Abstract
Purpose:
The purpose was to investigate outcomes of high-risk patients undergoing thoracoabdominal aortic aneurysm (TAAA) repair using fenestrated or branched endovascular aneurysm repair (F/BEVAR) devices at a single center in Canada.
Materials and Methods:
A retrospective review of all patients undergoing endovascular TAAA repair with F/BEVAR from June 2007 to July 2020. Imaging and clinical endpoints of interest including death, reintervention, and target vessel patency were reported.
Results:
Ninety-five consecutive patients underwent endovascular TAAA repair using F/BEVAR stent grafts (63 males, median age 74 [interquartile range 70, 78] years). Repairs included 81 elective and 14 urgent/emergent cases (6 ruptures and 8 symptomatic). Graft deployment was 100% successful. Intraoperative target vessel revascularization was successful in 336/355 (94.6%) vessels with the celiac having the lowest success rate 72/82 (87.8%). In-hospital mortality was 9.5% (7.4% elective and 21.4% urgent/emergent, p=0.125) and permanent paraplegia was 4.2% (3.7% elective and 7.1% urgent/emergent, p=0.458). In-hospital complications included stroke in 5.3%, acute myocardial infarction in 8.4%, and bowel ischemia in 5.3%. No patients required permanent dialysis or tracheostomy during their hospital stay. However, 22 (23.2%) patients required additional unplanned procedures for various indications (branch occlusion, endoleaks, realignment) during their hospital stay. Patients were followed up for a mean of 3.6 ± 3.0 years. Clinical follow-up was 100%, with 80/86 (93%) having surveillance imaging. On follow-up imaging, 43 (50%) patients had at least 1 endoleak identified and 337/341 (98.8%) of the target vessels were found to be patent. At 5 years, cumulative probability of reintervention was 46.3% (95% confidence interval [CI], 36.1-56.4). Survival at 5 and 8 years was 50.1% (95% CI, 38.4-65.4) and 34.4% (95% CI, 22.5-52.8), respectively. Progression of aneurysmal disease leading to rupture on follow-up was confirmed in 1 patient at 10 years.
Conclusion:
Endovascular TAAA repair provides a safe treatment option with a high technical success rate and low pulmonary and renal complications. Long-term survival is similar to previous literature; however, high rates of secondary reintervention reaffirm the need for ongoing patient follow-up and further technical improvements.
Clinical Impact
This study demonstrates that endovascular repair of TAAAs can be performed in a high-risk elderly population with acceptable rates of mortality, TALE and SCI, using evolving technology. The incidences of post-operative respiratory failure and renal dysfunction were lower in patients who underwent endovascular repair compared with open repair. Future technical and procedural refinements in addition to increasing surgical experience are expected to lead to further improvements in short- and long-term outcomes exceeding those of open repair.
Introduction
Thoracoabdominal aortic aneurysm (TAAA) repair using fenestrated/branched endovascular aneurysm repair (F/BEVAR) devices was initially described by Chuter and colleagues in 2001. 1 It provided an alternative treatment modality for patients who were deemed to be high surgical risk for open repair, even at high-volume centers.2–4 Successful trials of infrarenal endovascular aneurysm repair (EVAR) for AAAs in the 1990s 5 paved the way for minimally invasive techniques to treat more complex aneurysms. Multiple meta-analyses6,7 have identified an early survival benefit with infrarenal EVAR, with a higher rate of reintervention but unchanged long-term survival compared with open repair. 8 For TAAA, literature to date has found similar rates of midterm mortality, permanent paraplegia, and other comorbidities between open and endovascular repair.9–12
A systematic review has documented excellent outcomes following F/BEVAR for TAAA repair 10 ; however, there is a paucity of real-world data evaluating its midterm outcomes while including evolving techniques that minimize spinal cord ischemia (SCI). Furthermore, new reporting standards allow for more objectivity when comparing outcomes of endovascular repair between centers of different referral volumes. 13 This study aims to report a thorough and evolving endovascular TAAA experience from a single center.
Materials and Methods
Study Population
All 95 consecutive patients from a tertiary center treated for TAAA using F/BEVAR from the initiation of the program in November 2007 to June 2020 were identified from an institutional endovascular database and included. Patients with aortic aneurysms involving the renal-mesenteric arteries were seen in a multidisciplinary aortic clinic and physiological fitness was assessed by criteria adopted from Society for Vascular Surgery practice guidelines. 14 Our selection criterion for elective patients includes, but is not limited to, maximum aneurysm size >6 cm, no active malignancy, normal pulmonary function tests with forced expiratory volume >1 L, cardiologist clearance with left ventricle ejection fraction >40%, an agreement to attempt cessation/reduce smoking, and no active incidental pathologies found during investigations. All elective, symptomatic and ruptured aneurysms were included. During this time period, 62 open TAAA repairs were performed. All patients in this series were deemed to be high surgical risk for open repair and consented for endovascular repair. All computerized tomography (CT) relevant angiograms were reviewed for inclusion criteria, using Coral (hospital-based picture archiving and communication system) and TeraRecon (ConcertAI, Durham, North Carolina). The Crawford-Safi classification and the extent of aortic coverage were separately measured and recorded. 15 Indications for surgery were based on aneurysm size and rate of growth and the study was approved by the Institutional Research Ethics Board.
Procedure Details
This report includes the learning curve and experience of 8 surgeons over the 13 years. All procedures were undertaken in hybrid operating rooms, which began with Toshiba Aquilion Imaging (Tokyo, Japan), then a Siemens Artis Zeego (Munich, Germany), and most recently a Philips Azurion (Amsterdam, the Netherlands), that latter 2 systems had fusion imaging. Preoperative assessments were recorded including open or percutaneous procedures to facilitate endovascular TAAA repair. Intraoperative arterial access was via arterial cut-down initially, with occasional conduit use, and evolved to percutaneous access. Endografts were either customized fenestrated or branched grafts or an off-the-shelf T-branch (Cook Medical, Australia) graft. Directional branches were employed for longitudinally oriented target vessels and when the distance between the stent graft and aortic wall >5 mm, while fenestrations were employed for transversely originating target vessels where the main graft was planned to reach the aortic wall (<5 mm). Bridging stents began with Advanta V12 (Atrium Medical Corp, Hudson, New Hampshire), Gore Viabahn (W.L. Gore & Associates, Flagstaff, Arizona), and Fluency (BD, Franklin Lakes, New Jersey). This then transitioned to also include VBX (W.L. Gore & Associates) and Lifestream (BD). EPIC uncovered stents (Boston Scientific, Marlborough, Massachusetts) were occasionally used to prevent branch kinking. Twenty-five target vessels did not require a branch/fenestration due to prior occlusion (n=9), nephrectomy (n=4), renal atrophy (n=4), bypass (n=3), or use of scallop (n=5). If a planned target vessel was not successfully cannulated intraoperatively, it was either reattempted as a planned reintervention or rarely occluded with an Amplatzer Vascular Plug (St. Jude Medical, St. Paul, Minnesota) if collateral supply was adequate. Proximal thoracic EVAR (TEVAR) and distal bifurcated grafts were used in patients to obtain adequate seal zones depending on aneurysm extent to achieve a 20 mm sealing zone in healthy aortic tissue, and the former was also utilized if at least 2 stent overlap was required with a staged TEVAR. The current protocol for minimizing SCI has evolved to include staged TEVAR which began in 2012, minimally invasive staged segmental artery coil embolization (MISACE) which began in 2017 and has been previously described by our group, 16 permissive hypertension, intraoperative neuromonitoring of motor evoked potentials (MEPs) and somatosensory evoked potentials (SSEPs), early sheath removal with distal limb perfusion, baseline hemoglobin targets, hyperbaric oxygen therapy, and cerebrospinal fluid (CSF) drainage.17,18 Those who underwent both MISACE and TEVAR staging had MISACE first and TEVAR subsequently during a separate admission at least 2 weeks later.
