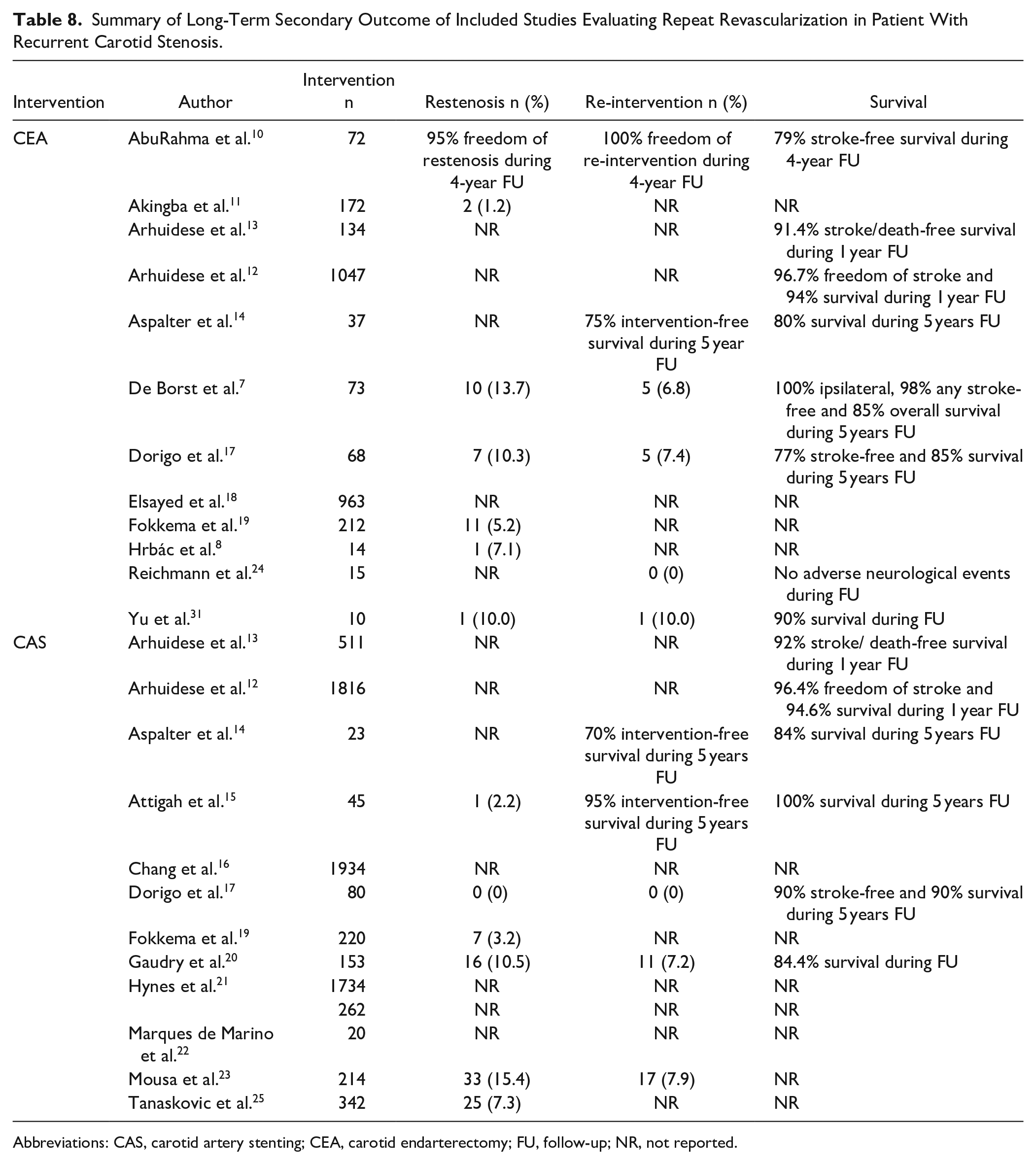
Abbreviations: CAS, carotid artery stenting; CEA, carotid endarterectomy; FU, follow-up; NR, not reported.
Discussion
The current review reports outcomes of CAS and CEA used as treatment for primary restenosis after prior CAS or CEA. Based on methodologically constrained studies, key results are as follows: (1) No difference in TIA/stroke or death between groups if unadjusted for symptomatic status; (2) Significant higher rate of TIA/stroke in symptomatic patients after CEA compared to CAS; (3) Significant higher rate of MI after CEA if unadjusted for symptomatic status; (4) No significant difference in other secondary outcomes between groups; (5) Stratification by index procedure did not affect conclusions.
