Abstract
Purpose:
Intravascular lithotripsy (IVL) has shown promising safety and effectiveness in calcified peripheral artery disease (PAD) in large trials and small real-world experiences. Real-world evidence from a larger cohort is lacking, so we aimed to evaluate the real-world acute performance of IVL in the treatment of calcified PAD.
Materials and Methods:
The Disrupt PAD III Observational Study (OS) is a prospective, multicenter, single-arm study. Patients with claudication or critical limb-threatening ischemia (CLTI) and at least moderate calcification were eligible. Independent predictors of procedural outcomes were assessed by multivariable analysis.
Results:
Between November 2017 and June 2021 across 30 global sites, 1373 patients with 1677 lesions (1531, 91.3% core lab evaluable) were enrolled. Diameter stenosis and lesion length was 80.6±17.6% and 93.5±74.3 mm, respectively. Target vessels included femoropopliteal (61%), iliac (15.8%), common femoral (10.7%), and infrapopliteal arteries (12.8%). Lesion characteristics included 31.1% chronic total occlusions (CTOs) and 19.3% long lesions (≥15 cm). At final assessment, residual stenosis was 23.8±11.3%, with 0.9% serious angiographic complications, no abrupt closures, distal embolization, no flow, or thrombotic events. Independent predictors of ≤30% residual stenosis were lesion length ≥15 cm (odds ratio [OR]=0.384), female sex (OR=1.850), age ≤75 years (OR=1.625), IVL balloon to artery ratio ≥1.0 (OR=1.538), and CTO lesions (OR=0.638). Lesion length ≥15 cm (OR=16.076) was an independent predictor of procedural complications.
Conclusions:
The Disrupt PAD III OS represents the largest assessment of IVL periprocedural outcomes in calcified PAD. It confirmed excellent procedural safety and effectiveness in complex lesions across multiple peripheral vascular beds.
Clinical Impact
This final analysis of the PAD III OS represents the largest report of peripheral IVL utilization in daily clinical practice. The outcomes of this study indicate that previously reported procedural results in clinical trial settings can be translated to a broader patient population. Treatment with peripheral IVL in severely calcified stenotic lower limb lesions demonstrated consistent acute safety and stenosis reduction, even in complex patients across multiple vessel beds. In addition, the importance of proper IVL balloon sizing to achieve excellent acute stenosis reduction was confirmed by multivariate analysis.
Introduction
Peripheral artery disease (PAD) contributes to functional limitation, decline in health-related quality of life, and an increased risk of cardiovascular morbidity and mortality in over 200 million patients worldwide. 1 The PAD is frequently complicated by the presence of severely calcified lesions, which can lead to a worse prognosis and is independently associated with higher rates of cardiovascular mortality and morbidity. 2 Severe calcification remains a treatment challenge, even for experienced endovascular interventionists and has been associated with higher rates of acute angiographic complications, including dissections, perforations, or distal embolization.3–5
Although conventional percutaneous balloon angioplasty (PTA) is the standard of care for atherosclerotic lesions, calcium can prevent effective dilatation and luminal gain and lead to complications such as dissection, perforation, or distal embolization during interventional treatment. Alternatives to PTA include stenting, mechanical debulking, and/or modification of the plaque with cutting/scoring balloons or atherectomy, each with relative risks and benefits. In some vessel segments like the iliac arteries, provisional stenting can be a preferred treatment to address calcific lesions as the risk of restenosis of a permanent implant is limited. However, in other segments including the infrainguinal circulation (eg, the common femoral artery [CFA], superficial femoral artery [SFA], or below the knee [BTK]), a strategy eliminating or at least minimizing scaffolding is generally preferred. 6 Avoiding vascular complications during endovascular treatment (EVT) of such segments should be paramount. Although high pressure PTA, specialty balloons, or atherectomy tools can address superficial calcium, they cannot target deep calcium. In addition, their use has frequently been associated with a risk for periprocedural complications, especially dissections and perforations.5,7,8
Intravascular lithotripsy (IVL) is a well-studied calcium modification tool that uses sonic pressure waves to target both superficial and deep calcium to improve vessel compliance, facilitate definitive treatment, and minimize risk to target vessels. 4 The technology uses multiple emitters mounted in a traditional angioplasty balloon catheter that provide pulsatile sonic energy. The generated pressure waves are atraumatic for soft tissue, while selectively disrupting superficial and deep calcium without creating emboli. 4 The effectiveness of IVL has been demonstrated in multiple studies and meta-analyses. 9 Consistently low rates of periprocedural complications with low residual stenosis in heavily calcified PAD were shown.10–16
The aim of the global Disrupt PAD III Observational Study (OS) was to evaluate the procedural safety and effectiveness of peripheral IVL in a real-world complex cohort of patients with calcified PAD across multiple vessel beds, including iliofemoral, femoral, popliteal, and infrapopliteal. In addition, we studied predictors of procedural safety and effectiveness to better understand how IVL treatment might be optimized.
