Abstract
Purpose:
To report a rare case of acute renal vein thrombosis (RVT) that was treated with endovascular thrombectomy and lysis, and discuss potential etiology and indications for catheter-directed management.
Case report:
A 21-year-old female athlete presented with sudden pain in her left flank and vomiting. A 3-phase computed tomography (CT) angiogram identified total occlusion of the left renal vein with no excretion from the swollen tender left kidney. Catheter-directed thrombolysis and thrombectomy were initiated 24 hours after onset of symptoms. Complete resolution of the RVT with normalized renal function was achieved. Post-operative Doppler ultrasound scan confirmed normal renal resistance and flow in the renal vein. The patient was discharged on Apixaban and remains well at 6 months. Combined hormonal contraception via an intra-vaginal ring and raised Factor VIII activity were the only identified risk factors.
Conclusion:
Acute complete RVT with impaired kidney function is rare. Combined hormonal contraception and increased Factor VIII activity were potential risk factors. Endovascular thrombectomy and lysis restored renal perfusion and function, and can be used effectively in the management of fit patients with acutely compromised kidney function from total renal vein obstruction.
Keywords
Introduction
Renal vein thrombosis (RVT) is rare. The incidence of both acute and chronic RVT have yet to be established. Acute manifestations typically include flank pain, nausea, hematuria, and fever. 1 Owing to its rarity, prompt diagnosis of acute RVT remains a challenge particularly in previously clinically healthy individuals without predisposing factors such as nephrotic syndrome or known coagulopathy. We present a case of acute RVT with complete obstruction of the vein and no renal excretion in a young athlete, that was successfully treated percutaneously with selective endovascular thrombolysis and thrombectomy.
Case Report
A previously healthy 21-year-old female elite swimmer presented a few hours after acute onset of vomiting, with severe pain in her left iliac fossa. She had been unwell for 3 days with dysuria and constipation. She had a history of irregular periods and was on combined hormonal contraception (NuvaRing intra-vaginal ring, Organon Pharma). She had received both doses of the mRNA-127s vaccine (Moderna, USA) 6 months prior to this incident. She was a non-smoker with no family history of thromboembolic disease.
The patient was tender in her left flank, with a urine dipstick positive for hematuria. Her white cell count was 10.7 × 109/L, CRP 91.2 mg/L, and serum creatinine 131 µmol/L. A computed tomography (CT) scan without contrast demonstrated a grossly oedematous left kidney with perirenal fat stranding and free fluid tracking along the perirenal space down to the left iliac fossa. The left renal vein was abnormally distended and hyperattenuating, suggesting left RVT. A subsequent contrast-enhanced 3-phase CT angiogram showed normal renal arteries but delayed left parenchymal enhancement and no excretion of urine (Figure 1). The distended left renal vein did not opacify, confirming the suspicion of total venous obstruction (Figure 2). The thrombus did not extend into the inferior vena cava (IVC). The right kidney was entirely normal. The patient was started on heparin infusion.
