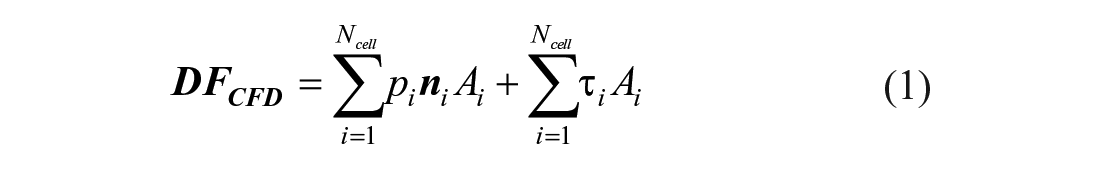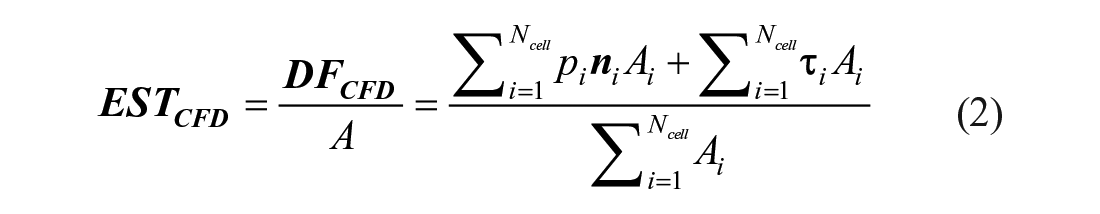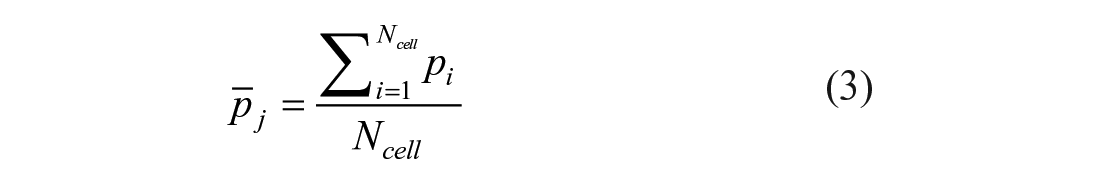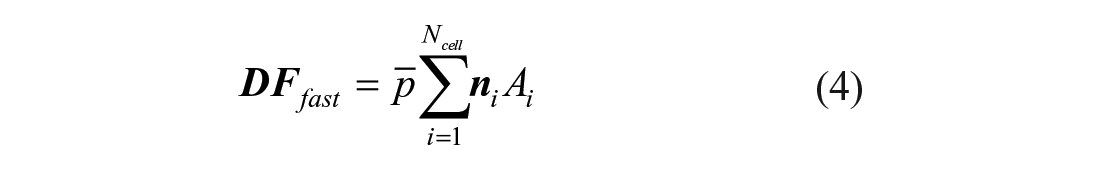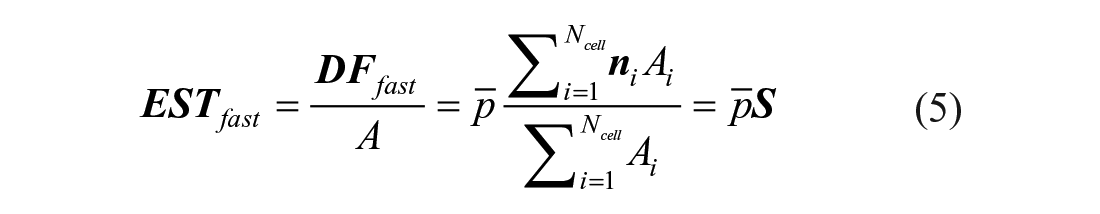Abstract
Purpose:
Displacement forces (
Materials and Methods:
We tested the fast-approximate approach against CFD gold-standard in 34 subjects with the “bovine” aortic arch variant. For each dataset, a 3-dimensional (3D) model of the aortic arch lumen was reconstructed from computed tomography angiography and CFD then employed to compute
Results:
Fast-approximate approach consistently reflected (r2=0.99, p<0.0001) the
Conclusion:
Requiring only a few seconds and quantifying clinically relevant biomechanical parameters of proximal landing zones for arch TEVAR, our method suits the real preoperative decision-making process. It paves the way toward analyzing large population of patients and hence to define threshold values for a future patient-specific preoperative TEVAR planning.
