Abstract
Introduction:
Abdominal aortic aneurysms (AAAs) and peripheral artery disease (PAD) are two vascular diseases with a significant risk of major adverse cardiovascular events and mortality. A challenge in current disease management is the unpredictable disease progression in individual patients. The VASCUL-AID-RETRO study aims to develop trustworthy multimodal predictive artificial intelligence (AI) models for multiple tasks including risk stratification of disease progression and cardiovascular events in patients with AAA and PAD.
Methods:
The VASCUL-AID-RETRO study will collect data from 5000 AAA and 6000 PAD patients across multiple European centers of the VASCUL-AID consortium using electronic health records from 2015 to 2024. This retrospectively-collected data will be enriched with additional data from existing biobanks and registries. Multimodal data, including clinical records, radiological imaging, proteomics, and genomics, will be collected to develop AI models predicting disease progression and cardiovascular risks. This will be done while integrating the international ethics guidelines and legal standards for trustworthy AI, to ensure a socially-responsible data integration and analysis.
Proposed Analyses:
A consensus-based variable list of clinical parameters and core outcome set for both diseases will be developed through meetings with key opinion leaders. Blood, plasma, and tissue samples from existing biobanks will be analyzed for proteomic and genomic variations. AI models will be trained on segmented AAA and PAD artery geometries for estimation of hemodynamic parameters to quantify disease progression. Initially, risk prediction models will be developed for each modality separately, and subsequently, all data will be combined to be used as input to multimodal prediction models. During all processes, data security, data quality, and ethical guidelines and legal standards will be carefully considered. As a next step, the developed models will be further adjusted with prospective data and internally validated in a prospective cohort (VASCUL-AID-PRO study).
Conclusion:
The VASCUL-AID-RETRO study will utilize advanced AI techniques and integrate clinical, imaging, and multi-omics data to predict AAA and PAD progression and cardiovascular events.
Clinical Trial Registration:
The VASCUL-AID-RETRO study is registered at www.clinicaltrials.gov under the identification number NCT06206369.
Clinical Impact
The VASCUL-AID-RETRO study aims to improve clinical practice of vascular surgery by developing artificial intelligence-driven multimodal predictive models for patients with abdominal aortic aneurysms or peripheral artery disease, enhancing personalized medicine. By integrating comprehensive data sets including clinical, imaging, and multi-omics data, these models have the potential to provide accurate risk stratification for disease progression and cardiovascular events. An innovation lies in the extensive European data set in combination with multimodal analyses approaches, which enables the development of advanced models to facilitate better understanding of disease mechanisms and progression. For clinicians, this means that more precise, individualized treatment plans can be established, ultimately aiming to improve patient outcomes.
Keywords
Introduction
Abdominal aortic aneurysms (AAAs) and peripheral artery disease (PAD) are prevalent vascular disorders with a large impact on patients’ health.1,2 AAAs are often asymptomatic until they rupture, which is highly lethal with a mortality of 85% to 90%. 3 Once diagnosed, current treatment guidelines advise surgical intervention based on aneurysm diameter (5.5 cm in men and 5.0 cm in women) or aneurysm growth rate (>10 mm/year).4,5 However, this “one-size-fits-all” approach fails for many individuals because small aneurysms can still rupture. 6 Besides, AAA patients have a high prevalence of myocardial infarction, stroke, heart failure, and coronary artery diseases.7,8 PAD is characterized by a spectrum of clinical manifestations, including claudication, impaired health-related quality of life, and functional limitations that can progress to critical limb-threatening ischemia. 9 In addition, PAD is a significant risk factor for major adverse cardiovascular events (MACE) such as myocardial infarction, stroke or cardiovascular death, and the need for revascularization procedures or amputation, ultimately increasing morbidity and mortality risks.9,10
One of the main challenges in current management of these two vascular diseases is the inter-patient uncertainty in disease progression, leading up to events such as aneurysm rupture in AAA patients, or the need for artery revascularization or amputation in PAD patients. Moreover, the proportion of patients with a high risk of MACE is estimated to be 25% 8 and 23% 11 in AAA and PAD, respectively. Secondary preventive measures for key modifiable risk factors (eg, diabetes, hypertension, and smoking) have proven their efficacy, but they remain to be adequately implemented in clinical practice.12,13 Previous research has indicated that implementing optimal cardiovascular risk management (CVRM) strategies could effectively reduce the 10-year risk of MACE in AAA patients from 43% to 14% 12 . Nonetheless, the tools to predict patient-specific progression of these diseases, prognosis, and outcomes of interventions are lacking. Therefore, the development of reliable methods to improve the implementation of evidence-based patient-specific management is paramount and could potentially lead to more commitment of patients and physicians for preventive disease management.