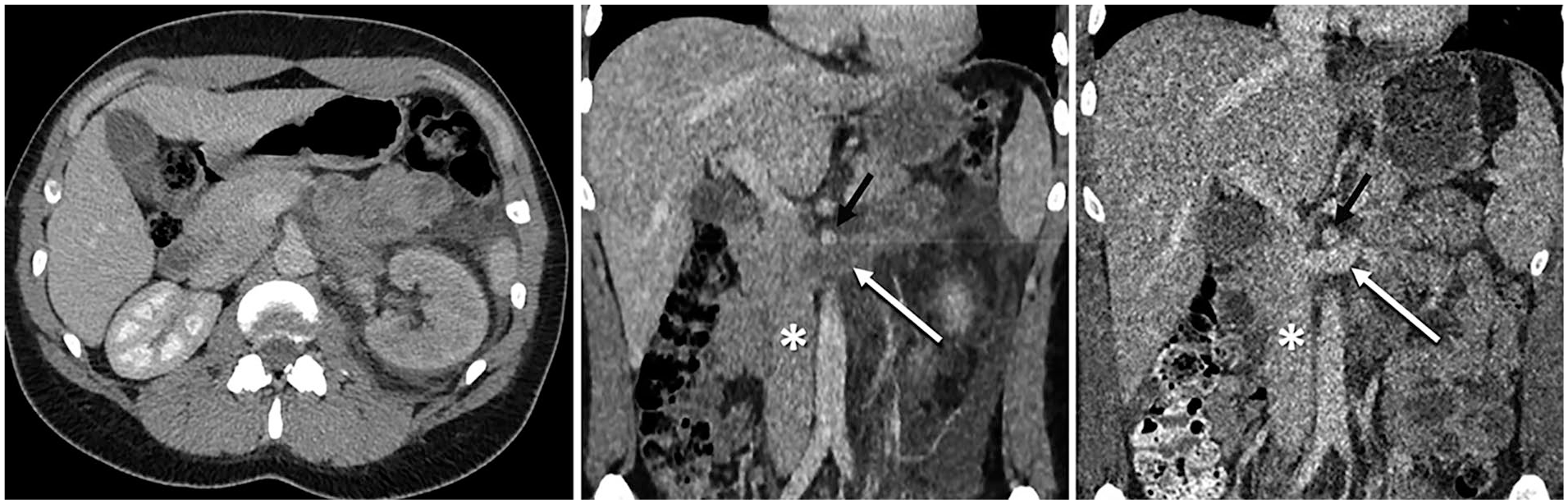
Venous phase computed tomography (CT) angiographies. Pre-operatively (left and center), the left kidney was poorly opacified and oedematous with perirenal fat stranding and total obstruction of the renal vein (white arrow). Post-operatively (right), the left renal vein is fully opacified. The inferior vena cava (*) and super mesenteric artery (black arrow) are marked.
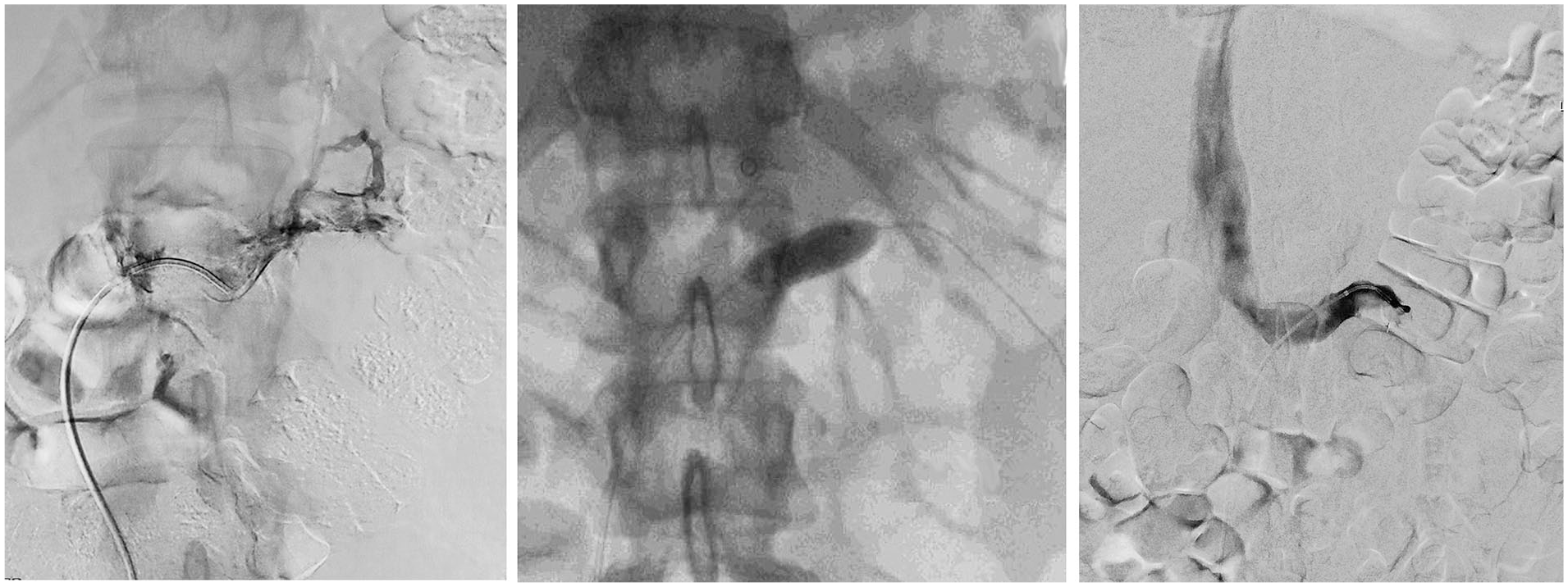
Initial venogram (left) demonstrates complete occlusion of the left renal vein. A 12 mm percutaneous transluminal angioplasty (PTA) balloon (center) was used for a Fogarty thrombectomy. Completion venogram (right) shows opacified left renal vein with flow into the caval vein.