Keywords
Introduction
Thoracic endovascular aortic repair (TEVAR) is a well-established therapeutic option in patients with adequate anatomic features 1 and an acceptable life expectancy. 2 Yet, while TEVAR of the descending thoracic aorta currently represents a first-line treatment, 3 TEVAR of the aortic arch is associated with relevant rates of postoperative clinical failure, 4 and still poses specific challenges related to the peculiar geometric configuration and unique fluid dynamic environment 5 of this aortic tract.
Current preoperative planning of TEVAR remains based on computed tomography angiography (CTA) imaging protocols, which disregard the fact that the aorta is a pressurized conduit subjected to displacement forces (
Despite the awareness of this limitation, however, the inclusion of
In the present work, to overcome the need of time-consuming CFD analyses, we sought to develop a fast approximate and reliable approach to compute
Furthermore, aortic blood pressure is reported to be only slightly heterogeneous along the aortic arch.14–16 Accordingly, we herein hypothesized that blood pressure space distribution within the aortic arch can be reasonably simplified with a single clinically pertinent value of pressure, eg, a noninvasive measure of the central aortic pressure. Based on this evidence, we hypothesized that
To test the validity of the proposed fast approximate approach against CFD reference analysis, we chose to prove our case in subjects (n=30) presenting with the so called “bovine” aortic arch, 17 which is the second most common anatomic variant after the standard arch configuration, and is characterized by a common origin of the innominate and left carotid artery (CILCA arch). 18 Notably, this anatomic pattern is highly prevalent among patients requiring TEVAR, 19 being a recognized risk factor for the development of thoracic aortic disease, 20 and it presents a consistent and peculiar geometric pattern. 19
Materials and Methods
Study Design
The newly introduced fast approximate approach was tested on 30 anonymized thoracic CTA scans of subjects with a CILCA arch configuration and a healthy aorta. In addition to this group, 4 more cases of patients with a CILCA arch and an aneurysmatic aorta, which were retrieved from a database of patients treated by TEVAR at our center, were also included. On these CTAs, both the new fast approximate approach and CFD analyses were performed to (1) confirm that in CILCAs the pattern of
The CTAs of 30 healthy aortas all respected the following criteria: thin-cut (1.0 mm), visible origins of the supra-aortic branches, patient age>60 years, thoracic aorta diameter<40 mm, arch radius of curvature>20 mm, no previous aortic surgery, and no radiologic signs of aortic pathology, including dissection, intramural hematoma, or penetrating aortic ulcer. CTAs were evenly selected among 3 anatomical phenotypes, ie, arch types I, II, and III,
21
based on the vertical distance from the origin of the brachiocephalic trunk to the top of the arch
22
: this distance is shorter than the left common carotid artery diameter (
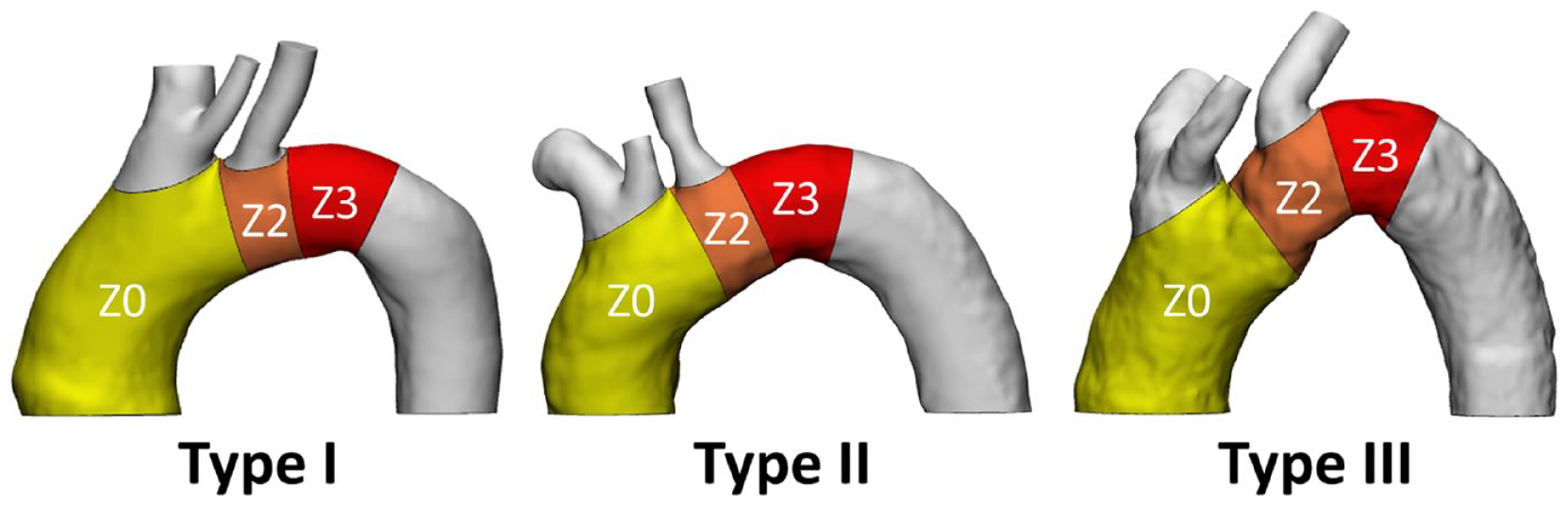
Aortic arch classification. Aortic arch classification in types I, II, and III and subdivision in the Ishimaru’s zones, ie, zones 0 (Z0), 2 (Z2) and 3 (Z3).
