Abstract
Objectives:
Electrosurgery has been long used in endovascular procedures, with only case reports in the aortic field. Our aim is to present a case series with the use of an electrified wire to perform catheter-based electrosurgery by applying external current through an electrocautery pen.
Methods:
Single-center retrospective case series of all patients undergoing complex aortic surgery from October 2020 to August 2023, in whom the electrified wire technique was used: (1) Perforation of a dissection flap or left subclavian artery (LSA) in situ endograft fenestration—a 0.014” polytetrafluoroethylene (PTFE) insulated guidewire is detached from the insulation with a scalpel at the end and a cautery pen is here attached with a clamp. A curved tip catheter or sheath is positioned against the aortic flap or the endograft (through a left brachial access in this case) and the wire pushed, crossing the flap by activating the electrocautery pen and (2) slicing a dissection flap (“powered cheese-wire technique”)—after same preparation as above, the middle section of the 0.014 guidewire is removed from the PTFE and bent into a V-shape. Once in the aorta, the guidewire crosses from the true lumen (TL) to the false lumen (FL) and a through-and-through access is obtained. Sheaths are positioned against the flap from both sides and moved up or down while the electricity is activated, slicing the flap and communicating both lumens. Technical success and technical-related complications were evaluated.
Results:
Eleven cases concerning aortic dissections and 1 case of aortic atresia were treated. Four patients presented urgently, whereas the rest were planned procedures. Seven cases underwent perforation of a dissection flap, 2 cases underwent the powered cheese-wire technique, in 2 cases for an LSA in situ fenestration, and in 1 case to cross an aortic atresia at the aortic isthmus. The technique was in all cases successfully applied. No complications related to the technique occurred.
Conclusions:
The “electrified wire” technique is a feasible and ready-available tool that can be safely used in complex aortic interventions, especially to perforate aortic tissue like dissection flaps or to perform in situ fenestrated repairs by perforation of the endograft fabric.
Clinical Impact
The electrified wire technique described herein is a straightforward technique that uses readily available tools to perform electrosurgery. We present its use in complex aortic procedures. However, it could be envisioned for any vascular procedure that requires crossing of the vessel or even prosthetic material. As we have described in this series, when used along with an adequate properative planning, it can be a safe tool of great utility, as has already been demonstarted in the field of the interventional cardiology.
Introduction
Through the use of electricity, electrosurgery achieves focused vaporization of tissue, obtaining an adequate incision and hemostasis. Despite having been invented more than a century ago, it continues to be a fundamental tool for surgeons of all specialties, having undergone multiple improvements such as its adaptation for laparoscopic or endoscopic surgery, its use as a localized treatment for cancer or the ability to achieve hemostasis in medium-sized vessels with the use of specialized devices, among others.1,2
In catheter-based interventions, electrosurgery has been part of the arsenal of interventional cardiology for decades with the use of specific radiofrequency catheters. With the widespread use of this technology in cardiac interventions, several procedures, such as BASILICA (Bioprosthetic or native Aortic Scallop Intentional Laceration to prevent Iatrogenic Coronary Artery obstruction) have demonstrated the feasibility and safety of catheter-based electrosurgery, 3 encouraging the development of novel techniques in other specialties, such as aortic interventions. Despite this great development, to date there is only limited evidence based on case reports in the aortic interventional field.4,5
We present a case series of patients treated with a simple method of performing catheter-based electrosurgery by applying external current through an electrocautery pen to a wire and using this “electrified wire” to traverse the aortic tissue or endograft fabric.
Methods
Single-center retrospective case series of all consecutive patients undergoing complex aortic surgery in an aortic center from October 2020 to August 2023, in whom the electrified wire technique was used.
Device Description and Standard Technique
Depending on the pathology and intended use, the technique of the electrified wire varies.
Dissection Flap Fenestration
For perforation of an aortic dissection flap, when traversing from one lumen to the other, the true lumen (TL) is preferentially accessed with the false lumen (FL) chosen as the target, as it is usually larger and therefore easier to reach (Figure 1). This fenestration technique may be necessary in cases of fenestrated-branched endovascular aortic repair (f/bEVAR) when an entry tear between the TL and FL cannot be identified and used to bridge the main graft located in the TL to a target vessel (TV) originating in the FL.