Materials and Methods
Study Device
The Peripheral IVL System (Shockwave Medical, Santa Clara, California) used in the PAD III OS is indicated for lithotripsy-enhanced, low-pressure balloon dilatation of calcified, stenotic peripheral arteries, and has been previously described. 10 The system15,17 consists of a generator, a connector cable, and an IVL catheter that houses an array of lithotripsy emitters enclosed in an integrated balloon.
Study Design and Patient Enrollment
The Disrupt PAD III OS was a prospective, nonrandomized, multicenter, single-arm study designed to assess the acute safety and effectiveness of the Shockwave Peripheral IVL System, in patients being treated for de novo calcified lower limb lesions in the iliofemoral, femoral, popliteal, and infrapopliteal arteries. The study design has been previously described.10,11,17 The IVL could be used in combination with other adjunctive devices, including PTA, specialty balloons, atherectomy, drug-coated balloon (DCB) or stents as determined by the operator’s discretion to allow a real-world representative view on the technology’s utilization in complex lesions. Study design and patient enrollment has been previously described, and the study was conducted in accordance with the Declaration of Helsinki, ISO 14155:2011 guidelines, and Good Clinical Practices. 10 An independent angiographic core laboratory (Yale Cardiovascular Research Group, New Haven, Connecticut) analyzed all angiograms; calcifications were graded by the laboratory using the Peripheral Academic Research Consortium (PARC) criteria. All safety events were adjudicated by a clinical events committee. The ethics committee or institutional review board of each site approved the study protocol and informed consent form, which was signed by all patients prior to study enrollment.
Study Procedures
All investigators were trained in the use of the Peripheral IVL System and adjunctive technologies, including drug-elution therapy and atherectomy; these technologies were used per physician discretion. 10 Vascular access, anticoagulation, introduction of guidewires, and catheter use were conducted according to each institution’s standard of care for endovascular procedures. Angiography (including runoff views) was performed after IVL and at the end of the procedure to assess results.
Statistical Analysis
No formal hypothesis testing was performed for safety and effectiveness outcomes. Continuous variables are expressed as mean ± standard deviation and range. Categorical variables are described as the percentage. Data were analyzed based on availability of core laboratory assessment (Figure 1). If core laboratory assessment could not be cross-referenced to site data, lesions and respective patients were excluded. To assess predictors of outcomes, we performed a stepwise multivariable logistic regression, that included patient, lesion, and procedural characteristics which were identified a priori based on their clinical relevance. The threshold for selection was p=0.2.

PAD III OS study flow. The PAD III observational study consort diagram. IVL, intravascular lithotripsy.
Results
Patient Population
Between November 2017 and June 2021, 1373 patients with 1677 lesions were enrolled across 30 sites in the United States and Europe. Of the 1677 lesions, 91.3% (1531/1677 in 1262/1373 patients, Figure 1) were core lab evaluable and cross-referenced with site-reported data. The complex patient population included 36% chronic limb-threatening ischemia (CLTI) and exhibited risk factors for calcification, including increased age (71.9±9.0), high rates of smoking, diabetes, and renal insufficiency (Table 1). Treated lesion locations included the iliac artery (15.8%), CFA (10.7%), SFA (48.6%), popliteal artery (12.1%), and BTK vessels (12.8%) (Figure 2); 31.1% were chronic total occlusions (CTOs) and 19.3% of lesions had a length of 15 cm or more. Baseline patient and lesion characteristics are summarized in Table 1.
Baseline Patient and Lesion Characteristics.