End Points
Primary endpoint was in-hospital and long-term mortality. Secondary outcomes include thoracoabdominal aortic aneurysm life-altering events (TALE), a composite of mortality, stroke, paraplegia, and permanent dialysis. 4 Spinal cord ischemia is graded according to the 4-level system (grade 0-3), and acute kidney injury is defined as doubling of baseline serum creatinine. 13 Primary technical success was defined as successful delivery and deployment of the aortic stent graft and its side branches on an intent-to-treat basis, target vessel patency on CT or duplex ultrasound (DU) imaging prior to discharge, and absence of type I and III endoleaks beyond 30 days. Assisted primary technical success included any unplanned in-hospital procedures required to meet technical success. Early postoperative period was to hospital discharge; events afterwards are included within follow-up.
Follow-up
Clinical and imaging follow-up with either CT or DU took place 1, 3, and 6 months after discharge, and annually thereafter at the index hospital or a regional center. Follow-up imaging studies were reviewed to evaluate target vessel patency and endoleaks. Graft branches intentionally occluded to treat endoleaks were not deducted from branch patency rates. Follow-up was obtained through a combination of in-person clinic appointments or researcher phone calls using an approved standardized questionnaire.
Statistical Analysis
All analyses were performed using the R project of Statistical Computing version 3.6.1. Continuous variables are described as mean ± standard deviation (SD) or median (interquartile range [IQR]), while categorical variables are represented as percentages. To evaluate differences between elective and urgent/emergent cohorts, student’s t test or Mann-Whitney tests were used for continuous variables, while the Fischer exact test was used for categorical variables. Time-to-event analyses for freedom from TALE and survival were performed using Cox proportional hazards models to obtain hazard ratios 8 years after surgery. Long-term rates of secondary procedures on the thoracoabdominal aorta or its branches were obtained accounting for death as a competing risk. Kaplan-Meier survival functions were estimated for survival and freedom from TALE. Cumulative incidence functions were generated for secondary interventions. A p<0.05 was deemed to be significant.
Results
Patient and Aneurysm Characteristics
Ninety-five consecutive patients (63% male; median age 74 [IQR 70, 78] years) underwent F/BEVAR repair for TAAA; 81 patients (85.3%) were treated electively, and 14 (14.7%) patients required an urgent/emergent procedure. Contained rupture was present in 6 (6.3%) patients and 8 (8.4%) were symptomatic (Table 1). Crawford extents were extent I: 3 (3.2%), extent II: 23 (24.2%), extent III: 27 (28.4%), extent IV: 37 (38.9%), and extent V: 5 (5.3%). Twelve patients (12.6%) had dissections and 2 (2.1%) presented with intramural hematomas.
Preoperative Patient and Aneurysm Characteristics.
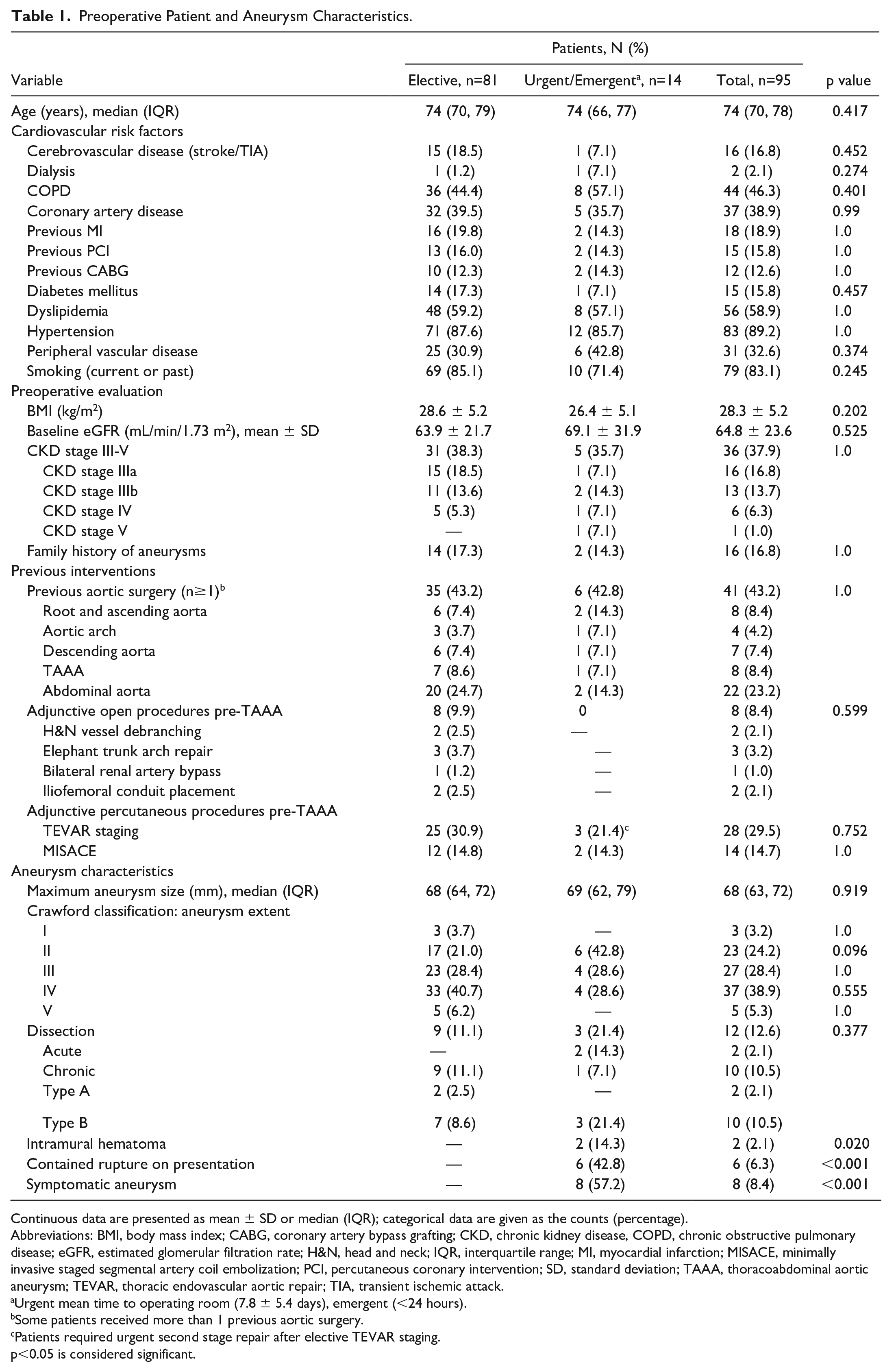
Continuous data are presented as mean ± SD or median (IQR); categorical data are given as the counts (percentage).
Abbreviations: BMI, body mass index; CABG, coronary artery bypass grafting; CKD, chronic kidney disease, COPD, chronic obstructive pulmonary disease; eGFR, estimated glomerular filtration rate; H&N, head and neck; IQR, interquartile range; MI, myocardial infarction; MISACE, minimally invasive staged segmental artery coil embolization; PCI, percutaneous coronary intervention; SD, standard deviation; TAAA, thoracoabdominal aortic aneurysm; TEVAR, thoracic endovascular aortic repair; TIA, transient ischemic attack.