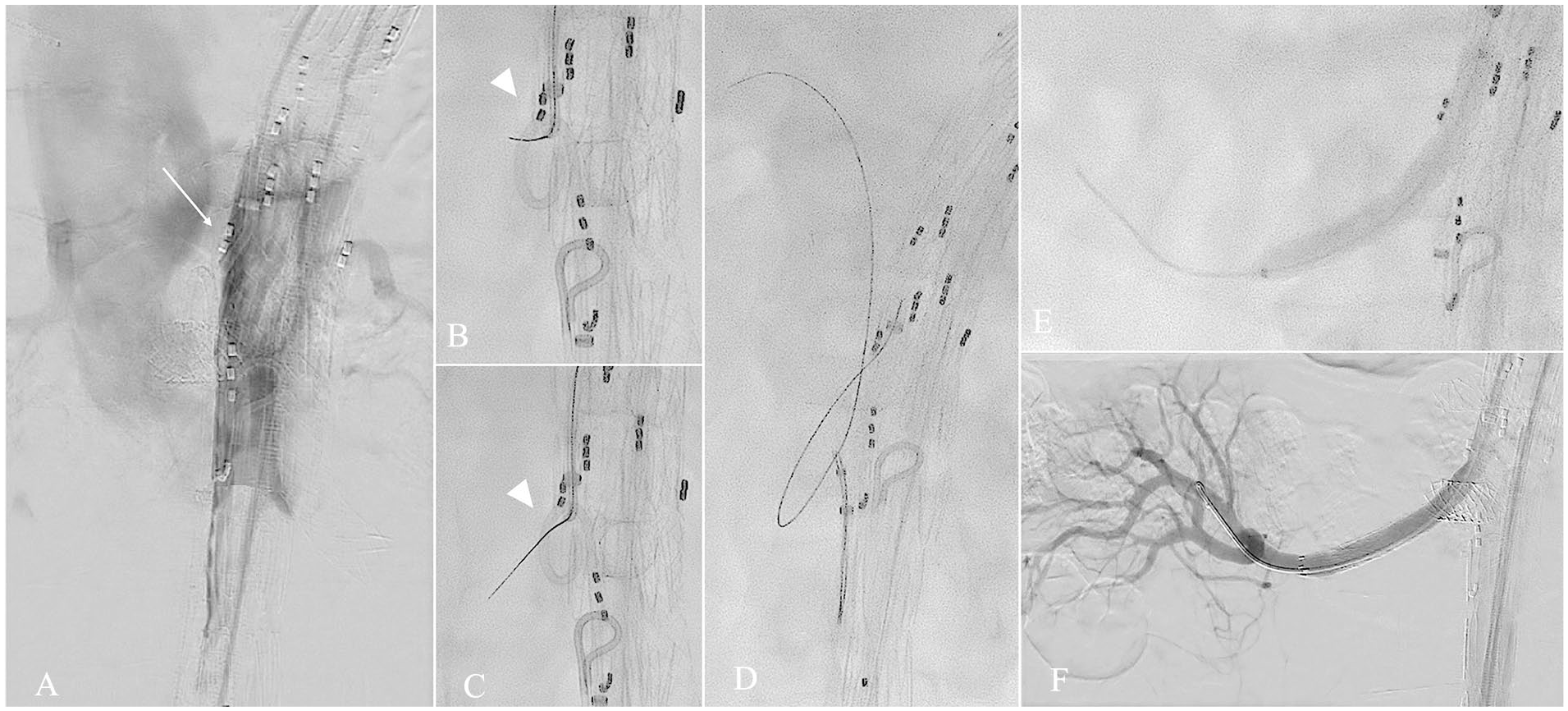
Angiogram showing the electrified wire flap fenestration (A). DSA showing a branched EVAR deployed in the TL. White arrow shows a Berenstein catheter lending over the dissection flap that separates the TL from the FL (B) (C). Fluoroscopy showing the electrified wire (white arrowhead) getting across the flap, using a Pigtail catheter as target (D). The wire is snared (E). Balloon dilatation after bridging cover stent deployed in the right renal artery (F). DSA showing successful incorporation of the RRA with the bridging stent passing across the flap.
As an example, after locating the TV ostium in the FL with angiography, the appropriate catheter configuration, frequently a 5F Berenstein (Cordis, Miami Lakes, Florida) is positioned in the corresponding antegrade branch, which lies against the dissection flap and the tip is pushed against the dissection flap (Figure 1A). A Tuohy-Borst valve is attached to the catheter, and a drip infusion of 5% glucose connected to the catheter is started to help dislodge blood and prevent blood clotting without interacting with the electricity. 3 A 0.014” Astato XS20 guidewire (Asahi-Intecc, Tokyo, Japan) is used, as it is a stable guidewire with good pushability and the ability to have the polytetrafluoroethylene (PTFE) insulator removed in selected areas, allowing transmission of electricity only in the non-insulated parts of the guidewire, as described in reports of cardiac interventions. 3 The distal end of the guidewire is then stripped of the PTFE insulation with a scalpel, and a mosquito clamp is attached to this end including the tip of the electrocautery-pencil, ensuring contact between the wire and the electricity delivered through the pen. The catheter is pushed against the flap, ensuring that the wire is optimally insulated from the blood by the nonionic glucose solution. The wire is then advanced until the tip of the catheter is reached. The correct direction of the wire is controlled using fluoroscopy in 2 projections, ensuring that it directs toward a pigtail-catheter or large snare serving as a target (Figure 1B). The electrocautery is set to 70 to 100 watts, and, in a coordinated motion, the electrocautery pen is activated in the cutting mode, whereas the wire is pushed against the flap. The wire usually advances with ease and can be seen moving freely into the FL. Electrocautery is stopped and the snare may be used to fixate the 0.014” wire or even externalize it in order to enhance the stability for subsequent catheter or balloon exchanges. Balloon angioplasty is then performed to dilate the communication with increasing diameters, reaching a diameter suitable to be crossed with a 6-8 mm covered stent. A first balloon usually needs to be 0.014” compatible, before the Astato wire can be exchanged for a larger and more stable wire. Once in the FL, the target vessel is catheterized and bridged with a covered stent.