Data represented as mean±standard deviation or rate in % (n/N) unless otherwise specified.
Abbreviations: ABI, ankle brachial index; CLTI, critical limb-threatening ischemia; CTO, chronic total occlusion; CVA, cerebrovascular accident; MI, myocardial infarction; TIA, transient ischemic attack.
Based on the PARC definition.
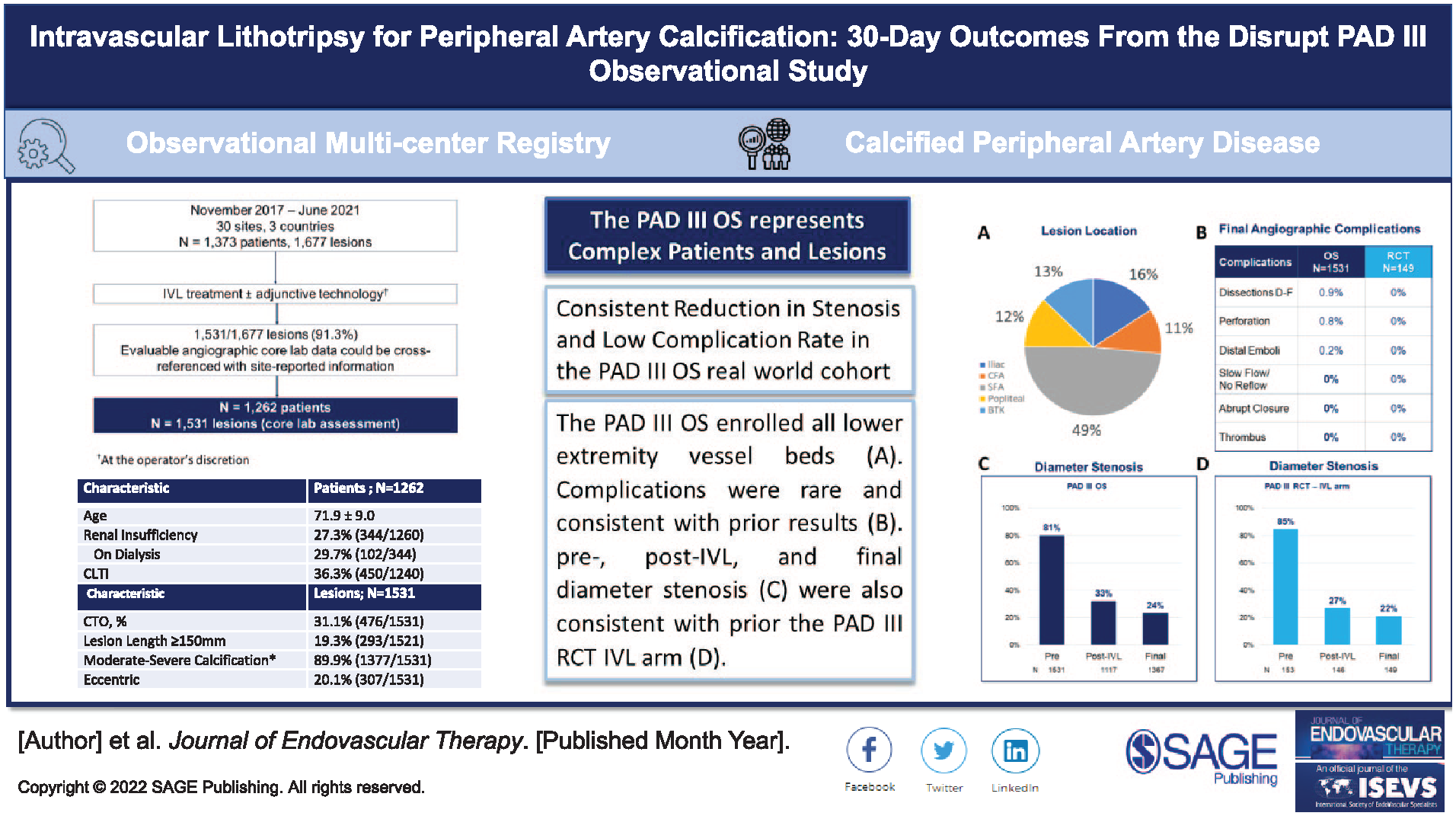
Consistent reduction in stenosis and low complication rate in PAD III OS real-world cohort. The PAD III observational study enrolled patients across all lower extremity vessel beds (A). Final Angiographic complication rates were low and comparable to previous results in the randomized controlled PAD III trial (B). Diameter stenosis pre- and post-IVL and at final assessment in the PAD III OS (C) were also consistent with the prior reported outcomes of the PAD III RCT IVL arm (D). BTK, below the knee; CFA, common femoral artery; IVL, intravascular lithotripsy; SFA, superficial femoral artery; OS, observational study; RCT, randomized controlled trial.
