Abstract
Purpose:
A variety of last-generation endografts are currently available for standard endovascular repair (EVAR) of infrarenal abdominal aortic aneurysms (AAAs). The purpose of this study is to report the preliminary clinical outcomes of the Minos trimodular stent-graft system, which was recently introduced to the European market.
Materials and Methods:
Between February 2020 and 2022, we treated 41 consecutive AAA patients (mean age 72.2±8.5, 37 males) with elective standard EVAR using the Minos. The mean maximum diameter of AAAs was 54.7±6.6 mm, the mean proximal neck’s (PN) diameter was 24.8±2.7 mm, while the relevant length and angulation were 16.0 mm and 21.7°, respectively. Overall, 22 (53.6%) patients presented with shorter and angulated PN, according to the stent-graft’s instructions of use, and in 6 (14.6%) patients the PN angulation >60° was combined with concomitant iliac angulation >60°. Eleven (26.8%) EVARs were performed with concomitant enormous iliac artery narrowing and tortuosity. Finally, in 19 (46.3%) AAAs, the distal iliac landing zone was aneurysmatic and they were treated with the bell-bottom technique in 17 patients and with limb extension to the external iliac artery in two cases. We evaluated technical and clinical success of the index procedures, which was based on the combination of five factors: freedom from EVAR-related mortality, from graft-related endoleak of any type, from migration at any part of graft as well the absence of notable increase AAA’s sac maximum diameter and the patency of bifurcated stent-graft and of access vessels.
Results:
Primary technical and clinical success of index procedures was 100%. During a median 12-month radiological follow-up the clinical success remained 100%. No type I or III endoleak, stent-graft migration, EVAR-related death, AAA rupture, or graft-related adverse events or reinterventions were documented. Four (9.8%) type II endoleaks were detected with stable AAA sac diameter. The overall incidence of sac regression was 34.1% (n=14).
Conclusion:
The preliminary results of our series showed that Minos provided excellent feasibility and safety features even through angulated and tortuous iliac vessels and in short and angulated PNs. The overall clinical success at 1 year suggests that performance of Minos follows very high standards. Further validation of these promising results with long-term data is acquired to complete the evaluation of this recently introduced stent-graft system.
Clinical Impact
The current study explored the clinical performance of a new in market ultra-low profile bifurcated abdominal aortic stent-graft, the MINOS. The early and 12-month results of study suggest that implantation of this stent-graft in standard EVAR, even in hostile proximal aortic neck and iliac vessels conditions, follows very high clinical standards and encourage the further clinical use of MINOS.
Introduction
Standard endovascular abdominal aortic aneurysm (AAA) repair (EVAR) was first introduced in 1991 and showed a remarkable and continuous evolution, achieving an extremely high safety and efficacy profile compared to the gold standard of open repair. In the last decade, EVAR has become an equally acceptable first-line treatment and the worldwide rate of EVAR’s utilization is over than 50%, especially in the elderly and high-risk patients.1,2 The key role in this exponential increase of EVAR’s utilization rate is the continuous evolution of endovascular techniques, materials and especially of stent-grafts which increased the applicability of EVAR. 3 Several stent-grafts have been introduced and withdrawn from market due to early or long-term failures, while other brands showed remarkable endurance overtime, which was based mainly in their successful evolution and redesign of previous platforms.4 –6 Additionally to the brands that have evolved over time, there are currently available on market several recently introduced last-generation stent-grafts. One of them is the developed by Endovasctec Minos stent-graft system (Shanghai MicroPort Endovascular MedTech Co., Shanghai, PRC). Minos was first introduced in China in 2019 and acquired the Conformité Européenne (CE) mark in September of the same year, and we performed the first implantation of this stent-graft outside China on February 20, 2020.
The availability of this wide variety of endografts provides multiple options in selecting the appropriate endograft, fit to any specific AAA anatomy and morphology. In experienced hands, the technical characteristics and mechanical properties of each device play the primordial role in feasibility and safety of the indexing procedure as well in the durability of outcomes and dictate the framework within the correct choice should be made, among all the available stent-graft options. The aim of this study is to evaluate the clinical performance and early results of Minos and to provide the data from our preliminary experience, in a series of patients treated with conventional elective EVAR using the Minos stent-graft system.
