Abstract
Purpose:
Radiation exposure for vascular interventionalists is still a concern. The aim of this study was to assess the value of advanced imaging guidance on radiation exposure and iodinated contrast volume during endovascular treatment of lower extremity arterial disease (LEAD).
Materials and Methods:
It was a prospective, randomized, monocentric, pilot, single-operator study, conducted from June 2018 to October 2019. Consecutive patients requiring a preoperative computed tomography angiography (CTA) for a symptomatic LEAD and scheduled for an iliac and/or femoropopliteal endovascular repair in a hybrid room were included. Patients were randomly assigned to the use of fusion imaging guidance (Vessel Navigator®, Philips) or not. The primary endpoint was the dose area product (DAP, Gy.cm²). Secondary endpoints were DAP for fluoroscopy, DAP for fluorography, Air Kerma, fluoroscopy time, volume of contrast, and number of digital subtraction angiography (DSA). Data were expressed in median [Q1-Q3].
Results:
In all, 64 of the 77 patients enrolled (34 in fusion group, 30 in control group, 82% men, 65.8 years [61–71]) were included. Groups were similar in terms of comorbidities, BMI (26 kg/cm2 [24–28]), but lesion location were not equally distributed (p=0.004). There was no significant difference between the groups regarding DAP (31.6 Gy.cm2 [23.4; 46.9] for fusion group vs 25.6[16.9; 34.0] Gy.cm2; p=0.07), Air Kerma (160 mGy [96;3365] vs 115 mGy [76;201]; p=0.12, fluoroscopy time (560 seconds [326;960] vs 454 seconds [228;1022]; p=0.44), contrast volume (60 ml [42;80] vs 50 ml [40;66]; p=0.10), or operative time (68 minutes [55;90] vs 46 minutes [30;80]; p=0.06). The median number of DSA was 14 [10–18] in the fusion group versus 11 [6–18]; p=0.049.
Conclusion:
Fusion imaging guidance does not affect radiation exposure and contrast volume during endovascular revascularisation of iliac and femoropopliteal occlusive disease in a hybrid room environment.
Introduction
Lower extremity arterial disease (LEAD) is a worldwide burden disease, with atherosclerosis as a leading cause. When supervised exercise therapy is not sufficient in symptomatic LEAD, endovascular revascularisation is performed as first line therapy in most of the cases. 1 However, the well-known drawback of this minimally invasive procedure using X-ray guidance, is the exposure of both patient and staff to radiation and patient’s exposure to nephrotoxic contrast.
Reducing both in order to decrease their respective toxicities must be a priority for the vascular interventionalists. As low as reasonably achievable (ALARA) principles and several studies, including guidelines, state basic safety standards for protection against X-rays and iodinated contrast exposure.2,3
One other way to reduce both radiation and contrast use is to develop alternative advanced imaging systems to X-rays, and to improve surgeon vision using 3D images. Echography, including transoesophageal echocardiography (TEE), intravascular ultrasound (IVUS), and contrast enhanced ultrasound (CEUS) are used to facilitate repair and assist endograft placement, as well as the Fiber Optic Realshape Technology (FORS) to assist the navigation. 4
Currently, the most common adjunctive technique is fusion imaging guidance. It allows overlay of a 3D version of the vascular mask from a preoperative computed tomography angiography (CTA) onto the live X-ray image creating a “3D roadmap.” 5 It has been proven that using fusion imaging guidance during endovascular aneurysm repair (EVAR) reduces both contrast and radiation dose,6 –8 especially if the registration protocol is contrast and almost radiation free 9 and during complex aneurysm repairs. 10
However, the literature is scarce about the impact of using fusion imaging guidance on radiation dose and contrast use, during LEAD endovascular procedures. The aim of this trial was to assess the impact of image fusion guidance during LEAD endovascular revascularisation on patient radiation exposure and iodinated contrast agent administration.
Materials and Methods
Study Design
The Safety Of Fusion (SOFT) Trial was an open pilot, single center, randomized controlled trial, conducted in accordance with the ICH-E6, French Good Clinical Practice guidelines, and appropriate regulatory requirements. The ethical committee approved the study for France (CPP Sud Ouest et Outre Mer III- Nantes 2018/19). Participation required informed consent, which was retrieved on the day before the procedure and compliance with the study inclusion and exclusion criteria. The registration number of the study on ClinicalTrials.gov was NCT03546881.
Patients
From June 2018 to October 2019, we prospectively screened for inclusion all consecutive patients who presented symptomatic LEAD, in a single university hospital. After informed written consent, we included all adults (age>18 years) patients with symptomatic LEAD (Rutherford stage 2 to 6) related to iliac and/or femoropopliteal atherosclerotic lesions, eligible for an endovascular repair and who had had a preoperative diagnostic Computerized Axial Tomography (CTA, required for the fusion mask). A preoperative CTA was required only in patients with aortoiliac lesions or expansive lower limb atherosclerosis, to optimize revascularisation strategy and anticipate stents’ sizing.
Exclusion criteria were females under the age of 50 years old (to avoid unknown pregnancy), patients requiring an associated procedure (below the knee or visceral revascularisation), patients with femoropopliteal stenosis on duplex examination who did not require a preoperative CTA and emergency surgery.