Procedural Details and Acute Outcomes
Patients received lithotripsy treatment with a mean 190.5±107.8 pulses per lesion. As shown in Table 2, the use of IVL with conventional balloon-based technologies was more common than IVL being used with other calcium-modifying therapy (defined as concomitant cutting/scoring balloons and/or atherectomy use, 22.1%). Concomitant cutting/scoring balloons and/or atherectomy use varied across vessel beds; it was lowest in the iliac arteries (6.2%) and highest in the CFA (32.3%). The SFA and popliteal showed similar adjunctive calcium-modifying technology use (26.2% and 24.3%, respectively), whereas BTK arteries had a rate of 15.8%. The use of DCBs accounted for most of the adjunctive therapy (77.7%), with stenting used 31.8% of the time; not surprisingly, stenting was frequently used in iliac arteries (69%) and rarely employed in BTK vessels (10.2%). Embolic protection filters were used in 5.7% of total cases, but only 1.3% of cases if IVL was not used with concomitant atherectomy or specialty balloon.
Procedural Characteristics.

Data represented as mean±standard deviation or rate in % (n/N) unless otherwise specified.
Abbreviations: IVL, intravascular lithotripsy.
Median and interquartile range.
Atherectomy and/or cutting/scoring balloon.
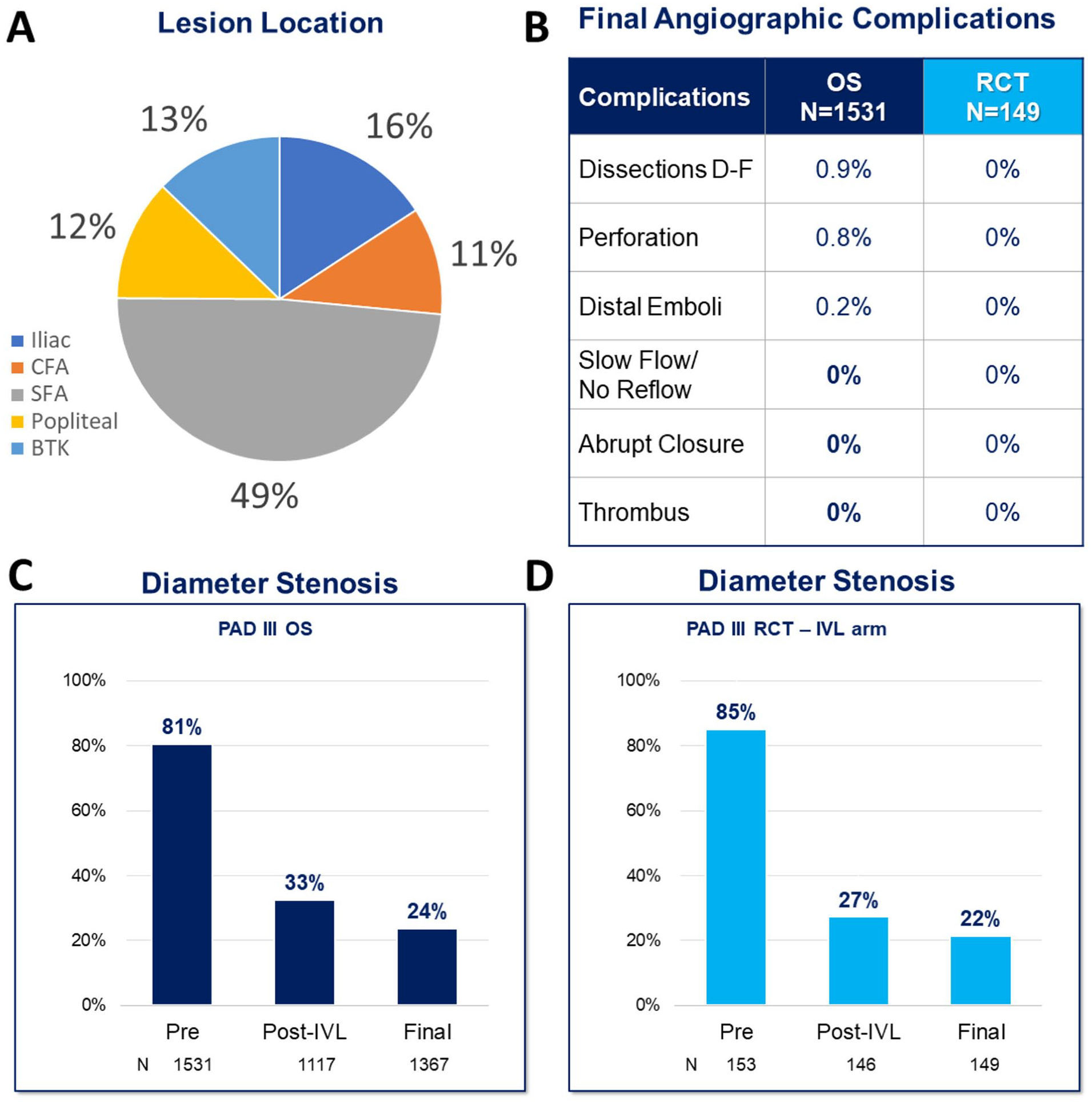
