Abstract
Purpose:
Vessel calcification is estimated to be present in 30% to 50% of patients with peripheral arterial disease (PAD) and is one of the main challenges in endovascular treatment. The popliteal artery is unique compared with other arteries due to its exposure to significant deformation and biomechanical stress during knee motion. Intravascular lithotripsy (IVL) is a novel technique that uses acoustic pressure waves to cause microfractures within the intimal and medial wall calcification. Intravascular lithotripsy is safe in femoropopliteal and infrapopliteal lesions, but follow-up studies are lacking. Therefore, the purpose of this study was to describe the first follow-up outcomes of IVL in popliteal and infrapopliteal arterial disease.
Methods:
This prospective, multicenter cohort study included all patients treated with IVL in the popliteal and infrapopliteal arteries at 4 sites. Standardized follow-up with duplex ultrasonography was scheduled at 6 to 8 weeks and 12 months. The primary safety endpoint was a composite of major adverse events (MAEs) at 30 days. Primary efficacy endpoints were primary patency, limb salvage, and amputation-free survival (AFS) at 12 months. Secondary endpoints were primary-assisted patency and freedom from target lesion revascularization (TLR). Endpoints were distributed for patients with chronic limb–threatening ischemia (CLTI) and intermittent claudication (IC) and estimated using the Kaplan-Meier method.
Results:
Between April 2021 and March 2023, 29 patients with 30 limbs were treated. Diabetes mellitus (DM) and CLTI were present in 62.1% and 80.0% of patients, respectively. Within the 32 treated lesions, severe calcification was present in 84.4% and bailout stenting was necessary in 12.5% of the lesions. Four MAEs occurred within 30 days: 1 closure device failure, 1 major amputation, and 2 deaths, neither of which was related to the study device. The primary patency, primary-assisted patency, freedom from TLR, limb salvage, and AFS at 12 months were 68.8%, 90.0%, 93.3%, 83.9%, and 57.1% for CLTI patients, respectively. No events occurred in restenosis, re-occlusion, TLR, major amputation, or mortality in patients with IC.
Conclusions:
This first-ever analysis on follow-up outcomes of IVL in the popliteal and infrapopliteal arteries demonstrated promising safety and efficacy outcomes with a low rate of bailout stenting.
Clinical Impact
Vessel calcification is a common feature in peripheral arterial disease (PAD) and is one of the main challenges in endovascular treatment. The popliteal artery is subjected to biomechanical stress during knee motion, which makes stenting unappealing and often leads to worse clinical outcomes. This study aimed to describe the first follow-up outcomes of IVL in popliteal and infrapopliteal arterial disease. As in line with previous studies, no relevant procedural complications were found and the rate of bail-out stenting was only 12.5%. Moreover, in a complex patient population, this study demonstrated promising safety and efficacy outcomes. The comparison of IVL with angioplasty alone or other vessel preparation devices for popliteal and infrapopliteal arterial disease is warranted.
Keywords
Introduction
Peripheral arterial disease (PAD) can manifest with lesions at different anatomical levels, with each level having its own challenges. Popliteal lesions are often evaluated together with superficial femoral lesions under the heading of femoropopliteal disease.1,2 However, the exposure to significant deformation and biomechanical stress during knee motion makes the popliteal artery unique from other arteries. 3 For this reason, the implantation of stents may be complicated by an unacceptable risk of stent fracture and restenosis or acute occlusion and should therefore be avoided in this mobile area.3,4
Vessel calcification is estimated to be present in 30% to 50% of PAD patients and is one of the main challenges in endovascular treatment. 5 Severe calcification causes difficulties to cross the lesion with wires or balloons, restricts vessel expansion leading to early elastic recoil, and impairs the antiproliferative effect of drug-coated devices.5–7 Furthermore, severe calcification is associated with an increased risk of technical failure, restenosis, clinically-driven target lesion revascularization (CD-TLR), amputation, and mortality after an endovascular intervention.8–11
The main risk factors for calcification comprise advanced age, diabetes mellitus (DM), and chronic kidney disease (CKD). 5 With an estimated increase in the prevalence of DM from 463 million today to 700 million in 2045, not only will the prevalence of PAD increase but also the percentage of patients manifesting with severe calcification. 12
Atherectomy was developed to overcome the problem of severe calcification and potentially decrease bailout stenting. However, atherectomy has the disadvantage of not being selective for the calcified plaque, leading to simultaneous removal of healthy intimal soft tissue, whereas deeper calcifications are not adequately treated. 13 Several trials reported perforation rates over 5%.14,15 Furthermore, atherectomy shows comparable results to plain old balloon angioplasty (POBA) or drug-coated balloon (DCB) angioplasty alone in popliteal and infrapopliteal arterial disease.16–18
Intravascular lithotripsy (IVL) is a novel and less aggressive technique that uses acoustic pressure waves to cause microfractures within the intimal and medial wall calcification. 19 Intravascular lithotripsy is safe in femoropopliteal and infrapopliteal lesions with minimal vascular complications.19–21 However, efficacy studies with follow-up are lacking, particularly those investigating popliteal and infrapopliteal lesions. 22 Therefore, the purpose of this study was to describe the first follow-up outcomes of IVL in popliteal and infrapopliteal arterial disease.