Artificial intelligence (AI) is a transformative avenue that enables more precise quantitative analyses within large patient data sets. AI-powered tools have the potential to improve predicting the risk of AAA growth and rupture, allowing for more targeted treatment approaches. 14 Earlier research has explored the contribution of machine learning algorithms to predict AAA growth using patient data and characteristics such as blood vessel elasticity and aortic diameter with promising results. 15 More recent studies developed AI algorithms that analyze patient clinical and imaging (biomechanical and morphological) data to estimate the risk of AAA rupture.16,17 These models showed promising value in predicting clinical outcomes, including disease progression of AAA patients.
In the field of PAD, the majority of AI models have focused on disease detection and diagnosis. Predictive models for PAD disease progression and outcomes are less explored in this field.18,19 Nonetheless, AI models for predicting in-hospital mortality 20 and long-term mortality21,22 have been reported. Particularly, the model presented by Ross et al 21 considered diverse modalities including demographic, clinical, imaging, and genomic data to stratify PAD patients with a high risk of mortality. Similarly, the evaluation of major cardiovascular events presents a significant challenge in PAD. In this regard, models exist that predict MACE 21 or major adverse limb events (MALE).23,24 A recent study by Li et al 24 has investigated MALE in a PAD patient population and achieved good model performance by combining demographic, clinical, and proteomic data in a multimodal AI model for risk prediction.
Despite the substantial progress in AI-powered diagnostics and risk stratification for AAA and PAD, key challenges remain. A significant hurdle is the lack of trustworthy AI development with large multimodal data sets and the actual integration of AI tools in clinical practice to aid clinical decision-making. Current models are frequently trained on data sets with a limited number of patients, potentially hindering their generalizability to more diverse populations and compromising external validity. Furthermore, the scope of existing models is often narrow and neglects the intricate interplay of factors contributing to AAA and PAD development, including genetic predispositions, lifestyle habits, and a broader spectrum of biomarkers. To achieve a more comprehensive understanding of these diseases, future AI models should prioritize multimodal data integration, incorporating information from diverse sources like electronic health records, imaging data, genetic data, and proteomic data. By addressing challenges and fostering robust, generalizable models in the vascular surgery field, AI has the potential to enhance the management of AAA and PAD.
The multidisciplinary European VASCUL-AID project is designed to overcome these challenges. The project aims to develop a platform for predicting disease progression and cardiovascular events in patients with AAA or PAD. The VASCUL-AID platform will support the creation of several AI models such as image analysis algorithms and risk prediction algorithms. In addition, the project includes the development of a mobile health app for patients and physicians to monitor their disease progression. In VASCUL-AID-RETRO, the retrospective study of this project, the aim is to develop predictive models based on data that has been previously collected. The study addresses key challenges in AI-powered diagnostics and risk stratification by utilizing a large multimodal patient data set. The inclusion of clinical and imaging data, alongside proteomics and genomics data, contributes to the comprehensive nature of the multimodal data set. Importantly, to achieve the establishment of trustworthy AI tools ready for the use in daily clinical care, ethical and legal considerations will be taken into account throughout the study. Through the extensive retrospective data set, the VASCUL-AID-RETRO study aims to develop trustworthy multimodal predictive AI models for multiple tasks including risk stratification of disease progression and MACE in patients with AAA and PAD. The developed models will be further adjusted by adding prospective data and will be internally validated in the future VASCUL-AID-PRO study.
Methods
Study Design and Study Population
The primary objective of this retrospective multicenter study is to develop and train algorithms to predict disease progression and risk of cardiovascular events in AAA and PAD patients by leveraging multimodal data from retrospective patient cohorts and biobanks. The secondary objective is to internally validate the developed algorithms using data from retrospective cohorts. An overview of the VASCUL-AID-RETRO study is shown in Figure 1.