Randomization and Procedures
Patients were randomly assigned in a 1:1 ratio to the fusion group or control group with the use of a web-based system on the day of the procedure. Due to the nature of the intervention, the operator was aware of study-group assignments. Patients were scheduled for an elective procedure in the hybrid room (Allura FlexMove Clarity®; Philips®) with a single operator (YG). The leading surgeon was qualified to the Philips fusion technique. Procedures were performed under fluoroscopic guidance with a 48 cm × 30 cm flat panel detector. Low-dose settings were set by default with a 7.5 image frame rate. In order to assess minimum radiation exposure, the leading operator applied the ALARA principles, minimizing fluoroscopy time, maximizing collimation, and limiting steep angulations. As standard of care in our institution, procedures were carried out under sedation (remifentanil) and local anesthesia. The patient’ legs were fixated in both groups to maintain accuracy of the fusion or roadmap imaging. As contrast agent, Xenetix 300 mg I/mL was used, except in case of chronic kidney disease (stage III or IV) where it was replaced by Visipaque 320 mgI/ml. The revascularisation technique and stent implantation were at the operator’s discretion.
If patients were randomized in the fusion group, before each procedure a bone and a vascular 3D model were reconstructed from the preoperative CTA on a workstation and then fused with live fluoroscopy (VesselNavigator®, Philips). The registration process was performed using bone landmarks visible on 2 fluoroscopic orthogonal shots (anterior-posterior and lateral). During the procedure, this layout was used to locate the area of interest and to identify the critical vessels’ origins. The registration could be refined at any time by the operator. The accuracy of the fusion mask was always checked prior to stent graft deployment by a digital subtraction angiography (DSA runs). Road mapping was used in patients randomized in the control group.
For every patient, if femoropopliteal lesions were diagnosed on the preoperative CTA, an ipsilateral diagnostic angiography bolus chase was performed prior to and after the intervention to obtain an accurate assessment of femoropopliteal and below-the-knee lesions and to verify the technical success of the procedure. Even if radiation consuming, this was justified by the poor quality of CTA to assess accurate vessel patency in below the knee calcified arteries. These images were only for diagnostic purpose and could not be used as a roadmap or to adjust the fusion mask.
Outcomes
Patients were followed-up until the discharge from the operative room. The primary endpoint was the total dose area product (DAP, expressed in Gy.cm²) defined by the entire amount of energy delivered to the patient with the beam. The measure was collected at the end of the procedure. The secondary endpoints included the following criteria about radiation exposure: number of DSA runs, Air Kerma (AK) (mGy), fluoroscopy time (minute), and the DAP related to a bolus chase. Other secondary endpoints were as follows: the volume of iodinated contrast agent volume (ml) and total operative time (minute).
General data protection regulation guidelines were followed and baseline data as patient and procedure characteristics, sex, age, BMI, cardiovascular risk factors, location of the targeted lesions (iliac/femoral/popliteal), TASC classification, access site, technical success, and total number of stents implanted, were collected through an internet-based data collection tool (eCRF).
Statistical Analysis
As a pilot study about fusion use during LEAD, we used the hypothesis that fusion imaging guidance should result in a 20% reduction in radiation exposure compared with the control group, based on other trials studying fusion during EVAR. We calculated that a sample of 88 patients, randomly assigned in a 1:1 ratio, would provide a power of 80% to reject the null hypothesis of no difference in the radiation exposure between the groups at a 2-sided alpha level of 5%. Demographic and co-morbidity data were recorded and compared between the 2 groups. For binary variables, counts, and percentages, 95% confidence intervals were calculated, and p-values presented. Pearson’s chi-squared test or Fisher’s exact test were performed when appropriate. For continuous variables, means and standard deviations or median with interquartile range or minimum and maximum were calculated, depending on distribution. Values were compared between the 2 groups with t-test or Mann-Whitney test. The first principal analysis on radiation exposure was analyzed without adjustment. The effects of DAP and radiation exposure were analyzed through linear regression models correcting for the following confounders: obesity, location lesion, TASC, and diabetes. A p-value of <0.05 was considered statistically significant. Data were analyzed with the SAS packages (SAS Institute Inc version 9.4, Cary, NC).
Results
Population and Procedure Characteristics
From June 2018 to October 2019, 77 patients were enrolled in the trial and 69 underwent randomization: Eight patients were not randomized due to technical reasons (unavailability of the hybrid room) and 5 patients were excluded in postrandomization because of nonrespect of exclusion criteria. Finally, the modified intention to treat population included 64 patients: 34 (53%) patients were assigned to the fusion group and 30 (47%) patients to the control group. The mean age was 65.8 years (SD=10.4), and most of the participants were male (n=53 [83%]) and Rutherford stage 2 to 3 (n=58 [90%]) (Table 1). Lesions length and localization were significantly different between groups: iliac lesion alone, n=3 (8.8%) in the fusion group versus n=10 (33.3%) in the non fusion group,; femoro-popliteal lesions alone, n=15 (44.1%) versus n=16 (53.3%), respectively,; and both iliac and femoro-popliteal lesions, n=16 (47.1%) versus n=4 (13.3%), respectively, p=0.0045. TASC distribution was not different between groups. More stents were implanted in the fusion group (3.2+/-1.6 versus 2.1+/-1.1, p=0.0050). The technical success was similar (94.1% in fusion group and 96.7% in control) (Table 2). One failure in each group was due to femoropopliteal re-entry and the second failure in the fusion group was due to an iliac rupture.