Urgent mean time to operating room (7.8 ± 5.4 days), emergent (<24 hours).
Some patients received more than 1 previous aortic surgery.
Patients required urgent second stage repair after elective TEVAR staging.
p<0.05 is considered significant.
Endovascular Repair
Twenty-eight patients (29.5%) received TEVAR staging, with the majority (n=22) in extent II/III aneurysms, placed a mean of 73 ± 94 days prior to primary TAAA repair, and landing a mean of 3.2 ± 1.4 cm above the celiac axis. Minimally invasive staged segmental artery coil embolization was performed beginning in 2017 in 14 (14.7%) patients at a mean of 51 ± 30 days prior to repair, embolizing on average 2.9 ± 1.4 segmental arteries per patient. Seven patients received both staged TEVAR and MISACE (Table 1).
Custom grafts were used in 79/81 elective patients, while T-branch grafts were used in 2 elective patients and 9/14 urgent/emergent patients (Table 2). Of the custom grafts, 45 (53.6%) were BEVARS, 27 (32.1%) FEVARS, and 12 (14.3%) F/BEVARs. A total of 355 visceral arteries were targeted with 231 (65.1%) directional branches and 124 fenestrations. Intraoperative proximal extensions were placed in 57.9% patients to ensure appropriate overlap. The total Crawford extent of coverage as defined per guidelines was extent I: 2.1%, extent II: 47.4%, extent III: 15.8%, extent IV: 31.6%, and extent V: 3.1%. Extent of graft coverage changed the classification in 31 (32.6%) cases. Graft delivery and deployment was 100% successful with no conversions. Target vessel revascularization was successful in 336/355 (94.6%) visceral arteries using a mean of 1.2 ± 0.4 (range: 1-3) covered bridging and 0.2 ± 0.4 (range: 0-2) self-expanding stents for kinks. No significant differences in target vessel revascularization were found between branches and fenestrations (94% vs 96%, p=0.43). Primary and assisted primary technical success was achieved in 72 (75.8%) and 82 (86.3%) patients, respectively. Assisted technical success was due to type I and III endoleak repair (n=8) and Celiac Artery (CA) revascularization (n=2). Unsuccessful visceral artery revascularization was responsible for the remaining technical failures in patients (n=13).
Procedural Details for Endovascular TAAA Repair.
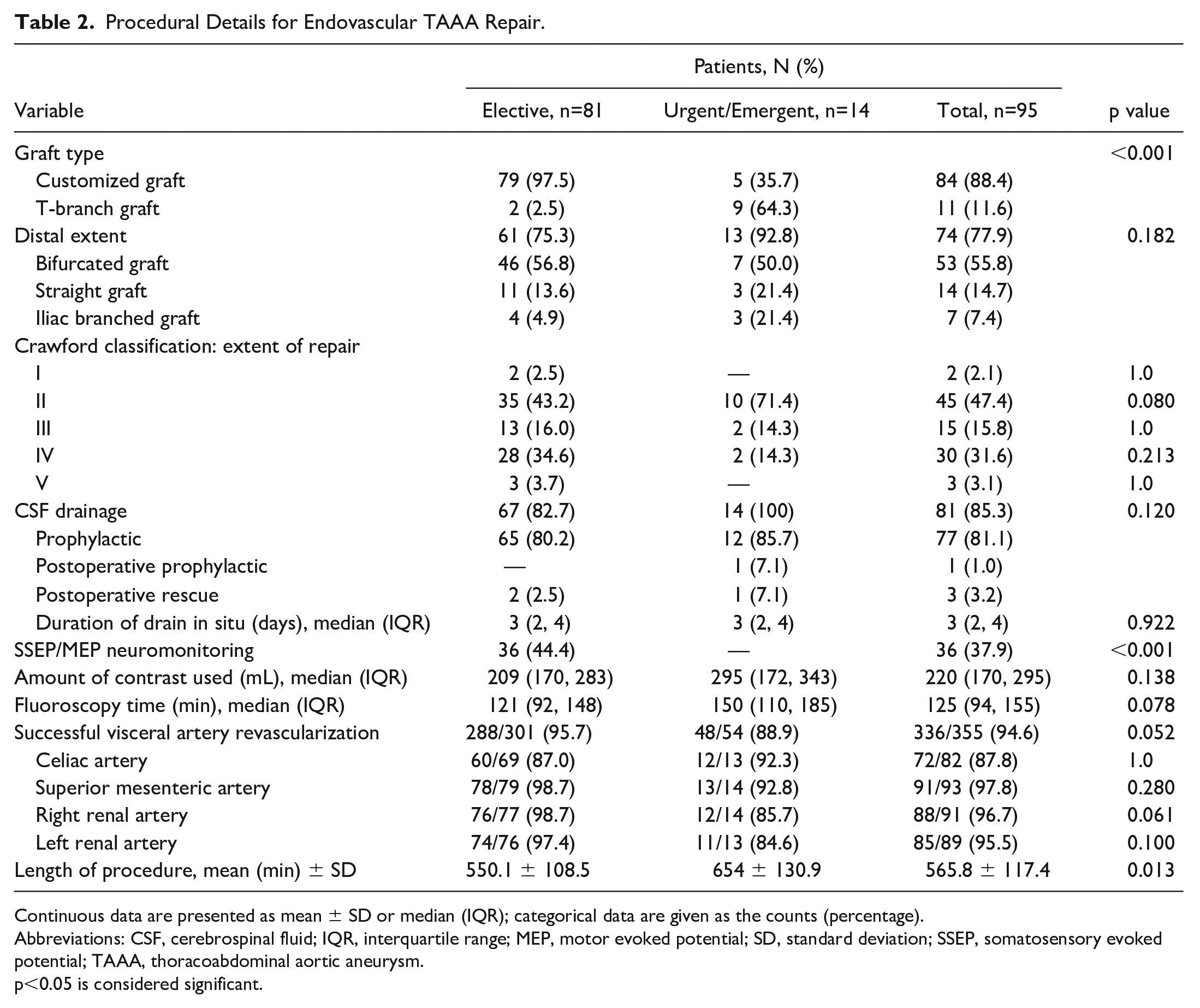
Continuous data are presented as mean ± SD or median (IQR); categorical data are given as the counts (percentage).
Abbreviations: CSF, cerebrospinal fluid; IQR, interquartile range; MEP, motor evoked potential; SD, standard deviation; SSEP, somatosensory evoked potential; TAAA, thoracoabdominal aortic aneurysm.
p<0.05 is considered significant.
Postoperative In-Hospital Course
In-hospital mortality was 9.5%: 7.4% in elective patients and 21.4% in urgent/emergent presenting patients (Table 3). Causes of overall in-hospital mortality included myocardial infarction (n=1), toxic megacolon (n=1), respiratory infection (n=1), stroke (n=2), multisystem organ failure with sepsis (n=1), intraoperative iliac rupture (n=1), and 2 patients ruptured postoperatively (1 from type Ia endoleak and 1 due to an unstented Superior Mesenteric Artery (SMA) [occluded between surgical planning and the procedure, rupturing the aneurysm prior to planned embolization]).
Perioperative Events and Requirements.