Materials and Methods
This study was a retrospective analysis of prospectively collected data. All consecutive patients treated with IVL in the popliteal and infrapopliteal arteries in 4 Dutch hospitals were included. Patients were eligible if they were treated for disabling intermittent claudication (IC) or chronic limb–threatening ischemia (CLTI), which corresponds to Rutherford stages 3 to 6, and if the lesion was calcified at the discretion of the treating physician. The level of calcification was assessed by the physician based on a preoperative computed tomography angiography (CTA) and perioperative digital subtraction angiography (DSA). Patients with simultaneous endovascular inflow procedures or hybrid procedures were also included. Informed consent was obtained from all patients before participation. Exclusion criteria were acute limb ischemia, distal embolization after a proximal intervention, aneurysmal disease, nonatherosclerotic arterial disease, and patients unable to give informed consent.
Study Device and Procedures
The Shockwave Peripheral IVL system (Shockwave Medical, Santa Clara, California) consists of a generator, a connecter cable, and an IVL catheter with lithotripsy emitters enclosed in an integrated balloon. The system delivers pulsatile sonic pressure waves that selectively disrupt and fracture calcium, altering vessel compliance, while minimizing injury and maintaining the integrity of the fibro-elastic components of the vessel wall. The length of the IVL balloon used was 60 mm, whereas the diameter ranged from 3.0 to 6.0 mm. A sizing of the IVL catheter of 1.1:1 relative to the reference vessel diameter was used. The balloon expanded to 4 atm delivers 1 pulse per second. After every cycle of 30 pulses, the balloon was inflated nominally to 6 atm to maximize luminal gain. The number of cycle repetitions depended on the vessel diameter obtained and was at the discretion of the treating physician.
Predilatation with undersized POBA was performed in patients with chronic total occlusions (CTOs) or near-occlusions to enable the crossing of the IVL catheter. After IVL treatment, postdilatation with POBA or DCB was performed in the case of a flow-limiting dissection or significant residual stenosis. The choice for POBA or DCB was at the discretion of the physician. If a flow-limiting dissection or stenosis persisted, provisional stenting was performed. A completion angiography was performed to assess the final procedural result and run-off. Postoperative antithrombotics were at the physician’s discretion. Standardized follow-up visits with duplex ultrasonography (DUS) and clinical evaluation were scheduled at 6 to 8 weeks and 12 months after the intervention.
Endpoints and Definitions
The primary efficacy endpoints were primary patency, limb salvage, and amputation-free survival (AFS) at 12 months. Primary patency was defined as freedom from TLR and a restenosis >50%, as determined with a peak systolic velocity (PSV) ratio >2.4 on DUS. Limb salvage was defined as freedom from a major amputation (above the ankle), whereas AFS defines the survival without major amputations.
The primary safety endpoint was the freedom from a composite of major adverse events (MAEs) through 30 days, defined as abrupt closure, distal embolization, perforation, emergency revascularization, major amputation, and death. Secondary endpoints were primary-assisted patency, freedom from TLR, and survival at 12 months. Primary-assisted patency was defined as the durability of an intervention that failed initially but not to the level of thrombosis and was retreated. Functional outcomes were improvement in Rutherford score (of at least 1 category) for all patients and wound healing for CLTI patients.
In all lesions, calcification was scored by 1 observer (M.J.N.) according to the peripheral arterial calcium scoring system (PACSS) and the tibial artery calcification (TAC) score proposed by Kang et al.5,8 In the PACSS score, grade 0 to 5 refers to the absence of visible calcium at the target lesion, unilateral calcification <5 cm, unilateral calcification ≥5 cm, bilateral calcification <5 cm, and bilateral calcification ≥5 cm, respectively. 5 In the TAC score, mild calcification was defined as a calcium mass ≤10 mm and a total extent of calcification <10% of the target vessel, whereas severe calcification was defined as linear calcification with a length >50 mm or diffuse calcification comprising >50% of the target vessel. Moderate calcification delineates calcification in between these 2 categories. 8
Statistics
Statistical analyses were performed with RStudio version 1.3 (Boston, Massachusetts). Continuous variables are presented as mean values±standard deviation, whereas categorical variables are given as counts and percentages. The MAE at 30 days and functional outcomes were reported as absolute percentages. Cumulative patency, freedom from TLR, limb salvage, and AFS were distributed for patients with CLTI and IC and were estimated using the Kaplan-Meier (KM) method. Limbs were included in the KM analysis only if the intervention was performed more than 3 months before the start of the analysis. The threshold for statistical significance was set at p<0.05.