Materials and Methods
Study Design
The study covers the period from February 2020 till February 2022 and is a retrospective analysis of prospectively collected in electronic form data of patients treated by conventional EVAR, using the Minos stent-graft, in our academic institution. The study protocol was approved from our institutional Ethical Committee, and it was in accordance with the principles of declaration of Helsinki. All patients gave their informed consent regarding the data storage, analysis, and presentation in this study as well for their willing to conform with the scheduled follow-up protocol.
All procedures were performed by a single senior vascular surgeon, using the same protocol of preoperative planning, procedure, and perioperative care. Inclusion criteria for the surgical treatment of fusiform infrarenal AAAs were considered the presence of a maximum aneurysm diameter ≥ 55 mm, while in saccular AAAs or in cases with a high annual growth rate (> 6 mm/year) the diameter threshold was set at 50 mm. EVAR procedure planning enabled aneurysm’s morphometrical analysis in preoperative computer tomography angiogram (CTA) using the center lumen line (CLL) technique. 7 Our protocol of conventional EVAR includes AAA with infrarenal proximal neck (PN) width ≤30 mm and length ≥10 mm, regardless the presence of PN’s angulation or other adverse morphological features, such as the PN’s bulge or conical shape and/or the thrombus or calcification circumference, as well notwithstanding in any adverse iliac arteries anatomy. 8
Definitions and Endpoints
Primary technical success (PTS) was defined as the completion of endograft’s implantation without any graft-related visible endoleak and with patency of both stent-graft’s limbs and access vessels. PTS was evaluated at the end of index procedure and at 30-day CTA follow-up. Assisted PTS was achieved in cases with adjunctive endovascular or open techniques during index procedure. The primary endpoint of study during follow-up was the clinical success, which was based on the concomitant presence of these five factors: the freedom from EVAR-related mortality; freedom from graft-related endoleak of any type; freedom from migration at any part of graft; the absence of notable increase in AAA’s sac maximum diameter and finally the patency of the bifurcated stent-graft and of access vessels. Graft related endoleaks included all types except from type II endoleak if it was unequivocally detected to be the one and only source of endoleak. As notable increase in AAA’s sac maximum diameter was defined the increase of sac diameter by ≥2.5 mm and as important if it was ≥ 5 mm. Secondary endpoint was considered the freedom from graft-related reintervention by endovascular means or by open conversion during follow-up.
As PN length we considered the infrarenal part of abdominal aorta with diameter at the level of lowest renal artery ≤30 mm (LRND) and with diameter increase at the end of neck length (END) ≤10%. For calculation of graft’s PN oversizing, we used the mean neck diameter (MND) value between the LRND and END. The presented iliac diameters are referred to the maximum diameter at the distal landing zone just before the iliac bifurcation. Total aortoiliac lengths were considered the distances between the lowest renal and iliac bifurcations using the respective central lumen lines. Diameters of external iliac and femoral arteries were measured in the narrowest vessels’ point. We analyzed the infrarenal neck’s angulation, which was the angle between the axis of the PN and the CLL axis of the aneurysm, and the iliac angulation which were calculated as the angle between the terminal aorta’s CLL and the respective iliac axes. Furthermore, we recorded the concomitant presence of iliac arteries narrowing ≤5 mm from a circumferential calcified lesion and iliac tortuosity index >1.5, which was defined as hostile iliac artery.9,10
EVAR Procedure
All patients were operated with locoregional anesthesia in a conventional for open repair operational theater, equipped with a digital mobile C-arm with vascular application abilities (BCA-plus, BMI Biomedical International, Curno, BG, Italy). In the absence of data favoring the percutaneous access and according to the surgeon’s preference, we used femoral cut-downs in all EVAR cases.11,12 All intraoperative angiograms performed with manual infusion of 15mL iodinated contrast medium with strength 300 mg I/mL. In patients with moderate or severe chronic renal failure (estimated glomerular filtration rate [eGFR] < 45 mL/min/1.73 m²) we reduced the number of intraoperative angiograms in just two. The first was taken with the stent-graft in the preplanned place in the infrarenal aorta, to tracking the renal arteries ostium and iliac bifurcations at the same time. The second angiogram was taken at completion of procedure to ensure the absence of graft-related endoleak, migration, or deployment failure. In all cases, the full deployment of bifurcated stent-grafts was followed by balloon formation using the Hercules (Shanghai MicroPort Endovascular MedTech Co., Shanghai, PRC) balloon inflation catheter. Perioperative medication included the bolus i.v. administration of 2.500 units of heparin just before the cannulation of the common femoral arteries and all patients received prophylactic administration of i.v. cefoxitin 1 gr at the induction of anesthesia. In postoperative hospital stay, all patients received Cefoxitin 1gr bid and thromboprophylaxis with tinzaparin (50 IU anti-Xa/Kg). The body temperature and vital signs were recorded every 3 h, at least for the first postoperative day, and regularly thereafter during hospital stay. A plain abdominal roentgenogram was performed in first postoperative day to confirm the correct positioning, the configuration of main body and limbs, and the metal skeleton integrity. During the patient’s hospital stay, a routine blood sample analysis was performed daily, with the addition of high sensitivity CRP, for the proper recognition of potential occurrence of even oligosymptomatic postimplantation syndrome (PIS). 13 If PIS was not diagnosed or suspected the patients were discharged the first postoperative day or when the PIS was clinically ameliorated. Furthermore, all patients were set in long-life single antiplatelet medication, unless it was indicated otherwise.