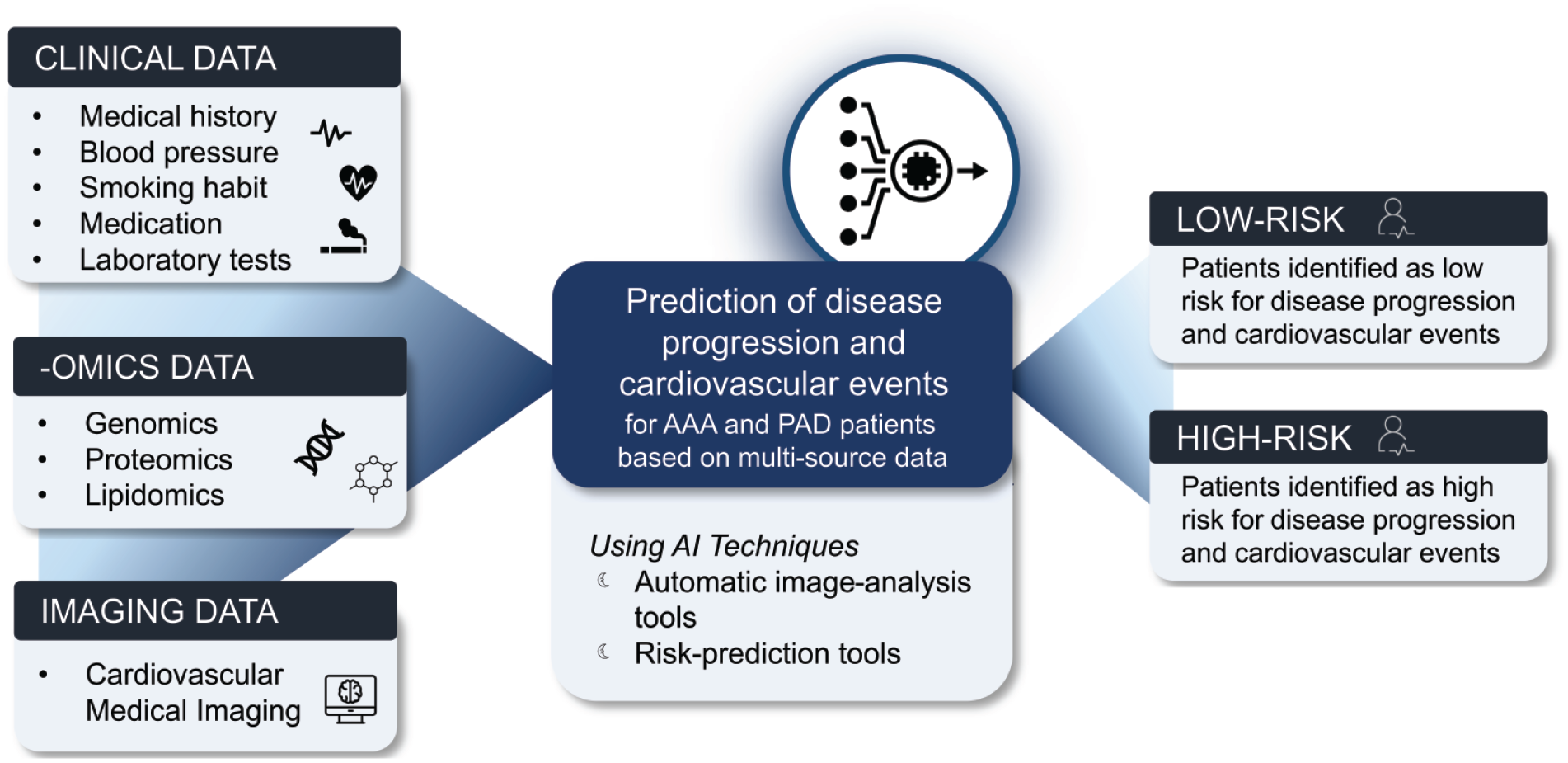
Overview of the VASCUL-AID-RETRO study.
For the development of the VASCUL-AID algorithms, patient data from electronic health records (EHRs) from European clinical consortium partners is available. The clinical partners of the VASCUL-AID consortium are Amsterdam University Medical Center (NL), Asklepios Kliniken Hamburg (DE), Centro Hospitalar e Universitário Sâo Joâo (PT), Helsinki University Hospital (FI), Oxford University Hospitals (UK), and Serbian Clinical Center (RS). All participating centers have obtained approval by ethical committees or other research bodies.