Results
Baseline and Procedural Characteristics
Between February 2021 and March 2023, 29 consecutive patients with 30 limbs and 32 lesions were treated with IVL. The baseline characteristics reflect a complex and diseased patient population (Table 1). Mean age was 73.9 years, coronary artery disease was present in 44.8%, and cerebrovascular disease was present in 31.0%. The main risk factors for calcification, namely, DM, CKD, and a history of smoking were present in 62.1%, 37.9%, and 82.8% of patients, respectively. Chronic limb–threatening ischemia was present in 80.0% of the limbs.
Baseline Characteristics.
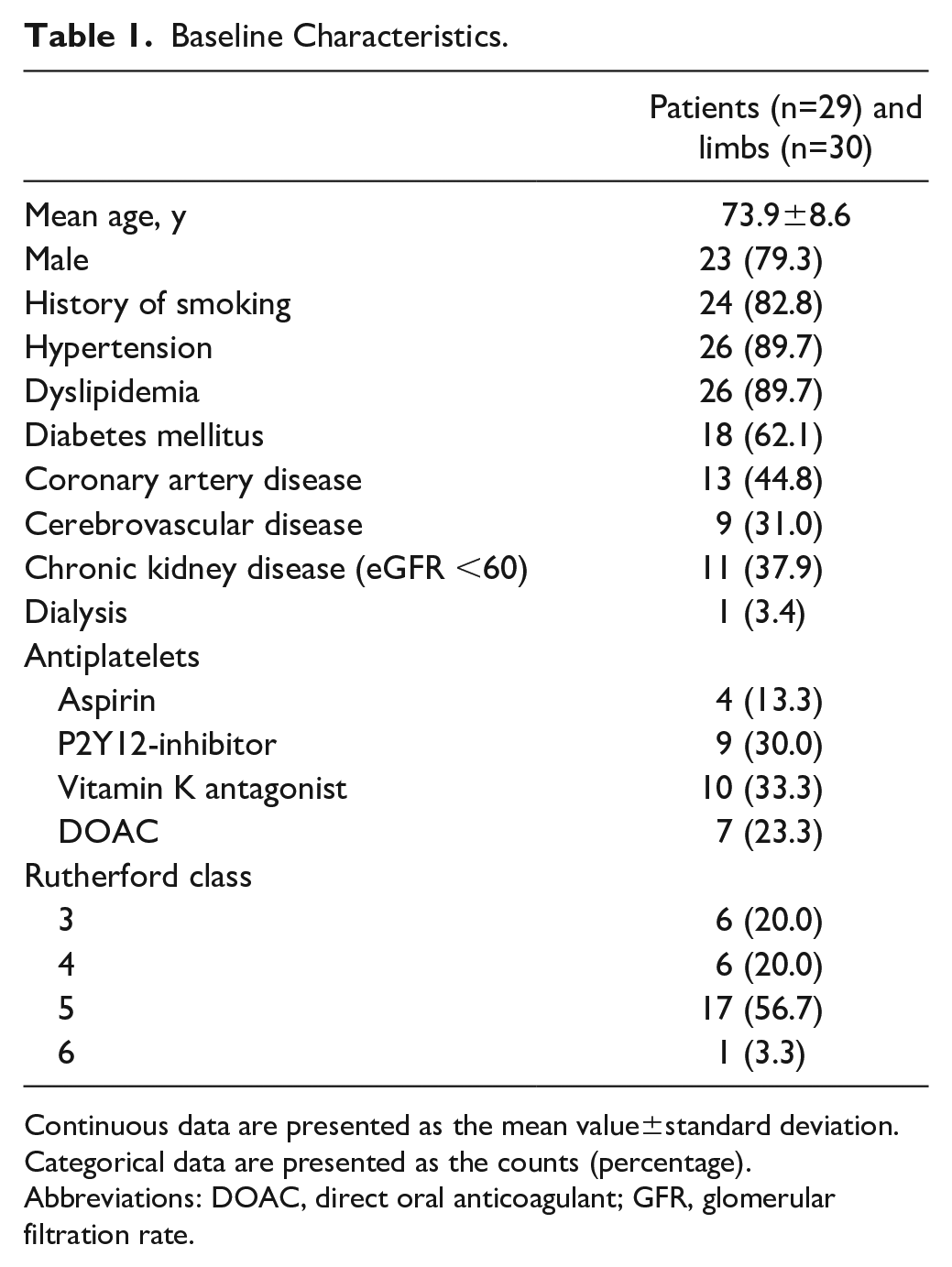
Continuous data are presented as the mean value±standard deviation. Categorical data are presented as the counts (percentage).