Patients’ Characteristics.
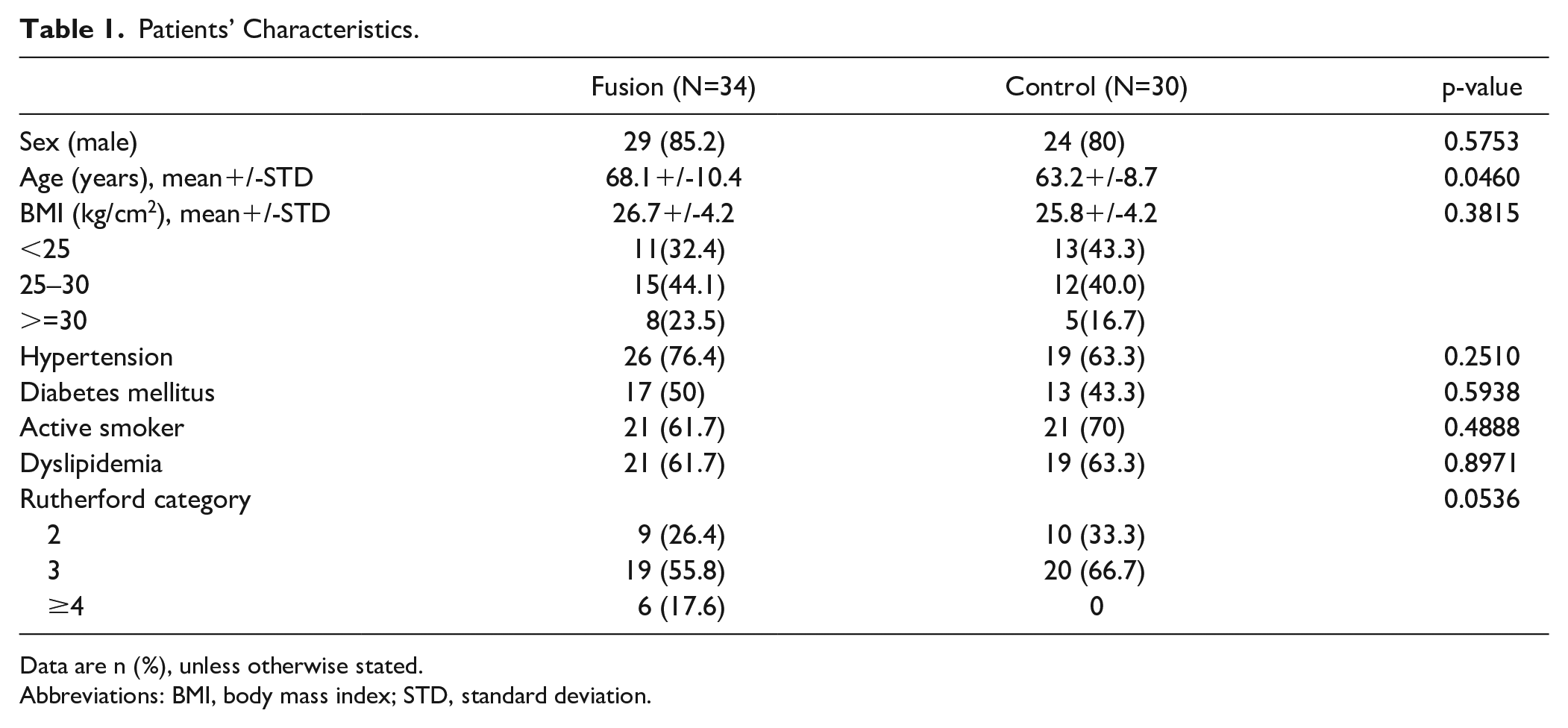
Data are n (%), unless otherwise stated.
Abbreviations: BMI, body mass index; STD, standard deviation.
Intraoperative Data.
