Abstract
Purpose:
To report the occurrence of acute postrenal kidney failure caused by external ureteral obstruction after iliac venous stent placement.
Case report:
A 73-year-old male patient presented with a chronic swelling and feeling of heaviness of his right leg. The presence of venous thrombosis was excluded by duplex ultrasound (DUS). However, DUS revealed a high-grade non-thrombotic stenosis of the right external iliac vein which was confirmed by computed tomography venography. This stenosis was attributed to local scarring following prostatectomy, lymphadenectomy, and adjuvant radiation therapy for prostate cancer 7 years before. According to the clinical presentation, the patient underwent catheter-based revascularization by implantation of a dedicated venous stent in the external iliac vein. One day after this procedure, the patient experienced acute kidney failure, which was attributable to an incipient right-sided grade II hydronephrosis. Subsequent ureterorenoscopy demonstrated a stenotic lesion of the intermediate part of the ureter at the same level as the venous stent. Finally, ureteral stent placement resulted in the restoration of urinary drainage as well as resolution of acute kidney failure. This case report highlights the importance of considering nonvascular complications after venous stent placement and it stresses the importance of postprocedural clinical and laboratory surveillance and follow-up.
Clinical impact
In recent years the number of endovenous stent implantations has significantly increased. In patients with clinically relevant iliofemoral obstructions, endovenous stent placement potentially reduces related symptoms and improves quality of life. However, endovenous stent placement may have an impact on peri-vascular structures, such as the ureters. This case report highlights the awareness of potential nonvascular complications, which might be caused by iliofemoral stent implantation.
Keywords
Introduction
A 73-year-old Caucasian male patient presented with chronic lifestyle-limiting swelling and the feeling of heaviness of his right leg (CEAP class C3, Villalta Score: 10 points) to the vascular outpatient clinic of a tertiary-care hospital. Acute deep vein thrombosis was excluded by duplex ultrasound (DUS). However, DUS showed a high-grade non-thrombotic stenosis of the right external iliac vein. A subsequent computed tomography (CT) venography of the pelvic veins confirmed a high-grade non-thrombotic stenosis at this location without suspicious lymph nodes, local inflammation, or signs of a tumor. Seven years before presentation, the patient had undergone prostatectomy and lymphadenectomy with adjuvant radiation therapy due to prostate cancer. The medical report of this surgical procedure reported an intraoperative vascular injury; however, precise anatomic information was missing. In both CTs from 2021 and 2023, no hydronephrosis could be detected. The patient’s history was free of any venous thromboembolic events, nor was there a history of any other previous pelvic surgical procedure. In particular, the patient had never received a central intravenous line via his femoral veins.
At initial presentation, the serum creatinine was 1.09 mg/dL and the estimated glomerular filtration rate (eGFR) was 72 mL/min/1.73 m2 (as calculated by the Chronic Kidney Disease Epidemiology Collaboration equation, CKD-EPI). Further blood tests, such as complete blood count and inflammatory markers, were unremarkable and the prostate-specific antigen was not elevated (baseline prostate-specific antigen showed to be 0.58 µg/L). The patient had undergone regular urological follow-up examinations without signs of recurrence of prostate cancer.
Aiming for symptom relief and restoration of iliac venous blood flow, the patient was scheduled for a catheter-based venography and stent placement. In preparation of the endovascular procedure, 1000 mL of isotonic crystalloid fluid was administered intravenously. Subsequently, the patient underwent catheter-based multiplane venographic imaging and intravascular ultrasound confirming a >90% stenosis of the patient’s right external iliac vein (Figure 1A and B). Both, intravascular ultrasound (IVUS) and venography, gave sufficient understanding of vascular injury and repair, but in this case, IVUS was the preferred modality in which we could quantify the stenosis directly to have factual support of our assumption of chronic venous obstruction in this segment.