The study was approved by the Ethics Committee of reference; informed consent was waived by the Ethics Committee because of the retrospective nature of the study and the analysis of anonymized images. The study was conducted according to the principles outlined in the Declaration of Helsinki.
Aortic reconstruction
Each CTA scan was segmented in Mimics Medical v21.0 (Materialise, Leuven) to reconstruct the thoracic aorta, from the aortic valve annulus to the diaphragm, and including the proximal tract of the innominate artery, the left common carotid artery, and the left subclavian artery (Figure 2a).
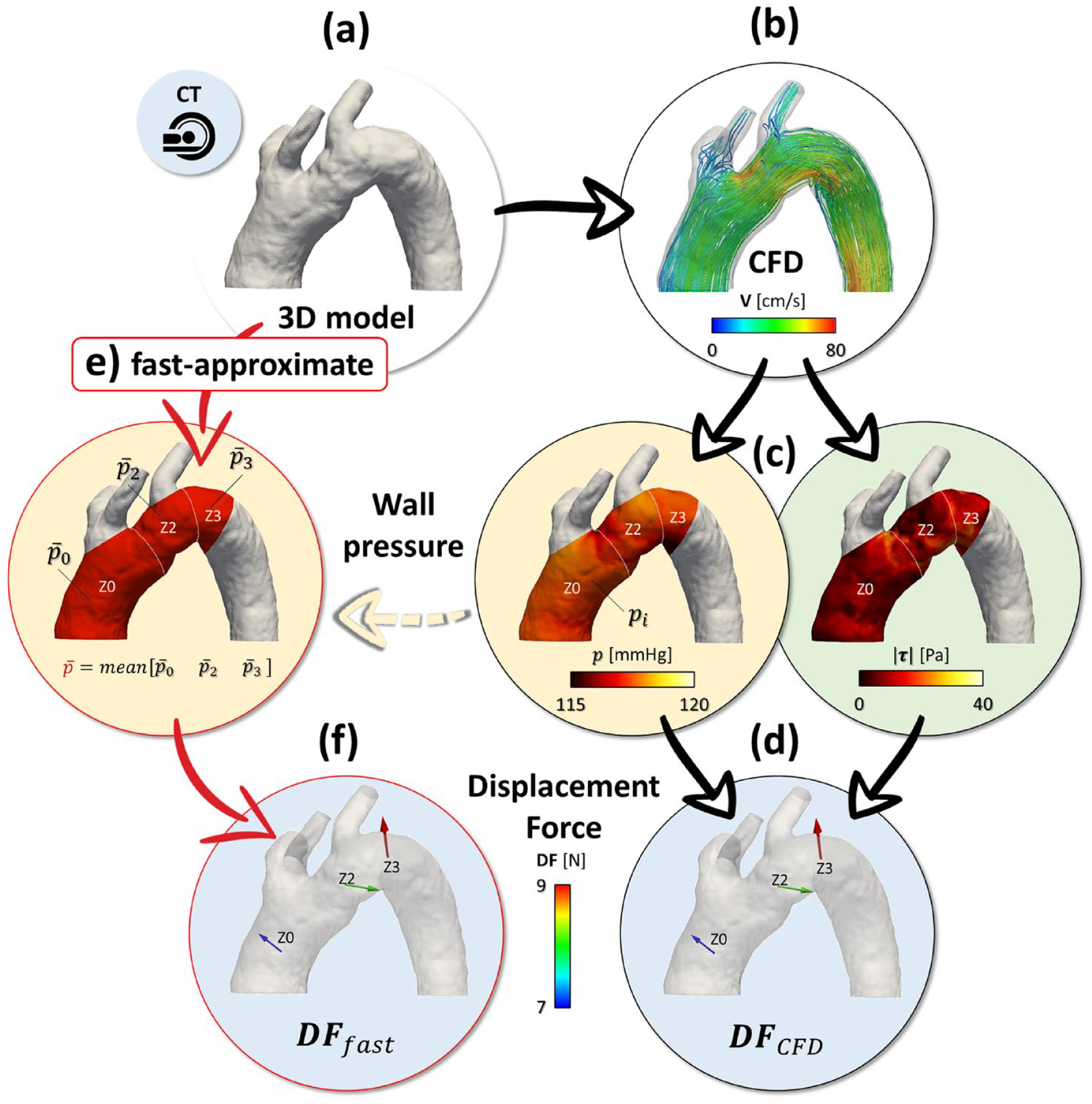
Study workflow and displacement forces computation. Patient-specific 3D geometry (a) used to simulate pulsatile aortic blood flow through CFD analysis (b); blood pressure and shearing stresses acting on the aortic wall surface (c) were extracted to compute displacement forces (d). Assuming a mean blood pressure acting on the aortic wall (e), we defined a novel fast-approximate quantification of aortic displacement forces (f). CFD, computational fluid dynamics; CT, computed tomography; DF, displacement force.
Subsequently, each 3D aortic geometry was imported in stereolithographic format (.stl) in the open-source library Vascular Modelling ToolKit (VMTK v1.4; www.vmtk.org). Straight flow extensions were added at both the inlet and the outlet faces of the domain to ensure a fully developed velocity profile at the inlet section and to minimize the influence of outlet boundary conditions.23,24 These fictitious extensions were not part of the aortic domain of interest for postprocessing analysis.
The final 3D model was then discretized to generate a volumetric grid suitable for CFD analysis within the aorta 25 ; an average cut-off of 2 million tetrahedral cells was considered for all the simulated anatomies, following a preliminary mesh convergence analysis. 5 The triangular faces of the cells that belonged to the aortic wall were tagged for subsequent processing.
CFD Analysis