Long Segment Dissection Flap Slicing (“Powered Cheese-Wire Technique”)
For the performance of the “powered cheese-wire technique”, 6 the Astato wire is selectively stripped of PTFE insulation along 4 to 5 mm of the guide wire length in a mid-segment of the wire. This section is then kinked so that the stripped portion of the guidewire is confined to the inside part of the bend, forming a “flying V” configuration. The distal end of the guide is also stripped as described above to connect to the electrocautery-pencil. After accessing the TL and FL separately, a through-and-through access across an existing cephalad entry tear or by creating a fenestration through the flap is obtained with the help of a snare. After closely approximating catheters and/or sheaths from both sides of the dissection flap, the “flying V” segment of the guidewire is placed in contact with the flap. With a 5% glucose infusion connected to both catheters, the electrocautery is activated, and the catheters/sheaths moved coordinately together with the wire toward distal, allowing the electricity to cut the flap, achieving unobstructed flow between the 2 lumens.
Left Subclavian Artery Endograft In Situ Fenestration
For the creation of left subclavian artery (LSA) in situ fenestrations on thoracic endografts, a left brachial access is obtained and through this access, a sheath is approximated to the LSA ostium (Figure 2). After deployment of the endograft, the brachial sheath, with or without a catheter depending on the morphology of the LSA, is advanced in close proximity with the endograft in a near 90 degrees position. To achieve a beneficial angulation, an angulated or steerable sheath may be used. This is especially valuable in steep angulations as frequently present in type III arches. 7 Through a femoral access, a snare is placed inside the endograft, confronting the sheath positioned at the LSA. The modified Astato wire is brought, and the 5% glucose drip initiated. With the electrocautery activated, the wire is advanced, and caught by the snare, creating a stable through-and-through access. Through this access, balloon dilatation is performed, and a balloon expandable covered stent (BES) is deployed, bridging the LSA.
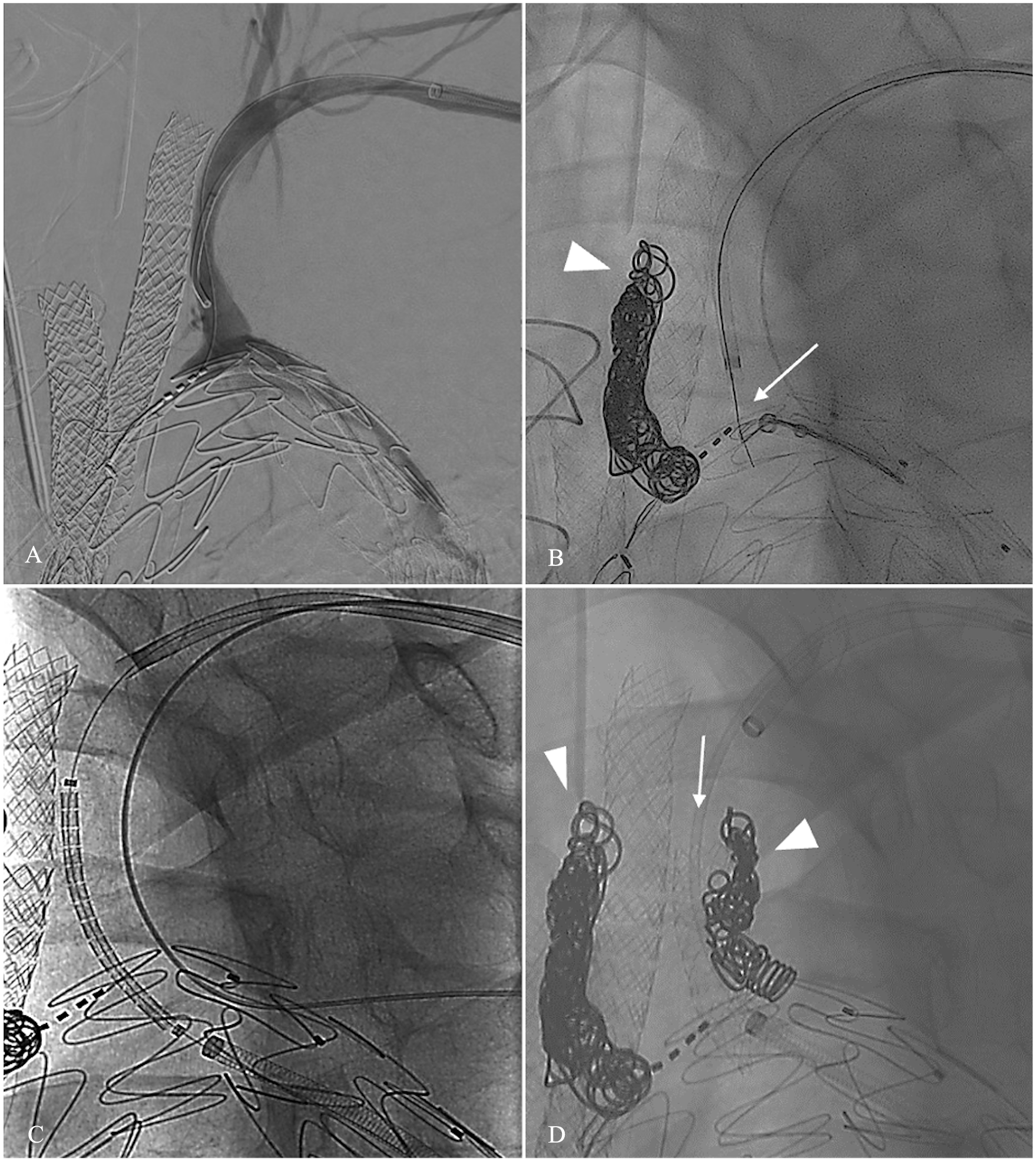
Angiogram showing fenestration of a branched TEVAR, with planned branches to the innominate (IA) and left common carotid (CCA) arteries (A). Digital substraction angiogram showing an almost 90° between the origin of the left subclavian artery and the endograft (B). Astato wire traversing the endograft, with the snare capturing it (white arrow). Arrowhead shows coils in the left CCA false lumen (C). After dilating the hole, a sheath was advanced, and a balloon expandable covered stent positioned. The sheath is then withdrawn, and the stent is ready to be deployed (D). LSA already deployed (white arrow). White arrowheads showing coils in the FL of IA and LCCA.