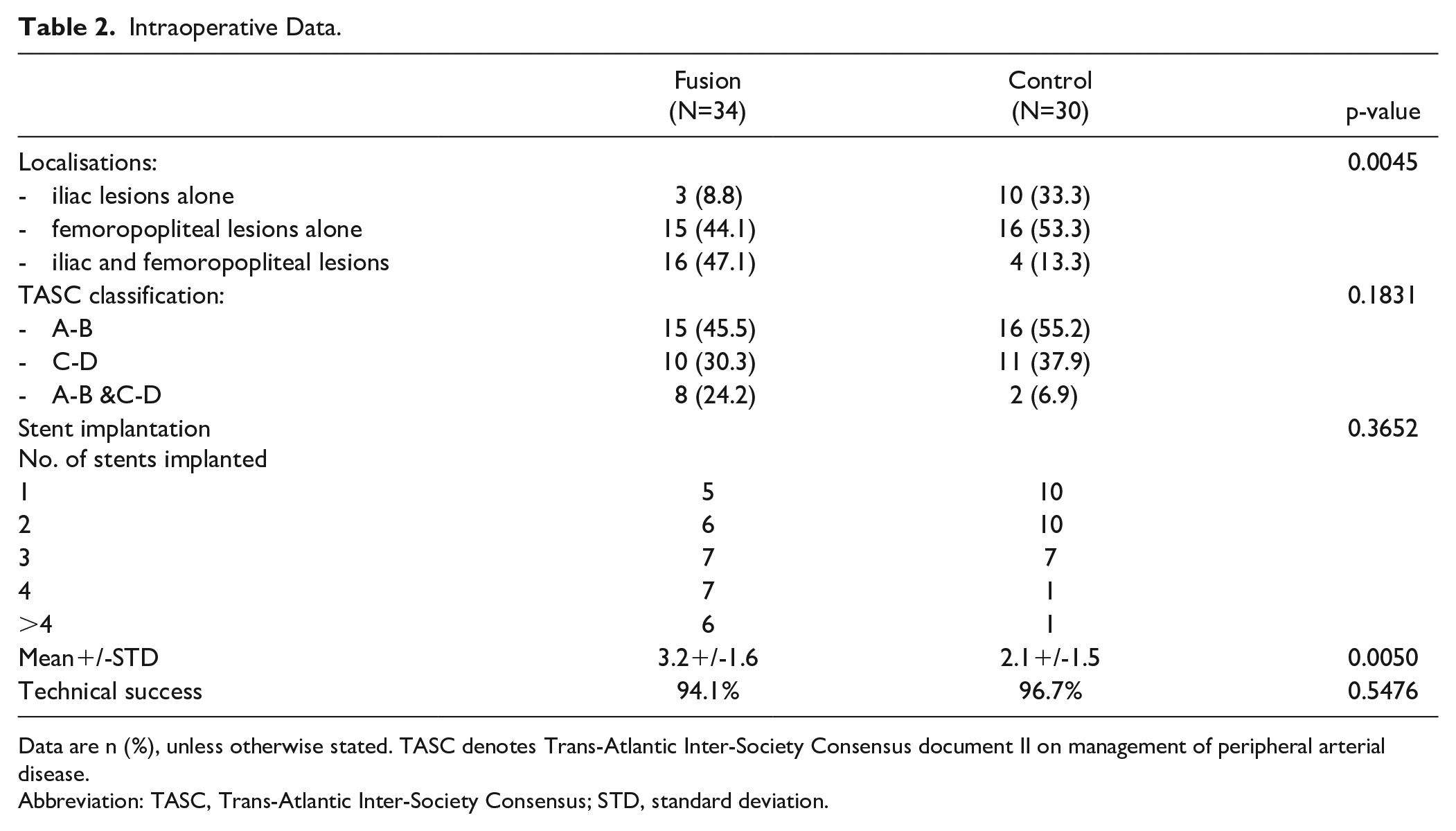
Data are n (%), unless otherwise stated. TASC denotes Trans-Atlantic Inter-Society Consensus document II on management of peripheral arterial disease.
Abbreviation: TASC, Trans-Atlantic Inter-Society Consensus; STD, standard deviation.
Radiation Exposure Parameters
The total median DAP was 31.6 Gy.cm² [23.4; 46.9] in the fusion group, versus 25.6 Gy.cm² [16.9; 34.0] in the control group (p=0.07). Subtracting the exposure associated with the bolus chase, the median DAP of the procedure was 22.1 Gy.cm² [9.3; 32.4] versus 10.5 Gy.cm² [6.1; 29.9], respectively (p=0.11) (Figure 1). The number of DSA per procedure was significantly higher in the fusion group (14 [12; 18] versus 11 [6; 18], p=0.049). Other exposure parameters were similar between the 2 groups (Table 3).