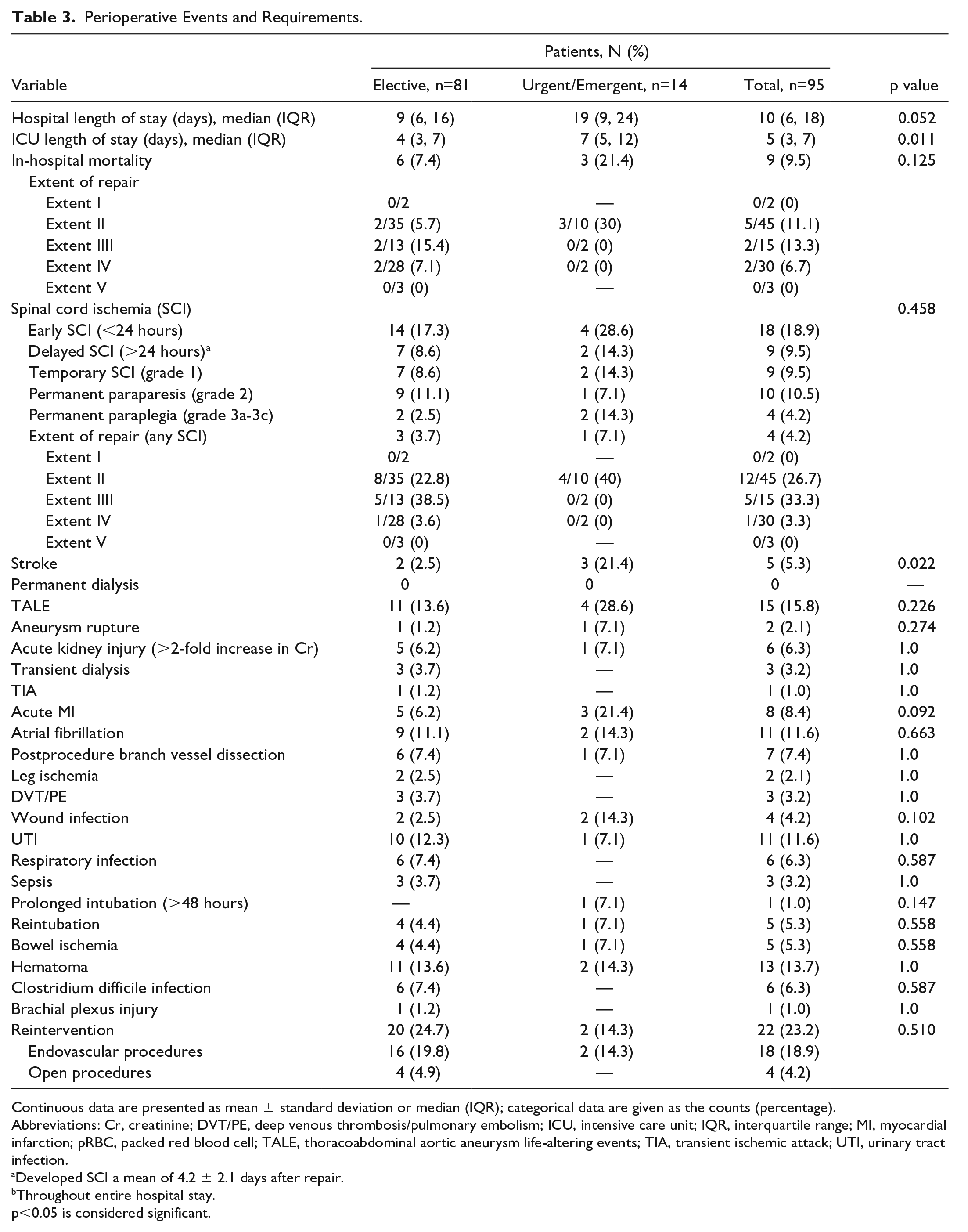
Continuous data are presented as mean ± standard deviation or median (IQR); categorical data are given as the counts (percentage).
Abbreviations: Cr, creatinine; DVT/PE, deep venous thrombosis/pulmonary embolism; ICU, intensive care unit; IQR, interquartile range; MI, myocardial infarction; pRBC, packed red blood cell; TALE, thoracoabdominal aortic aneurysm life-altering events; TIA, transient ischemic attack; UTI, urinary tract infection.
Developed SCI a mean of 4.2 ± 2.1 days after repair.
Throughout entire hospital stay.
p<0.05 is considered significant.
Eighteen (19%) patients developed SCI postoperatively with 4 (4.2%) permanent paraplegia (grade 3a-3c) cases (Table 3). Of the patients who received staged TEVAR, SCI developed in 21.4% (6/28; 1 permanent paraplegia and 1 permanent paraparesis) compared with 17.0% in those without staging (8/47; 1 permanent paraplegia and 3 permanent paraparesis, Figure 1A). SCI developed in 2 (14.3%) patients who received preoperative MISACE, with 1 case of permanent paraparesis and no permanent paraplegia. Spinal cord ischemia was observed in 6 (20.0%) non-MISACE patients, of whom 2 had permanent paraparesis and 1 had permanent paraplegia (Figure 1B). In-hospital mortality was observed in 4/18 patients (22.2%) who suffered SCI.
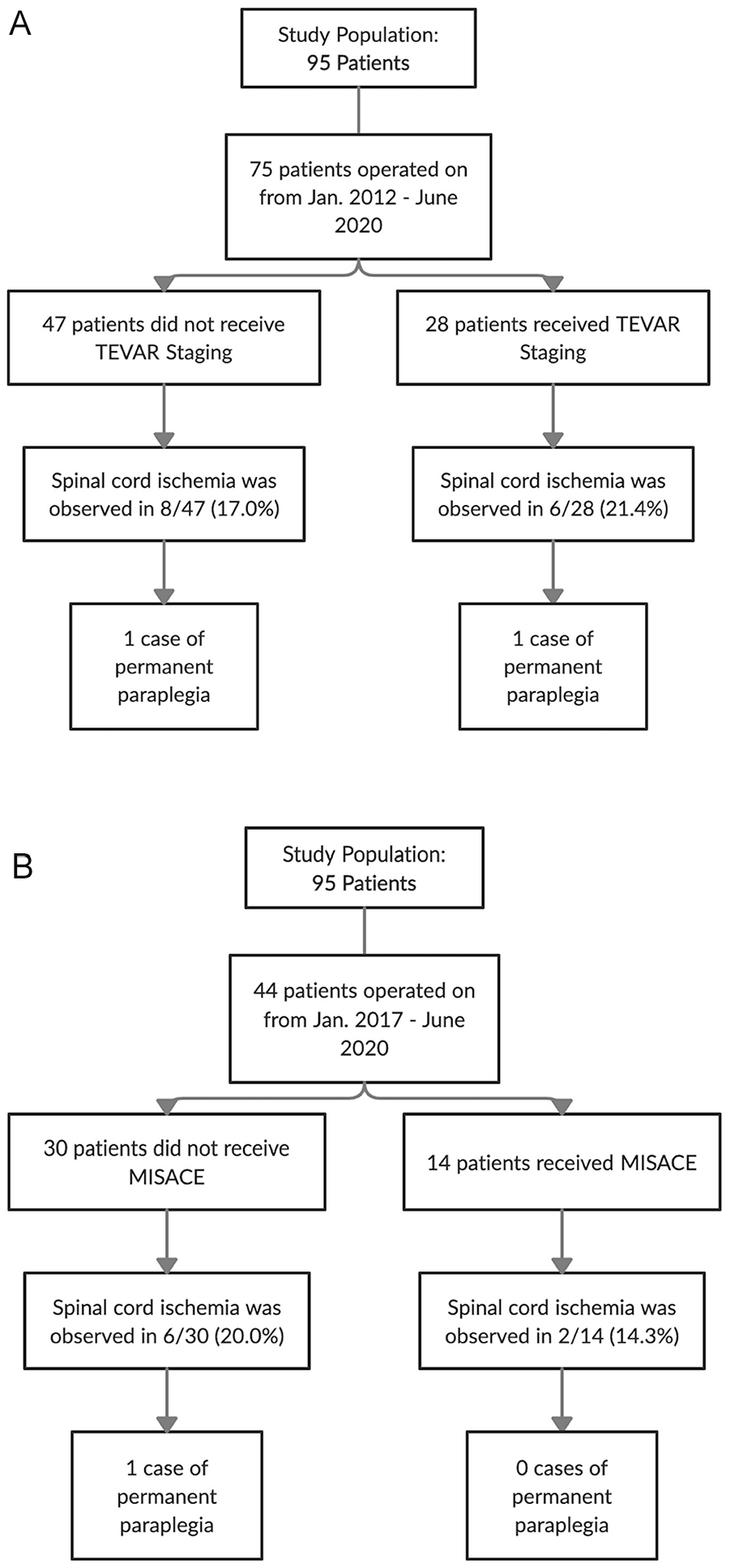
Flowchart of spinal cord ischemia and permanent paraplegia incidence in patients (A) with and without TEVAR staging and (B) with and without MISACE. TEVAR, thoracic endovascular aortic repair; MISACE, minimally invasive staged segmental artery coil embolization.