According to the local ethical committee, ethical approval was not deemed necessary for this retrospective study using anonymized data. All interventions were performed in a hybrid operating room (Phillips Allura Clarity), with fusion computed tomography angiography (CTA) (Vessel Navigator, Philips Healthcare, Best, The Netherlands). Electronic records, preoperative and postoperative CTAs, and the imaging of the procedures were reviewed, including measurement of flap thickness in cases of aortic dissection. Demographics, medical history, indications for repair, and reason for using the electrified wire were recorded. The primary outcomes were the rate of successful perforation with the electrified guidewire and the absence of technique-related complications.
Continuous variables are expressed as medians with ranges. Categorical variables are presented as percentages.
Results
During the study period, 12 patients underwent aortic intervention with the help of the electrified wire technique. The median age of the cohort was 66 (51-80), and 67% (8/12) of the patients were male. Table 1 shows the demographic data and presentation.
Demographic Data.
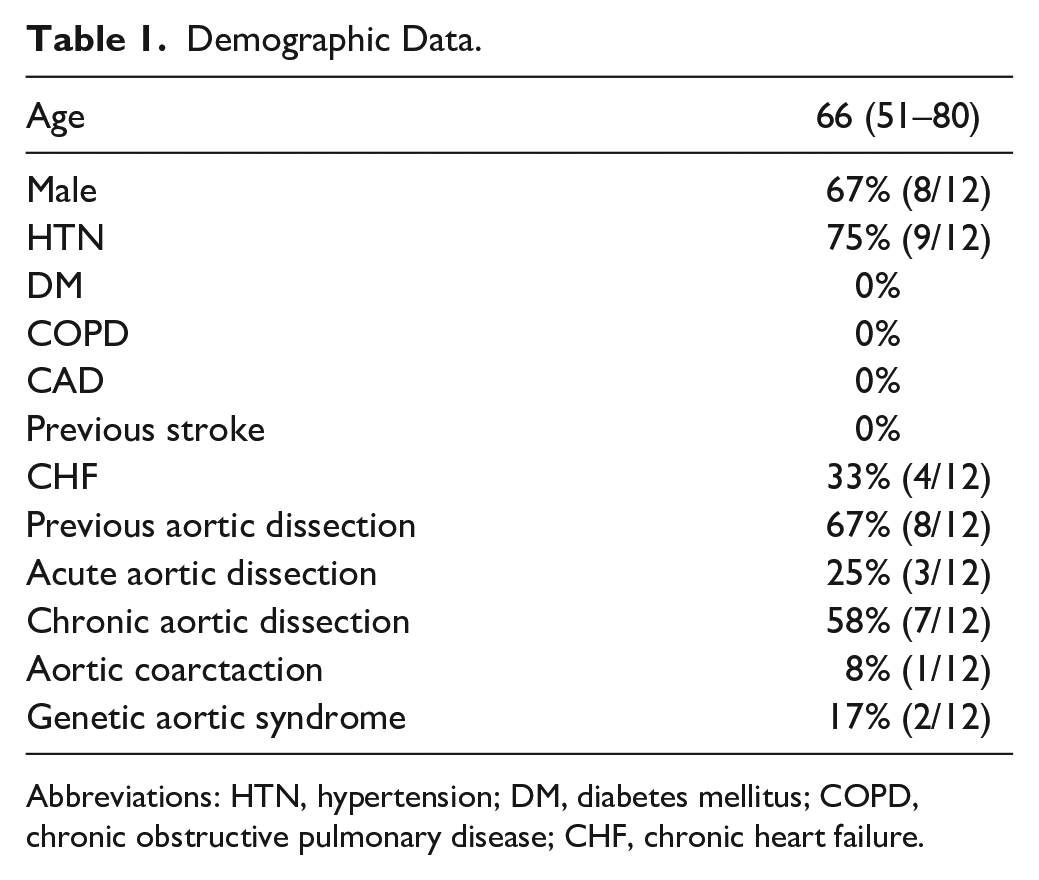
Abbreviations: HTN, hypertension; DM, diabetes mellitus; COPD, chronic obstructive pulmonary disease; CHF, chronic heart failure.
