Abstract
Purpose:
After complex endovascular aortic repair (cEVAR), long-term surveillance is advocated to monitor for potential (stent-related) complications. Although various imaging modalities are used, computed tomography angiography remains the standard in current clinical practice worldwide. However, radiopaque markers can cause metal artifacts and scattering, hampering assessment of patency of side branches. The maximal systolic acceleration (ACCmax) is a relatively new duplex ultrasound (DUS) parameter measured distal to a stenosis, avoiding in-stent assessment and scattering. The aim of this article is to illustrate the potential diagnostic utility of the ACCmax after cEVAR and visceral artery stenting in general.
Technique:
The ACCmax is measured at the maximal slope of the upstroke during the systolic phase in arterial flow. By manually connecting 2 points, a tangent line is created in which the maximal acceleration is automatically calculated and expressed in m/s2. A higher value reflects better arterial perfusion proximal to its measurement point. One measurement of a visceral artery takes about 5 minutes.
Conclusion:
ACCmax measurements can be a useful addition during DUS follow-up after cEVAR or visceral artery stenting, possibly decreasing radiation exposure, nephrotoxicity, and health care costs.
Clinical Impact
Long-term surveillance after complex endovascular aortic repair (cEVAR) is necessary to monitor for potential (stent-related) complications. Although CTA is the most common modality for imaging, metal artifacts and scattering can hamper the assessment of stent patency. The maximal systolic acceleration (ACCmax) is a duplex ultrasound based parameter that reflects arterial perfusion proximal to its measurement point. Due to its noninvasive nature, it can be a useful addition during follow-up after cEVAR or visceral artery stenting, possibly decreasing radiation exposure, nephrotoxicity and health care costs as well.
Keywords
Introduction
In recent decades, there has been a growing trend toward endovascular treatment strategies for abdominal arterial disease.1,2 This trend applies not only to aneurysm repair, but also to mesenteric artery disease. In addition, thoracoabdominal aortic aneurysms (TAAs) are increasingly managed with endovascular techniques, which involve the use of fenestrated and branched endografts to include the visceral segment and arch. 3 After endovascular treatment, long-term surveillance is advocated to monitor potential (stent-related) complications. Although various imaging modalities are used for follow-up, computed tomography angiography (CTA) is currently the standard in clinical practice worldwide. 4 The CTA is well suited to detect aneurysm sac enlargement, endoleak, and stent migration (European Society for Vascular Surgery [ESVS] Guidelines). However, assessment of patency of mesenteric side branches with CTA imaging following complex endovascular aortic repair (cEVAR) can be difficult. Although metal reduction techniques in CTA are available, radiopaque markers on endografts can cause artifacts and scattering obscuring patency of side branches.5,6 Also, the effects of ionizing radiation and possible contrast nephropathy are unavoidable in CTA. Finally, CTA is expensive and has a substantial impact on hospital resources.
Duplex ultrasound (DUS) imaging is also used as diagnostic tool to evaluate the mesenteric circulation.7,8 As DUS is noninvasive and readily available, it provides advantages as potential follow-up tool. Currently, velocity parameters such as the peak systolic and end-diastolic velocity are used to evaluate visceral arteries after endograft treatment. However, it is known that these parameters can be influenced by respiration and the collateral mesenteric network.9,10 Also slightly higher velocities are seen in stented arteries in comparison with native vessels. 11 In the assessment of peripheral arterial disease (PAD) of the lower extremity, the maximal systolic acceleration (ACCmax) is a relatively new parameter measured distal to a potential stenosis. 12 Applicability of this parameter would have a major advantage in the assessment of visceral branches after endovascular treatment, as its diagnostic performance is not influenced by metal artifacts, scattering, respiration, and the collateral network. The aim of this article is to illustrate the potential diagnostic utility of the ACCmax after cEVAR or primary visceral artery stenting. This concept will be illustrated by 2 patient cases.