Patients followed a common strict follow-up protocol, with clinical evaluation and color duplex ultrasound (cDUS) every 6 months. Scheduled CTAs included a scan at first postoperative month to confirm the 30-day technical and clinical success as well as annually thereafter. Interim CTAs beyond the scheduled follow-up were performed in any case of clinical or cDUS evidence of potential adverse outcome.
Stent-Graft System Technical Presentation
Minos is a new generation, independently developed, abdominal aortic stent graft system and according to its instructions of use (IFU) is offered for EVAR case with PN length no less than 15 mm. It has a hydrophilic delivery system with ultra-low (14F–16F) outer sheath profile, enabling the ability to perform EVAR in patients with vascular access stenosis and tortuosity. Minos system features a three-piece design that enables in situ customization, allowing the intraoperative adjustment of the total stent-graft length from the lowest renal to iliac bifurcation, through a fluctuating stent overlapping by 25–35 mm or 25–55 mm, for the contralateral and ipsilateral limbs, respectively (Figure 1A). It has a suprarenal fixation with 8 laser-cut M-bare stents and integrated barbs (Figure 1B). The main body is offered with proximal diameter up to 34 mm, to keep the outer sheath’s profile low. For cases with wide neck there is available a proximal cuff extension with diameter up to 36 mm, but with an inevitable increase of outer sheath profile at 18Fr. The endoskeleton of main body is constructed by multinitinol wire braided struts and is covered with seamless woven graft. The iliac limbs are made by single wire in helical spiraling pattern nitinol stents and the same woven graft. The iliac limbs offer distal diameter in a range from 10 to 24 mm and in lengths up to 140 mm, allowing the completion of EVAR with avoidance of redundant iliac extensions. In the field of radiopaque markers, the main difference with the other similar stent-graft platforms is the presence of a marker with the shape of 7 at the level of main body’s bifurcation, in the outer line of the contralateral limb, that corresponds to the frontal axis of the stent-graft. During the stent-graft’s deployment, the 7-shaped marker should be depicted as I and, in that case, the contralateral stamp will be deployed as usual in conventional EVAR, at the frontal plane. The unwanted twisting or the intentional rotation of stent-graft and depiction of a 7 corresponds to the deployment of the contralateral stamp in an anterior sagittal position, while the depiction of the marker as the Greek capital letter gamma (Γ) means that the stamp will be deployed in a posterior sagittal position (Figure 2). The process of graft’s deployment has two steps, a clockwise rotational mechanism for the main body’s liberation from sheath and a separate contra-clockwise rotational mechanism for the final deployment of the suprarenal fixation after a potential downward repositioning of the graft, if it is required (Figure 1C).