Numerical simulations (Figure 2b) were carried out, by solving the unsteady Navier-Stokes equations in the region of interest through the open-source finite element solver LifeV (www.lifev.org) following a previously employed and well-established CFD approach.5,7,26 To obtain periodic pulsatile flow conditions, 6 consecutive heartbeats with a 1 s period were simulated in unsteady conditions with a constant time step of 0.001 s. CFD data computed in the last cardiac cycle were postprocessed.
Blood was treated as a Newtonian fluid with constant viscosity (μ=3.5 cP) and density (ρ=1060 kg/m3); aortic wall was assumed rigid and no-slip (ie, null velocity) boundary conditions were prescribed on the luminal surface.
Given the retrospective nature of the study, patient-specific hemodynamic data were not available, and the same inflow and outflow boundary conditions were applied in all cases. 5 Specifically, on the inflow section, a pulsatile aortic flow waveform with a cardiac output of 4.88 L/min was imposed as representative of ascending aortic flow waveform and cardiac output for adult patients. This flow waveform, derived from phase contrast magnetic resonance imaging on a previous cohort of patients investigated at our study hospital, was extracted as the mean flow waveform measured at the level of the ascending aorta. At each outflow section, a 3-element Windkessel circuit was coupled to the 3D discretized domain to reliably mimic both compliance and resistance of the distal aortic vasculature, 26 thus resulting in a physiological range of the average simulated blood pressure. 27
CFD Postprocessing
CFD results were postprocessed at peak systole in Paraview v5.5.2 (Kitware Inc, Clifton Park, NY), using dedicated Python scripts to quantify 3D displacement forces (
Displacement forces - The displacement force
where
Equivalent surface traction (
where
Fast-Approximate DF Quantification
Fast-approximate DF
First, we neglected the wall shear stress contribution to DF since blood pressure has already been demonstrated to be several orders of magnitude larger than the corresponding
Second, the CFD-derived blood pressure distribution acting on the aortic wall surface was approximated. To this purpose, blood pressure distribution was averaged within each aortic landing zone as
with
pulling
Subsequently,
where
Statistical Analysis
Continuous variables were expressed as mean±standard deviation (SD) after Shapiro-Wilk normality tests.