Technique
Maximal Systolic Acceleration
The DUS of the mesenteric circulation was performed in supine position with the probe in an angle below 60° (usually 0°), aiming primarily at the celiac trunk and superior mesenteric artery (SMA). Then, the ACCmax was measured in the systolic phase of an arterial flow. It is calculated at the maximal slope of the upstroke by manually connecting 2 points at the start and end of the systolic pulse (Figure 1). The acceleration of this tangent line is automatically calculated in m/s2 and defined as the maximal systolic acceleration. The higher the value of the ACCmax, the lower the probability of a hemodynamic significant stenosis proximal to the measuring point. In other words, the ACCmax reflects the arterial perfusion proximal to its measurement point. Currently, a value below 5.5 m/s2 is used as cutoff to diagnose PAD in peripheral arteries. 13 One measurement of a visceral artery takes about 5 minutes and no additional software is required to obtain the ACCmax. Previous studies indicated a very low interobserver variability.12,14 In the following case reports, vascular technicians performed DUS testing.
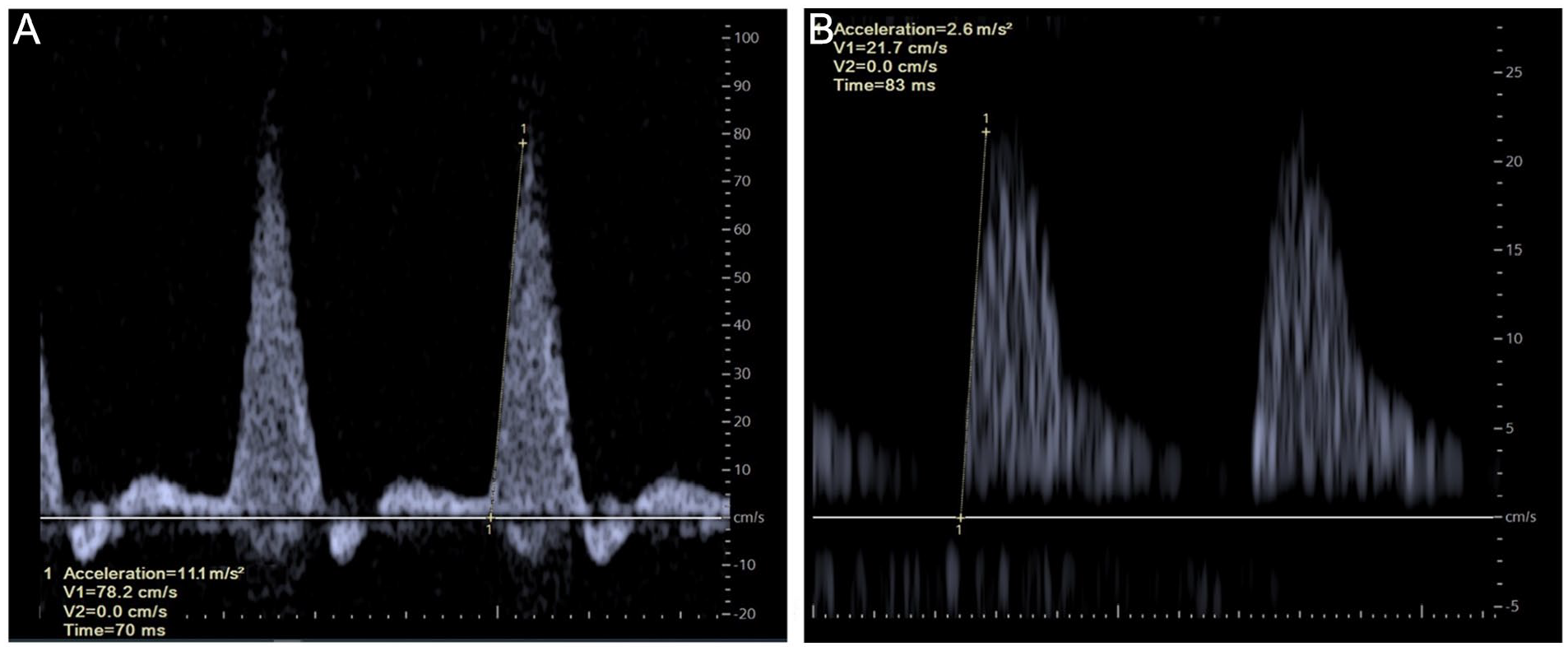
(A) Normal triphasic waveform showing an ACCmax measurement of 11.1 m/s2, calculated at the maximal slope in the systolic phase of an arterial flow (tangent line indicated by 1). (B) Abnormal waveform showing an ACCmax measurement of 2.6 m/s2, calculated at the maximal slope in the systolic phase of an arterial flow (tangent line indicated by 1). Note the differences in scales between the figures.