Results and angiographic complications after IVL and at the end of the procedure are summarized in Table 3. The average acute gain at the end of the procedure was 3.3±1.4 mm (4.4±1.8 mm in iliac arteries, 3.4±1.3 mm in CFA, 3.2±1.1 in SFA, 3.0±1.0 in the popliteal arteries, and 2.0±0.7 mm in BTK arteries). The final mean residual stenosis was 23.8%±11.3%, with very consistent results across vessel beds (Figures 2 and 3) and the majority of reduction in stenosis following IVL treatment (Figure 2).
Core Laboratory Adjudicated Lesion Outcomes.

Data represented as mean±standard deviation or rate in % (n/N) unless otherwise specified.
Stenosis reduction based on lesion location. Pre-procedure, post-IVL, and final diameter stenosis across vessel beds in the PAD III Observational study. BTK, below the knee; CFA, common femoral artery; SFA, superficial femoral artery.
Predictors of Acute Procedural Outcomes
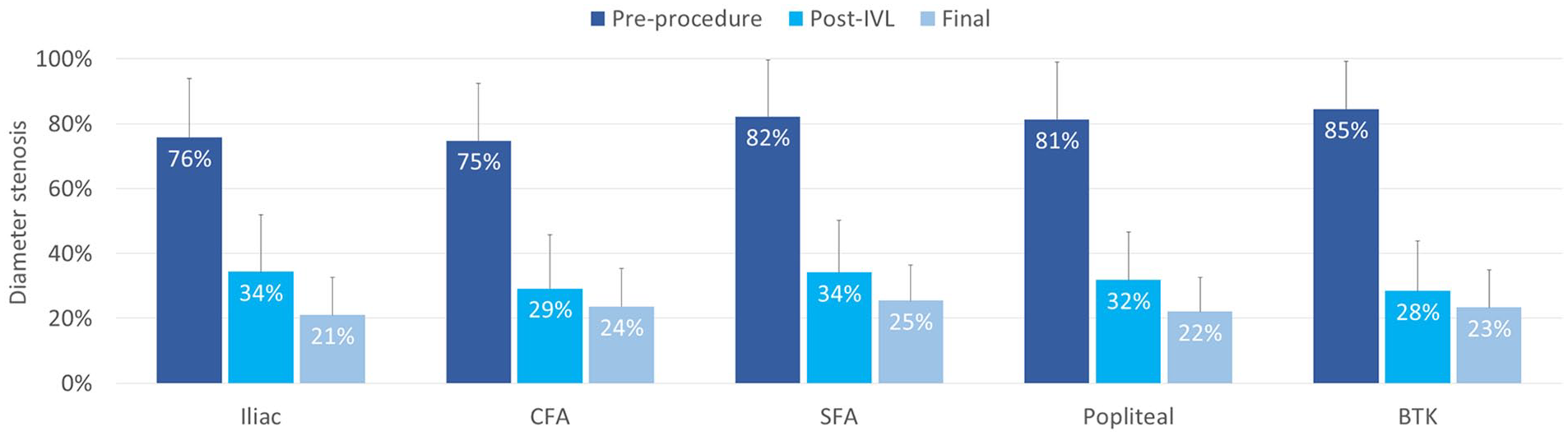
To better understand the patient, lesion, or procedural characteristics that might influence acute outcomes, we performed multivariable logistic regression, for final stenosis ≤30%, and angiographic complications. Independent predictors of final residual stenosis ≤30% are listed in Table 4. For stenosis reduction, lesion length ≥15 cm (odds ratio [OR]=0.384 [0.283-0.521]), female sex (OR=1.850 [1.376-2.489]), age ≤75 years (OR=1.625 [1.247-2.117]), IVL balloon to artery ratio ≥1.0 (OR=1.538 [1.189-1.989]), and CTO lesions (OR=0.638 [0.482-0.844]) lesions were found to be independent predictors. The only independent predictor for angiographic complications was lesion length ≥15 cm (OR=16.076 [3.394-76.152]).