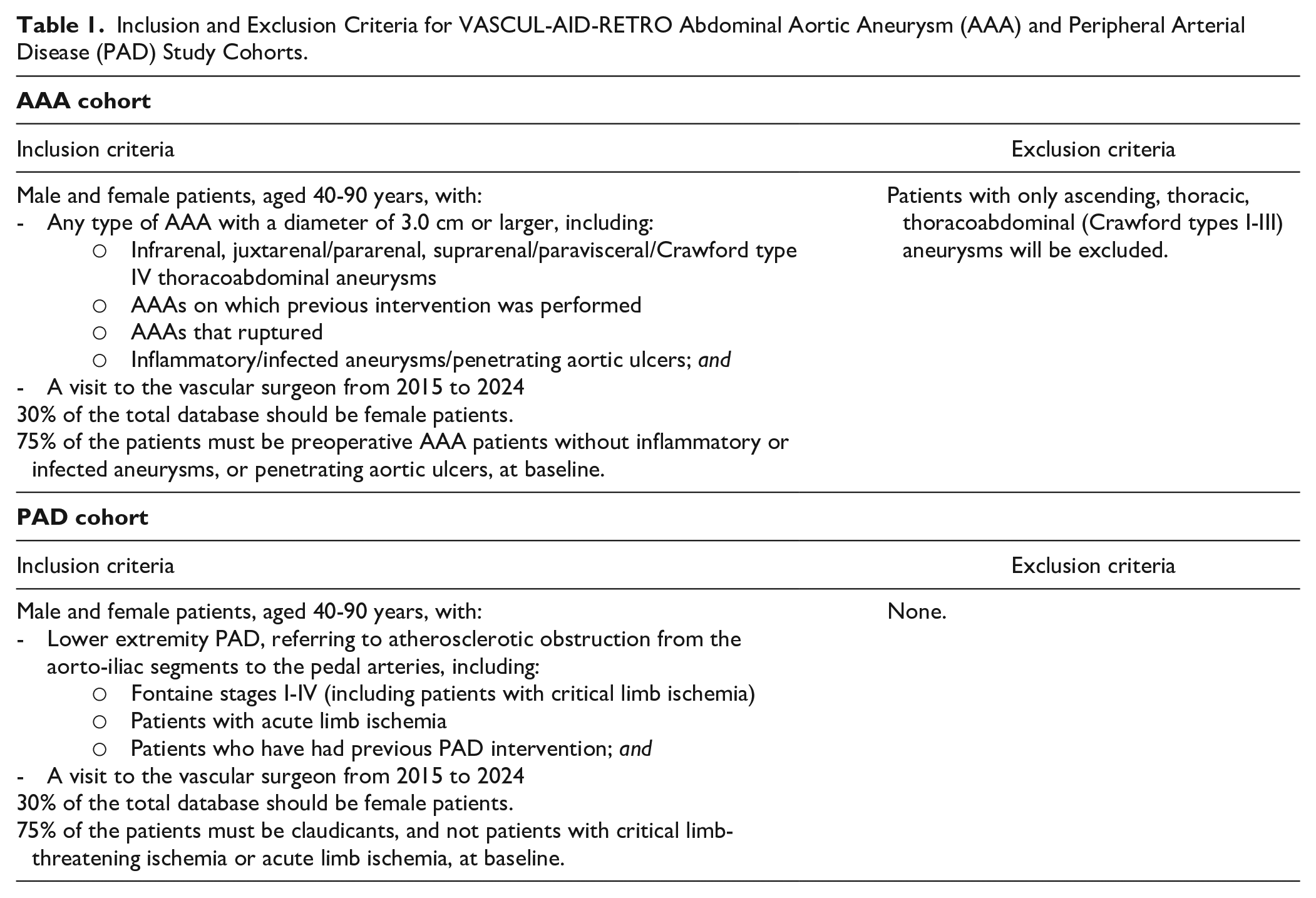
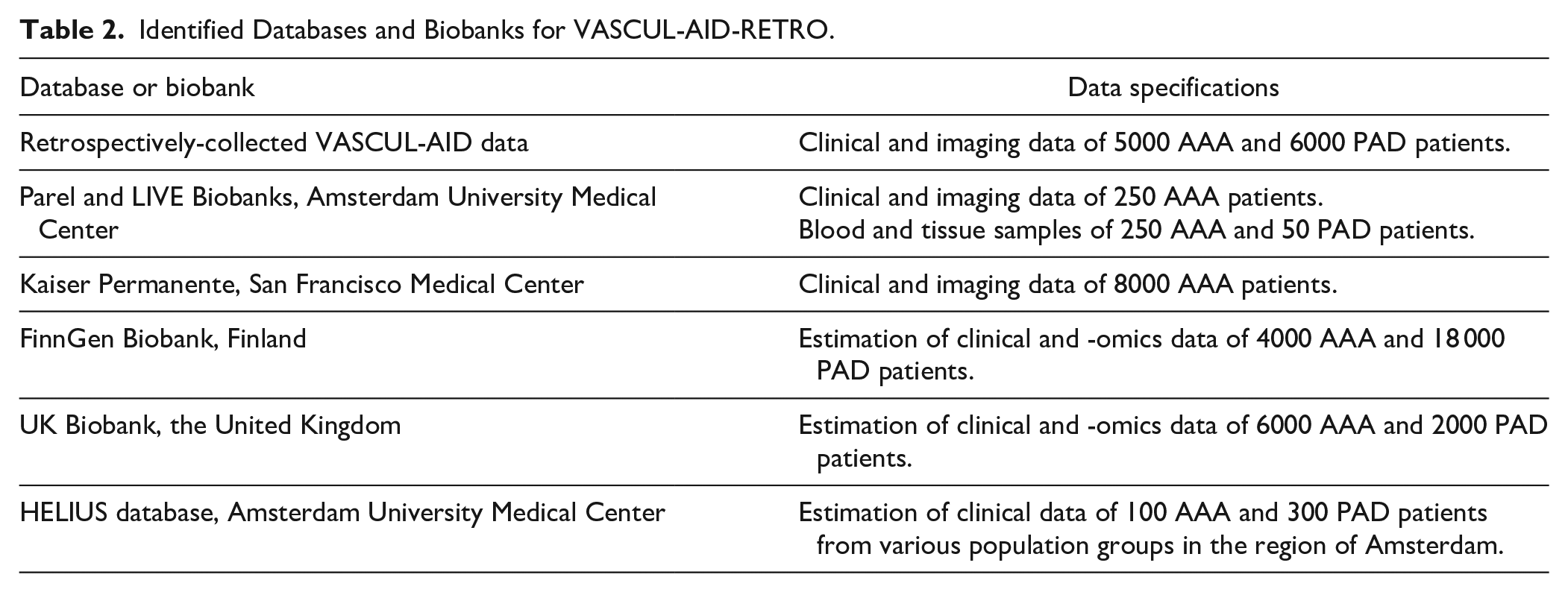
In this retrospective study, imaging and (reported) clinical EHR data of 5000 AAA and 6000 PAD patients will be collected from European consortium partners. Inclusion and exclusion criteria of patients are depicted in Table 1. Furthermore, blood samples of a subset of patients will be extracted from existing biobanks to perform multi-omics analyses. In addition, this consortium has access to large cohorts and registries with data for the envisioned study that will be used to enrich the retrospectively-collected VASCUL-AID data. All this data will be used to develop and train the algorithms for the predictions of cardiovascular events and AAA or PAD progression. An overview of the complete identified database including registries and biobanks that will be used in this study is described in Table 2. During the study, other data from new collaborations could be added as input to the prediction models.
Inclusion and Exclusion Criteria for VASCUL-AID-RETRO Abdominal Aortic Aneurysm (AAA) and Peripheral Arterial Disease (PAD) Study Cohorts.
Identified Databases and Biobanks for VASCUL-AID-RETRO.
During the entire VASCUL-AID-RETRO study, ethical and legal issues involved in developing trustworthy AI tools will be addressed. A Health Ethical Legal and Social Aspects (HELSA) framework of AI will be established early in the project and updated annually, to ensure compliance with evolving international laws and standards for responsible AI development. In this framework, relevant ethical, legal, and social issues and concerns for the VASCUL-AID-RETRO study will be discussed, and integrated, if necessary through guidelines. The HELSA issues include the fairness and inclusivity of AI (avoiding bias), data privacy, and exercising autonomy by patients. Within the HELSA context, potential liability risks of introducing AI tools in PAD and AAA care will be investigated, followed by recommendations on how to avoid these risks and what safeguards are needed in terms of use and oversight.
Sample Size Calculation
To identify important predictive features for disease progression and cardiovascular risk, clinical input variables for AAA and PAD will be collected. The amount of clinical input variables will depend on a clinical variable list that will be established through expert consensus. These clinical variables will be enriched with imaging and multi-omics variables. To get to the final input parameters for the predictive AI models, principle component analyses or feature selection will be performed to select the maximum amount of variables that satisfy the rule of at least 10 events per predictor parameter 25 depending on the data distribution within the cohort of 5000 AAA and 6000 PAD patients.