Eleven cases corresponded to patients with acute or chronic aortic dissections: 1 acute type A aortic dissection (TAAD), 2 acute type B aortic dissection (TBAD), and 8 chronic aortic dissections (1 TBAD, 7 residual dissections after TAAD surgery).
The nondissection case corresponded to a 46-year-old patient with severe heart failure and aortic atresia of the descending aorta at the aortic isthmus, in whom the electrified wire was used to cross the aortic occlusion. A 12 mm balloon expandable covered stent was deployed and postdilated to 16 mm 8 (Figure 3).
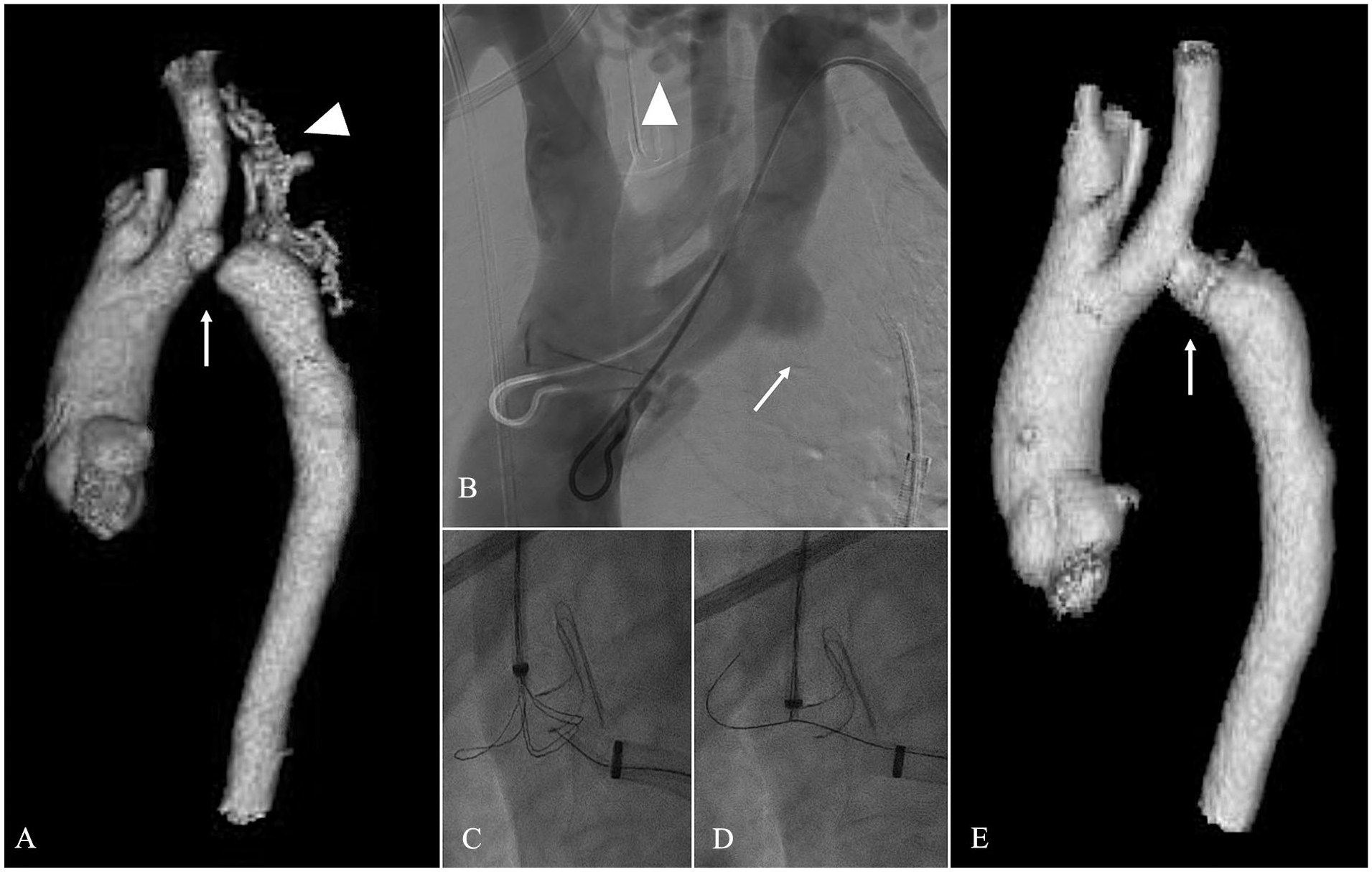
(A). CTA 3D reconstruction (A–E) and angiograms of a patient with aortic atresia (white arrow), there is an extense collateral network supplying the distal aorta (white arrowhead) (B). DSA showing the aortic occlusion (white arrow) and the collateral network (white arrowhead) (C) (D). Electrified wire crossing the aortic occlusion and being snared (E) CTA 3D reconstruction of the aorta after covered stent placement across the occlusion.