Abbreviations: DOAC, direct oral anticoagulant; GFR, glomerular filtration rate.
The popliteal artery was treated in 87.5% of the lesions, whereas the tibioperoneal trunk and proximal one-third of the anterior tibial artery were treated in 3.1% and 9.4%, respectively. Mean lesion length was 75.8±69.4 mm, whereas moderate and severe TACs were present in 15.6% and 84.4% of the lesions, respectively (Table 2).
Angiographic Characteristics.
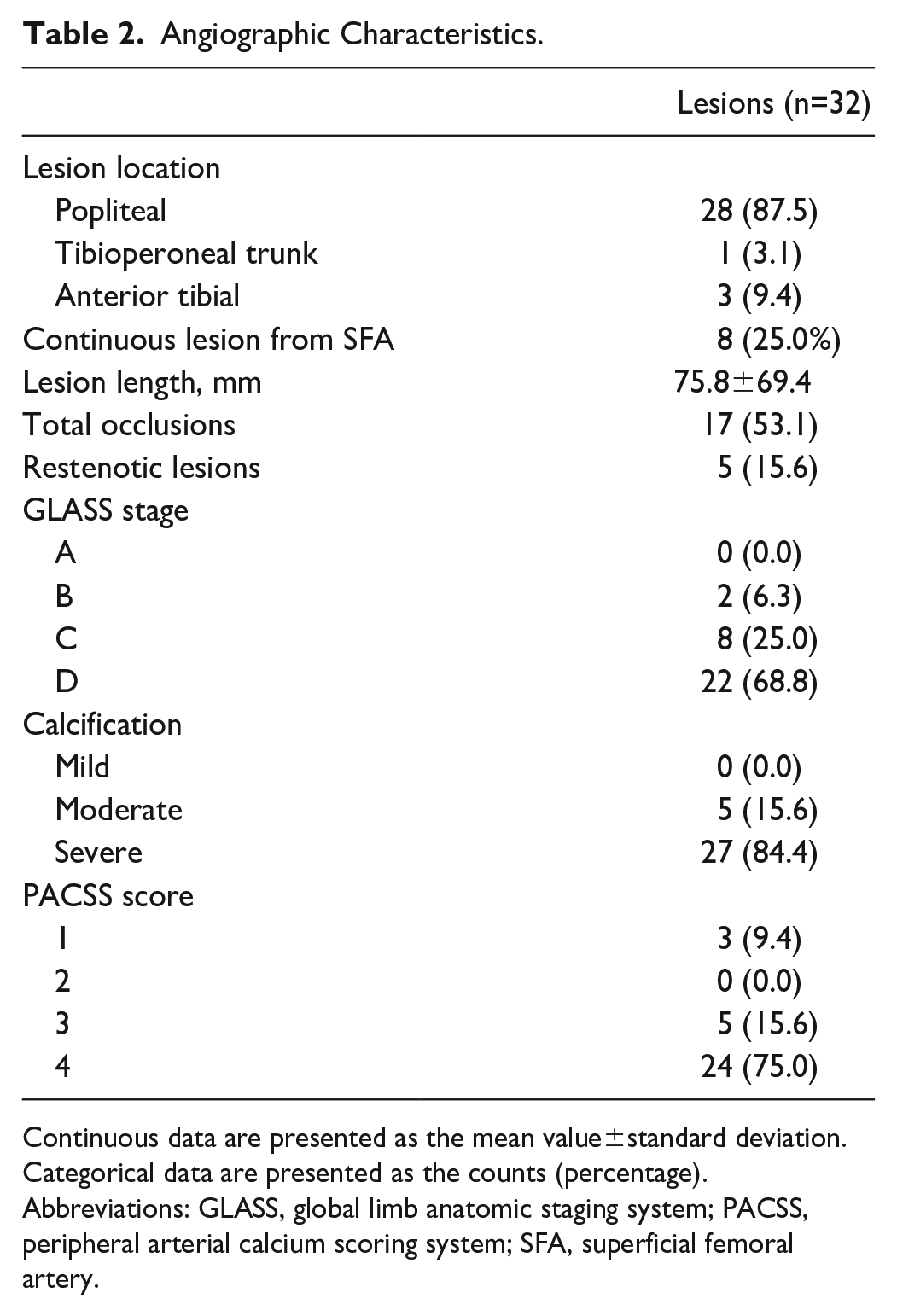
Continuous data are presented as the mean value±standard deviation. Categorical data are presented as the counts (percentage).