Results
Magnitude of
and
The magnitude of fast-approximate displacement forces, ie,
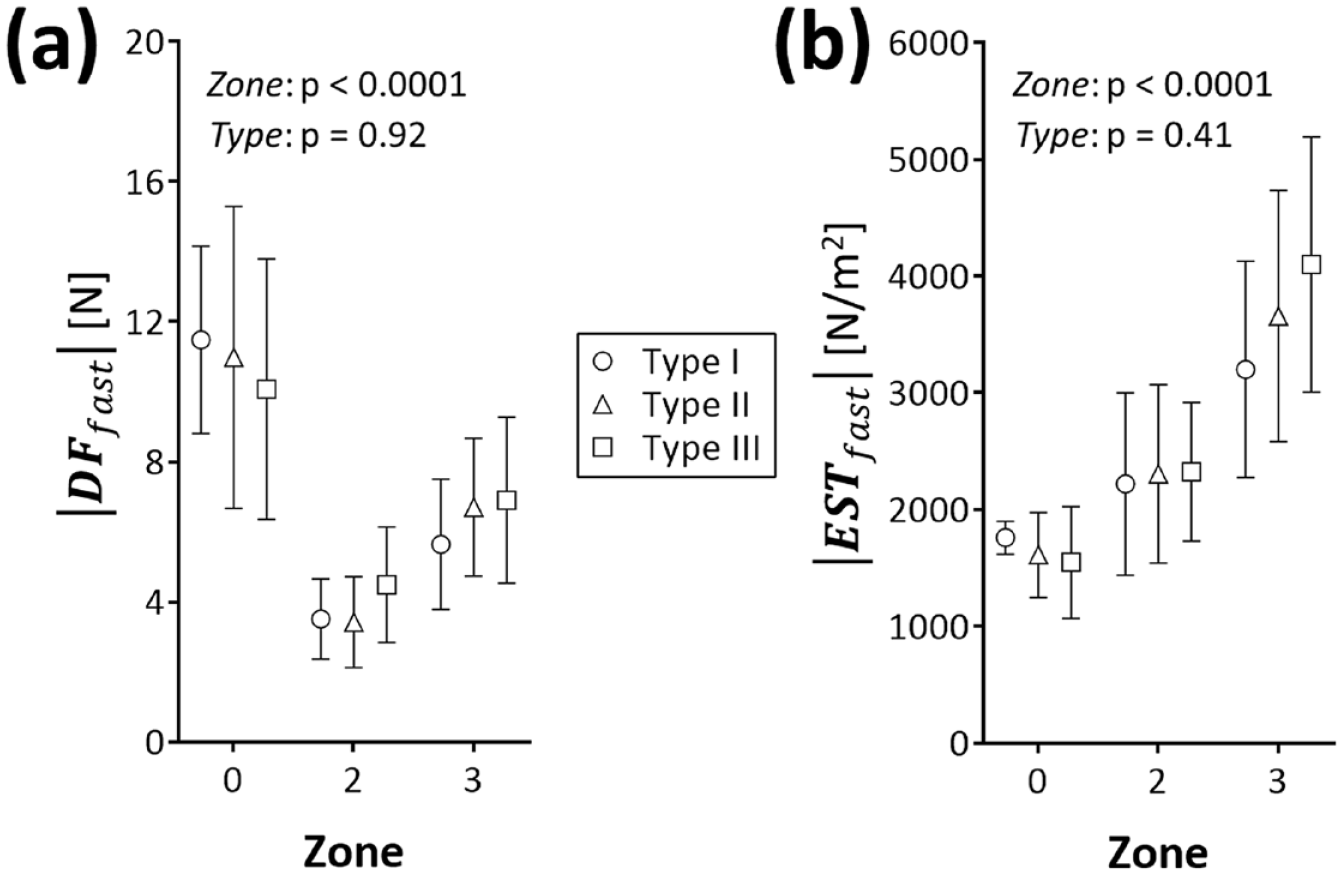
Fast-approximate magnitude quantification of displacement force
The normalized forces
Differences remained negligible within each landing zone when comparing