Renal dysfunction occurred in 6.3% of patients; 3.2% required temporary dialysis but no patients required permanent dialysis. Respiratory insufficiency necessitated 5 (5.3%) reintubations, whereas no patients required a tracheostomy. The incidence of the postoperative TALE was 15.8% (Table 3). Branch vessel dissection prior to discharge was observed in 10 arteries in 7 patients (7.4%) (n=1 each for left subclavian, CA, right hepatic, SMA, RR (Right renal), and left external iliac; n=2 for LR (Left Renal) and right external iliac each). Five of these required in-hospital reintervention with stenting.
In-hospital reintervention for the aorta and/or its branches was required in 22 patients (23.2%, 25 procedures: 21 percutaneous and 4 surgical). Surgical interventions included an axillary-axillary bypass to mitigate SCI (n=1) and 3 open artery repairs (left iliofemoral, right radial, and right external iliac artery repairs for iatrogenic injuries). Percutaneous procedures included creation of a type 1b endoleak to reverse SCI (n=3, successful in 1), restenting for branch instability 8.4% (stenosis, kink, occlusion, dissection, n=8). One patient required stenting of celiac, SMA, and LR arteries on his side after an incomplete emergent repair due to class III obesity that prevented imaging of those branches in the supine position. Two (2.1%) patients required branch coil embolization for bleeding (splenic artery and LR mid-pole artery) and 8 patients required endoleak repair (type 1b, n=4; type 3, n=4).
Follow-up
Clinical follow-up was 100% complete, with a mean follow-up of 3.6 ± 3.0 (range: 0-12) years during which 28 (32.6%) patients died. There was 1 confirmed aneurysm-related death: a rupture due to proximal aneurysm progression at the site of an unstented CA fenestration within the neck, 10 years after index repair in a patient that declined further intervention. Long-term survival for elective patients was 52.6% (95% confidence interval [CI], 40.1-69.0) at 5 years and 41.5% (95% CI, 28.4-60.9) at 8 years (Figure 2), whereas survival for the urgent/emergent cohort at 5 years was significantly lower, 33.7% (95% CI, 12.0-97.0; log-rank test, p<0.05). Freedom from all-cause mortality was 50.1% (95% CI, 38.4-65.4) at 5 years and 34.4% (95% CI, 22.5-52.8) at 8 years (Supplemental Figure 1). Table 4 highlights major complications in our series compared with open and EVAR literature.
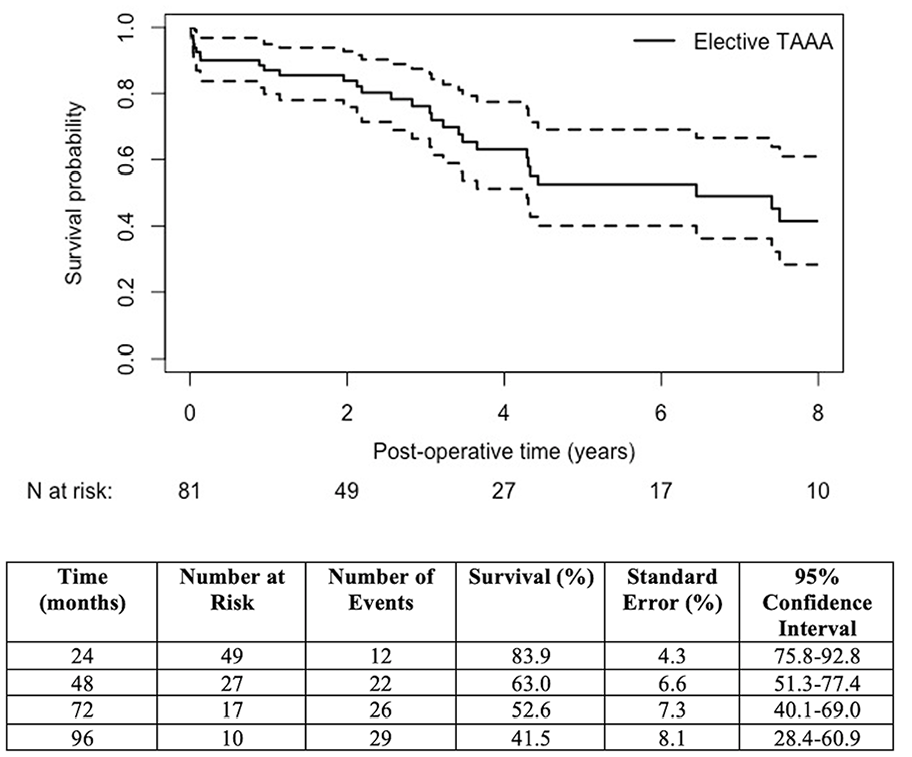
Kaplan-Meier estimates of elective patient survival after endovascular TAAA repair. TAAA, thoracoabdominal aortic aneurysm.
Comparison of Postoperative Outcomes Between Current Series and Contemporary Open/Endovascular TAAA Data.