Setting of Presentation
Four patients (33%) presented as urgency, 2 patients with acute mesenteric ischemia due to TL collapse in the aortic visceral segment, 1 case treated by flap slicing with the powered cheese-wire plus stent deployment in the superior mesenteric artery and the other case, a patient with an acute TAAD, was treated with single flap fenestration with the electrified wire and dilatation up to 14 mm, allowing unobstructed flow to the TL as bridge therapy to a frozen elephant trunk performed 2 days later. The other 2 urgencies underwent electrified wire in situ graft fenestrations, 1 in the setting of a 9 cm symptomatic arch post-dissection aneurysm undergoing endovascular repair with a custom-made branched TEVAR (bTEVAR, Figure 2) which was initially designed for other patient but fitted the aortic arch anatomy of this urgent case, and the other presented with rapid diameter increase FL dilatation in a TBAD and underwent TEVAR, both with LSA in situ fenestration.
Among the elective cases, 7 patients underwent a single flap fenestration while 1 underwent slicing of the flap with the powered cheese-wire technique, in all the cases to catheterize a vessel from the FL without a re-entry in the vicinity (Figure 1). Table 2 presents patient characteristics and technical details.
Patient Characteristics and Technical Details.
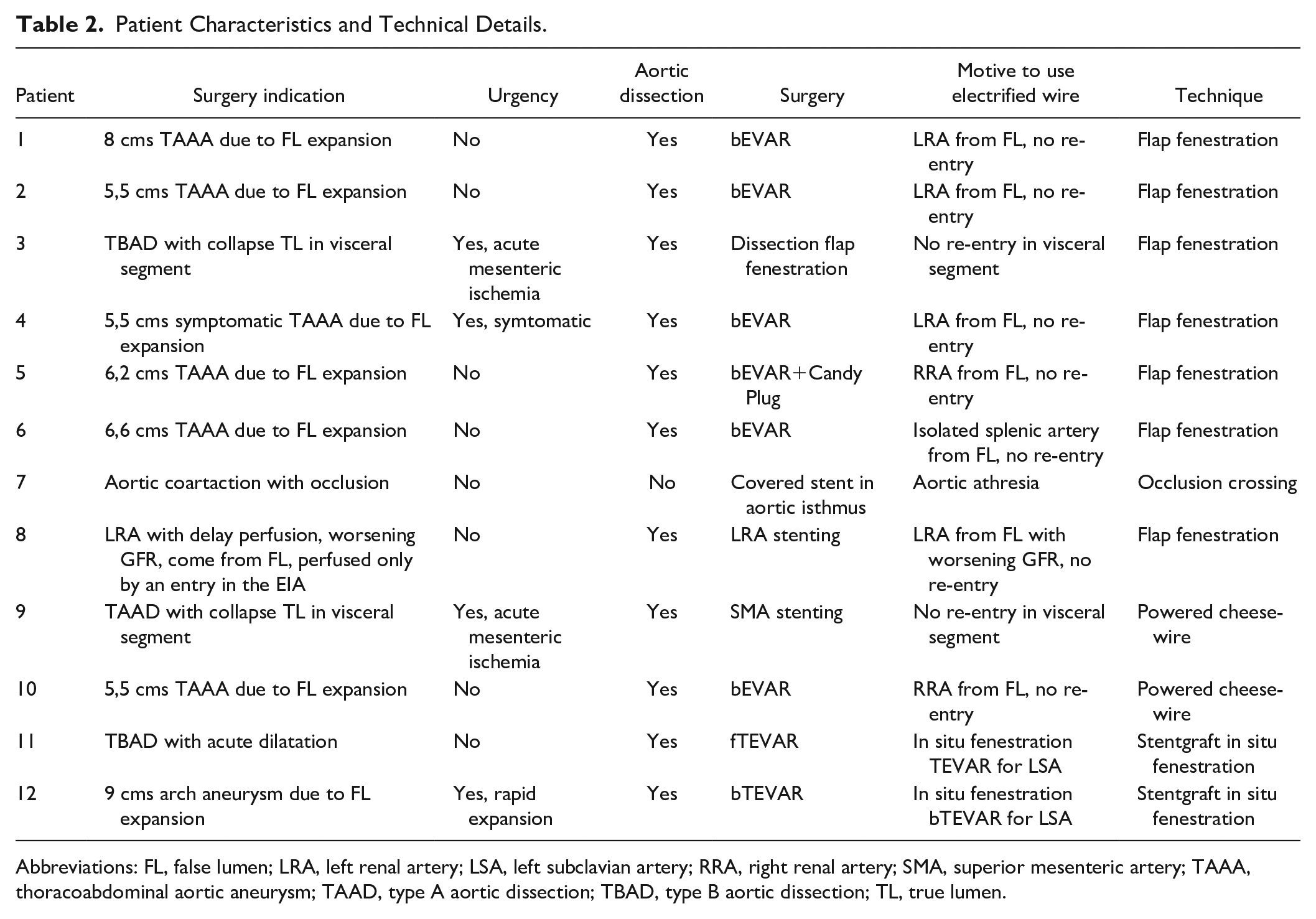
Abbreviations: FL, false lumen; LRA, left renal artery; LSA, left subclavian artery; RRA, right renal artery; SMA, superior mesenteric artery; TAAA, thoracoabdominal aortic aneurysm; TAAD, type A aortic dissection; TBAD, type B aortic dissection; TL, true lumen.