Abbreviations: GLASS, global limb anatomic staging system; PACSS, peripheral arterial calcium scoring system; SFA, superficial femoral artery.
Procedural success was achieved in 100% of the interventions. Endovascular inflow and hybrid procedures were performed in 60.0% and 30.0% of the interventions, respectively. Adjunctive POBA and DCB angioplasty were used in 81.3% and 28.1% of the lesions, respectively. In 12.5% of the lesions, bailout stenting was deemed necessary (Table 3).
Procedural Characteristics.
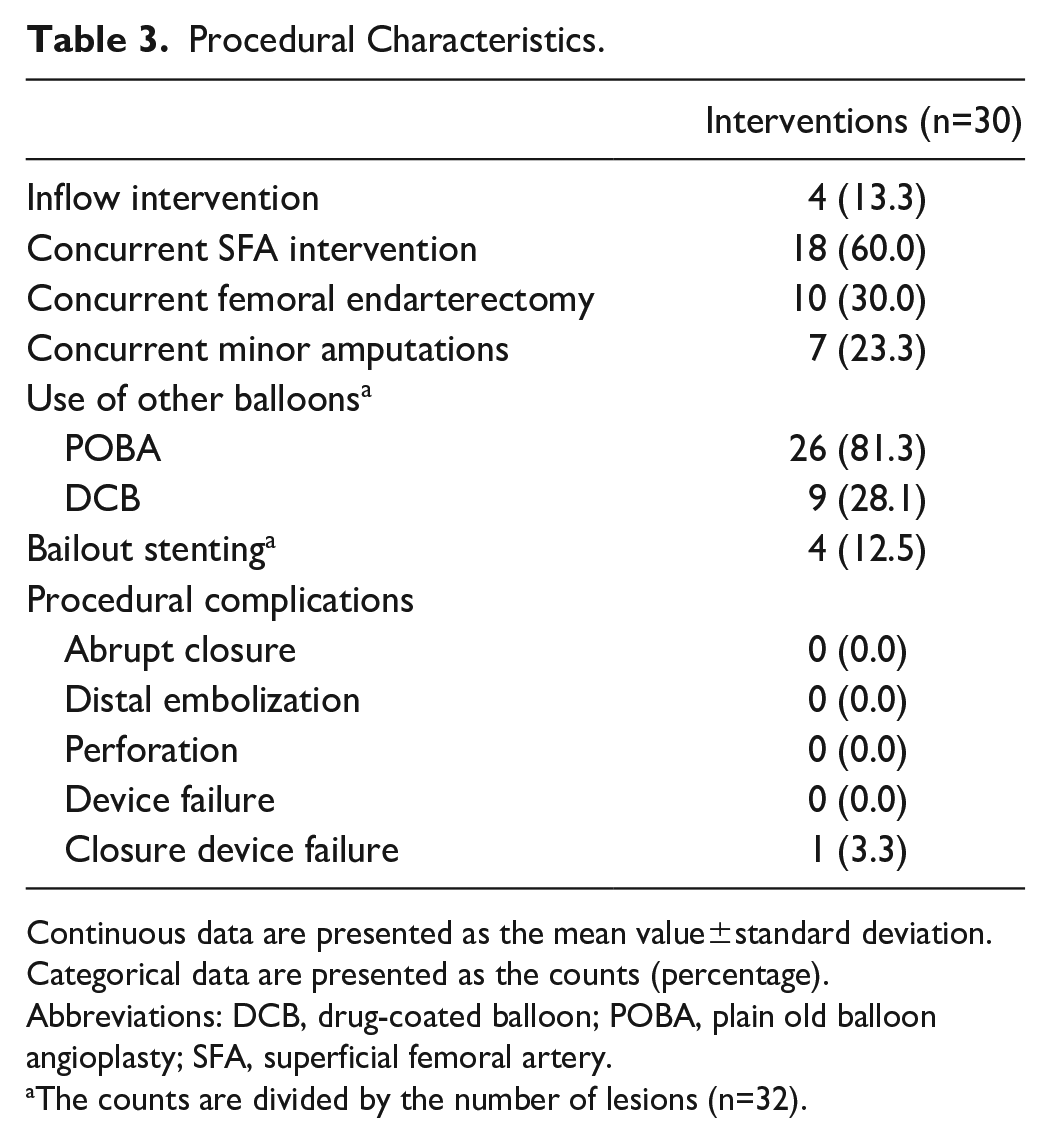
Continuous data are presented as the mean value±standard deviation. Categorical data are presented as the counts (percentage).
