Abstract
Purpose:
To present a rare cause of type III endoleak via the left renal artery (LRA) fenestration following fenestrated endovascular aneurysm repair (FEVAR) and to describe a successful reintervention for treating this endoleak.
Technique:
The patient presented with a type IIIc endoleak following FEVAR, due to inadvertent placement of the LRA bridging balloon expandable covered stent (BECS) via the superior mesenteric artery (SMA) fenestration, but deployed outside the SMA fenestration. The proximal part of the BECS was positioned outside of the main body. This caused a type IIIc endoleak via the open LRA fenestration. Reintervention was performed by relining the LRA with a new BECS. First, access to the lumen of the previously placed BECS was gained using a re-entry catheter, followed by placement of a new BECS via the LRA fenestration. Completion angiography, and computerized tomography angiography (CTA) at 3 months follow-up showed total obliteration of the endoleak and patency of the LRA.
Conclusion:
Placement of a bridging stent via an incorrect fenestration during FEVAR is a rare cause of type III endoleak. In certain cases, successful treatment of such an endoleak could be achieved by perforation and relining of the misplaced BECS via the correct fenestration of the target vessel.
Clinical Impact
To our best knowledge, a type IIIc endoleak following fenestrated endovascular aneurysm repair, due to placement of a bridging covered stent through an incorrect fenestration and deployed short of the fenestration, has not been described before. Reintervention was performed with perforation of the previously placed covered stent and relining using a new bridging covered stent. The technique presented here was successful for treating the endoleak in this case and could help guide clinicians when dealing with this or similar complications.