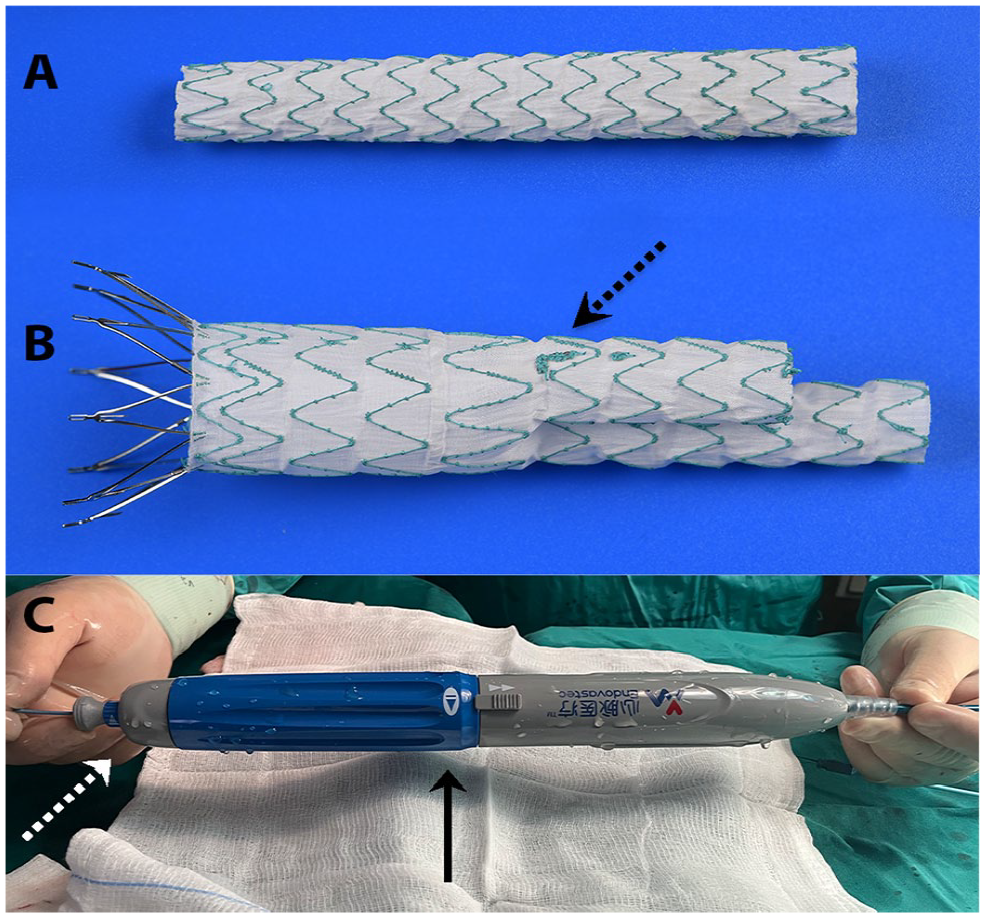
Minos technical characteristics. (A) contralateral limb, (B) Main body, the dotted black arrow shows the radiopaque marker in shape of 7, (C) The deployment device, the black arrow shows the clockwise rotational mechanism for the main body’s liberation from sheath and the white dotted arrow the separate contra-clockwise mechanism for the final deployment of the suprarenal fixation.
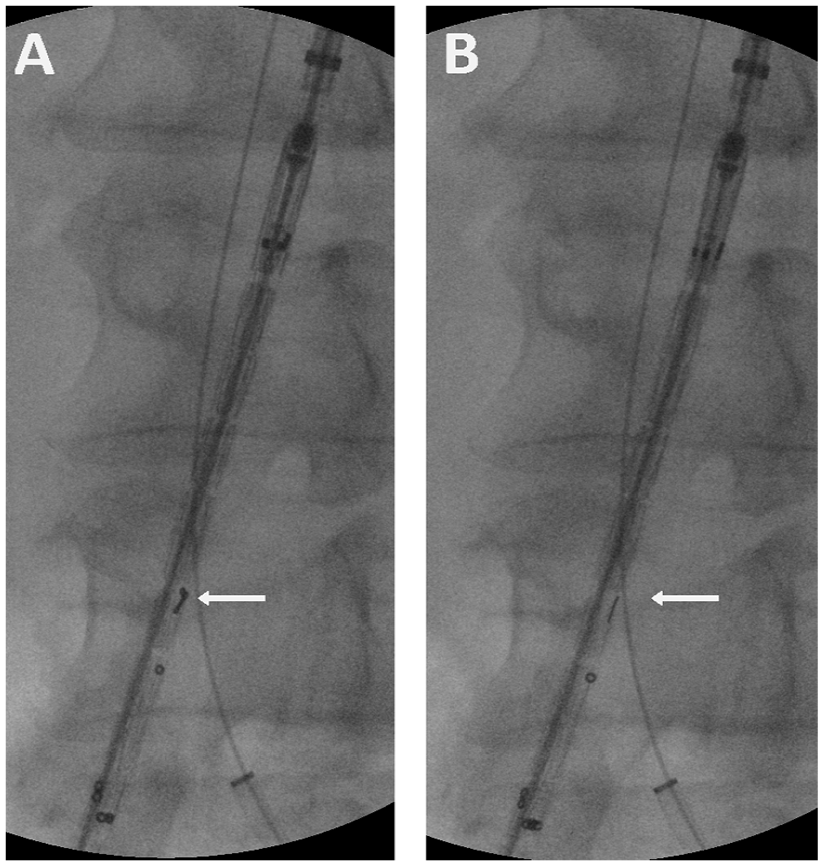
Intraoperative C-arm images showing the orientation of Minos stent-graft before the deployment. The arrows show the 7-shaped radiopaque marker. (A) The marker is depicted as 7 and deployment in this orientation would result in the contralateral limb stump’s position in an anterior sagittal plane. (B) The marker is depicted as I and this corresponds with deployment of the contralateral limb stump in the predesigned left frontal plane.