Abbreviations: DCB, drug-coated balloon; POBA, plain old balloon angioplasty; SFA, superficial femoral artery.
The counts are divided by the number of lesions (n=32).
Efficacy Outcomes
Twenty-five limbs with a mean follow-up of 11 months (range=0-23) were included in the KM analysis. The primary patency, limb salvage, and AFS for CLTI patients were 68.8%, 83.9%, and 57.1% at 12 months, respectively (Figure 1). During follow-up, 3 major amputations were performed due to progressive foot ulceration without infection, all within 3 months of intervention. A total of 5 patients died, whose causes of death were acute coronary syndrome (ACS), acute mesenteric ischemia, and in 3 patients a palliative course, including 1 due to progressive foot ulceration.
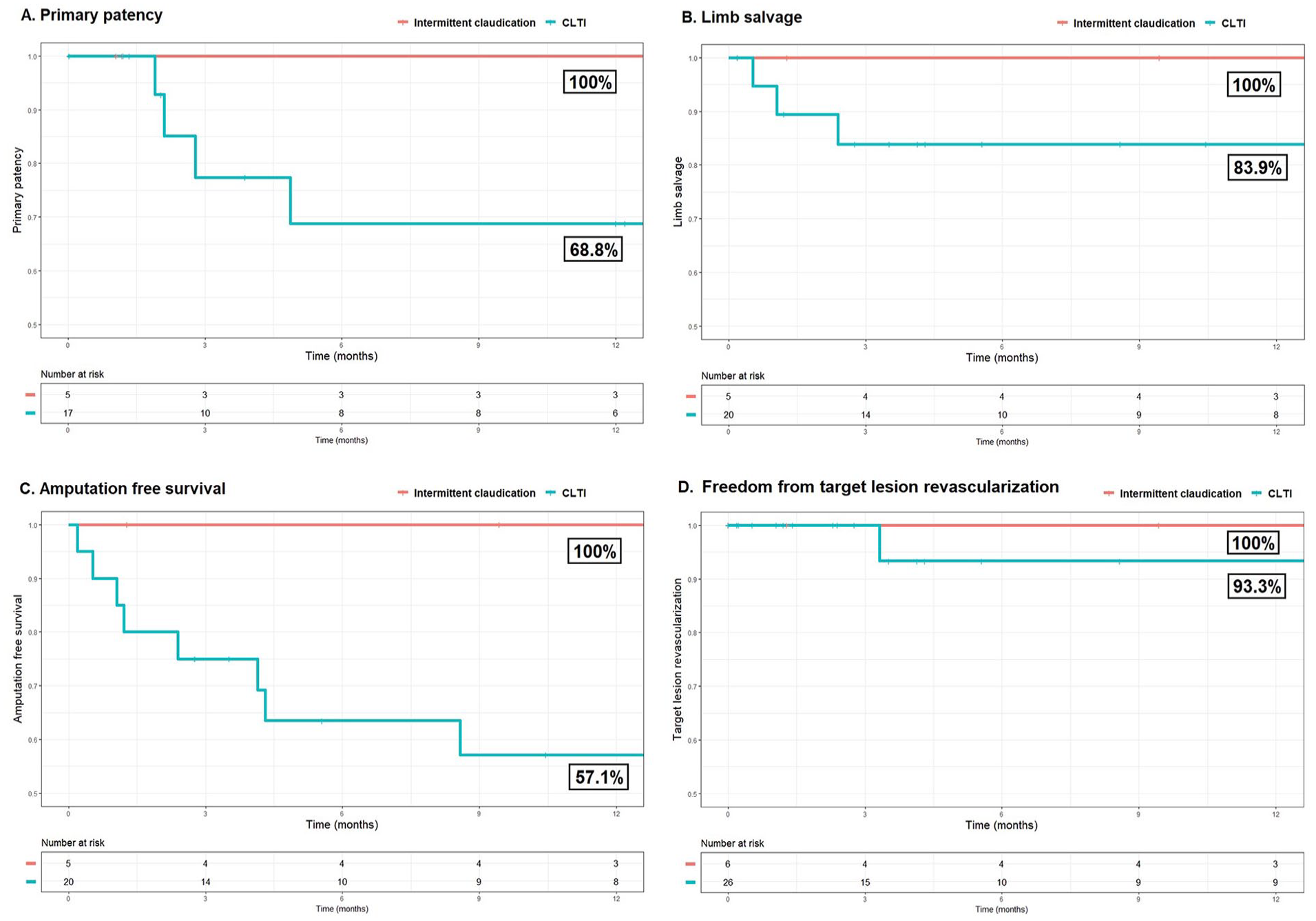
The 12-month primary patency (A), limb salvage (B), amputation-free survival (C), and freedom from target lesion revascularization (D) divided between patients with intermittent claudication (IC) and chronic limb–threatening ischemia (CLTI).