Keywords
Introduction
Fenestrated endovascular aneurysm repair (FEVAR) is an effective treatment for complex abdominal aortic aneurysms (AAA). Although short-term outcomes are comparable to open reconstruction, reintervention is more commonly seen using an endovascular approach, most notably due to a type I or II endoleak. 1 Type I and III endoleaks require prompt intervention, due to the risk of aneurysm expansion and rupture, thereby necessitating frequent follow-up with computerized tomography angiography (CTA).2–6 A type III endoleak is defined as leakage due to detachment between or defects of stent components. A distinction is made between inadequate attachment of the aortic-aortic/bifurcated components (type IIIa), iliac limb components (type IIIb), branch components (type IIIc), and graft tears, perforations, and fractures (type IIId). 7 This report presents a rare cause of type IIIc endoleak via a renal fenestration, due to inadvertent placement of the renal bridging covered stent via the superior mesenteric artery (SMA) fenestration, but deployed short of the SMA fenestration. A successful reintervention for treating this complication is described.
Technique
The patient, an 82-year-old male with a past medical history including hyperhomocysteinemia, heterozygous factor V Leiden and chronic obstructive pulmonary disease, was previously diagnosed with a 50-mm juxta-renal AAA and a 42-mm common iliac artery aneurysm. The 4-FEVAR with a left sided iliac branched device was indicated. Target vessels were stented using a 7 mm x 32 mm Advanta V12® (Atrium Medical