Multivariable Analysis Outcomes for <30% Residual Stenosis and Procedural Complications.

Procedural complications include dissections, perforation, thrombus, distal emboli, or acute flow limitation.
Abbreviations: CTO, chronic total occlusion; IVL, intravascular lithotripsy.
The independent predictors of <30% residual stenosis, and procedural complications, at the end of the procedure were determined by multivariable logistic regression using stepwise selection with a p<0.2 threshold for entry and a p<0.05 level of significance to stay in the final model. The following variables were entered into the models: presence of CTO, lesion length (≥150 mm), performance of postdilatation, IVL balloon to artery ratio (≥1.0 vs <1.0), calcification (by PARC, non/mild vs moderate/severe), reference vessel diameter (RVD) (≥5.4 vs <5.4), number of pulses delivered (≥180 vs <180), BTK lesion location, Rutherford group (claudicant vs CLTI), current dialysis, lesion eccentricity (eccentric vs concentric), diabetes mellitus, sex, smoking status (current vs former), and age (≤75 years vs >75 years). Importantly, long lesions still had low rates of angiographic complications (8.2%, n=16 post-IVL; 3.2%, n=9 final), which were mostly limited to severe dissections (7.3%, n=14 post-IVL, and 2.9%, n=8) and rarely perforations (1.0%, n=2 post-IVL; 0.4%, n=0.4%). There were no occurrences of abrupt closures, acute thrombus, or no/slow flow.
Discussion
This analysis from the Disrupt PAD III OS represents the largest real-world clinical experience with the treatment of severely-calcified lesions in lower limb PAD. The PAD III OS confirmed the safety and effectiveness of IVL in a complex patient and lesion cohort. The series of Disrupt PAD studies (PAD I, PAD II, PAD BTK, and PAD III randomized controlled trial) previously-reported safety and effectiveness of IVL in various patient groups.14,18–20 The PAD III OS extended those findings in a wider, “real-world” setting, highlighting that IVL can be safely used to treat calcified iliac to tibial arteries. The acute outcomes of peripheral IVL in the PAD III OS were also consistent with earlier studies. Results showed excellent stenosis reduction with low rates of periprocedural complications, despite the inclusion of additional vessel beds, more complex patients with more challenging lesions, and the use of adjunctive technologies.
The all-comer PAD III OS enrolled patients with iliac (16%), CFA (11%), SFA (49%), popliteal (12%), and BTK (13%) lesions, with a great proportion of long lesions (19%), CTOs (31%), and high rates of CLTI (36%) and diabetes (56%). Moreover, patients with renal insufficiency (27%), of whom 30% were on dialysis were also included. With the presence of severely-calcified lesions, and the inclusion of complex comorbidities, patients such as those in PAD III OS are frequently excluded from other contemporary peripheral studies. The high degree of comorbidity in this real-world IVL cohort is not surprising, given that severe calcium, especially medial calcium, is a frequent challenge in patients with advanced age, diabetes, CLTI, and/or renal insufficiency. 21 Importantly, even in these challenging patients, results with IVL therapy were consistent with previously-published acute outcomes from observational and randomized controlled studies (Figure 2). 18 Atherectomy is not indicated in iliac arteries, whereas surgical intervention via endarterectomy remains the gold standard for CFA treatment. 22 The successful use of IVL in large vessels (CFA and iliac) in the PAD III OS cohort offers unique insights and an important treatment option for these hard-to-treat vessel beds in which endovascular options were previously-quite limited.