Technical Details
Of the 9 cases in which a flap fenestration was performed, in eight, the fenestration was performed from the TL targeting toward the FL, because the FL was larger. In 1 case, it was performed from the FL toward the TL as there was little space in the FL (Figure 4).
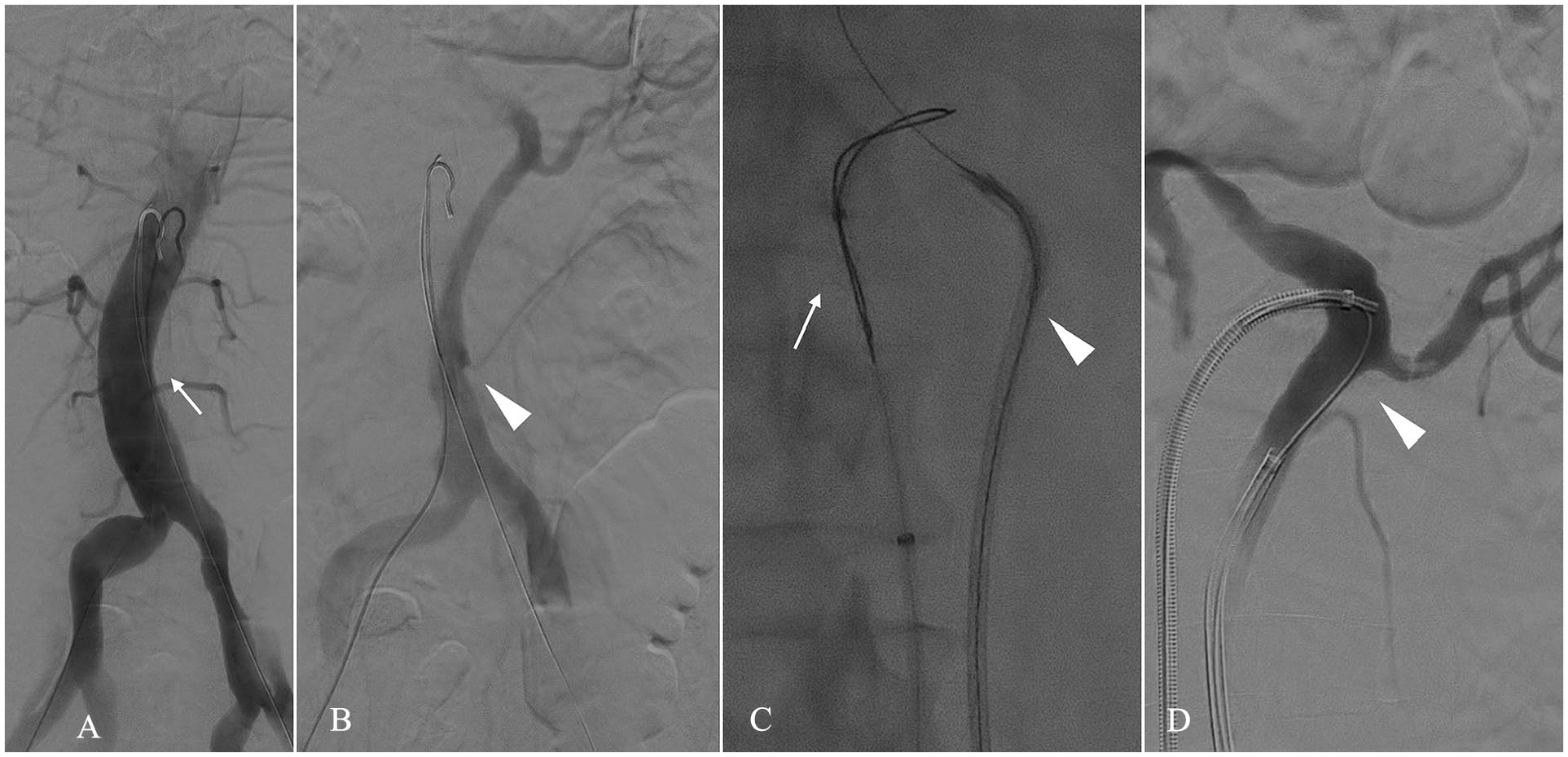
Angiography showing the electrified wire technique crossing from false to true lumen. (A). DSA showing the true lumen (TL) (B). DSA showing the false lumen (FL), with the left renal artery supplied by the FL (C). Electrified wire crossing from the FL (white arrowhead) to the TL (white arrow) (D). DSA showing through-and-through maneuver that allowed stable access to FL (white arrowhead).
In 50% of the cases, after the wire crossed the flap, a snare was used to capture the wire to perform the powered cheese wire or to enhance stability for balloon advancement. After passage of the guidewire to the FL, all cases with single fenestration underwent balloon dilatation to at least 6 mm and then a covered stent was successfully deployed across the flap.