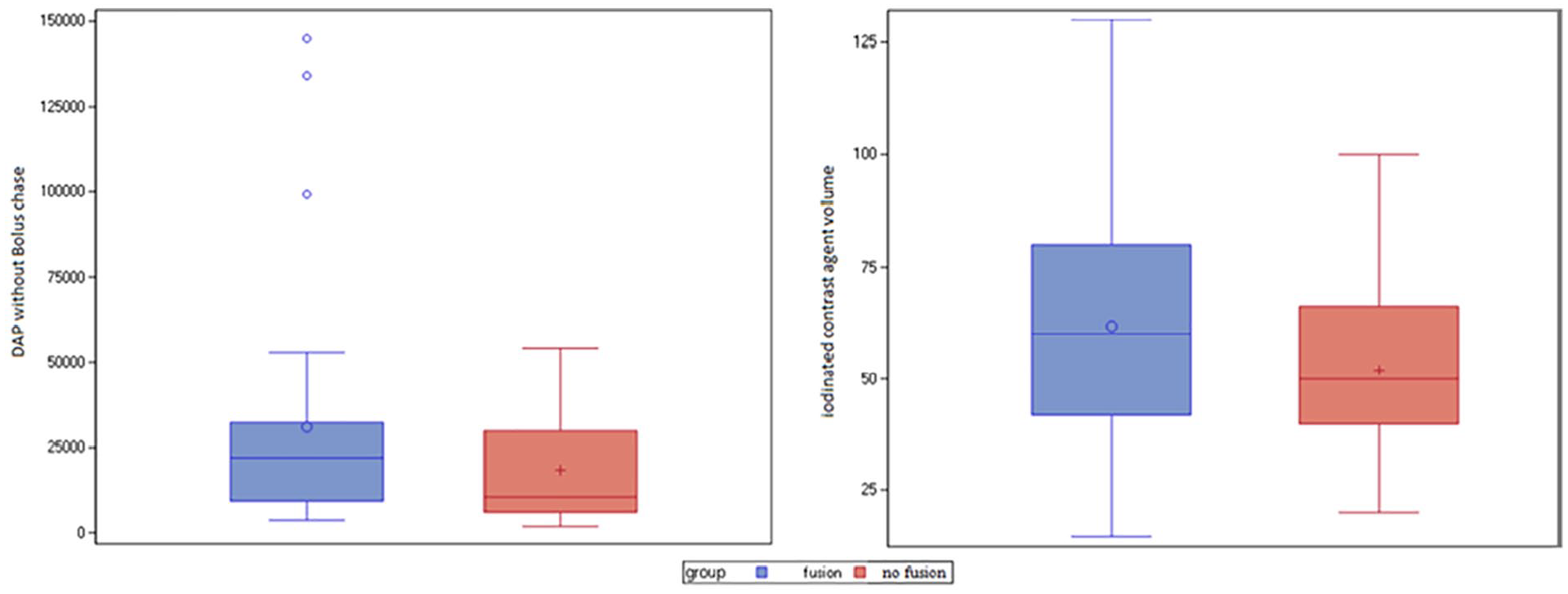
Mean radiation exposure (left) and contrast volume (right) in each group. DAP, dose area product.
Radiation Exposure According to Patients’ Group.
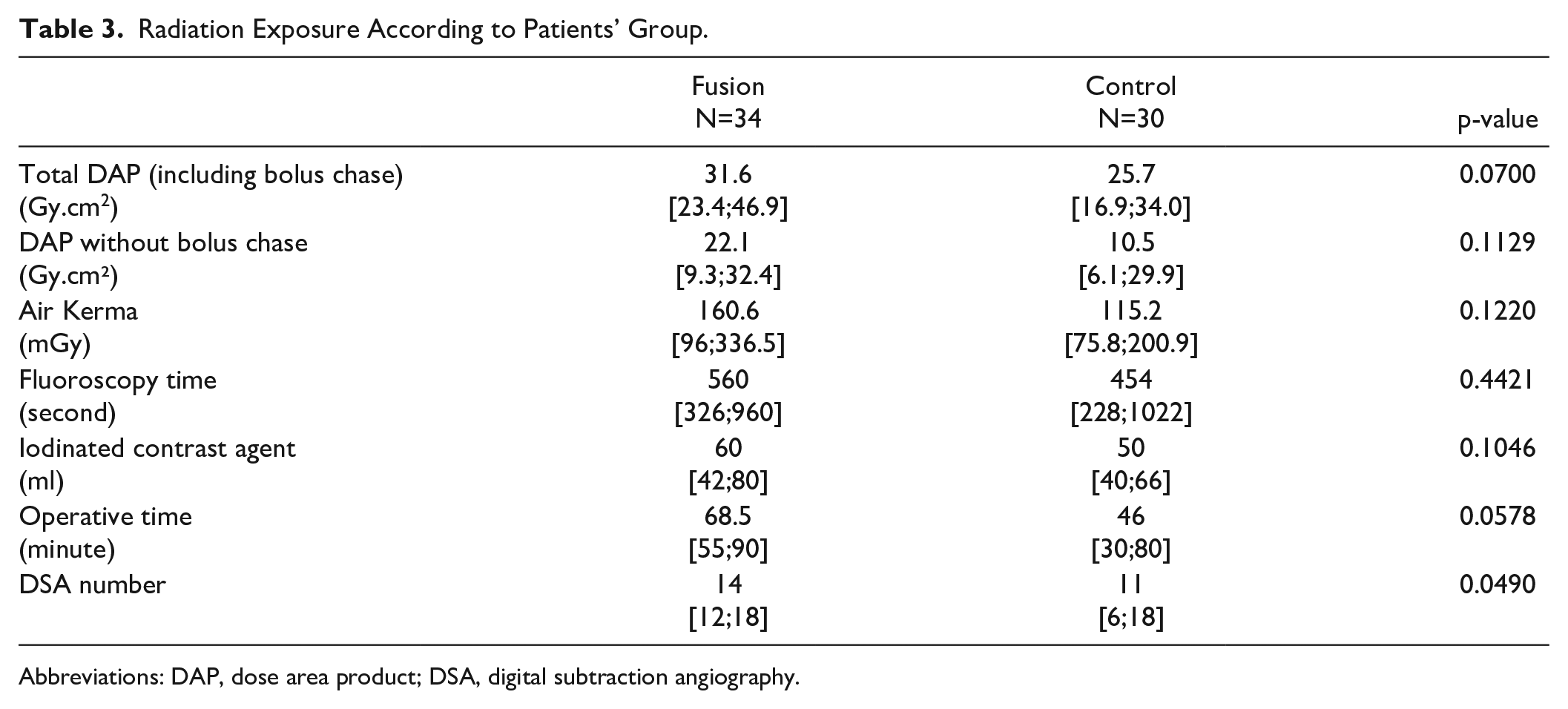
Abbreviations: DAP, dose area product; DSA, digital subtraction angiography.