Patient Cases
Case 1
A 49-year-old man, known with peripheral artery disease, presented with chronic postprandial abdominal pain. A CTA was performed, which revealed high-grade stenoses at the origins of the celiac trunk and SMA. Percutaneous transluminal angioplasty (PTA) with stenting of the SMA was performed with good angiographic and clinical result. Initial follow-up was uneventful, but re-stenting had to be performed due to in-stent stenosis 10 years further. One year after this procedure, the patient complained of recurrent abdominal pain. The CTA showed a possible restenosis in the stent, with pickup of contrast distally. Due to metal artifacts, reliable assessment of luminal patency was not possible (Figure 2). The DUS was performed, which showed an ACCmax measurement of 1.2 m/s2 distal of the stent, implying a high-grade in-stent stenosis. Digital substraction angiography (DSA) confirmed this finding (Figure 3), and PTA with a 6 mm drug-eluting balloon was performed. Postintervention ACCmax increased to 19.0 m/s2, indicating a technical successful revascularization. Eight years after this procedure, no recurrentstenosis has occurred.
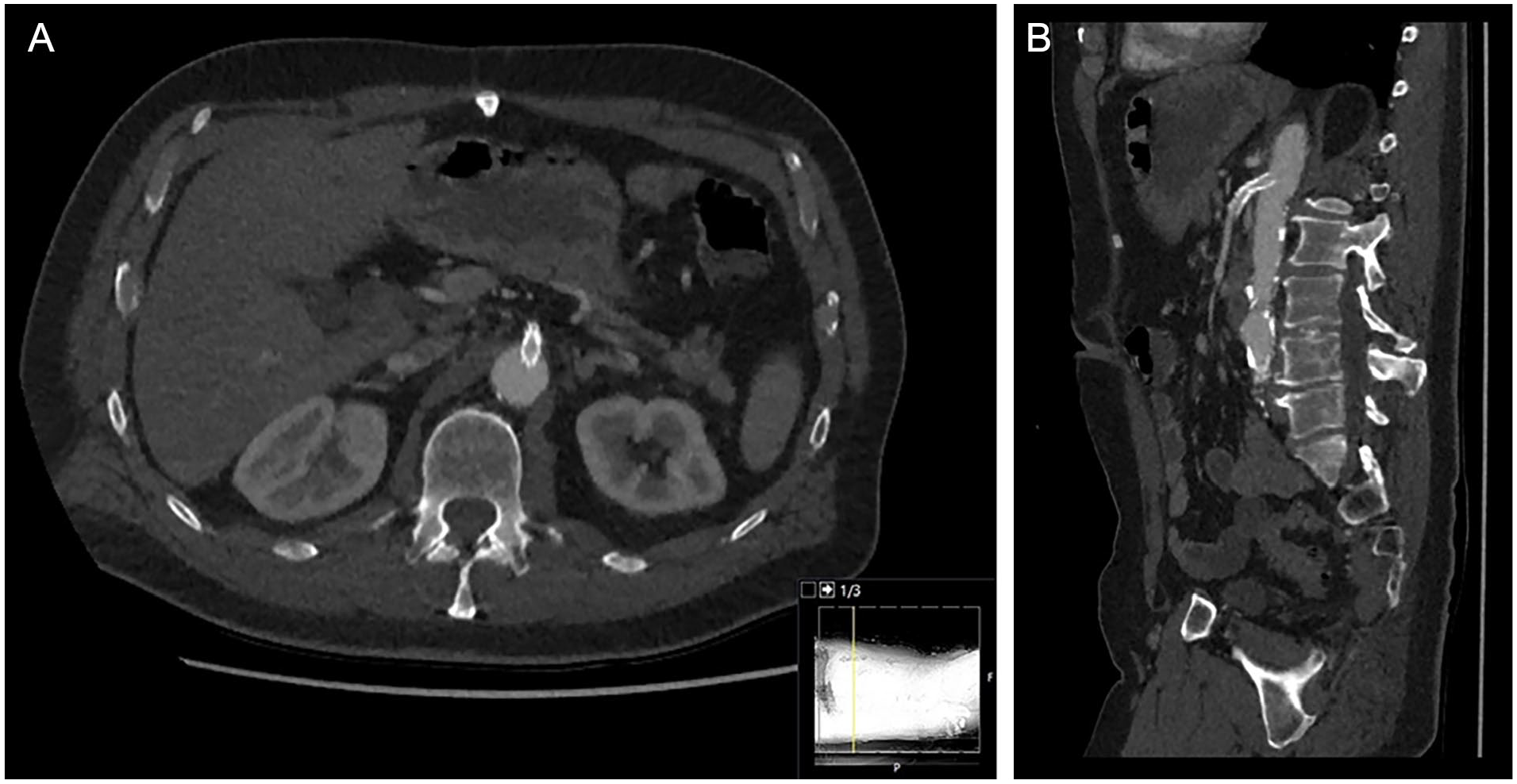
Computed tomography angiography (CTA) of the abdominal aorta (A = axial plane, B = sagittal plane). The CTA showed a possible in-stent restenosis with pickup of contrast distally. Due to metal artifacts, reliable assessment of luminal patency was not possible.