Statistical Analysis
All data were entered into an electronic database, and the statistical analysis was performed using the IBM SPSS Statistics program—version 27.0 for iOS (IBM, NY, USA). The significance level of the performed tests was defined at p<.050. Categorical variables are presented as counts and percentages. Continuous variables that followed the normal distribution are presented as mean ± standard deviation, whereas those which did not follow the normal distribution are presented as median and interquartile range. Assessment of difference in preoperative vs postoperative AAA’s maximum sac diameter was based on paired two-tailed Student t-test.
Results
Within the study’s time frame, a total of 110 elective conventional EVAR procedures were performed and in 41 of those cases we used the Minos stent-graft. The single selection criterion for use of Minos stent-graft was the availability of this stent-graft in our hospital. The exclusion criteria, which were particularly applicable to the inclusion in the Minos cohort, were the diagnosis of malignancy, autoimmune, inflammatory, or of other severe systematic diseases that could limit the survival or exogenously affect the prospectively collection of clinical outcomes of the studied endograft.
The mean age of 41 included patients was 72.2±8.5, and 37 (90.2%) of them were male in gender. Table 1 shows the baseline anatomical morphological and perioperative data of patients. In 22 (53.6%) patients, the PN’s length was <15 mm and/or the angulation was >60°. These patients were out of the MinosTM IFU but were within the limits of our conventional EVAR protocol as well within the usual clinical praxis. Furthermore, in 9 (22.0%) patients, the iliac angulation was >60° and in 6 (14.6%) both the PN and iliac angulation were >60°. The rate of hostile iliac arteries was 26.8% (n = 11).
Preoperative Anatomical Morphological and Perioperative Data.
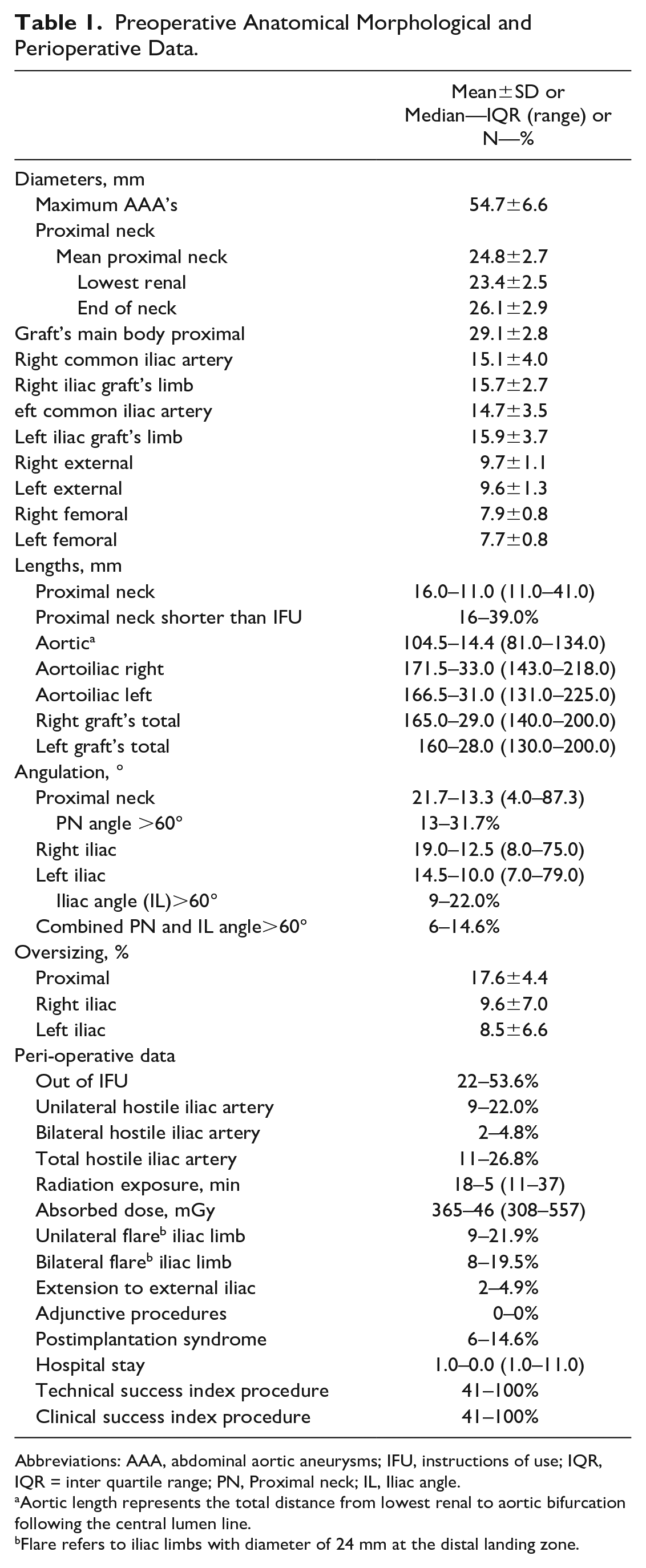
Abbreviations: AAA, abdominal aortic aneurysms; IFU, instructions of use; IQR, IQR = inter quartile range; PN, Proximal neck; IL, Iliac angle.