Univariate linear regression models revealed a significant correlation between BMI (<25, 25–30, and >30) and radiation exposure (6.1 Gy.cm² [3.1; 13.9], 12.7 Gy.cm² [4.4; 29.1] and 20.7 Gy.cm² [8.6; 27.7] respectively, p=0.017); a significant correlation between lesion location (femoropopliteal only, iliac only, both) and radiation exposure (p<0.001) (Table 4); a significantly higher radiation dose in complex lesions (TASC A and B 10.7 Gy.cm² [5.2; 27.8] versus TASC C and D 23.4 Gy.cm² [93; 31.7], p=0.02) (Table 5); and a significantly higher radiation dose in diabetic patients with a DAP of 30.2 Gy.cm² [10.0; 45.3] versus 11.3Gy.cm² [6.2; 25.7] in nondiabetic patients.
Radiation Exposure According to Lesion Location.
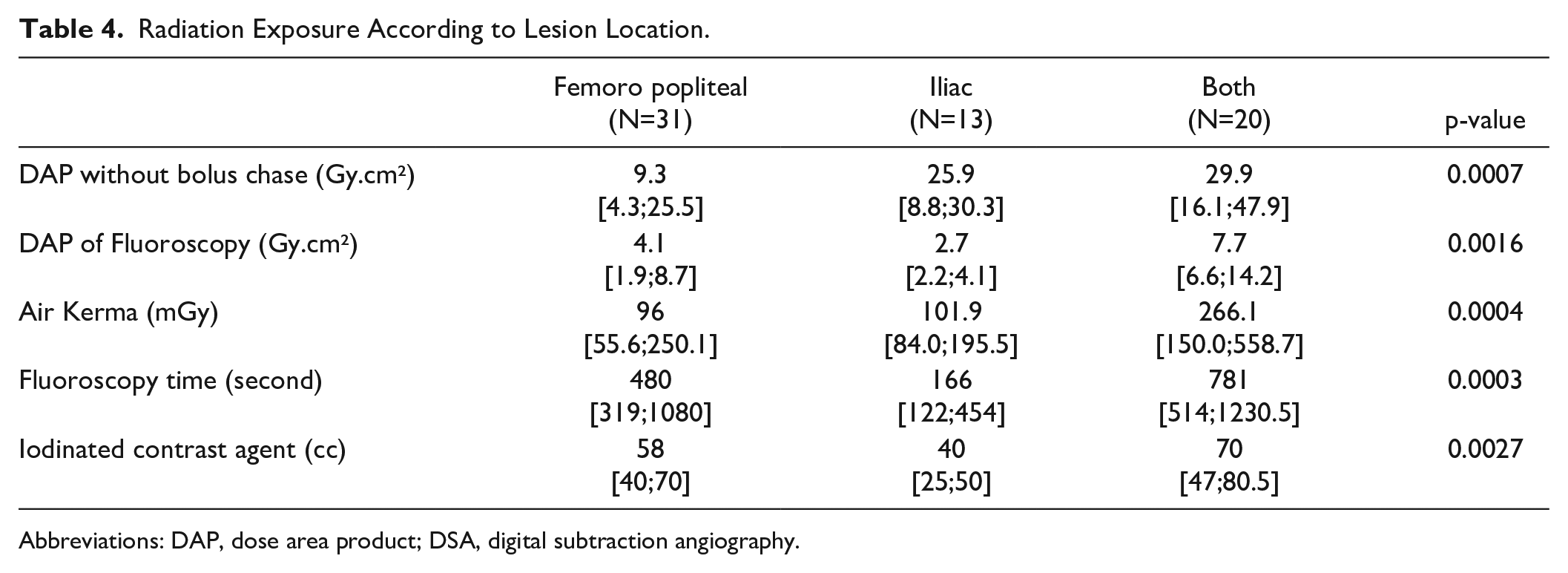
Abbreviations: DAP, dose area product; DSA, digital subtraction angiography.
Radiation Exposure According to TASC Classification.