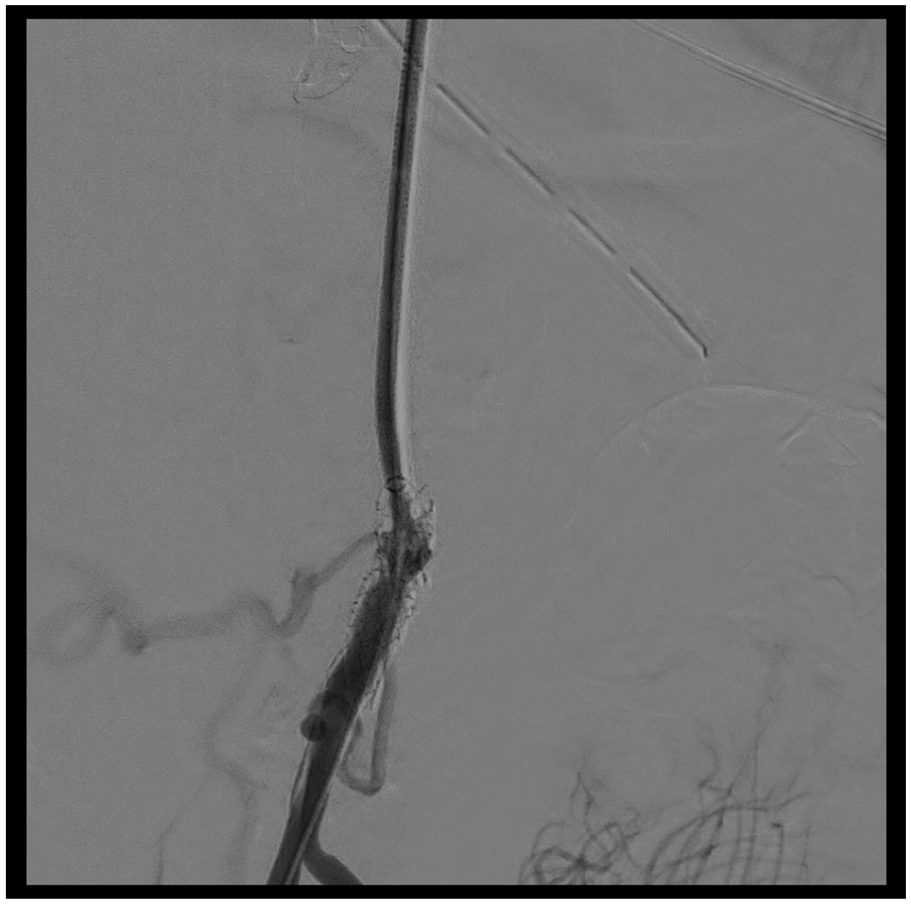
Digital substraction angiography (DSA) of the abdominal aorta. The DSA revealed an in-stent stenosis in the superior mesenteric artery. A PTA with 6 mm drug-eluting balloon was performed.