Corporation, Merrimack, NH, USA) balloon-expandable covered stent (BECS) for both celiac trunk and SMA. The right renal artery was stented with a 5 mm x 22 mm BeGraft® Peripheral BECS (Bentley InnoMed, Hechingen, Germany) distally and extended proximally into the fenestration with a 6 mm x 22 mm BECS. For the left renal artery (LRA) a 5 mm x 22 mm BeGraft® BECS was used. The total procedure length was 215 minutes, with a fluoroscopy duration of 51 minutes, a dose area product (DAP) of 147 Gy·cm2, and 120 mL of contrast agent were used. Completion angiography demonstrated patency of all target vessels and an endoleak (Figure 1). At first, it was thought to potentially be a type II endoleak via the inferior mesenteric artery (IMA) or lumbosacral arteries. The postoperative abdominal X-ray showed no abnormalities (Figure 2). A close look at the procedural 1-month follow-up CTA showed the proximal part of the LRA 5 mm x 22 mm BeGraft® stent positioned outside and ventrally of the main body (Figure 3). It was hypothesized that the stent was placed via the SMA fenestration into the LRA, but deployed outside the SMA fenestration. This caused a type IIIc endoleak via the open LRA fenestration. Patency of the LRA stent was presumably caused by flow through the LRA fenestration to the aneurysm sac and into the open end of the LRA stent. It was then decided to try to reline the LRA with a new BeGraft® BECS via the LRA fenestration, by perforating the previously placed BeGraft® BECS. Under local anesthesia, access was gained through the right common femoral artery. First, 2 ProGlide® Suture-Mediated Closure Systems (Abbott Vascular, Redwood City, CA, USA) were placed, followed by placement of a 12-F Dryseal® sheath (W.L. Gore & Associates, Flagstaff, AZ, USA) over an 80 cm Amplatz® guidewire (Cook Medical Inc, Bloomington, IN, USA). Using a 7-F Destino Twist® steerable guiding sheath (Oscor, Palm Harbor, FL, USA) access was secured through the still open LRA fenestration. The BeGraft® BECS was punctured under 30° RAO projection using a 4-F Berenstein® catheter (Cordis Corporation, a Cardinal Health company, Milpitas, CA, USA) and a 5.9-F, 120 cm Outback® Elite re-entry catheter (Cordis Corporation, Miami Lakes, FL, USA). Flow detection through the Outback® catheter confirmed successful access to the lumen of the stent. An 0.014’’ Command® guidewire (Abbott Vascular, Redwood City, CA, USA) was advanced into the LRA. Subsequently, predilatation of the puncture hole in the stent graft with a 3 mm micro balloon was carried out. After switching to a Rosen® guidewire (Infiniti Medical, Malibu, CA, USA), predilatation was repeated using a 6 and 8 mm balloon (Advance Low-Profile PTA Balloon; Cook Medical, Bloomington, IN, USA), respectively. After switching to an 8.5-F Destino® steerable sheath with an 80 cm 6-F flexor sheath, the new 5 mm x 22 mm BeGraft® BECS was placed successfully. Following flaring with an 8 mm x 20 mm balloon, the endoleak was still minimally present. Endoleak via the SMA was excluded with a lateral angiogram. It was therefore decided to reline the LRA again, using a 6 mm x 22 mm Advanta V12® BECS, and after flaring using a 10 mm x 20 mm balloon (Advance Low-Profile PTA Balloon; Cook Medical), the endoleak was no longer detectable (Figure 4). The puncture site in the groin was closed by securing the ProGlide® sutures. The total procedure length was 112 minutes, with a fluoroscopy duration of 40 minutes, a DAP of 197 