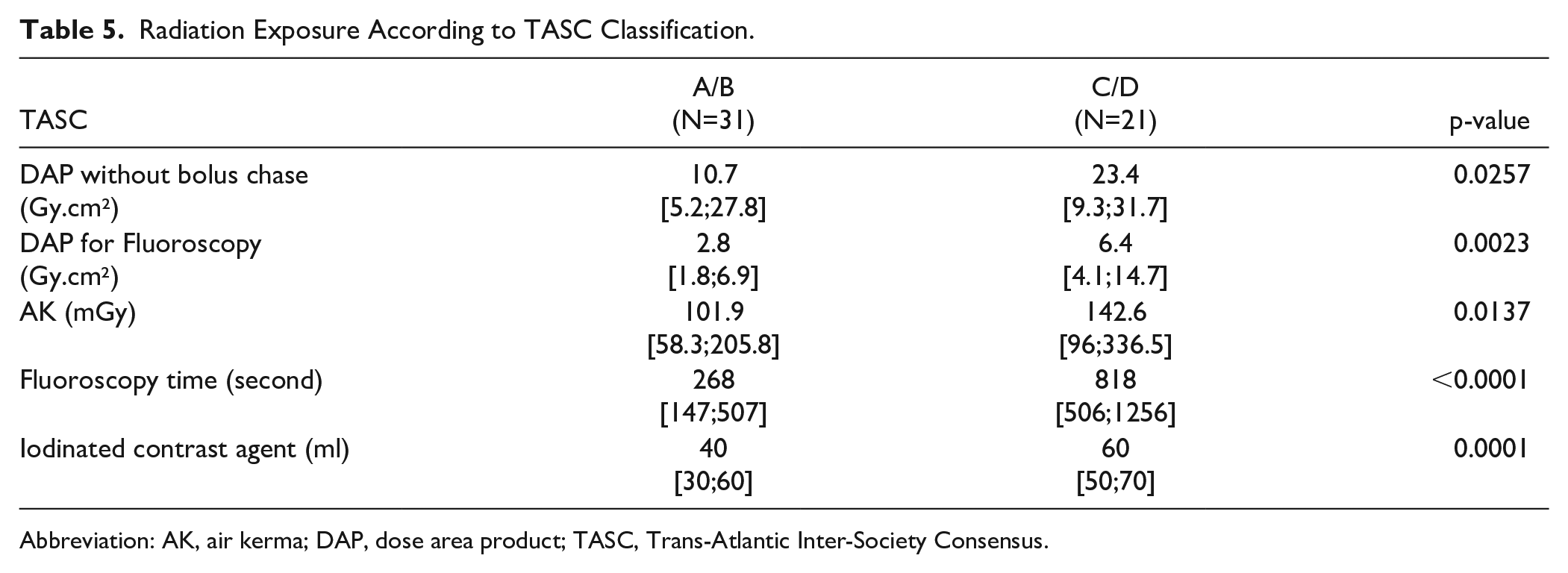
Abbreviation: AK, air kerma; DAP, dose area product; TASC, Trans-Atlantic Inter-Society Consensus.
The DAP was not significantly different between groups in multivariate linear regression model adjusted on lesion location, obesity, diabetes, and TASC classification (Table 6).
Factors Associated With Dose Area Product With Adjustment.
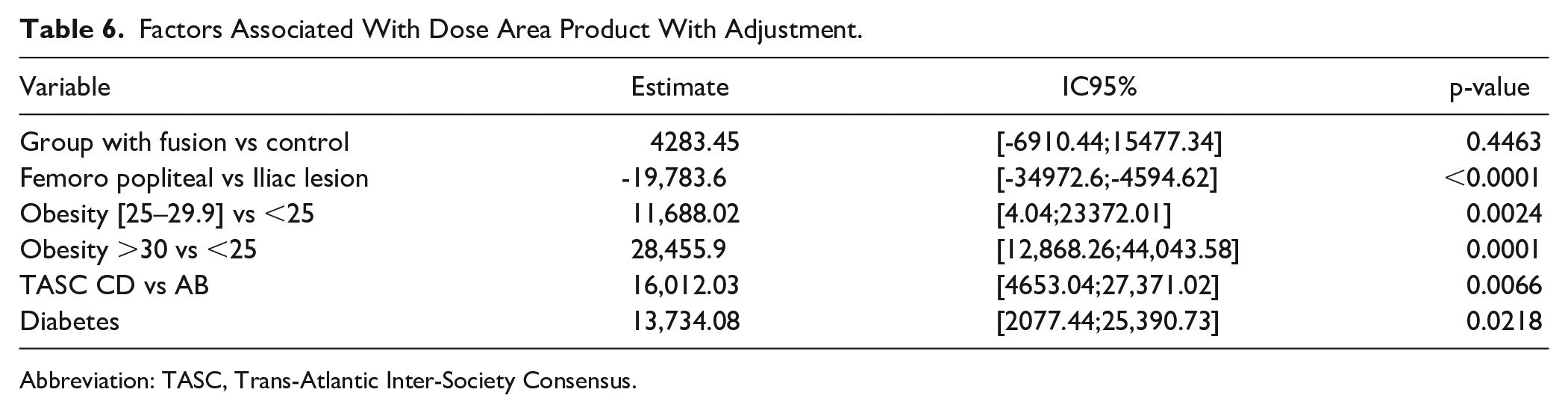
Abbreviation: TASC, Trans-Atlantic Inter-Society Consensus.