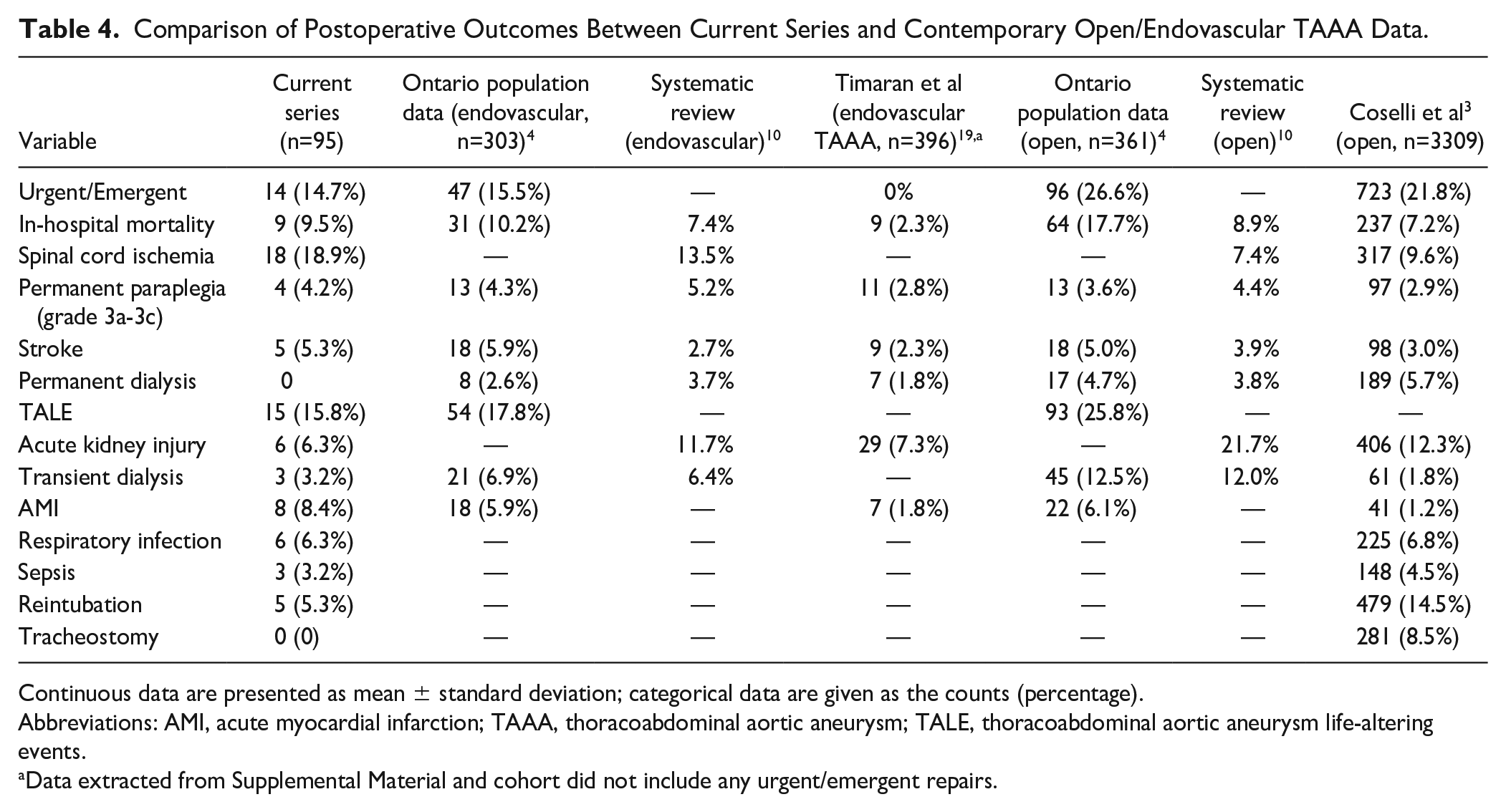
Continuous data are presented as mean ± standard deviation; categorical data are given as the counts (percentage).
Abbreviations: AMI, acute myocardial infarction; TAAA, thoracoabdominal aortic aneurysm; TALE, thoracoabdominal aortic aneurysm life-altering events.
Data extracted from Supplemental Material and cohort did not include any urgent/emergent repairs.
During follow-up, 5 (5.8%) patients suffered a stroke on average 3.1 ± 2.2 years after repair and 3 (3.5%) patients required dialysis a mean of 1.3 ± 1.2 years after repair, none having required transient postoperative dialysis. Of the patients who required dialysis on follow-up, 2 had occluded an RR artery stent (1 had a solitary right kidney) and 1 following a subsequent open arch repair. Freedom from the composite TALE outcome was 47.1% (95% CI, 35.5-62.6) and 32.3% (95% CI, 20.8-50.1) at 5 and 8 years, respectively (Supplemental Figure 2).
Surveillance imaging was performed on 80/86 surviving patients (93%), with a mean time to last surveillance imaging postrepair of 2.6 ± 2.7 years. Stable aneurysm sac size was noted in 27 (33.8%) patients, decreased aneurysm sacs (<5 mm) in 36 (45%), and sac expansion (>5 mm) was noted in 16 (20.0%). In this latter group, 15 received reinterventions to resolve sac perfusion and 1 was treated conservatively. Postdischarge target vessel kink or dissection was observed in 6 (6.9%) patients with 4 requiring restenting. Long-term primary patency for successfully revascularized target vessels was 97.7% (333/341). On follow-up, 4 patients required reintervention for target vessel stenosis/thrombosis resulting in a secondary patency of 98.8%. Freedom from branch instability was 45.5% (95% CI, 33.8-61.3) at 5 years (Supplemental Figure 3).
Forty-three patients had at least 1 endoleak identified on follow-up imaging, of which 31 (36.0%) received 44 procedures to resolve them. In total throughout follow-up, 35 (40.7%) patients required 53 late reinterventions (52 percutaneous and 1 open) for their aorta and/or visceral branches (Figure 3). Cumulative probability of reintervention was 46.3% (95% CI, 36.1-56.4) at both 5 and 8 years (Figure 4). Thirty day mortality after late reintervention was observed in 3 (8.5%) patients.
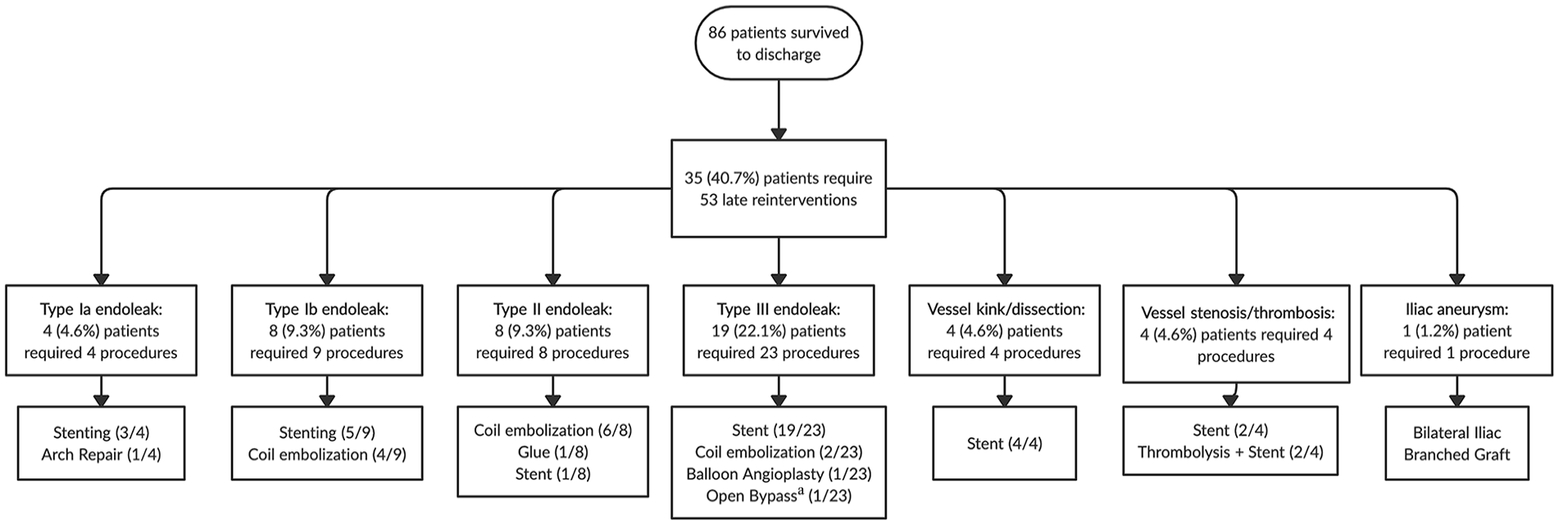
Flowchart of late reintervention indication and types of procedures executed, for a total of 53 (52 percutaneous and 1 open) reinterventions.