Case 2
A 71-year-old woman, with a medical history of hypertension, presented with acute and progressive abdominal pain since 3 days. The patient was conscious and had normal vital parameters without fever. On abdominal examination, diffuse tenderness on palpation in the epigastric region was present. Laboratory tests showed a hemoglobin (Hb) level of 13.2 g/dL (reference interval [RI] = 12.1–16.1 g/dL), a creatinine of 92 μmol/L (RI = 49–90 μmol/L), and a C-reactive protein of 180.0 mg/L (normal <5.0 mg/L). The CTA revealed a type III TAA of 6 cm with a small retroperitoneal hematoma. As open reconstruction was considered unsuitable given the age of the patient and the high risk of major complications, an endovascular approach was chosen. A branched EVAR procedure was performed using an off-the-shelf 4-branched device (T-Branch; CookMedical – Bloomington, USA), from which the patient recovered uneventfully. During routine surveillance after 3 months, CTA (with artifact reduction) showed adequate position of the branched device and visceral stents. There was good patency of the celiac trunk and both renal arteries. Due to metal artifacts of the stent at the origin of the SMA, visualization of patency was not possible (Figure 4). The DUS was performed with flow measurements of the visceral branches to assess arterial perfusion, which showed ACCmax measurements of 5.5 and 7.5 m/s2 for the celiac trunk and SMA, respectively. Both renal and common iliac arteries were patent with good ACCmax measurements (right interlobar artery 7.5 and left interlobar artery 7.0 m/s2). Combining both diagnostic modalities, adequate patency of the visceral stents was confirmed and no further diagnostic analysis using DSA was needed. To date, no restenoses have occurred in 3 years follow-up.
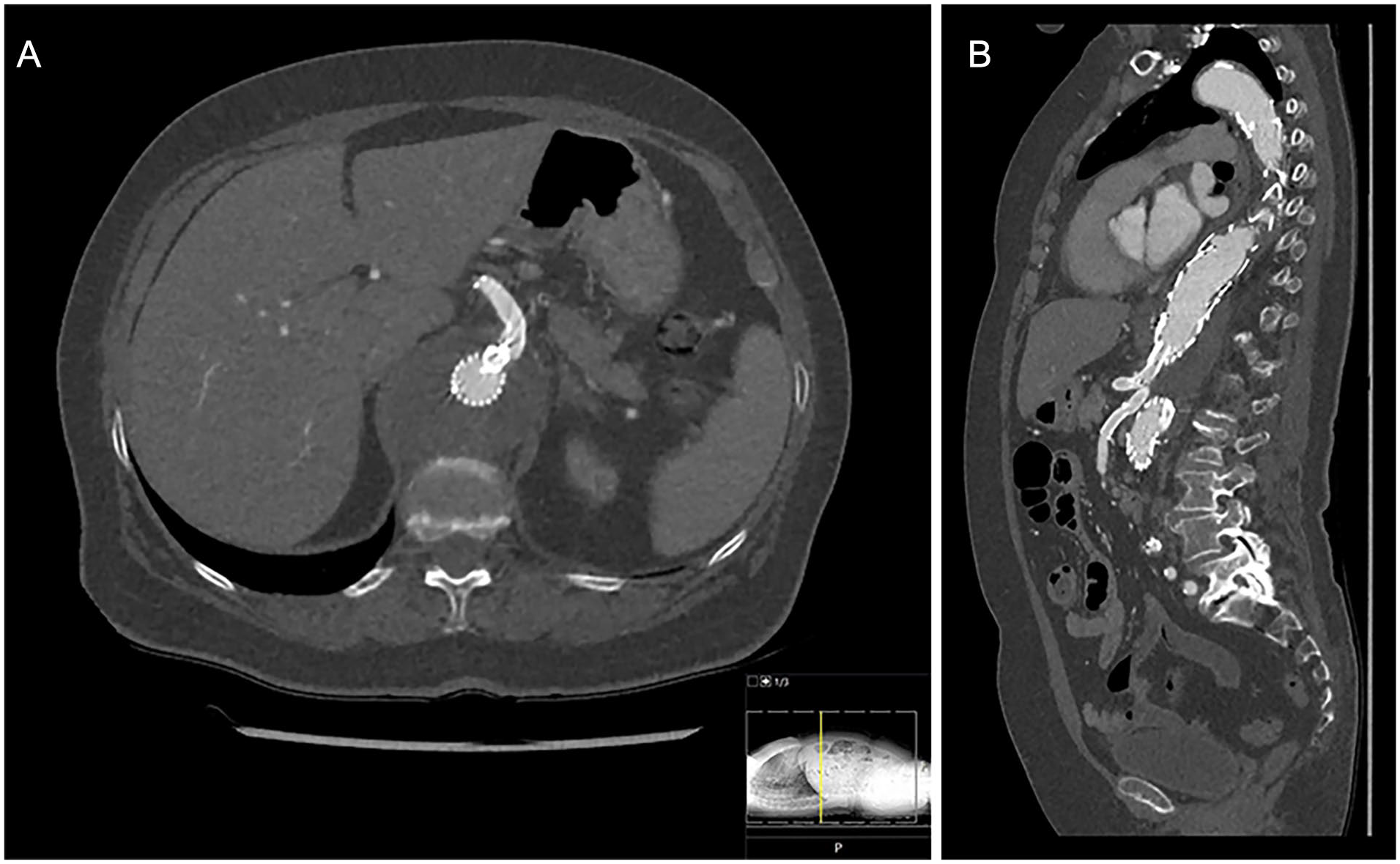
Computed tomography angiography (CTA) of the abdominal aorta (A = axial plane, B = sagittal plane). The CTA showed adequate position of the branched device and visceral stents. There was good patency of the celiac trunk and both renal arteries. Due to metal artifacts of the stent at the origin of the superior mesenteric artery, visualization of patency was not possible.
Discussion
As illustrated in the patient cases, DUS examination with ACCmax measurements could have additional value during follow-up of patients after cEVAR or primary visceral artery stenting. As measurements are performed distal to the endograft or stent, scattering or metal artifacts does not interfere with its diagnostic reliability.
Regular CTA imaging for follow-up after endovascular treatment is associated with cumulative radiation exposure, nephrotoxicity, and high financial health care costs. Therefore, clinical guidelines recommend DUS as an alternative after standard EVAR if neither aneurysm sac enlargement nor endoleak is observed after 1 year.15,16 These guidelines do not address whether this strategy could be applied in surveillance after cEVAR or mesenteric stenting as well. Zierler et al already described an extended DUS protocol for follow-up used at the University of Washington after complex aneurysm repair. 7