Aortic length represents the total distance from lowest renal to aortic bifurcation following the central lumen line.
Flare refers to iliac limbs with diameter of 24 mm at the distal landing zone.
The median radiation exposure time and absorbed dosage were 18΄–5΄ and 365–249 mGy, respectively. The PTS of index procedures and at 30-day follow-up was 100%. In 2 (4.9%) patients, the right iliac bifurcation diameter was 28 mm, and we extended the distal landing zone at the external iliac using tapered iliac limbs with nominal distal diameter at 13 mm. Additionally, we used the bell-bottom technique, with unilateral or bilateral flare design of iliac limbs, in 9 (21.9%) and 8 (19.5%) patients, respectively. In one patient with hostile left iliac artery, we intentionally deployed the contralateral limb in ballerina (ballet-type) configuration (Figure 3). No contralateral limb misplacement was noted in any of our cases. No adjunctive endovascular or open techniques were performed at index procedures.
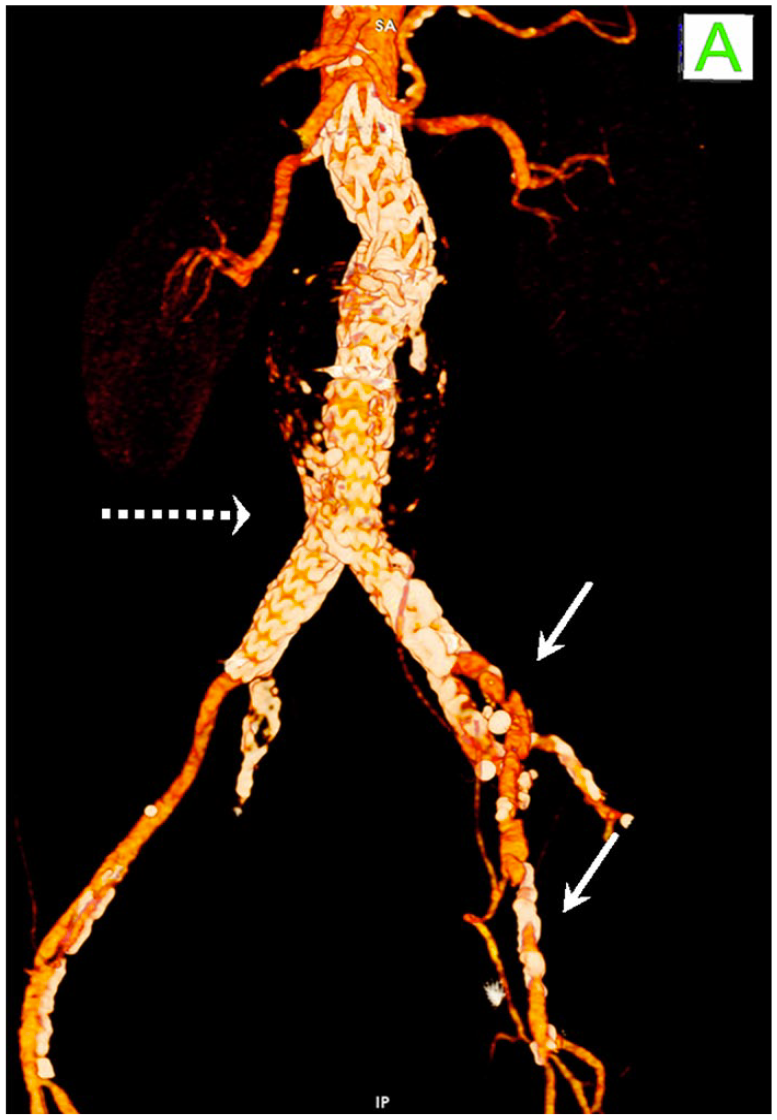
Anterior computer tomography angiogram (CTA) view of implanted Minos stent-graft in 3D reconstruction. White block arrows show the narrow and heavily calcified left iliac artery and the white dotted arrow shows the ballerina (ballet-type) configuration of the bifurcated endograft.
The median hospital stay was 1 day (range: 1–11 days). One patient presented acute cardiac ischemia and required a prolonged (for a total of 11 days) hospital stay in coronary heart disease unit. Two patients (4.9%) experienced urinary tract infection and 6 (14.6%) of them developed a mild and self-limited postimplantation syndrome. No mortality was recorded in index procedures as well as during the 30-day period. PTS remained 100% at 30-day follow-up and the same rate was observed in 30-day clinical success. CTA at first month revealed 3 (7.3%) cases with type II endoleak, which remained visible during the entire follow-up and with stable AAA’s sac diameter so far.