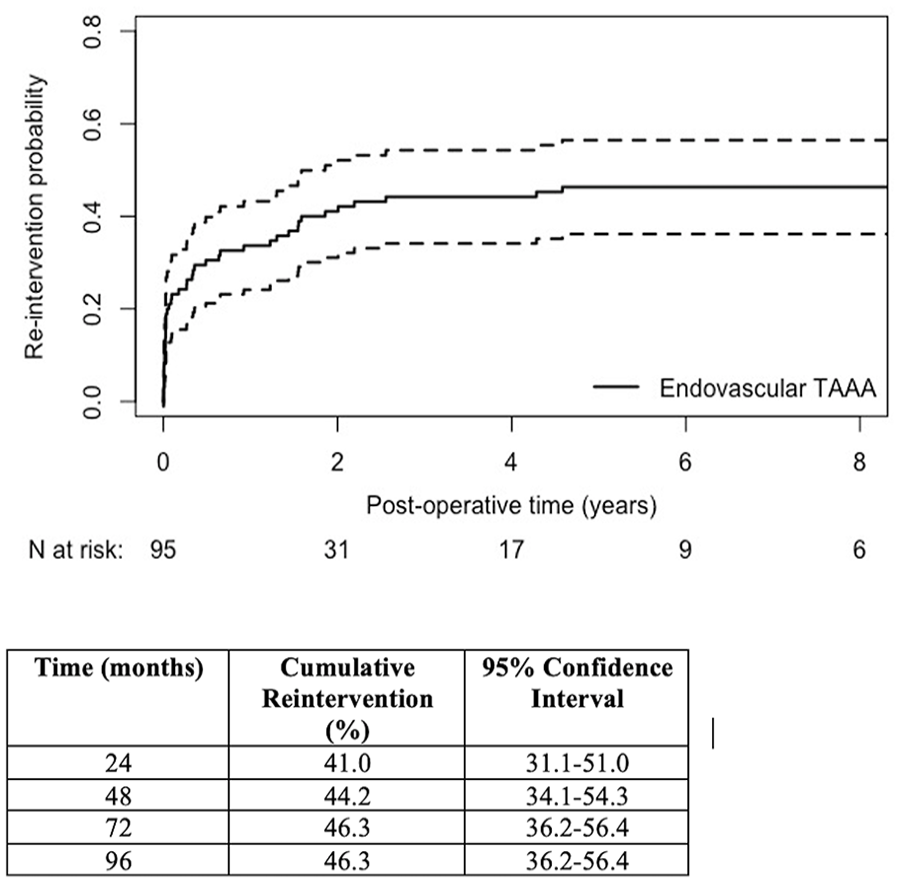
Cumulative probability of reintervention on the aorta and/or its branches after endovascular TAAA repair. TAAA, thoracoabdominal aortic aneurysm.
Discussion
Endovascular TAAA repair remains complex and technically challenging with published single-center series follow-up ranging between 1.0 and 3.0 years.10,20–22 We present a 13 year experience with endovascular TAAA repair using F/BEVAR. Our cohort is a comorbid, older population who have all been assessed and turned down for open repair. Importantly, our data include the evolution of endovascular devices and stents, constant revision of procedural techniques, and patient selection, and include all urgent/emergent repair—displaying the adoption of evolving endovascular techniques by multiple surgeons for high-risk patients at a single center.
Endovascular TAAA repair has excellent technical success, with the celiac artery accounting for the largest number of unsuccessful target vessel revascularizations (7/13), also noted by others. 23 As reported in literature, we too found target vessels to be occluded or stenosed between preoperative planning and the actual procedure, 24 resulting in a decreased primary technical success rate. This did lead to a postoperative death due to rupture in a patient as the SMA was identified as occluded intraoperatively and could not be stented intraoperatively.
Elective in-hospital mortality was 7.4%, similar to reports from other high-volume endovascular centers and a pooled proportion value of 7% from a contemporary meta-analysis of endovascular TAAA repair (Table 4).10,25,26 Moreover, a 5% to 10% operative mortality rate has been shown in large open repair series, making F/BEVAR a competitive treatment option.3,10 An Ontario, Canada, population-based propensity-matched analysis comparing open versus endovascular TAAA repair reported a higher early mortality in the open cohort (17.4% vs 10.8%), further supporting the use of F/BEVAR. 4 A 21.4% mortality rate was observed for urgent/emergent repairs; however, this rate is consistent among other reports of F/BEVAR for acute complex aortic disease. 27
Long-term survival is superior in the elective cohort versus the urgent/emergent cohort, with overall 5 year survival numbers consistent with centers of excellence.17,23,25 Our mean follow-up is 3.6 years, with an 8 year overall survival of 34.4% with F/BEVAR, comparable to the 10 year survival of 36.8% observed after open repair from Coselli et al’s 3 landmark study, despite the present series patients being on average 7 years older. At 8 years, there is a 7% higher survival rate in the elective cohort compared with the overall group, highlighting an important difference between cohorts operated electively and a consecutive cohort. In these older/comorbid patients, we demonstrate respectable long-term results, especially when compared with open repair literature.
Spinal cord ischemia remains a significant complication as endovascular series report a wide incidence between 4% and 33%. 10 Our overall SCI rate was 18.9% with the vast majority in type I to III and type V repairs. Several reports have found extent of aneurysm, single-staged procedures, and occlusion of hypogastric arteries to be predictive of SCI. 28 In the present series, 63.2% received extent II and III coverage (including appropriate seal zones), with 17/18 SCI cases being observed in this group. Increased aortic coverage as a risk factor for SCI was also reported by Kitpanit and colleagues. 29 The rate of SCI in our urgent/emergent cohort was 28.6%, with a 40% SCI rate in those requiring extent II repair. These high rates can be attributed to symptomatic or ruptured aneurysms with hemodynamic instability and extensive aortic coverage performed in a single procedure. Nonetheless, only 4 patients suffered permanent paraplegia in the entire cohort. This rate is comparable to the 2.9% permanent paraplegia rate seen by Coselli et al 3 after open TAAA repair and the 4.3% endovascular repair rate observed in the province of Ontario (Table 4). 4
Minimization of SCI after endovascular TAAA repair is an ongoing area of research. Universally adopted techniques such as the use of prophylactic CSF drainage are now being questioned due to rates of serious complications 30 and there is certainly a large amount of variance in techniques used to prevent SCI, even among high-volume centers. 31 Nevertheless, a trend toward improvement is noted using a combination of preoperative MISACE, TEVAR staging, permissive hypertension, prophylactic CSF drainage, and intraoperative neuromonitoring.
O’Callaghan and others suggested that staging may reduce the incidence of SCI.32,33 Paradoxically, we had a higher rate of SCI (21.4%) in the staged compared with the nonstaged (17.0%) cohort. However, 24/28 (86%) of the staged patients received extent II coverage, which may have increased the risk of SCI and permanent paraplegia. Thoracic EVAR staging itself comes with a risk of interstage mortality. 23 In our series, 2 patients presented with contained rupture and 1 with a symptomatic aneurysm requiring emergent repair with a T-branch graft between stages. The potential benefit of reduced SCI needs to be weighed against the risk of rupture for patients receiving staging. Data with larger sample sizes are needed to thoroughly evaluate this adjunctive technique in minimizing postoperative complications, compared with the use of MISACE.