In general, a follow-up modality should provide relevant clinical information including aneurysm sac diameter, the presence of endoleaks, and the evaluation of organ perfusion. 3 It is well established that DUS provides good performance in the assessment of sac enlargement and the detection of endoleaks. 4 ACCmax measurements can be used for reliable evaluation of organ perfusion as well. This parameter has already been used for assessing renal artery stenosis, in which a negative predicting value of 95% was found. 17 As ACCmax is measured distal to a stent, difficulty regarding scattering or obscured vision by the stent is prevented. Therefore, ACCmax could be highly valuable for detecting origin or in-stent stenosis of the visceral arteries, as these locations are often not measurable with conventional DUS. Currently, there are no clear cutoff values for the use of ACCmax in visceral arteries. Previous research has shown that measurements below 5.5 m/s2 detect a >50% stenosis with high certainty in PAD, 13 yet these measurements were obtained from the anterior and posterior tibial arteries at the level of the ankle. Although both of these studies are promising, it is unclear whether these results can be extrapolated to detect target vessel stenosis after cEVAR or primary visceral stenting. Therefore, we currently suggest that ACCmax could be of particular interest in cases where CTA is not able to reliably evaluate lesions around stents or endografts.
There are a few important considerations to take into account when DUS is used as a follow-up modality. Although ACCmax measurements can be performed on every location after the stent, abdominal imaging of the aorta and its branches can be challenging in obese patients due to an excess of abdominal fat preventing visualization of mesenteric arteries. Also, overlying bowel gas can make it difficult to accurately visualize vascular structures, which can be reduced if the patient fasts for several hours prior to examination. Furthermore, detection of endoleak and stent dislocation are difficult skills that are dependent on the experience of the vascular ultrasound technician. The CTA and occasionally DSA will therefore remain a useful adjunct for adequate evaluation in the follow-up of cEVAR.
Conclusion
ACCmax measurements can be a useful addition during DUS follow-up for cEVAR and potentially decrease radiation exposure, nephrotoxicity, and health care costs. Further research should focus on detecting relevant stenoses in visceral arteries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