In view of complete venous obstruction with progressive pain and acute kidney injury, the patient was offered an attempt to recanalize the renal vein endovascularly. After 24 hours of the onset of her symptoms, a 4 F catheter was gently negotiated through the fully thrombosed left renal vein into the hilus of the kidney from a percutaneous femoral approach. The left renal vein was completely thrombosed and no significant outflow by any collateral veins could be observed either. Infusion of recombinant tissue plasminogen activator (rTPA) was initiated via the catheter at 1 mg/h and continued overnight. A lysis check at 18 hours showed partial recanalization of the renal vein with restoration of some flow. Massive thrombi persisted in the vein, and the patient was still in pain. A Fogarty maneuver was therefore carried out with a 12 × 40 mm2 balloon catheter over the wire (Figure 2). A subsequent completion venography demonstrated brisk venous flow and the patient reported immediate pain relief. However, significant thrombi still persisted and to avoid re-thrombosis, local lysis was continued for another 24 hours. A final completion venogram showed complete resolution of the RVT with brisk flow into the IVC. No predisposing stenosis or external compression of the vein was observed. Clinically, the patient became completely pain-free and polyuric, suggesting recovery of the left kidney. She was hydrated on IV fluids and immediately anticoagulated. Repeat 3-phase CT angio on the following day showed opacification of the left renal vein, with only minor thrombi remaining in the renal hilum. Reconstructions of the CT scan did not suggest compression of the renal vein, such as in nutcracker syndrome. A Doppler ultrasound scan 3 days later confirmed normal renal resistance as well as unobstructed flow in the left renal vein. Extensive coagulation, renal, hepatic, and rheumatologic screens proved unremarkable initially, but subsequently increased Factor VIII activity was identified when screening for less common thrombophilia. The patient was discharged on Apixaban 5 mg twice daily. Her hormonal contraceptive therapy was discontinued. She remains asymptomatic at 6 months with normal renal function. She is back to swimming.
Discussion
To the best of our knowledge, this is the first report of acute RVT associated with the intra-vaginal ring, NuvaRing. It illustrates a potential thromboembolic risk even with non-oral combined hormonal contraceptives (NOCHCs), particularly in patients with unrecognized concomitant coagulopathies, such as the increased Factor VIII activity in our patient.
The use of the combined oral contraceptive pill (COCP) is associated with a 2- to 4-fold increase in venous thromboembolism (VTE) risk, but is rarely the sole precipitant of acute RVT. The risk of VTE associated with NOCHC, such as NuvaRing, is less known.2 –6
Increased Factor VIII activity is independently associated with an increased risk of recurrent VTE in a dose-dependent manner. 7 Cases of RVT in women using the COCP, with underlying raised Factor VIII activity, have previously been documented. 8 Our report highlights a potentially similar risk for such patients using NOCHC alternatives. However, to date, increased Factor VIII alone has not been shown to be associated with acute RVT without other predisposing risk factors.
Coagulopathies which are more commonly associated with acute RVT include Protein C and S deficiencies. Other common risk factors for acute RVT include nephrotic syndrome in membranous glomerulonephritis, transplant malfunction or anastomotic stenosis post-renal transplantation, renal malignancy, and other mechanical factors such as compression of the left renal vein by the superior mesenteric artery, as described in the Nutcracker syndrome.1,9
Given the widespread use of combined hormonal contraception, pre-prescription blood tests for coagulopathies may not be feasible. However, in previously healthy women presenting with rare VTEs with combined hormonal contraception being the only identified risk factor, it may be beneficial to offer screening for common and rare coagulopathies to manage the risk of future VTE events.