The median clinical follow-up (Table 2) was 12.4–12.0 months, and the respective CTA follow-up was 12.0–15.0 months. No graft-related endoleak was recorded during follow-up. Conversely, in one patient, a previously nonvisible type II endoleak was detected at 6th month’s cDUS and confirmed by CTA, with no increase in AAA’s sac diameter. No graft-related morbidity, mortality or reintervention was recorded during follow-up. Finally, one patient was diagnosed at 6 months with in situ gastric adenocarcinoma that was missed in the initial clinical evaluation. He underwent open upper gastrointestinal surgery and died from cardiac infarction in the perioperative period.
Outcomes.

Abbreviations: CTA, computer tomography angiogram; IFU, instructions of use.
Difference represents the difference between the preoperative and postoperative maximum AAA’s sac diameter. The p value of paired Student t-test between preoperative and postoperative maximum AAA’s diameter was <.001.
Definition of stable sac included all differences within the ±2.5 mm range.
The mean maximum AAA’s diameter at last CTA follow-up was 50.8±7.2 mm, and the comparison with respective preoperative values revealed that the difference was high statistically significant (p<.001, Table II). In 27 (65.9%) patients, the maximum AAA’s diameter showed decrease ≤2.5 mm and was considered as stable, while in 14 (34.1%), the absolute decrease was ≥ 2.5 mm. Finally in 6 (14.6%) patients, the sac AAA’s sac regression was important (≥5 mm). Notably, an increase in sac diameter was not recorded in any of our patients so far.
Discussion
In Greek, mythology Minos was the son of Europe and Zeus. In contemporary endovascular surgery of AAAs, Minos is a new in European market bifurcated abdominal aortic stent-graft, attempting to take his place in treatment options of AAAs. Assessment of a relatively new in market stent-graft should answer in the following queries. The first aspect is the technical feasibility and safety of the index procedure. The second is the technical and most importantly the clinical success in 30-day follow-up. The third and ultimate issue remains the procedure’s clinical success in the mid and long-term follow-up. Our preliminary results of using Minos showed that it performed excellent and showed high levels of safety, feasibility as well of durability in the first-year follow-up.