In addition to providing a more comprehensive review that includes a larger number of patients with more recent literature, the review offers new insights into restenosis. Unlike previous studies, it uniquely investigates the outcomes of repeat revascularization (CAS and CEA) independently of the index procedure, hypothesizing that factors such as patient characteristics and plaque-specific features are more decisive than the initial surgical technique. The results support this hypothesis since a sensitivity analysis revealed that stratification by the index procedure did not alter conclusions, confirming it was not a substantial confounder. While lower TIA/stroke and mortality rates were observed in patients treated with CAS for both primary and secondary procedures, these findings were not significant and should be interpreted with caution due to the limited data available regarding patients treated with CEA after previous CAS.
To the best of our knowledge, two systematic reviews and meta-analyses comparing CAS (2174 and 2485 procedures) and CEA (2402 and 1655 procedures) after prior CEA, as well as one review (comprising 894 CAS and 163 CEA procedures) comparing these interventions after prior CAS, have been published in the past decade.32–34 Regarding patients who underwent revascularization after prior CEA, both reviews demonstrate that CAS and CEA are safe for treating recurrent stenosis after CEA, with the exception of more frequent CNIs following CEA.32,33 One review, based on its included noncomparative studies, also found a higher incidence of MI after redo CEA, while more recurrent restenosis was reported after CAS during follow-up, which was subsequently associated with a higher tertiary intervention rate. Based on the included comparative studies, the CEA group demonstrated a higher freedom from restenosis at 36 months. 32 In contrast, the other review found less recurrent restenosis of ≥60% and ≥70% in favor of the CAS group. 33 No differences were observed in 30-day stroke rates when adjusted for symptom status.32,33 For patients who underwent revascularization after prior CAS, the review demonstrated that both CEA and redo CAS are equally safe and effective in the short- and mid-term. 34 Our results are consistent in terms of the higher frequency of CNIs and MI (unadjusted for symptomatic status) after CEA. However, they differ in several key aspects: (1) A significantly higher rate of TIA/stroke following CEA compared to CAS was observed in our analysis when adjusted for symptomatic status; (2) A higher incidence of restenosis after CAS could not be demonstrate; (3) No evidence of a higher tertiary intervention rate following CAS was found; and (4) Correction for the index procedure did not impact the conclusions.
Based on this review, a repeat revascularization for patients with primary restenosis after primary CAS or CEA appears to be safe, as the 30-day risk of death/stroke does not exceed the threshold of 3% and 6% of primary intervention for asymptomatic and symptomatic patients, respectively.1,4 However, the risk tends to increase in symptomatic patients after both CAS (1.4%–3.2%) and CEA (2.2%–3.7%). This increased risk may be attributed to symptomatic patients being predisposed to more vulnerable plaques, and thus increased risk of adverse events. 35 Highly calcified plaques are independent risk factors for restenosis in the first year after both CEA and CAS. Lipid-rich plaques are associated with lower risk for restenosis.35–37
Technical success rate of 99.0% (23,923 out of 24,172) after primary transfemoral CAS is reported in the literature. 38 In our review, the technical success of CAS as a repeat revascularization remained equivalent with a percentage of 99.0%. Long-term technical success is determined by the incidence of restenosis. One study which included 2836 CAS procedures and 2909 CEAs, found no difference in severe primary (≥70%) restenosis rate after CAS (7.0%) and CEA (5.6%) in patients with asymptomatic and symptomatic carotid stenosis. Moderate (≥50%) or severe (≥70%) primary restenosis was observed in 30.3% (309 out of 1019) and 21.1% (231 out of 1096) of cases during follow-up after CAS and CEA, respectively, favoring CEA (OR: 2.0; 95% CI: 1.12–3.6). 2 Secondary restenosis (≥50%) rate of 6.5% and 6.7% was observed after CAS and CEA, respectively. Due to variety in the reporting of severity of the stenosis and follow-up period, caution is required in the interpretation of these findings. Two of the included studies compared CAS for de novo lesion with CAS for primary restenosis after previous CEA. There was no significant difference in the occurrence of restenosis.23,25 Another study comparing primary CEA (265 patients) and redo-CEA (124 patients) also did not find a difference in the rate of primary and secondary restenosis, respectively. 39 A secondary restenosis might require treatment. A third intervention rate of 8.0% and 8.3% was found after CAS and CEA, respectively. Little is known on this topic and is beyond the scope of this review.