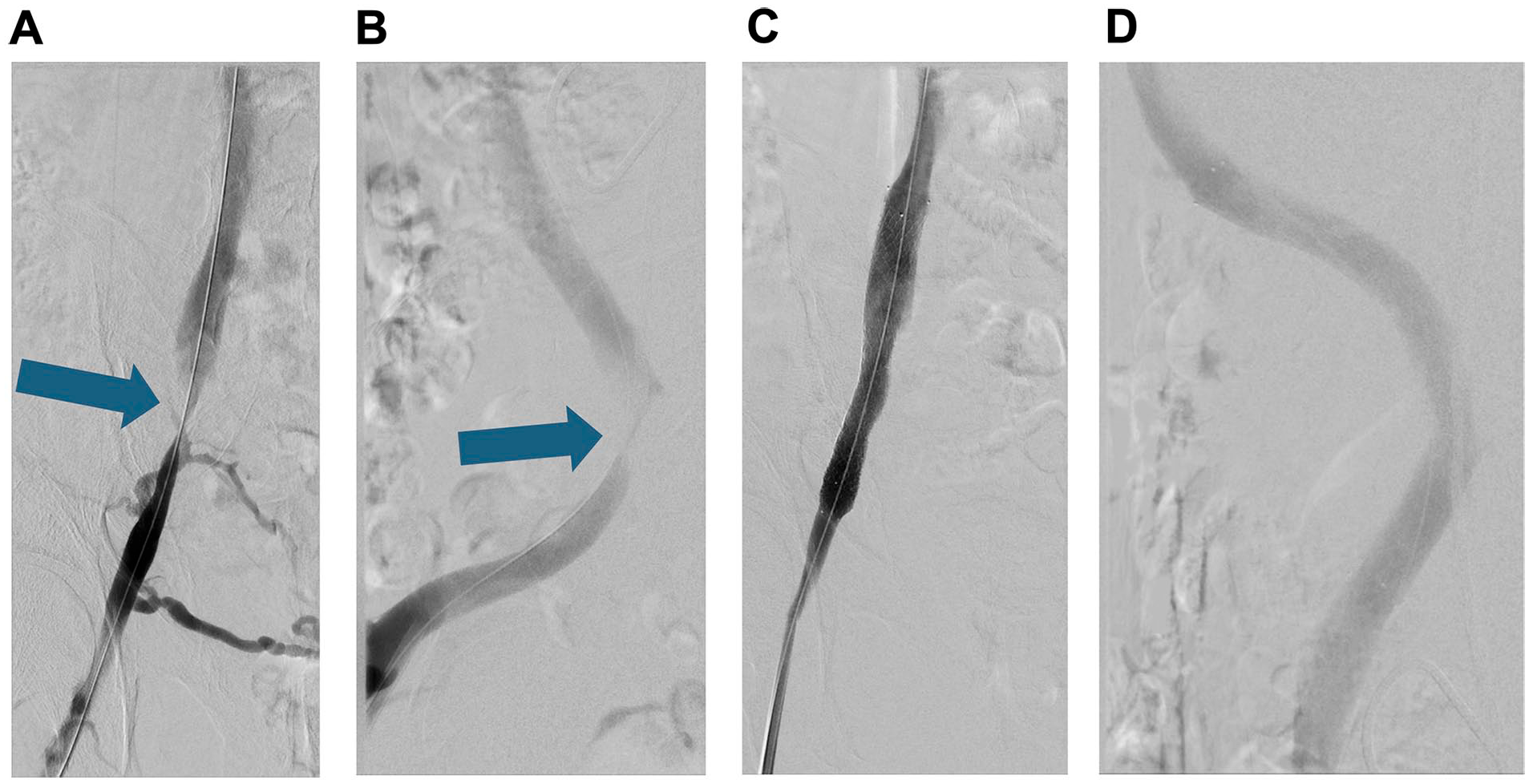
Catheter phlebography via femoral access of the right external iliac vein showing a high-grade non-thrombotic iliac vein lesion with collateral vein filling in posterior-anterior projection (A), and lateral view (B). Catheter phlebography after percutaneous transluminal angioplasty and stent implantation in the right external iliac vein in posterior-anterior projection (C), and lateral view (D).
Iliac vein recanalization was successfully performed after determining stent size by using IVUS by initial angioplasty with a 12/40 mm non-compliant high-pressure balloon catheter (Atlas Gold, Bard BD) followed by implantation of a 14/120 mm dedicated venous stent (Venovo, Bard BD). Control venography, intravascular ultrasound, as well as external DUS confirmed a satisfying morphological and hemodynamic result immediately after the procedure (Figure 1C and D). In total, 70 mL of iodinated contrast media (Iopamidol 300 mg/mL) was administered during the recanalization procedure. Following stent placement, anticoagulation was initiated with a therapeutic dose of low-molecular-weight heparin followed by apixaban 5 mg bid.
One day after stent placement, DUS confirmed sustained restoration of iliofemoral venous blood flow with respiratory flow modulation and adequate flow velocities. Clinically, the leg swelling and feeling of heaviness were regressive after iliac venous stent placement and the patient did not report any unusual pain.