Data Collection VASCUL-AID-RETRO Database
A clinical variable list will be established through expert consensus to select relevant variables from a clinical and ethical perspective and to minimize selection bias. Ethical considerations will be addressed through the involvement of ethical AI specialists that have assessed the variable list in accordance with the European AI Act that has been adopted in 2024. For the VASCUL-AID-RETRO database, clinical variables and imaging data will be collected from multiple follow-up visits with a vascular surgeon between 2015 until the latest reported available data in 2024. In addition, relevant EHR data from other specialties (such as cardiology, internal medicine, and nephrology) will be consulted to collect information about comorbidities and overall health status.
To collect the data, a data infrastructure is established that facilitates systematic data collection from all six participating clinical centers. The data infrastructure incorporates Castor EDC 26 for clinical data collection and the open-source eXtensible Neuroimaging Archive Toolkit (XNAT) 27 version 1.7.5 hosted by Health-RI 28 for imaging data storage and retrieval. The Castor EDC structure will specifically be designed for the VASCUL-AID-RETRO study and will incorporate the clinical variables determined with the expert consensus. The VASCUL-AID consortium partners will directly input their EHR data into Castor. Data from external databases and registries will be mapped to the VASCUL-AID clinical variables to enable integration of this data into Castor. Consequently, all data from different centers and external databases will be harmonized using the Castor designed for VASCUL-AID as a blueprint to obtain a uniform data set. Imaging of the VASCUL-AID consortium partners, as well as imaging from external databases, will be uploaded to the XNAT. The AI researchers will extract the data from these platforms for analyses.
For the multi-omics analyses, blood, plasma, and tissue samples of 250 AAA and 50 PAD patients will be collected from the existing Parel biobank and LIVE biobanks in Amsterdam University Medical Center. Blood plasma and tissue samples will be shipped to consortium partners in Porto for proteomics and lipidomics analyses. Targeted protein-resolved analyses will be performed to compare and validate the protein and lipid findings from blood and tissue samples, ensuring a comprehensive correlation between the two types of samples. To uncover the contribution of genetic variants related to AAA, PAD, and cardiovascular events, whole blood samples will be sent to consortium partners, VINS, in Serbia for genome-wide analysis. Genotyping will be performed using Infinium Global Screening Array-24+ v3.0, Illumina and scanned on Illumina iScan platform. This array provides extensive coverage (654 027 fixed genetic markers) and was commonly used in genome-wide association studies and meta-analyses. 29 Further implementation of imputation methods can enhance the resolution and accuracy of genotype data, enabling comprehensive genetic analyses.
Figure 2 illustrates the overview of the VASCUL-AID-RETRO data collection infrastructure.
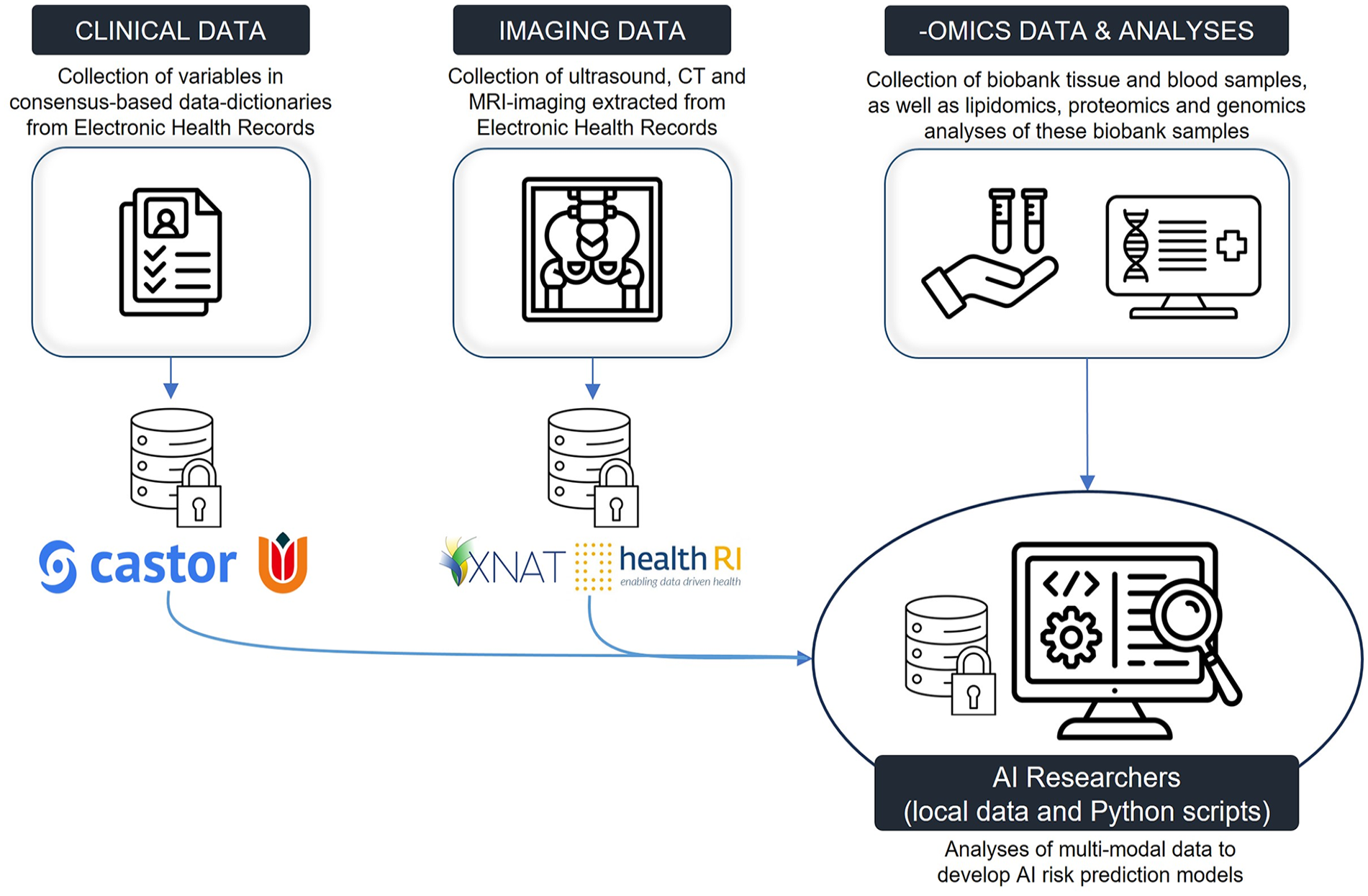
Schematic overview of VASCUL-AID-RETRO data infrastructure with Castor as the electronic data capturing system for health record data and an XNAT server from Health-RI for storage of imaging data.