Owing to its rare occurrence, best management of acute RVT remains undetermined. Currently, systemic anticoagulation and catheter-directed endovascular approaches form the 2 main stems of treatment. Systemic anticoagulation is often preferred due to its less invasive nature, and this has proven useful in symptom-relief and restoration of renal function in cases of acute RVT. 9 However, the degree of venous obstruction in these cases has not been well-described, and few reports offer follow-up imaging to ascertain vessel patency.10,11
Endovascular treatment involves percutaneous catheter-directed thrombolysis, thrombectomy, or a combination of both. It is advocated in select cases where rapid clot resolution is key, due to complete renal vein obstruction with manifestations of acute kidney injury. 12 Preferably, a dedicated over-the-wire thrombectomy catheter would have been used. These balloons are compliant and shorter. In the absence of such a catheter, the ordinary percutaneous transluminal angioplasty (PTA) balloon catheter also proved successful. Potentially, a dedicated thrombectomy device would have been used, rather than extracting the thrombi into the caval vein. We felt, however, that such existing devices of sufficient caliber may be too stiff to navigate safely the curve into the renal vein. This is partly because the renal vein is very short, which makes it impossible to advance the guidewire sufficiently into the kidney, to provide firm support for such a stiff device. Furthermore, we felt that the volume of the fragmented thrombus that was extracted from the short renal vein into the caval vein was too small to cause a potentially significant pulmonary embolus. There is extensive experience of similar maneuvers for thrombosed dialysis access shunts in the upper extremity. For this reason, we also refrained from using a suprarenal caval filter which in itself is a potentially dangerous maneuver with a risk for dislocation of the filter. 13
Our patient was a good candidate for endovascular treatment, given her relative low risk for treatment and the emergent state of her left kidney, which was compromised by complete obstruction of venous outflow. Patency of the left renal vein was swiftly restored with catheter-directed thrombectomy and thrombolysis. Subsequent patency was confirmed at follow-up CT venography and renal Doppler ultrasound. Successful endovascular management of acute RVT involving complete occlusion has also been documented in patients with pre-existing nephropathy. Kim et al 12 reported 6 cases of acute RVT with total occlusion, 5 of which were treated with catheter-directed thrombolysis and thrombectomy and 1 with catheter-directed thrombectomy only. Venography demonstrated patency of the thrombosed veins in all cases, following a mean duration of treatment of 22.1 (±21.0) hours, and return of baseline renal function at a mean of 6.67 days after percutaneous therapy. There were no relevant adverse events. 12
In view of the relatively invasive nature of endovascular treatment, systemic anticoagulation remains first-line therapy in partial venous obstruction that does not appear to jeopardize kidney function. 9 There are a few considerations to be made with regard to an endovascular approach. The left renal vein typically has multiple collaterals and can safely be ligated in open aortic aneurysm repair, as long as the ligation is medial to the lumbar and gonadal veins. The endovascular approach appears favorable in total obstruction of the renal vein with poor collateral runoff. 12 It has also been advocated in partial occlusion refractory to systemic anticoagulation, 14 and in patients with contraindications to long-term anticoagulation. 15 Endovascular treatment offers additional therapeutic options such as venoplasty with stenting, such as in patients with the Nutcracker syndrome, and insertion of an IVC filter in patients at risk of massive pulmonary emboli.12,15 To date, however, there has been no formal comparison between systemic anticoagulation alone and endovascular treatment of acute RVT.
Conclusion
This is the first report of NuvaRing-associated acute RVT with total obstruction of the left kidney, in an extremely fit patient with previously unrecognized increased Factor VIII activity. Rapid recanalization of the completely thrombosed left renal vein and subsequent resolution of symptoms was achieved with catheter-directed thrombectomy and thrombolysis. The patient is well, with normal renal function at 6 months. Further evaluation of the risk and benefits of endovascular venous thrombectomy of RVT versus systemic anticoagulation alone is warranted. Until such information becomes available, we propose the use of catheter-directed thrombolysis and/or thrombectomy in fit patients with acutely compromised kidney function from total renal vein obstruction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