The consistency in acute performance and safety between the PAD III OS and randomized controlled trial (RCT) cohorts further reinforces the broadly generalizable applicability of this technique when treating calcified lesions. In contrast, other real-world studies of calcific PAD often report high rates of procedural complications. 21 A study on the impact of calcium on DCB treatment in PAD, for instance, reported 8.3% flow-limiting dissections and a reduced DCB effectiveness with a higher degree of circumferential calcium. 23 In addition, another recent multicenter prospective DCB study assessing 3165 femoropopliteal lesions reported severe dissections in 4.6%, slow flow in 2.8%, no reflow in 1.1%, acute occlusion in 0.6%, and distal embolization in 0.6% of lesions post-DCB. 24
Importantly, the unrestricted use of adjunctive therapies in the Disrupt PAD III OS cohort provided new insight into the incorporation of IVL into broader clinical practice. Atherectomy or specialty balloons were safely used in combination with IVL in 22.1% of lesions in this study but were associated with a higher rate of embolic protection use. Of note, when IVL was used without any adjunctive calcium-modifying therapy, embolic protection was used in just 1.7% of cases. Atherectomy devices are designed to mechanically debulk plaque but are limited to the treatment of superficial calcium and are subject to wire bias. Using them in combination with IVL could create a potential synergy, as has been described in the coronary setting. 25 Ultimately, treatment strategies for severely-calcified lesions will likely evolve and incorporate individualized approaches.
To better understand how operators can optimize procedural outcomes with IVL, we performed a multivariate analysis, and included patient, lesion, and procedural characteristics. We found that the balloon-to-artery ratio was the only procedure-related significant predictor of residual stenosis <30%. Based on these findings, we recommend optimizing device sizing with a balloon diameter to RVD ratios of 1.1:1. Patient and lesion characteristics that impacted residual stenosis were sex, age, presence of CTO, and long lesions (Figure 4 and Table 4). Interestingly, although women have historically experienced more complications and overall worse outcomes after EVT, 26 female sex was a positive predictor for stenosis reduction in the PAD III OS and did not affect acute safety in our data set. The severity of calcium, presence of CLTI, and the target lesion location did not significantly predict residual stenosis, highlighting the consistent effectiveness of IVL in challenging settings across vessel beds. This is noteworthy as clinicians can expect successful angioplasty in even the most difficult lesions with the highest burden of calcification when applying IVL across vascular territories ranging from the iliac to infrapopliteal circulation. Importantly, although both CTO and long lesions were predictive of residual stenosis, IVL results in both subgroups were acceptable with a final residual stenosis of 30% in long lesions and 27% in CTO (Figure 2). The only characteristic that impacted the risk for angiographic complications was lesion length, confirming the consistent safety of IVL across vessel beds, and in challenging patients and lesions.
Independent predictors of procedural outcomes. Analysis of independent predictors of <30% residual stenosis (A). Identified the presence of long lesions (B), which was also a predictor of procedural complications (C), and total occlusions (D) as negative predictors. Optimized IVL balloon sizing, age≤75 years, and female sex were positive predictors for low (<30%) residual stenosis (A); CTO, chronic total occlusion.
Limitations
Limitations are inherent to the PAD OS design as a single-arm study without any control and have been previously described. 10 Although the study’s purpose was to assess acute IVL outcomes in a real-world setting, only acute procedure results are reported in this study, no follow-up will be available on these patients. Although many characteristics were site-assessed, angiographic outcomes required core laboratory assessment. Matching the site data with core laboratory data led to a loss of about 8% of lesions during the analysis. Finally, although comparisons across studies are generally not recommended, in the case of PAD III OS, it is especially hard to draw comparisons with other works, as the included patients are usually excluded from clinical trials.