The PTS of index procedure achieved in all 41 cases and adjunctive endovascular or open techniques were avoided. The hydrophilic and low-profile outer sheath allowed the uncomplicated passage of stent-grafts even in hostile iliac vessels with concomitant tortuosity and narrowing. Minos is a trimodular endograft with the advantage that the availability of long limbs precludes or reduces the cost of procedures with avoidance of using extra limb extensions. In our experience, the use of limb extensions was not required even in cases with extremely long aortoiliac length or with extension to the external iliac artery. On the other hand, for cases with wide (>30 mm) PN, the offered proximal graft’s diameter is limited to 34 mm, which may be not enough, and implantation of a 36 mm proximal cuff might be necessary for granting the proximal sealing. In our experience, we noticed that the presence of the 7-shaped radiopaque marker along with the several ordinary radiopaque markers facilitated the proper orientation of the main body and the alignment of the contralateral limb stamp to the desired and predesigned position, with obvious benefits in reducing the radiation exposure time. Furthermore, the stent-graft’s device showed precise and accurate deployment, even in cases with short and angulated PN, as well in more than half of our patients that were considered out of IFU. Notably, the incidence of postimplantation syndrome in our series was 14.6%, which in our experience lies at the lower bound of range of PIS’s incidence compared with the other similar platforms of contemporary trimodular endografts, constructed by nitinol wires and woven polyester liner. 13
Several experimental studies have shown that the PN’s and iliac limbs’ angulation >60° and large diameters have been associated with higher displacement forces and in greater predisposition to migration.9,10,14 Twenty-two of our cases were considered out of IFU according to the PN morphology and in 19 cases the iliac landing zones were aneurysmatic, thus were prone to migrations and type I or III endoleaks. 15 Despite these marginally hostile anatomical conditions, the excellent performance of Minos at index procedures was followed by great 30-day technical and clinical success rates, which maintained at the highest level of 100% during the entire (1-year, median) follow-up. Definition of hostile PN anatomy varies significantly among studies as well in IFUs and in reported EVAR experience with other endografts. However, in real world of daily praxis, the clinical demands often push in lowering the limits of PN length and in increasing the acceptable neck’s diameter and angulation. In our protocol, although we expanded the inclusion criteria out of the specific graft’s IFUs, we excluded the AAAs with PN length <10 mm. We considered that it would be premature, rush and beyond the ethics of our study to use a new and with no previous experience stent-graft, in these extreme anatomically EVAR cases or in advanced endovascular techniques, such as the chimney or the endostapling.
Another important field in assessment of EVAR outcomes is the aneurysm’s sac regression. 16 Despite the short duration of follow-up in our series, we recorded a regression in more than a third of our cases, whereas the rest of AAAs remained stable. With these data, we consider that preliminary outcomes of Minos are promising so far. However, mirroring the previous experience with other trimodular last-generation stent-grafts it would be important to explore and determine the long-term performance of this stent-graft in both, inside the IFU and in hostile PN conditions as well in pararenal aortic pathologies and in the setting of advanced endovascular techniques.17,18
Main limitation of our study remains the lack of randomization of included in our series patients, which implies a potential selection bias. Additional issues are the small number of patients and duration of follow-up. However, the purpose of this preliminary study was to explore and to present the performance of a new in market stent-graft and we believe that these aims were achieved with as much objectivity as it was feasible in the real world of daily clinical demands. Additionally, although half of the included in our cohort AAAs had challenging and severely angulated necks as well hostile iliac anatomy, the small number of patients prohibits the generalization of our impressive results in such anatomies. It is obvious that larger clinical studies with longer follow-up are needed to evaluate satisfactory the clinical performance of this new in-market endograft.
Conclusions
The Minos abdominal aortic stent-graft presented easy navigation even through hostile iliac vessels and provided precise deployment, optimal proximal fixation, and powerful sealing even in short and angulated necks. The results of this preliminary experience in standard EVAR, within as well out of the stent-graft’s IFU, are promising with excellent feasibility, safety, and effectiveness at first year. The overall clinical performance appeared to be in very high standards. However, the long-term performance of this new in-market endograft merits further evaluation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