The primary-assisted patency, freedom from TLR, and survival were 90.0%, 93.3%, and 72.2% for CLTI patients, respectively. No events occurred in restenosis, re-occlusion, TLR, major amputation, or mortality in patients with IC.
Safety Outcomes
The rate of MAE at 30 days was 13.3%. In 1 patient, the closure device failed and led to an acute occlusion, after which a femoral endarterectomy was performed to remove the closure device. Another patient was amputated after 16 days due to progressive foot ulceration. Two patients died within 30 days after the intervention because of an ACS and a palliative course due to treatment-requiring multimorbidity and lack of perspective. All MAEs occurred in patients with CLTI, but none of them were related to the study device.
Functional Outcomes
The Rutherford score improved at 6 weeks in all 5 patients with IC (100%) and in 6 of 17 patients (35.3%) with CLTI at baseline. At 1 year, all 3 IC patients (100%) and 7 of 8 CLTI patients (87.5%) who completed follow-up improved in the Rutherford score.
Eight of the 15 limbs (53.3%) with Rutherford 5 or 6 at baseline completely healed after a median follow-up of 127 days. Three limbs underwent major amputation, 1 patient died due to wound progression or infection, and 2 other patients died before wound healing could take place (1 ACS and 1 palliative course due to severe multimorbidity). One limb was still in the process of healing.
Discussion
This first-ever analysis of follow-up outcomes after IVL in popliteal and infrapopliteal arteries demonstrates promising safety and efficacy outcomes with a low rate of bailout stenting in a complex patient population.
In this cohort, the only procedural complication was a failing closure device. These promising procedural outcomes are consistent with the results of other femoropopliteal and infrapopliteal IVL studies.19–22 In contrast, studies on atherectomy in popliteal and infrapopliteal arteries show higher rates of distal embolization, abrupt closure, and perforation.16,17,23–26
This study demonstrated a bailout stenting rate of only 12.5% after IVL. In the previous literature, the rates of bailout stenting of 16% to 45% after POBA or DCB angioplasty alone in the popliteal artery have been described.16,17,27 To date, the precise clinical implications of significant deformation and biomechanical stress of the popliteal artery during knee motion are not well understood.28,29 However, stent placement could lead to impaired arterial remodeling and interfere with future surgical options. Also, stent fracture rates of 3.4% to 7.1% have been described in the popliteal artery at 12 months.27,30 Lastly, it seems that severe calcification leads to an increase in maximal curvature of the popliteal artery during knee motion. 28 To conclude, avoidance of a stent in the popliteal artery should be pursued until more conclusive literature is published.
As previously described, vessel calcification limits procedural and clinical outcomes after revascularization. For that reason, patients with severe calcification are often excluded from clinical trials.31,32 Atherectomy was developed to overcome the problem of severe calcification and prepare the artery for the definitive angioplasty treatment. A recent meta-analysis on atherectomy for femoropopliteal disease found a reduction in flow-limiting dissections and need for bailout stenting, but no difference in primary patency, TLR, and mortality, compared with angioplasty alone. 33 However, studies on atherectomy in popliteal arteries describe perforations in 5%, abrupt closures in 2.5%, and distal embolization in 5% to 22% of the patients.16,17 Unlike atherectomy, IVL can selectively differentiate between calcium and soft tissue, allowing intimal and medial calcium to be addressed with minimal vascular injury. Both this study and previous studies on IVL in femoropopliteal and infrapopliteal disease did not report any case of perforation, abrupt closure, or distal embolization.19–21
Despite promising patency and TLR rates at 12 months, the rate of AFS was relatively low in this study. All patients in this study were treated for moderate or severe peripheral calcification. Furthermore, two thirds of patients were treated for multilevel disease; 60% and 30% of limbs were undergoing concurrent proximal endovascular intervention or femoral endarterectomy, respectively. Previous studies have shown that severe peripheral calcification is an independent predictor of low AFS with a 3- and 5-fold higher risk of amputation and all-cause mortality, respectively.8,34
In this study, the choice for postdilatation with POBA or DCB was at the discretion of the treating physician, which led to DCB use in only 28.1% of the cases. Previous studies on follow-up outcomes after IVL in femoropopliteal disease describe primary patency rates of 55% and 81% after postdilatation with POBA and DCB, respectively (Table 4).35–37 Furthermore, in a meta-analysis on 13 trials, DCB consistently reduced late lumen loss, binary stenosis, and TLR compared with POBA alone in the treatment of femoropopliteal disease. 38 However, these trials included a vast majority of patients with IC, and it is therefore unclear if these results can be extrapolated to all patients with PAD. 39 Nevertheless, based on the previous literature on femoropopliteal disease, the low rate of DCB use in our cohort may have limited optimal endovascular treatment and therewith optimal patency outcomes.