Gy·cm2, and 80 mL of contrast agent were used. Duplex ultrasound at 6 weeks follow-up showed patency of the LRA and no endoleak present. Post-operative CTA at 3-months follow-up demonstrated patency of the LRA and elimination of the type III endoleak (Figure 5).
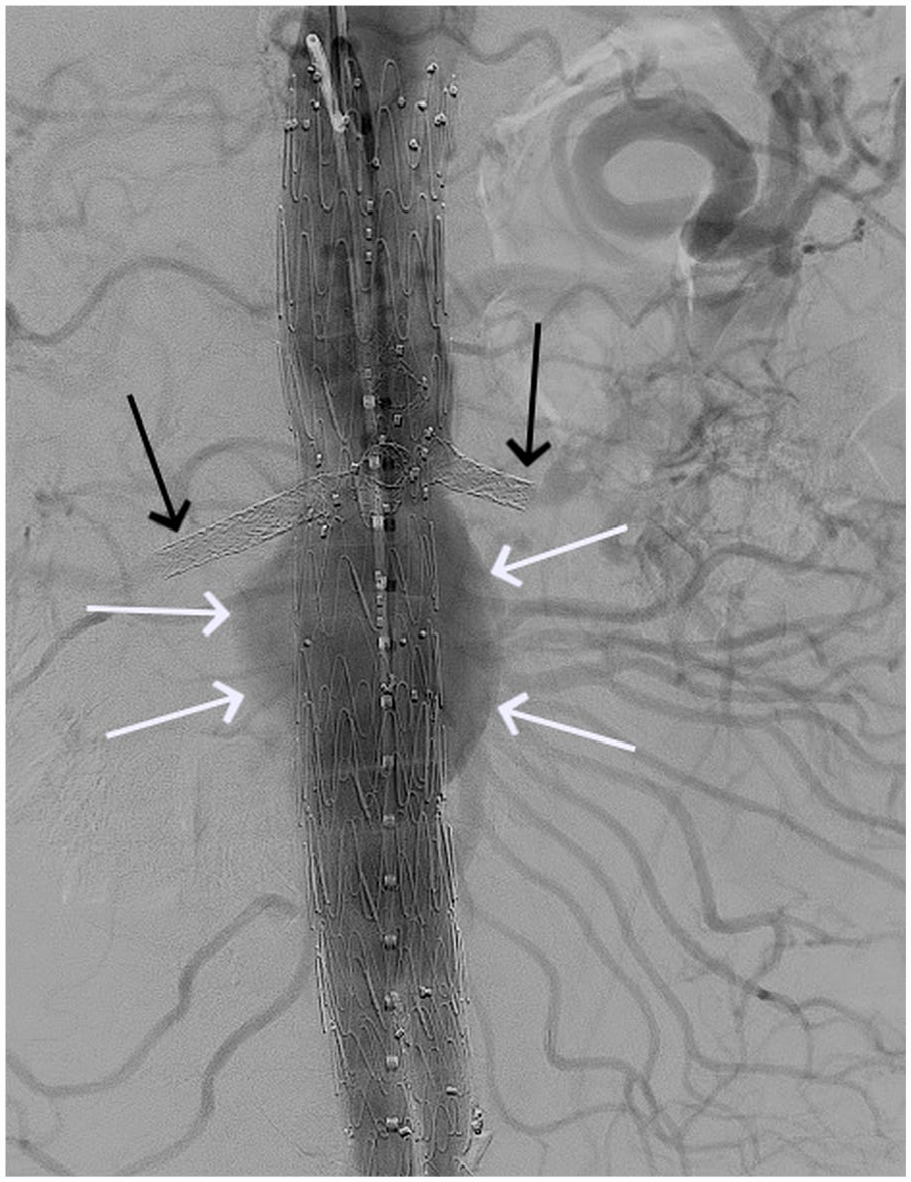
Completion angiography after fenestrated endovascular aneurysm repair demonstrating patency of the renal artery bridging stents (black arrow) and an endoleak (white arrow).
Postoperative abdominal X-ray showing no abnormalities.
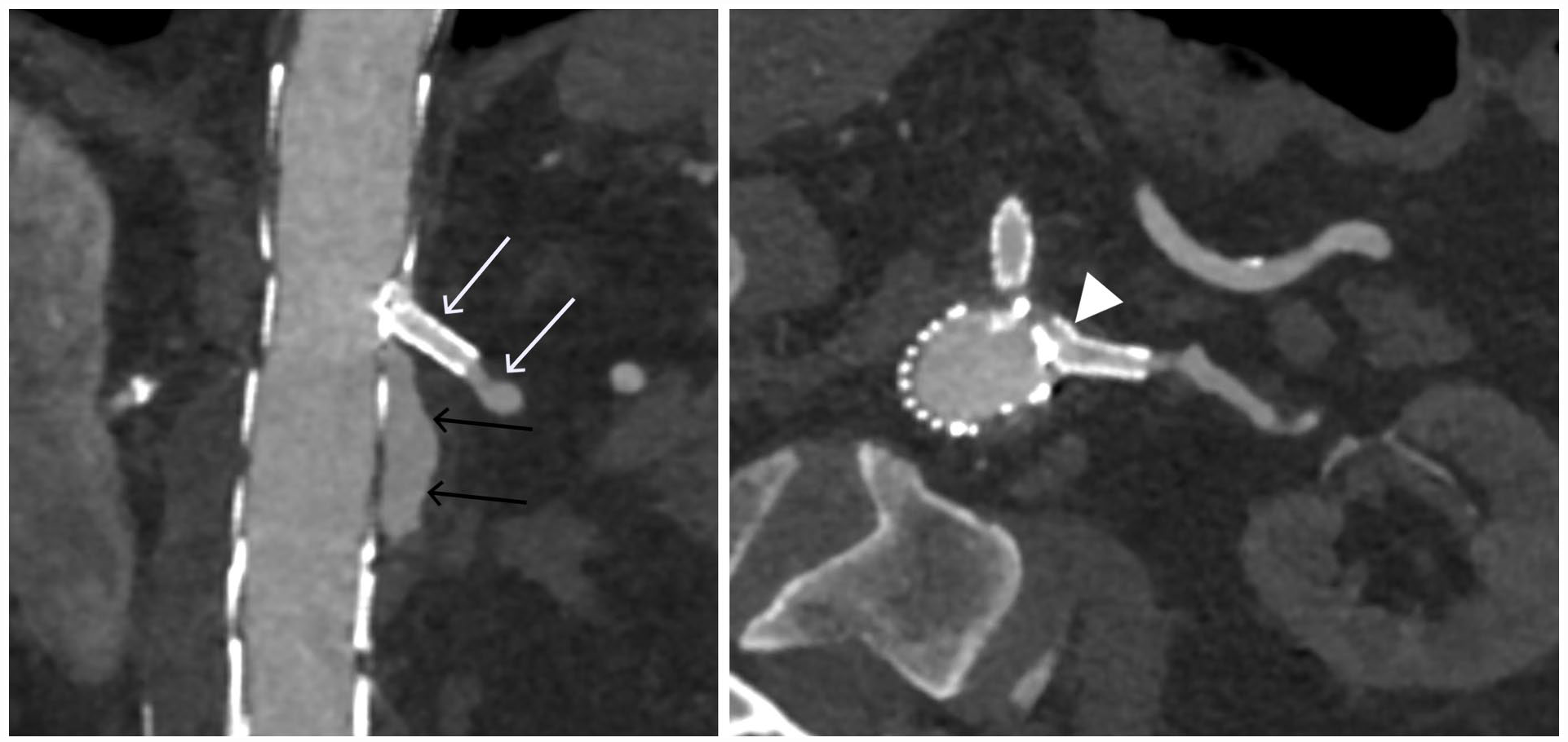
One-month follow-up computerized tomography angiography (CTA) after the initial procedure showing a type III endoleak (black arrow), a patent left renal artery (LRA) bridging stent (white arrow) and the proximal part of the LRA bridging stent positioned outside and ventrally along the main body (arrowhead).
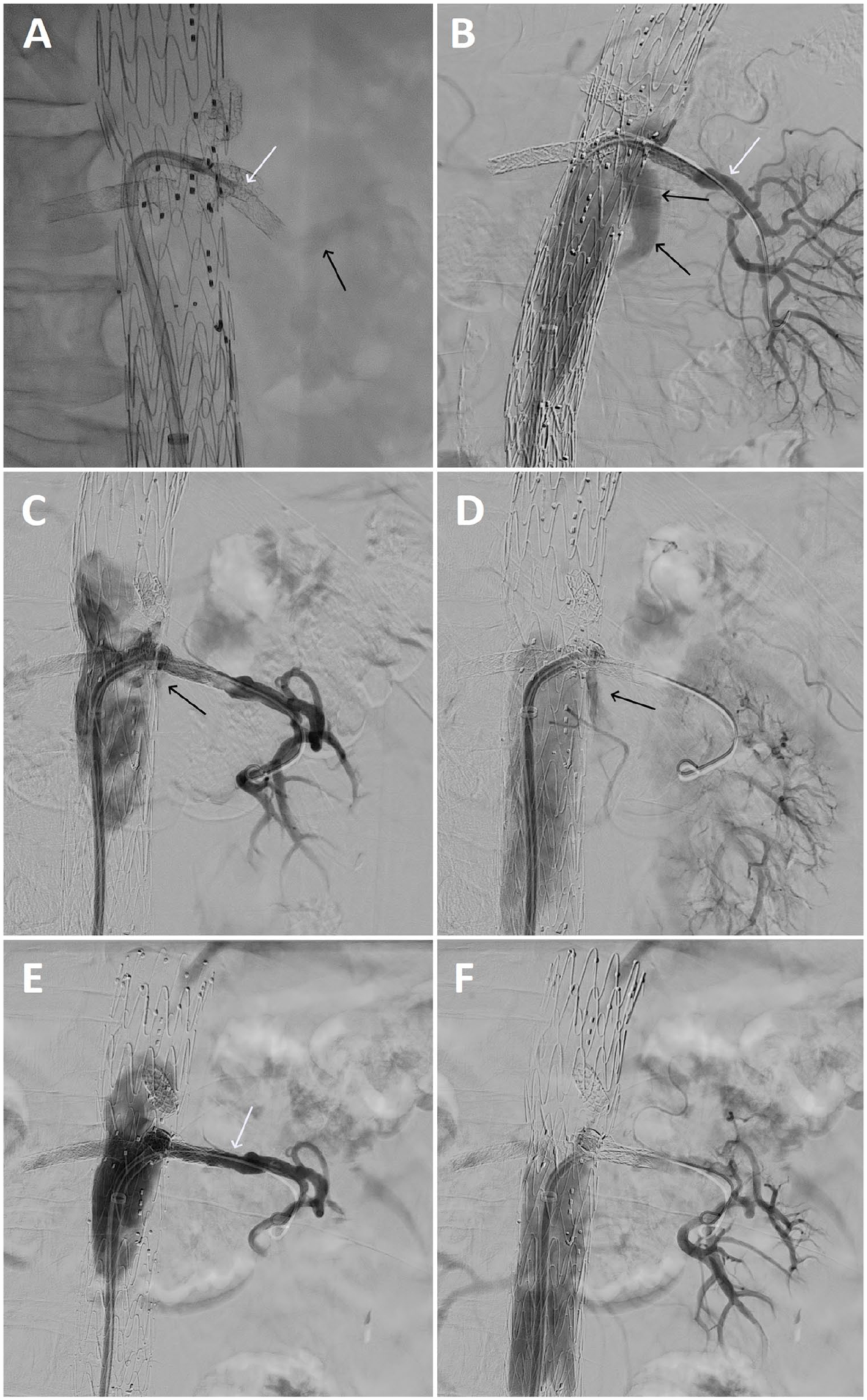
Angiography during perforation and relining of the left renal artery (LRA) bridging stent. (A) Perforation of the previously placed stent with the sharp needle (white arrow) on the Outback® re-entry catheter and a flow check through the needle (black arrow). (B) Type III endoleak (black arrow) before relining and a patent LRA (white arrow). (C+D) Angiography after placement of the BeGraft® stent, showing a patent stent with a minimally present endoleak (black arrow). (E+F) Angiography after placement of the Advanta V12® stent, showing obliteration of the endoleak and a patent LRA (white arrow).
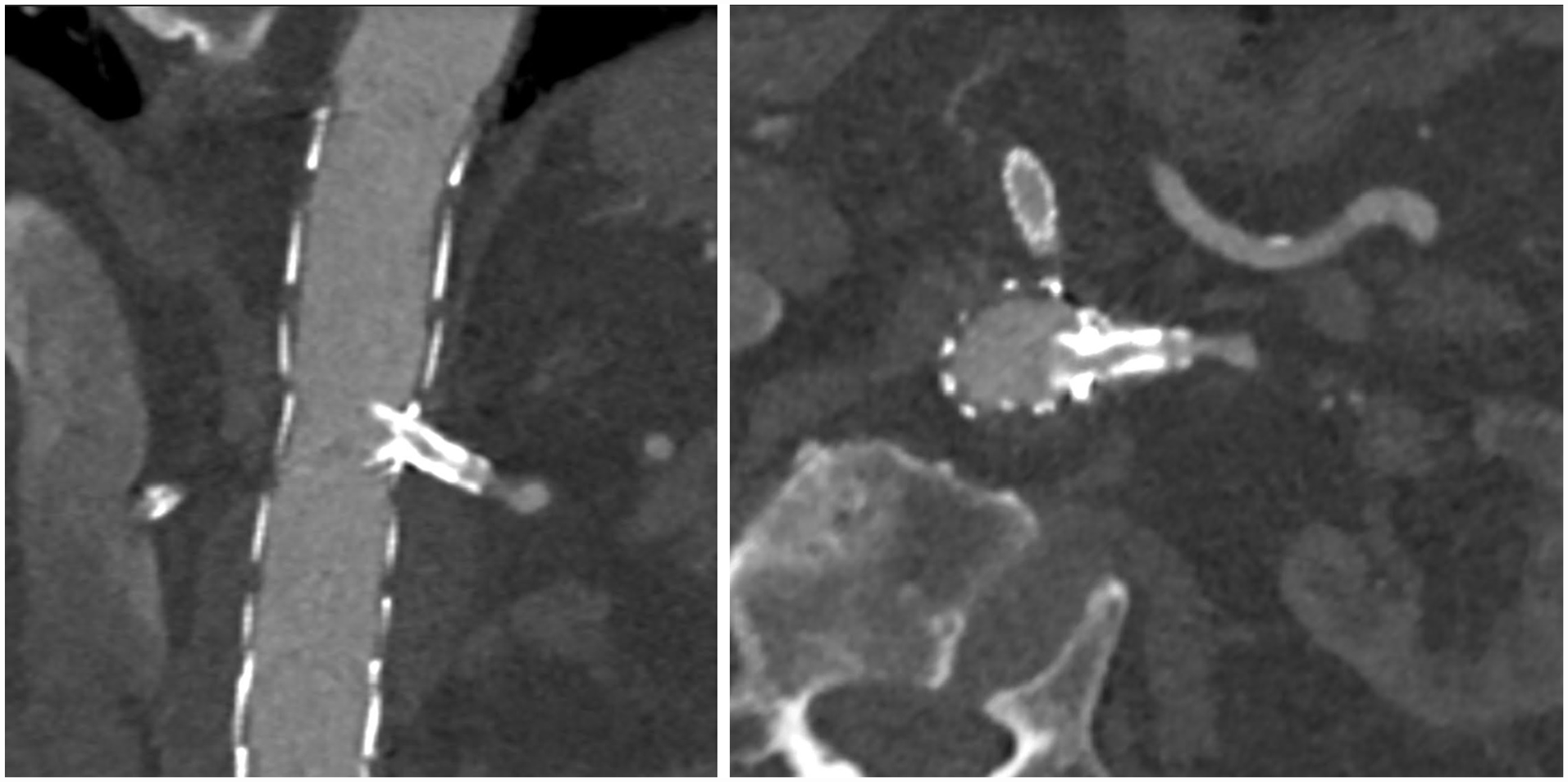
Three-months follow-up computerized tomography angiography (CTA) after the reintervention, showing a patent left renal artery (LRA) and elimination of the type III endoleak.