Additional data extracted from the 4 anatomies with aneurysmatic CILCA arch (Supplementary Tables 5 and 6) provided
Orientation of
and
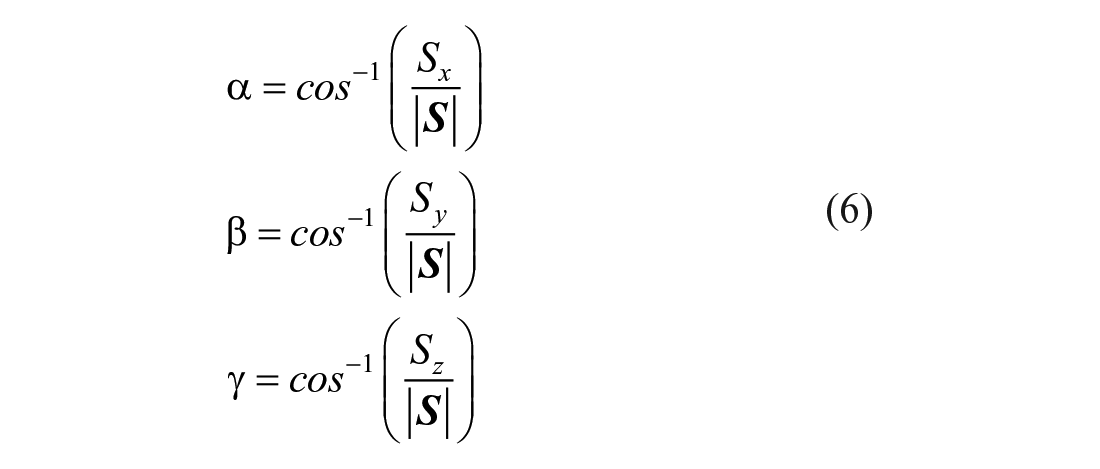
: The Aortic Shape Vector
The present fast-approximate approach allowed to represent the spatial