Overview of the Published Studies on Follow-up Outcomes After Intravascular Lithotripsy in the Femoropopliteal Arteries.
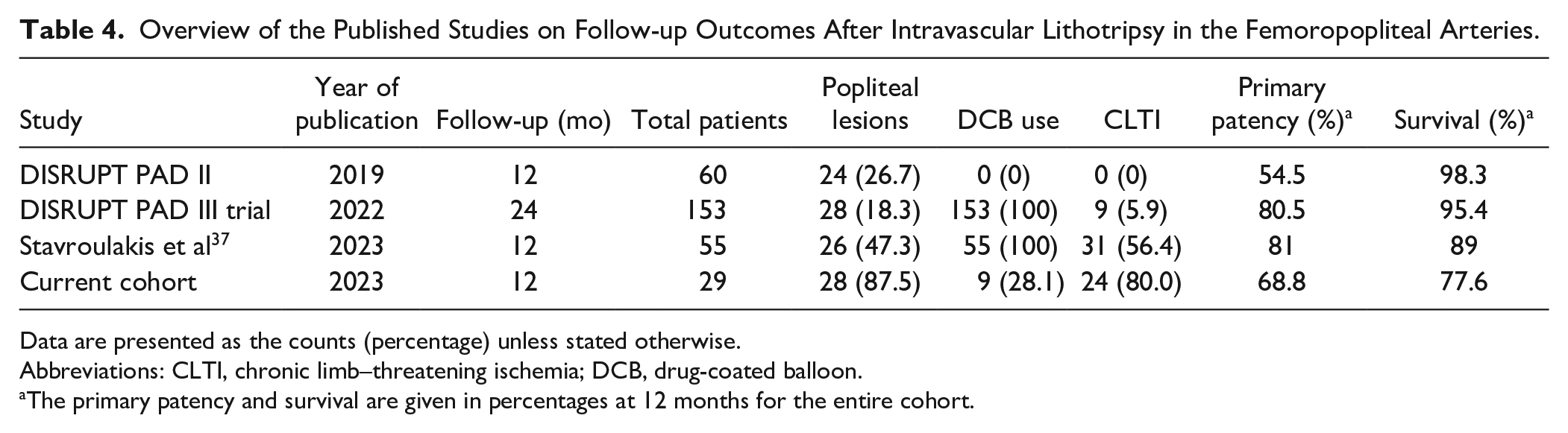
Data are presented as the counts (percentage) unless stated otherwise.
Abbreviations: CLTI, chronic limb–threatening ischemia; DCB, drug-coated balloon.
The primary patency and survival are given in percentages at 12 months for the entire cohort.
Despite the prospective nature of this study, it is limited by a low patient number, heterogeneity in postdilatation technique, the lack of a control group, and the lack of an independent core laboratory adjudication. The low patient number may be explained by the specific patient population that was studied, namely, severely calcified lesions around the knee joint. As previously mentioned, most studies evaluate popliteal lesions together with superficial femoral lesions, even though both arteries have different anatomical and dynamic characteristics and consequently different outcomes after intervention. A final limitation in all studies on calcified lesions is the lack of a consistent definition for calcification. To cover this problem, we decided to score the severity of calcification based on 2 common definitions.
As this study comprises the first-ever analysis on follow-up outcomes after IVL in popliteal and infrapopliteal arterial disease, it is clear that more follow-up studies are warranted. A handful of studies published IVL outcomes in the femoropopliteal artery, but they did not differentiate between femoral and popliteal lesions.35–37 Randomized trials are warranted to compare IVL with angioplasty alone or other vessel preparation devices in popliteal and infrapopliteal disease. Then, large population–based registries, such as the Disrupt PAD III observational study, are needed to test these outcomes in daily practice. 20
Conclusions
In a complex patient population, IVL seems to be a safe and effective endovascular treatment option for calcified popliteal and proximal infrapopliteal lesions with a low rate of procedural complications and bailout stenting.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prior Presentation
None.