Discussion
Causes of type III endoleak include inadequate attachment of stent components or defects of stent components. We observed and treated a rare cause of type IIIc endoleak via the LRA fenestration following 4-FEVAR, due to inadvertent placement of the LRA bridging stent via the SMA fenestration. The proximal part of the LRA bridging stent was therefore positioned ventrally of the main body, but outside the SMA fenestration, as seen on CTA.
Inadvertent placement of a bridging stent via an incorrect fenestration has been described before. 8 To our knowledge, the combination of the stent placed through an incorrect fenestration and deployed short of the fenestration, indicating this technique, has not been described before.
Cannulation of an incorrect fenestration is a known complication while performing FEVAR, especially when fenestrations lie near or in line with each other. In this patient, the SMA was located in close approximation, and slightly distal to the LRA. Extra caution is warranted in such cases, by verifying placement of the guidewire inside the appropriate fenestration in multiple radiographic projections.
Because the LRA bridging stent placed via the SMA fenestration was deployed short of the fenestration, it was still possible to place the SMA bridging stent via the SMA fenestration. Therefore, the misplaced LRA bridging stent initially went unnoticed. Due to flow via the endoleak, the LRA bridging stent still appeared patent on completion angiography, emphasizing the importance of looking for this type of endoleak on CTA.
Due to kinking of the misplaced LRA bridging stent, in combination with flow via the endoleak through the non-stented fenestration, the stent was at risk of becoming occluded with a subsequent loss of renal function. This and similar complications going unnoticed on completion angiography can therefore risk target vessel loss. Fortunately, the left kidney was still perfused when the endoleak was detected 1 month after the procedure. A valuable option to aid in prompt diagnosis and treatment of such an endoleak is performing cone beam computed tomography (CBCT) during the primary procedure. CBCT allows for accurate detection of technical problems such as kinks or stent graft compression requiring additional treatment.9,10 Routinely performing CBCT scans at the end of FEVAR procedures may in comparable future cases prevent target vessel loss, by early detection of such technical failure.
Perforation of the previously placed stent and relining using a new bridging stent, is a successful way of eliminating this cause of type IIIc endoleak. This can be achieved by using a Berenstein® catheter and a crossing or re-entry catheter to gain access to the lumen of the already placed stent via the correct fenestration, and placing a new stent graft after predilatation. Re-entry catheters are typically used during recanalization of chronic total occlusions (CTO). This reports demonstrates the possibility of using a re-entry catheter to perforate covered stent graft material and gain access to the lumen of the stent. An alternative technique could be to try to gain access between stent graft and target vessel wall with a new BECS along the previously placed bridging stent graft. However, this was considered a more dangerous technique as it is prone to dissection of the vessel wall.
Conclusion
Endoleaks are the most common cause of reintervention following endovascular aortic repair. Inadvertent placement of a bridging stent through an incorrect fenestration is a rare cause of type III endoleak. Patency of the target vessel on completion angiography, due to flow via the endoleak, highlights the importance to screen for this cause of type III endoleak during CTA follow-up. Reintervention with perforation of the previously placed stent via the correct fenestration and relining using a new bridging stent could be a successful treatment, causing total obliteration of the endoleak in this case.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