Proposed Analyses
To obtain a comprehensive variable list, the consortium will collaboratively develop a VASCUL-AID key opinion leader (KOL) consensus-based clinical variable list. The initial list will be set up based on the literature30,31 and shared with clinical and ethical VASCUL-AID consortium partners and external KOLs for evaluation. To reach a consensus on variables and definitions, consensus meetings will be held to discuss which variables and definitions should be included. It is expected that two expert consensus meetings will be sufficient to reach agreement. In case two physical meetings will not be sufficient, the remaining outcomes will be discussed through mailing. Extensive discussions involving physicians involved in the treatment of these patients, such as vascular surgeons, vascular internal medicine specialists, and cardiologists, will cumulate in a finalized list of variables. The KOLs will be from the VASCUL-AID consortium as well as external specialists in the field. The main categories of the consensus-based variable list will comprise demographics, vascular disease-related information, cardiac history, comorbidities, social/economic status, substance abuse, general fitness, vital parameters, family history, blood test and microbiology results, medication, diagnostic tests, and imaging data.
In addition to the variable list, an expert consensus-based core outcome set will be developed as part of the VASCUL-AID project. This outcome set will include clinical and patient-centered core outcome measures for AAA and PAD patients, covering outcomes related to general health or disease status, disease progression, such as components of MACE or MALE, treatment and treatment complications, or general disease status. To obtain a long list of potential outcomes, a systematic literature review and focus groups with patients and health care workers will be performed. Second, relevant stakeholders (patients, patient representatives, and health care workers) from consortium countries will be invited to participate in the Delphi study, which will conclude with a consensus meeting to determine the final core outcome sets, for AAA and PAD separately.
Following the collection of imaging, multi-omics data, and (reported) clinical EHR patient data of the AAA and PAD patients, baseline characteristics will be assessed using descriptive statistics. Results will be reported as proportions with percentages for categorical variables, and as mean with standard deviation or as median with interquartile range for continuous variables, as appropriate. Categorical data will be presented in numbers and percentages. Continuous variables will be presented with means and standard deviations or 95% confidence intervals or medians and the interquartile range, depending on their distribution. Survival analyses such as Kaplan-Meier estimation and Cox proportional hazards models will be performed to evaluate cardiovascular events and disease progression of the retrospective data set. The data set will comprise repeated measurements over time to perform longitudinal data analysis techniques, such as mixed-effects models and generalized estimating equations. Statistical analysis will be performed using R, SPSS, and Python.