orientation of aortic displacement forces, being the same for both
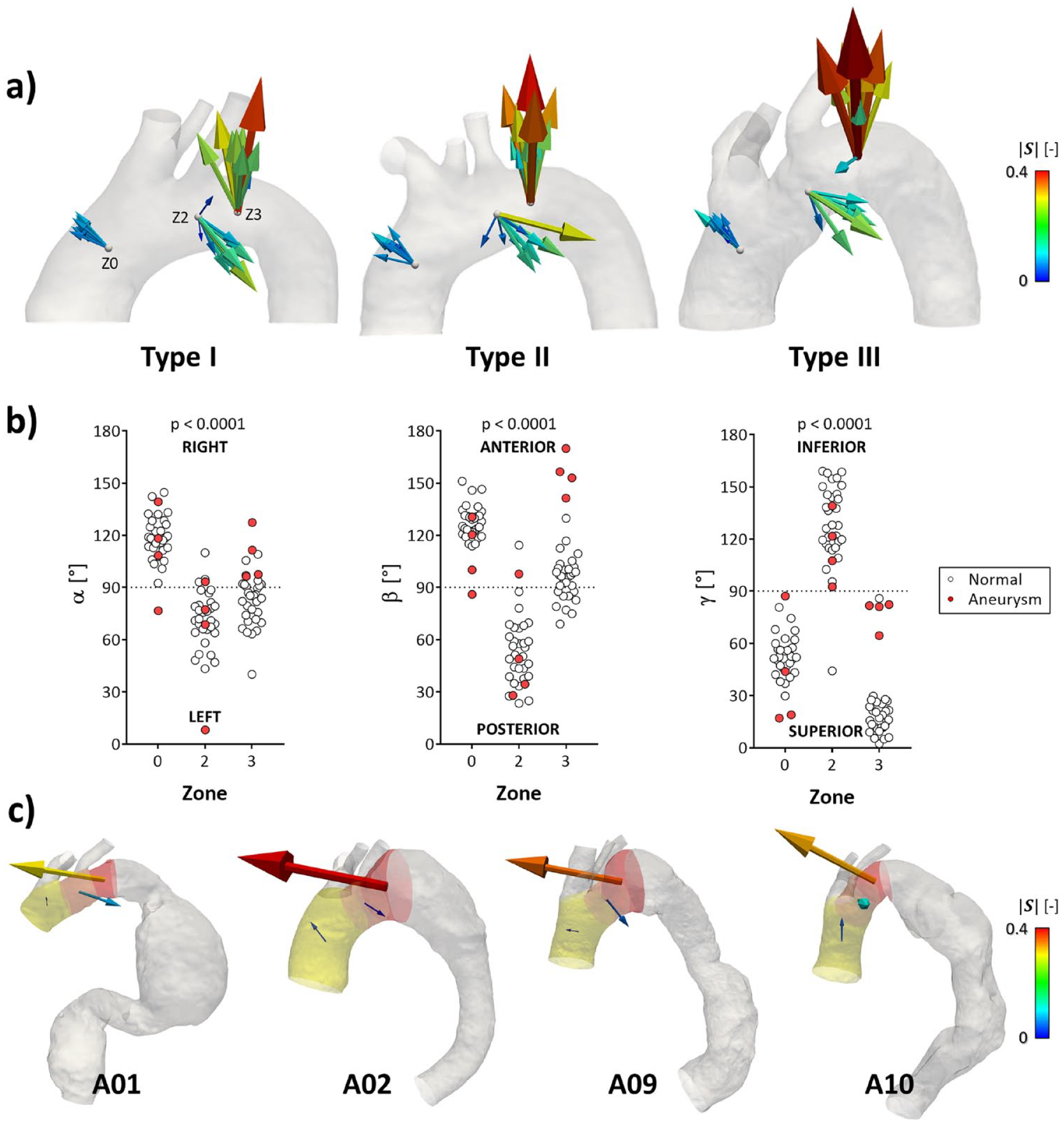
Magnitude and direction of the aortic shape vector
In the pathological aortic anatomies (Figure 4c, Supplementary Table 10), though
As clear from equation (5),