The Astato guidewire was used in all 12 cases. In 6 cases (50%), the wire was supported by a Berenstein catheter, in 2 patients, a Vanschie-3 catheter (Cook Medical, Bjaeverskov, Denmark) was used and in patients who underwent powered cheese wire and in situ graft fenestrations only the sheaths were used.
Operative Outcomes
In all cases but one the crossing was achieved at the first attempt with immediate fenestration in a fraction of a second. In the exemption, during an in situ fenestration of a bTEVAR, the electric generator power was set at 40 watts and after 30 seconds without success, it was raised to 70, achieving immediate passage of the wire. The median flap thickness was 3.5 mm (1.4-6).
There was no intraoperative false advancement of the wire outside the aorta and no bleeding related to the electrified wire. Postoperatively, 1 patient electively treated with complete coverage of the aorta due to a thoracoabdominal aortic aneurysm, developed complete paraplegia and died 2 weeks later due to low cardiac output syndrome. One patient, with a flap fenestration in a bEVAR (patient 4), suffered a mild stroke and another a cerebrospinal fluid leak after spinal drainage, which was treated with a blood patch. No technique-related complications were observed on postoperative CTAs.
Discussion
We present a series of cases of patients undergoing different complex aortic interventions assisted with a 0.014” wire coupled to external electrical current that allowed us to transverse dissection flaps, an aortic occlusion and endograft fabric for in situ fenestration.
The catheter-based use of “electrically powered” devices to access through tissue was described more than 30 years ago, with the use of radiofrequency for ablation procedures, pulmonary valve recanalization in cases of atresia and to achieve trans-septal access in electrophysiological interventions.9–11
In the field of aortic interventions, the use of an “electrically powered” device was first reported in 2008 by Almashham et al 12 performed to recanalize an aortic atresia. A year later, Murphy et al 13 described an in situ fenestration of an LSA in a TEVAR assisted with a laser catheter. Subsequently, in 2012, Ricci et al 14 reported the use of radiofrequency to perforate a dissection flap in a chronic aortic dissection.
Although these devices have been shown to function adequately within the aorta,13,15,16 this technology involves the use of special catheters with specific power generators, which are expensive and may not be available in every hybrid operating room. In contrast, the technique presented here can be performed using readily available materials, such as an additional 0.014 (Astato) wire connected to an electrocautery.
By connecting the electrocautery to the wire and activating the cutting mode, direct alternating currents are delivered at high voltages for a short time, allowing tissue vaporization. Within the aorta, it is especially important to minimize the contact of the current waves with the blood because of the possible occurrence of coagulation. A 5% dextrose drip through the emitting catheter helps to displace the blood and prevents blood clotting. 3
Some authors have recommended isolating the denuded part of the electrified wire with microcatheters to increase the focused energy delivered and avoid the compromise of a zone in the surrounding tissue (penumbra). 3 Khan et al 3 studied with microscopy the size of the damage left by Astato wire connected to a 50-watt electrocautery in a porcine aorta, finding a hole of less than 0.5 mm and a penumbra of less than 1 mm. In our experience, given that the dissection flaps are thin (median 3.5 mm in this study), and taking into account the small penumbra produced by this technique, isolation did not appear necessary and only commonly used sheaths or 5 Fr catheters were used to support the electrified wire.
In our series, most patients had aortic dissection. In chronic stages of aortic dissection, more than 60% of patients may develop a TAAA, 17 with 18% to 50% of renal arteries originating from the FL.18,19 In case of the need for complex endovascular repair with f/bEVAR, an entry tear communicating both lumens should be catheterized to incorporate vessels originating from the FL; however, when no tear is identified or a tear cannot be catheterized, a communication between both lumens should be artificially established. Williams et al 20 reported in 1990 the first fenestration of an aortic dissection flap with a rigid guidewire, communicating the TL and FL. Subsequently, fenestrations of dissection flaps have been performed, among others, with rigid wires, TIPS or trans-septal needles, and even re-entry devices.21,22 The “electrified wire technique” can be particularly useful in this scenario, as it allows rapid establishment of inter-lumen communication with the use of widely available supplies. In cases of acute aortic dissection with TL collapse and malperfusion syndrome, it can also be very helpful in the creation of big fenestrations or the performance of the “powered cheese-wire technique,” allowing blood flow to access the collapsed TL. Importantly, this technique not only works well with a straight wire in cases of antegrade upper extremity access, but also with transfemoral access using steerable sheaths, which has become our standard approach for bEVAR. 23
Apart from the aforementioned uses, there are many other scenarios within endovascular aortic interventions in which this technique could be useful, such as the creation of fenestrated devices in situ, crossing occlusions or even facilitating transcaval access to the aorta, among others.
Despite the absence of complications in our series, it is important to note that it is an off-label use of the devices involved, which are not originally designed or approved for use with electric current. For a safe use of the technique, interventionalists need to be skilled in orienting themselves with the different angular projections of the C-arm, always keeping in mind the 3-dimensional structure of the aorta, in order to avoid a false passage of the wire.
Conclusions
The “electrified wire” technique is an easily reproducible and readily available tool that should be considered as part of a preplanned procedure or as bail-out in complex aortic interventions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.K. is consultant, proctoring, IP, royalties, research, and travel grants with Cook Medical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