Modality-specific analyses will be performed next. Automated image segmentation models will be developed using images collected from the following imaging modalities: computed tomography (CT), ultrasound (US), and magnetic resonance imaging (MRI). These models will be used to segment structures relevant to AAA and PAD, such as the vascular geometry, artery lumen, thrombus, calcifications, and perivascular fat. The AI models will then be trained on the segmented AAA and PAD artery geometries to estimate hemodynamic parameters such as pressure, velocity, and wall shear stress. Furthermore, mass spectroscopy-based proteomic and lipidomic analysis of plasma samples will be undertaken to identify differentially secreted proteins and classify them into high vs low disease progression. Targeted genotyping will be done to identify the single nucleotide polymorphism information at previously identified risk loci. To help identify key variables or imaging features associated with cardiovascular events or disease progression, feature selection techniques may be utilized, such as recursive feature elimination or lasso regression (L1 regularization), in the development of the prediction models.
Initially, risk prediction models will be developed for each modality type (imaging, multi-omics, and reported clinical variables) separately. Various machine learning algorithms will be employed to create these individual prediction models. Finally, clinical data from the medical patient files, anatomical and hemodynamic data from medical imaging, and multi-omics data from patient blood samples will be integrated to be used as input to multimodal prediction models. Multiple AI models based on general machine learning (eg, random forest, XGBoost) or deep learning models (eg, convolutional neural networks, graph neural networks) will be assessed for optimal performance using suitable metrics (eg, sensitivity, area under the receiver operating curve) for its application. In the future VASCUL-AID-PRO study, these developed AI models will be further refined and internally validated using prospectively collected data. All results will be published in open-source vascular or data-/AI-related journals, and the algorithms will be made publicly available on GitHub.
Risks and Challenges
Even though multimodal AI models have emerged as an effective method for studying and analyzing heterogeneous data, multiple challenges remain. Ethical and legal challenges arise with the development of the AI models including data security, privacy, handling data, and ethical regulations and policies per country. 32 These challenges should be overcome to ensure trustworthy analysis and interpretation. In this project, emphasis is placed on maintaining transparency concerning data sources, collection methods, and preprocessing steps in adherence to relevant laws, regulations, and ethical standards. Transparency and adherence to relevant regulations are ensured through collaboration with our consortium partners specialized in this field, who will set up the HELSA framework for the VASCUL-AID project. As data could be imbalanced, heterogeneous, or even missing at all,32,33 it is important to review the VASCUL-AID data set and adjust it for imbalances that may arise. Also, data quality should be ensured by assessing the completeness and reliability of the data set.
Multiple techniques will be utilized to try to minimize the risks and to overcome these challenges. By utilizing a consensus-based variable list, most important variables will be included. Moreover, a single harmonized database is created in Castor EDC to store homogenous data sets from all participating partners. An Out-of-Distribution (OOD) filter will be implemented to identify and exclude patients who significantly deviate from the general population in our retrospective cohort. Implementing an OOD filter ensures that the data set used for the development of the models is representative of the target population, increasing the integrity and robustness of the models. Furthermore, to handle missing data techniques such as XGBoost and graph representation learning will be utilized. 34 Moreover, within the VASCUL-AID project, a study will be performed that will focus on the explainability of the developed algorithms through visualization to make the models as transparent as possible.
Conclusion
The VASCUL-AID-RETRO study will develop multimodal AI models, combining clinical, imaging, and multi-omics data, to predict AAA and PAD progression and associated risk of cardiovascular events in these patients for the first time. Following the VASCUL-AID-RETRO study, prospective VASCUL-AID studies will start in 2025 to collect prospective data and to test the algorithms in a pilot-setting (VASCUL-AID-PRO study). By integrating multimodal data sets, the final AI models have the potential to provide accurate predictions and more insights into disease progression, aiming to personalize treatments for improved patient outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.K.Y., MD, PhD, is Editor in Chief of the Journal of Endovascular Therapy. To avoid conflict of interest, an alternate member of the Editorial Board handled this submission. H.M. is co-founder and shareholder of Nicolab, TrianecT, and inSteps.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the European Union Horizon Europe Health program (HORIZON-HLTH-2022-STAYHLTH-01-two-stage) under the VASCUL-AID project (grant agreement ID: 101080947).