Discussion
The SOFT pilot study is the first randomized controlled trial assessing the impact of fusion guidance for reducing radiation exposure during endovascular treatment of LEAD. In our trial, the use of image fusion guidance failed to show a significant benefit regarding patient who underwent iliac and/or femoropopliteal endovascular procedures. This study also shows that radiation dose is higher when iliac arteries are involved, and increases proportionally to the complexity of the procedure and to the patient’s BMI, as already described in literature. 2 In the study, diabetic patients had greater radiation exposure, but we assumed that there are likely to have higher BMI and most complex arterial lesions. Nowadays, it is well known that repeated exposure to small doses of radiation exposes staff members to a stochastic effect that may increase the risk of solid cancers and leukemia, and radioprotection is of utmost importance. 11
Fusion imaging guidance is now available in most hybrid rooms as well as on mobile systems, using an automated or semi-automated patient-based image guidance process that will facilitate the broad use of this technology.12,13 Several methods are described to register the 3D vascular mask on the live image. In our center, the vascular 3D model is reconstructed from the preoperative CTA on a workstation (Philips VesselNavigator®), and then fused with live fluoroscopy (Philips Allura FlexMove®) through 2 orthogonal shots. At any time during the procedure, the registration can be refined by the operator from the table side.
This advanced imaging technology has been associated in literature with up to 70% reduction of the procedure’s radiation reported in complex EVAR, a lower fluoroscopy time and a decreased volume of iodinated contrast agent. 14 However, little is known about the impact of fusion imaging guidance on radiation and contrast dose during lower limb revascularisation.
A recent retrospective analysis reported that routine use of fusion imaging guidance during LEAD endovascular treatment is associated with low radiation exposure, high technical success, and reduced need for re-entry systems as compared to literature. 15 However, we did not confirm these results in our prospective randomized study.
Several hypotheses may explain our results. First, in modern angiography systems, patient thickness is estimated to automatically adjust dose production settings (such as peak kilovolt and amperage) in order to decrease exposure and to optimize signal-to-noise ratio. This allows constant image quality at the lowest dose and explains that abdominal procedures are more X-ray consuming than limb procedures. Smaller radiation doses during endovascular procedures for lower extremity arterial disease may require more patients in the study to highlight a significant benefit of fusion guidance. Second, the wide range of procedure complexity may have had an impact on our results, because the groups were not stratified on TASC lesion severity, nor on iliac and/or femoropopliteal lesions. More patients with iliac and femoropopliteal lesions were included in the fusion group, and the fluoroscopy time—which reflect the complexity of the procedure—was nonsignificantly higher in the fusion group (560 seconds. versus 454 seconds, p=0.44). This probably explains the high number of DSA in the fusion group, related to the procedure complexity. Besides the leading operator made the choice to use digital subtraction angiography (DSA) to assess fusion accuracy prior to stent implantation and to assess the result. This choice may have contributed to a high radiation dose in the fusion group. Modern imaging systems enable digital storage of fluoroscopic loops—far less radiation consuming—that can be used to assess arterial patency and adjust the fusion mask. This has been reported by Sailer et al 16 with among 17 patients and 47% of procedures performed using only fluoroscopic loops and a fusion mask without DSA. Stahlberg et al 17 also reported a significant reduction of radiation (of 6 Gy.cm2) and contrast medium (45 ml versus 120 ml) during iliac occlusive procedures. This suggests that during the treatment of LEAD, the use of fusion imaging guidance does not facilitate the procedure enough to have an impact on radiation exposure, but requires a change in work habit to be associated with X-ray dose reduction. The reduction of DSA runs when using fusion guidance seems to be of utmost importance.
Our trial had several limitations. It was not possible to blind the operator to the patient group. Our pilot study was underpowered because the original sample size calculation overestimated the DAP reduction dose in this nonaortic population, and several recruited patients were excluded before analysis because of violation of eligibility criteria. Moreover, the study was stopped prematurely due to an internal reorganization of the department which finally led to the termination of the study after 16 months. Finally, the randomization system failed to ensure equal assignment of patients presenting iliac and femoropopliteal lesions versus patients with only one stage lesions, which could have disadvantaged the interventional group.
Conclusion
To conclude, the SOFT pilot trial failed to demonstrate a reduction of radiation exposure and iodinated agent use when fusion imaging guidance is used during endovascular procedures for lower extremity arterial disease. Further prospective studies with larger stratified groups upon anatomic lesion and BMI are required, to assess the impact of fusion imaging guidance on patient outcome.
Footnotes
Authors’ Note
Abstract presented in fast-track sessions during the preconference days of the 2020 ESVS Month, September 15, online.
Abstract presented during the virtual SCVE meeting 2021, on the June 25, 2021, online.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: There is no source of outside support for research, and no financial support by industry.
B.P., T.L.C., L.B., P.C., B.G., and I.F.: No conflict of interest
B.N.: consulting fees from Medtronic, Biotronik, and W.L. Gore
Y.G.: Research funding from Abbott, General Electric, WL Gore. Personal fees and grants from Abbott, Bard, Biotronik, Boston Scientific, Cook, General Electric, Medtronic, Penumbra, Terumo, Veryan, WL Gore (medical advisory board, educational course, speakingconsulting fees)
B.M.: consulting fees from Cook Medical, W.L. Gore and Philips
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