Our practice further evolved in 2017 when the promising initial results of MISACE by Branzan et al 34 showed no SCI after endovascular TAAA repair. We observed a trend in SCI improvement after MISACE with 2 cases of paraparetic SCI out of 14 MISACE-treated patients (14.3%), with no permanent paraplegia, while the rate of SCI in the non-MISACE cohort treated from 2017 onwards was 20.0%. Our current standard approach to reduce SCI in patients that require extensive coverage (<4 cm of above celiac axis) is a 3-stage approach of MISACE, TEVAR, followed by endovascular TAAA repair with spinal cord drainage, neuromonitoring, and permissive hypertension. Spinal cord ischemia rates require further improvements to reduce the postoperative SCI burden.
The low postoperative renal and pulmonary morbidity in our series are key advantages of endovascular TAAA repair. Despite the 38% incidence of preoperative stage III to IV chronic kidney disease (CKD), there were no cases of permanent dialysis and only 3 patients required transient dialysis, in keeping with rates of permanent dialysis of 0% to 8% reported by other centers. 10 Conversely, high-volume open TAAA repair is associated with a 7.6% rate of postoperative dialysis, 5.7% of which is permanent. 3 While renal dysfunction is a major morbidity of open repair, 35 a low rate (3.5%) of dialysis in our long-term follow-up means F/BEVAR shows promise in operating on patients with preexisting CKD and reducing dialysis-dependent renal failure. Despite our significant incidence of COPD, no patients required a tracheostomy and only 5.3% required reintubation. In contrast, Coselli et al 3 noted a 14.5% reintubation rate and a tracheostomy incidence of 8.5%. Our pulmonary-renal complication rates are similar to the ones seen at high-volume centers and confirm the analysis from multiple national database studies that suggest F/BEVAR leads to lower rates of perioperative morbidity, especially as it relates to renal and pulmonary complications.17,36,37
Postoperative rates of TALE, a composite outcome used to measure successful repair and recovery, were 15.8%, consistent with the 17.8% rate of postoperative TALE after endovascular repair in Ontario and are considerably lower than after open repair (25.8%) (Table 4). 4 Long-term freedom from TALE was 47.1% at 5 years and 32.3% at 8 years and parallels freedom from mortality at 50.0% and 34.4%, respectively. Kang et al 38 reported a lower “good outcome” (77%), their own measure of repair and recovery, compared with survival (88%) at 1 year in their endovascular TAAA cohort. Both terms are important because their components are viewed by patients as treatment failures; however, our longer follow-up data exhibit that major complications are rare in the follow-up period and postdischarge TALE is driven by mortality. Durability of TAAA repair by F/BEVAR was excellent with only 1 case of late aneurysm rupture identified at 10 years after primary repair, demonstrating that late mortality is infrequently due to aneurysm-related pathology.
Secondary procedures are common after endovascular TAAA repair, likely secondary to the multiple components required and our policy to aggressively treat endoleaks associated with aneurysm growth over 5 mm.4,17,23 Reinterventions were 98% percutaneous and 81% of them were to treat endoleaks, divided equally among fenestrations and branches. Despite the need for reintervention, long-term vessel patency is excellent at 98.8% overall at follow-up with only 4 patients requiring intervention for occlusion and/or stenosis. Moreover, most reinterventions occurred on the renal arteries for type III endoleaks, consistent with Mastracci’s report where the renal arteries were noted to be the most unstable with a high reintervention rate that did not affect long-term clinical outcomes. 39 Rocha and colleagues 4 reported a 14% 30 day mortality after reintervention for endovascular TAAA repair in Ontario, highlighting 1 of the limitations of endovascular repair over open repair; however, we observed this in only 8% of cases.
In the 20 years since the initial description of endovascular TAAA repair, similar results are achieved and even improved results compared with the 70 year evolution of open repair. A summary of TAAA outcomes from population-based studies, systematic reviews, and the largest open series is compared in Table 4. While patient cohorts differ with respect to the incidence of connective tissue disorders and comorbidities, it is clear that endovascular TAAA repair has overcome its early technical challenges and outcomes are likely to continue to improve with better bridging stents and strategies to reduce SCI.
Limitations of this study include the retrospective nature, technical evolution of the procedure, and the “learning curve” associated with a complex procedure with several surgeons. While the mean follow-up was only 3.0 years due to inclusion of recent repairs (June 2020), we demonstrate some of the longest follow-up to date, a maximum of 12 years and Kaplan-Meier estimates to 8 years. However, further long-term data with close imaging follow-up are needed to better characterize durability and need for late reintervention. The biggest limitation is the modest and diverse sample size.
Conclusion
This series demonstrates a single-center evolution of endovascular TAAA repair. Mortality and SCI, while higher compared with referral centers, are on par with systematic reviews and compare favorably to population-based open/EVAR TAAA repair data from Ontario. Importantly, we demonstrate the renal and pulmonary advantages of endovascular TAAA repair. While further refinements in techniques continue, long-term survival and branch patency was excellent, with very few branch occlusions. We demonstrate safety in high-risk patients, even as the procedure and technology evolved. Further improvements in short-term and long-term outcomes of endovascular TAAA repair rival open repair and should be subject to prospective comparative studies.
Supplemental Material
sj-tiff-1-jet-10.1177_15266028241229005 – Supplemental material for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe
Supplemental material, sj-tiff-1-jet-10.1177_15266028241229005 for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe by Daniyal N. Mahmood, Rodolfo Rocha, Maral Ouzounian, Kong Teng Tan, Samantha M. Forbes, Jennifer C-Y. Chung and Thomas F. Lindsay in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-2-jet-10.1177_15266028241229005 – Supplemental material for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe
Supplemental material, sj-tiff-2-jet-10.1177_15266028241229005 for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe by Daniyal N. Mahmood, Rodolfo Rocha, Maral Ouzounian, Kong Teng Tan, Samantha M. Forbes, Jennifer C-Y. Chung and Thomas F. Lindsay in Journal of Endovascular Therapy
Supplemental Material
sj-tiff-3-jet-10.1177_15266028241229005 – Supplemental material for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe
Supplemental material, sj-tiff-3-jet-10.1177_15266028241229005 for Thoracoabdominal Aortic Aneurysm Repair Using Fenestrated and Branched Endovascular Grafts for High-Risk Patients: Evolving yet Safe by Daniyal N. Mahmood, Rodolfo Rocha, Maral Ouzounian, Kong Teng Tan, Samantha M. Forbes, Jennifer C-Y. Chung and Thomas F. Lindsay in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors would like to acknowledge Leah Gabriel for her assistance with patient follow-up results and Dyo Ramcharan for his diligent tracking of institutional endovascular procedures.
Authors’ Note
This study was presented in the Virtual Poster Competition at the 2020 Vascular Annual Meeting of the Society for Vascular Surgery, Toronto, Canada, June 17 to 20, 2020.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.O. received speaker honoraria from Edwards, Terumo, and Cryolife and is a member of the advisory board in Medtronic. K.T.T. is a clinical trials site principal investigator for Boston Scientific, Cook, BD Medical, and Gore. T.F.L. is a principal investigator for trials and proctoring for Cook Canada; however, all funds are paid to the Department of Surgery Scholarship Funds, University of Toronto. The other authors declare no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