Conclusions
This final analysis of the PAD III OS represents the largest report of IVL utilization in daily clinical practice. Treatment with peripheral IVL in severely-calcified stenotic lower limb lesions continued to demonstrate consistent acute safety, even in complex patients across multiple vessel beds. The importance of proper IVL balloon sizing to achieve excellent acute stenosis reduction was confirmed by multivariate analysis. Possible future comparative studies with longer-term follow-up and detailed treatment algorithm, including specified final therapy, could further investigate if these favorable acute results translate to a longer-term clinical benefit in these hard-to-treat patients.
Footnotes
Acknowledgements
The authors thank Maureen J Ostaff, PhD, for medical writing and Ryan Shields, MS, Suzanne Wilson, MS, and Padma Seethamraju, MS, for statistical support.
Authors’ Note
These data were presented on podium by Dr Ehrin J Armstrong as part of the Late-breaking Clinical Trial session at VIVA 2022.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.J.A. is a consultant to Abbott Vascular, BD Bard, Boston Scientific, Gore, Medtronic, Philips, and Shockwave Medical. S.A.P. receives institutional research support from Abbott Vascular, Boston Scientific, Shockwave Medical, TriReme Medical, Surmodics, Veryan Medical, MedAlliance, Concept Medical, and Acotec. He serves as an advisor to Abbott Vascular, Boston Scientific, BD, Cordis, Medtronic, Philips, and R3 Medical. He is a consultant to Terumo, Penumbra, Inari, and Canon and has equity in Advanced Nanotherapies, eFemoral, and Encompass Vascular. G.A. is a consultant and has education responsibilities for Shockwave Medical, Abbott Vascular, Phillips, and is CMO of Cordis. P.A.S. is a consultant to AngioSafe, Boston Scientific, Cordis, Endologix, Shockwave Medical, and W.L. Gore. He is on the Advisory Board of AngioSafe, Endologix, and Shockwave Medical. He receives institutional research support from Boston Scientific, Contego Medical, Endologix, MedAlliance, Micromedical Solutions, Philips, Reflow Medical, Shockwave Medical, SoundBite, and W.L. Gore. S.S.M. is a consultant for Shockwave Medical, Philips, Cardiovascular Systems Inc., and Medtronic. N.W.S. receives educational and research grants from Angiodynamics, Boston Scientific, Bard/BD, and Abbott and is an investigator on several Shockwave Medical studies. He is a consultant to Cardiovascular Systems, Inc, Abbott, Angiodynamics and Bard. A.M. is a consultant and trainer for Abbott, Phillips, and a speaker for Janssen. B.B. is a consultant and trainer for Medtronic Vascular, and a speaker for Shockwave Medical. W.A.G. is a consultant for Alucent, Boston Scientific Corporation, Edwards Lifesciences, efemoral, Medtronic, Phillips, W.L. Gore and Associates, Inc, and Shockwave Medical. He is performing contracted research for Boston Scientific Corporation, Edwards Lifesciences LLC, Medtronic, Phillips, and Shockwave Medical. G.T. is on the advisory board for BSC, Medtronic, and receives study support from B.Braun, Bold, BSC, Biotronic, Verian, Philips, and Shockwave Medical. E.Y.W. has no disclosures. J.F.M. has no disclosures. A.H. is a Medical Advisory Board Member for Medtronic, Gore, Philips, and Boston Scientific. He is a clinical investigator for Abbott, Bard-BD, Biotronik, Boston Scientific, Cagent, Cook, Efemoral, Endologix, Endospan, FluidX, Gore, Medtronic, Nectero, Philips, Reflow Medical, Shape Memory, Shockwave, Terumo, and TriReme. S.A.P. is a Clinical Trial Investigator for Abbott Vascular, Acotec, Boston Scientific, Concept Medical, MedAlliance, and Shockwave Medical. He is consulting or part of advisory boards for Abbott Vascular, Boston Scientific, Cordis, Medtronic, and Philips; receives grants or research support from Abbott Vascular, Shockwave Medical, Surmodics, and TriReme; conducts contracted research for Acotec, Concept Medical, and MedAlliance; a speaker for Inari, Penumbra, Shockwave Medical, and Teruma; and he is part of a DSMB for Boston Scientific.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shockwave Medical Inc (Santa Clara, California) provided financial support for this research.