However, the patient reported a restriction of urine excretion and subsequent blood tests revealed an increase in serum creatinine to 1.9 mg/dL and a drop of his eGFR to 37 mL/min/1.73 m2 (Chronic Kidney Disease Epidemiology Collaboration equation [CKD-EPI]). Over the course of that day, no improvement in urine excretion and serum creatinine levels was observed despite continuous intravenous crystalloid fluid resuscitation. Finally, a kidney ultrasound revealed an incipient grade II hydroureteronephrosis of the right kidney without urinary stasis of the bladder. An immediate urologist consultation was followed by ureteroscopy, in which retrograde pyelography revealed a narrowing directly at the level of the of the external iliac venous stent, resulting in a hydroureter. This confirmed the origin of the postrenal acute kidney failure (acute kidney injury, AKI) and the location of the problem.
The subsequent endourological drainage with a double-J ureteral stent insertion (Ruesch, Superglide DD-tumor-stent, Teleflex) at the crossing of the ureter and the external iliac vein resulted in urinary drainage (Figure 2).
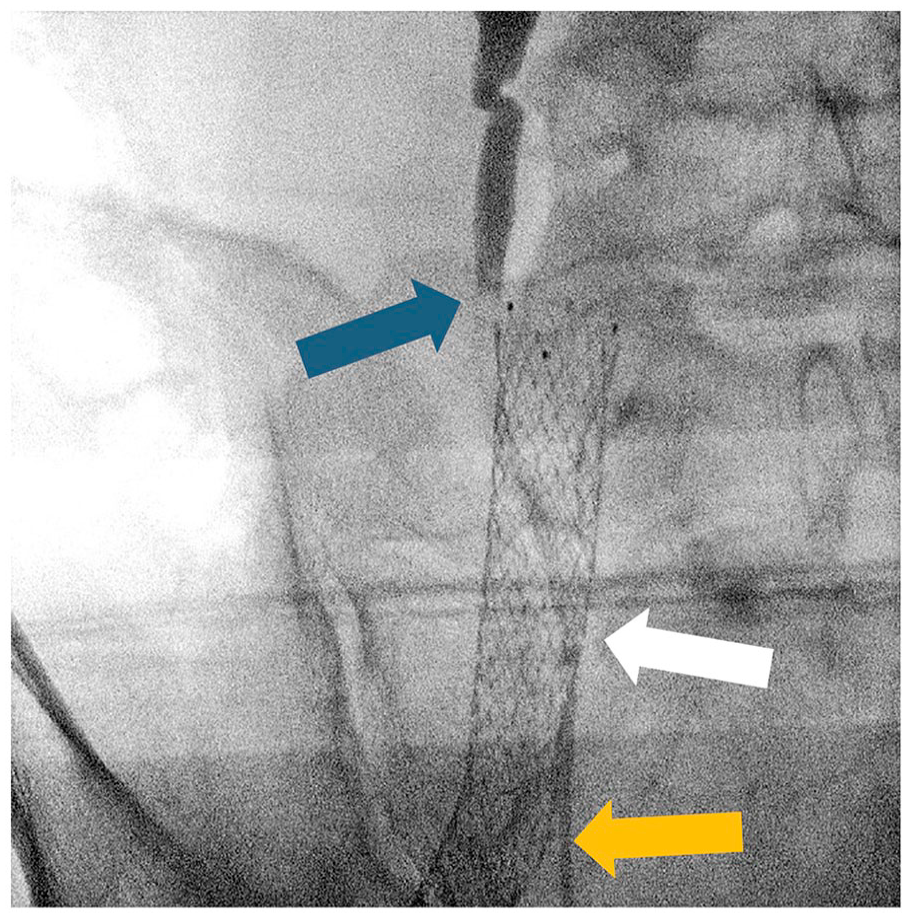
Ureteroscopic image revealing the blocked urine drainage in the right ureter (blue arrow) on the level of the iliac vein stent (white arrow). The post-stenotic part of the ureter showed a normal diameter (orange arrow).
One day after ureteral stent insertion, urinary excretion normalized and respective kidney function parameters began to recover (serum creatinine 1.33 mg/dL, eGFR 58 mL/min/1.73 m2, CKD-EPI) with normalization within the following days.
Over the following 6 months, the patient was followed periodically by vascular and urological assessments. Clinical signs and symptoms of venous congestion of the patient’s right leg resolved. Urine excretion sustained within the normal range and no further increase in serum creatinine was observed. A removal attempt of the double-J ureteral stent is routinely scheduled 1 year after insertion. If this proves unsuccessful, annual stent replacements would be the long-term solution.