CAS and CEA demonstrate comparable safety profiles in terms of TIA/stroke/death. Therefore, local complication rates could make a difference in choosing a treatment strategy. In this review a similar rate of hematoma, arterial occlusion, (pseudo)aneurysm and dissection was found. According to a multicentre randomized trial, primary CAS and CEA are associated with a local complication rate of 3.1% (8 out of 261) and 1.2% (3 out of 259), respectively. 40 Based on this review, comparable results can be achieved for CAS as second carotid intervention, with a complication rate of 2.8%, while CEA as a repeat revascularization technique showed a higher complication rate of 5.3% compared to the aforementioned literature.
CNI is another important factor to consider. Considering that CNI only occurs after CEA, CAS could be considered a more compelling intervention. In this review, a CNI incidence of 7.6% was found. One study compared primary CEA with repeat CEA. CNI was observed in 17% (21 out of 124) after repeat CEA versus 5.3% (14 out of 265) in the primary CEA group (p < 0.001). Important to note that permanent CNI occurred in only 1.6% (2 patients) versus 0.4% (1 patient), and the difference was not statistically significant. 39 Similarly, based on 3 out of 8 included studies that reported on and categorized the severity of CNI as transient or permanent, CNI appears to be mostly transient after CEA (84.2%).10,14,31
Limitations
The limitations of this review are an inevitable result stemming from the inherent limitations of the included studies. First, the methodological quality of the included studies was limited. All but one study were retrospective cohort studies, creating potential for allocation bias and confounding. Another limitation of this study concerns the heterogeneity of the included subjects, which hampered comparison. Both asymptomatic and symptomatic patients were treated, while current evidence questions the indication of treatment of asymptomatic restenosis; therefore, subanalysis of only asymptomatic patients was restrained. Furthermore, the grade of (re)stenosis was not uniformly assessed by the NASCET or ECST method. Our strict inclusion and exclusion criteria, aimed at achieving a direct comparison between CEA and CAS as secondary revascularization techniques, may have introduced selection bias and decreased the generalizability of our findings. However, we believe that our approach provides the most accurate results by minimizing confounding factors related to other reintervention techniques. Additionally, some studies adapted their stenosis cut-off values based on the intervention, introducing further heterogeneity. In addition, stroke could not be categorized into ischemic versus haemorrhagic stroke, which presents different entities. This differentiation should be made in future studies. Lastly, the inclusion period of included studies extends back to approximately 40 years ago. During this period, CAS in particular has improved (better stents, guidewires, and protection devices), which could potentially influence the outcome.
Conclusion
Derived from methodologically limited studies, this review demonstrated that both CAS and CEA represent safe treatment techniques in patients with either asymptomatic or symptomatic primary restenosis. The perioperative outcomes in terms of TIA, stroke, and death appear to be comparable between the groups if unadjusted for symptomatic status. Symptomatic patients have a higher TIA/stroke risk after CEA compared to CAS. Furthermore, the higher risk of local complications (i.e., CNI, which is usually transient) and MI are crucial factors to be taken into account. The primary revascularization technique did not influence the outcome of repeat revascularization. This could suggest that other factors, such as plaque composition, may play a crucial role in determining outcomes and guiding the choice of treatment modality. In conclusion, irrespectively from the initial revascularization technique, both CAS and CEA are reasonable and safe treatment options for primary restenosis. However, based on the obtained results, CAS appears to be preferable. Patients should be critically evaluated in a multidisciplinary team and further research is desirable.
Supplemental Material
sj-docx-1-jet-10.1177_15266028251325054 – Supplemental material for A Systematic Review and Meta-Analysis of Outcome After Repeat Revascularization for Primary Carotid Artery Restenosis
Supplemental material, sj-docx-1-jet-10.1177_15266028251325054 for A Systematic Review and Meta-Analysis of Outcome After Repeat Revascularization for Primary Carotid Artery Restenosis by Mert Kök, Franziska Röder, Reinoud P.H. Bokkers, Maarten Uyttenboogaart, Barzi Gareb and Clark J. Zeebregts in Journal of Endovascular Therapy
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