Discussion
Iliac venous stent implantation is increasingly performed in patients with iliocaval outflow obstructions, which can be caused by acute iliofemoral deep vein thrombosis and post-thrombotic or non-thrombotic iliocaval lesions to restore venous blood flow. In experienced vascular centers, iliocaval stent implantation may sustainably reduce clinical signs and symptoms of venous hypertension and is usually associated with low complication rates. 1
Considering adverse outcomes after iliocaval stent implantation, the major concern is the loss of stent patency caused by restenosis or re-occlusion of the treated venous segment. 2 Venous stent patency primarily depends on hematological factors, venous hemodynamics, and the underlying lesion type showing the best patency rates in patients with non-thrombotic types of lesions.3,4 In this case, the patient presented with a non-thrombotic obstruction, which was most likely attributable to prior prostate surgery and radiation of the respective region causing perivascular scarring.
Focusing on nonvascular complications following venous stent placement, acute kidney injury may be considered as a potential consequence of the administration of iodinated contrast media. Contrast-associated acute kidney injury affects approximately 7% to 8% of patients following peripheral vascular interventions. 5 Estimations on the risk of contrast-associated acute kidney injury in peripheral vascular interventions primarily derive from the arterial field; however, recent data analyses suggest a similar proportion of contrast-associated kidney injury in patients undergoing deep venous stenting. 1
In the present case, no pre-existing chronic kidney disease, which would have been regarded as an individual risk factor for contrast-associated kidney injury, was present. Nevertheless, isotonic crystalloids had been administered before and after the endovascular procedure and the amount of contrast media was within the regular range of these procedures. Viewing these preconditions together with the absence of other risk factors for contrast-associated acute kidney injury, such as diabetes and atherosclerotic cardiovascular diseases, the likelihood of an early nephrotoxic reaction to contrast media administration was low. Finally, it should be noted that the increase in kidney function parameters was detected within 24 hours after the endovascular procedure, which would have been unusually early for contrast-associated acute kidney injury. Most contrast-associated kidney injuries occur 3 to 5 days after the respective exposure to contrast media. 6
Regarding other nonvascular complications following venous stent placement, the compression of external perivascular structures caused by stent forces may be considered. In this context, appropriate stent dimensioning is crucial, and the question of venous stent sizing has been a matter of debate in recent years. According to hemodynamic considerations, recommended stent diameters are 16, 14, and 12 mm for the common iliac, external iliac, and common femoral vein. 7 Especially in non-thrombotic iliac vein lesions, a slight oversizing is commonly used to minimize the risk of venous stent migration. 8 On the contrary, stent erosion through the vessel wall needs to be considered in case of too pronounced oversizing of venous stents. 1 To optimize stent sizing and to adequately assess venous lumen dimensions and the extent of iliac vein lesions, the use of IVUS is the most appropriate tool in the scope of iliac venous stenting procedures, especially for non-thrombotic iliac vein lesions.9,10 Oversizing of iliac venous stents may result in chronic low back pain, primarily caused by compression of adjacent vertebral structures or nerves. In individual cases, this even may lead to surgical explantation of the respective venous stent.8,11
In the present case, we therefore used IVUS in addition to multiplane catheter angiography and accordingly chose a 14 mm stent for treatment of the obstruction of the mid part of the external iliac vein. Referring to the prompt development of ureteral obstruction, we rather believe in ureteral obstruction due to external compression and local scarring than immediate stent erosion.
Up to now, little is known about potential risks of venous stent placement on external ureteral obstruction. In general, vasculoureteral complications may arise as a consequence of surgical procedures, trauma, or congenital anomalies. A report of 2 cases demonstrates the occurrence of acute kidney injury in patients with retroperitoneal fibrosis undergoing aortic stent graft implantation. 12 These observations illustrate the risk of perivascular fibrotic tissue following an inflammatory process, which may result in ureteral obstruction following vascular realignment by stent placement. Vice versa, surgical procedures and arising inflammation as well as scarring may provoke the development of uretero-arterial fistula, a rare but serious complication where an abnormal connection forms between the ureter and a nearby artery, potentially causing severe hemorrhage.12,13
In summary, it remains unclear whether precautious measures could have prevented the occurrence of ureteral obstruction after venous stent placement. This case emphasizes the need to incorporate “nonvascular considerations” into the management of vascular patients.
Conclusion
This case highlights the importance of considering potential nonvascular complications when performing iliocaval stent placement and it stresses the role of periprocedural surveillance and follow-up after venous stenting.
Footnotes
Author Contributions
All authors were involved in the drafting of this case report. All authors have read and approved this version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Written informed consent was obtained from the patient. This is a case report, which does not require an ethical vote.