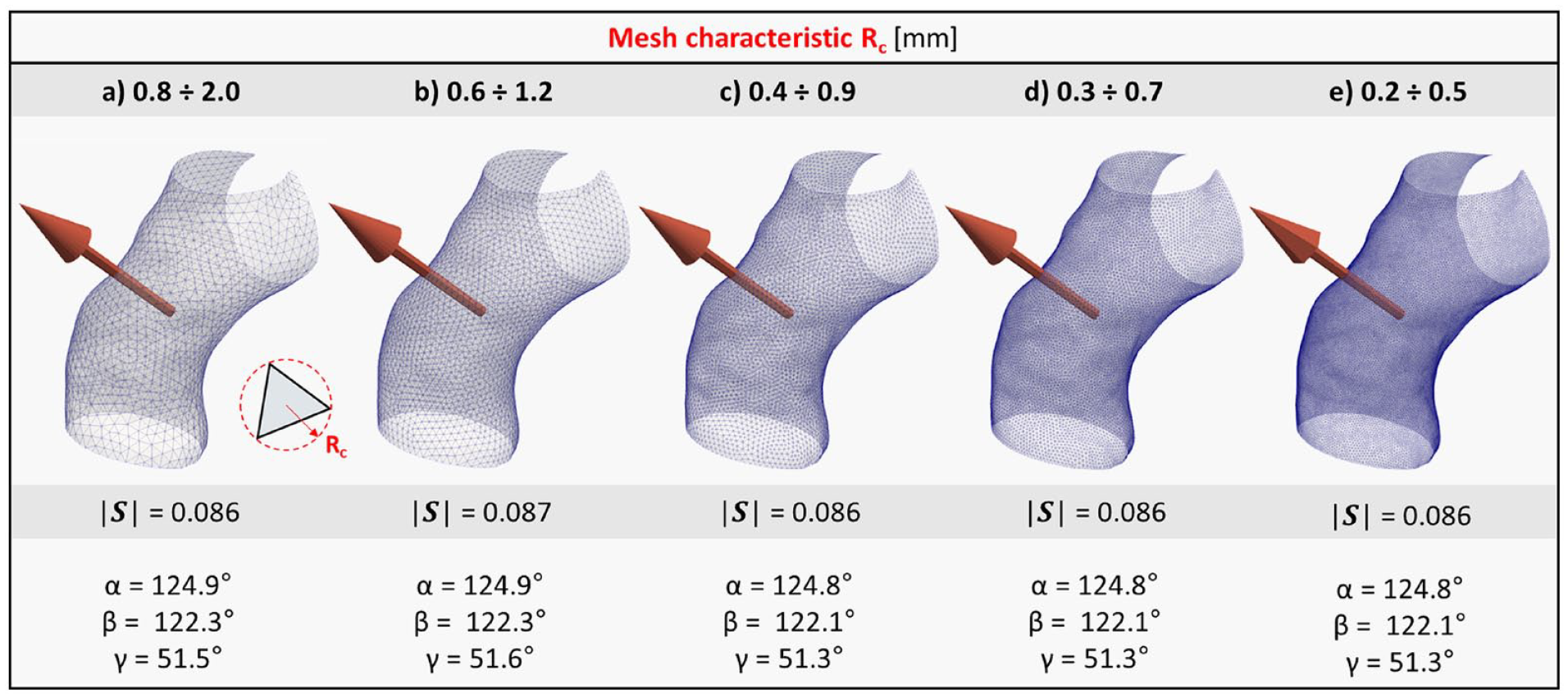
Sensitivity of
Fast-Approximate Approach vs CFD Gold-Standard Analysis
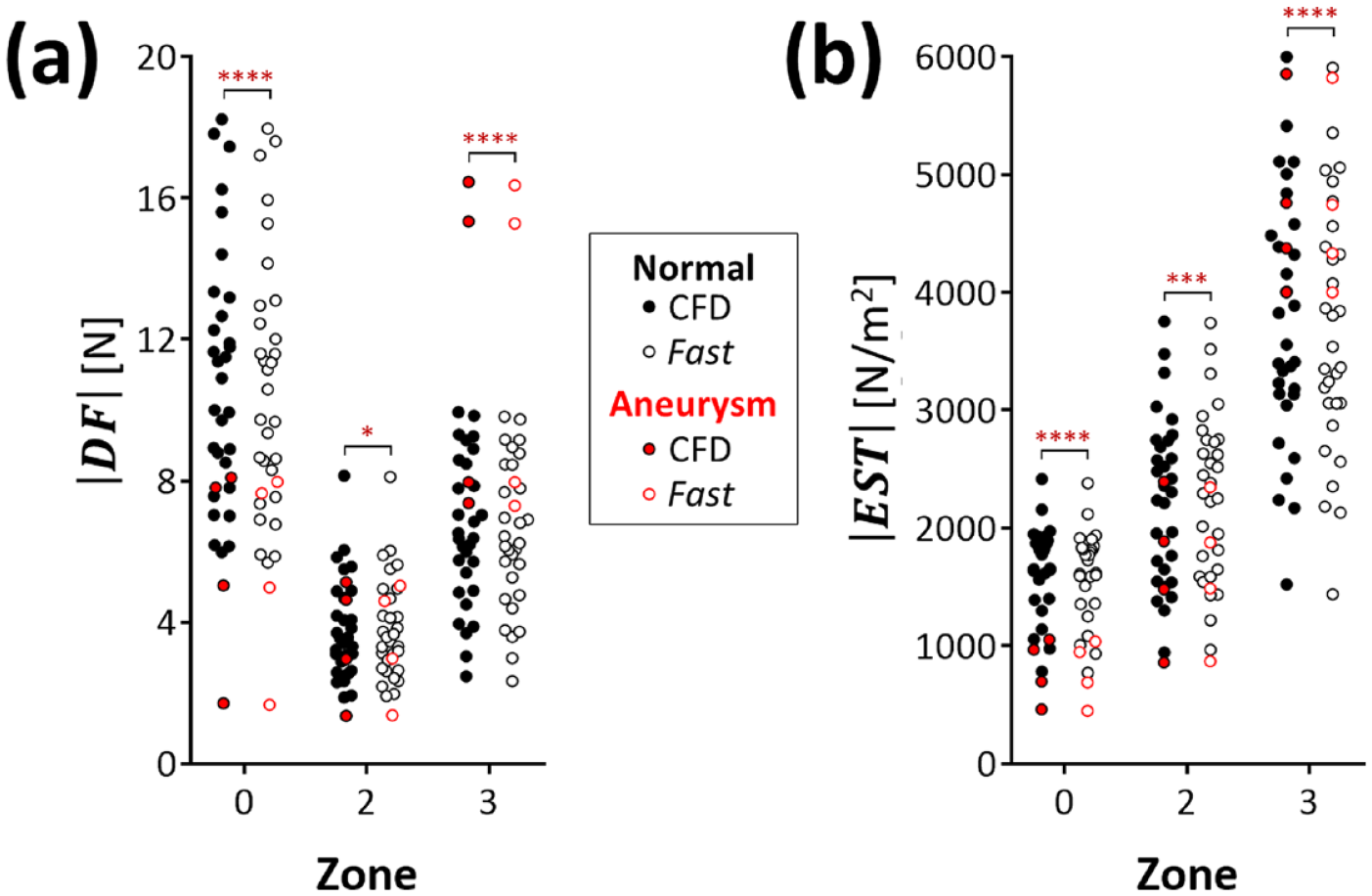
The fast-approximate quantification of displacement forces consistently reflected the pattern obtained by CFD analysis (Figure 6).
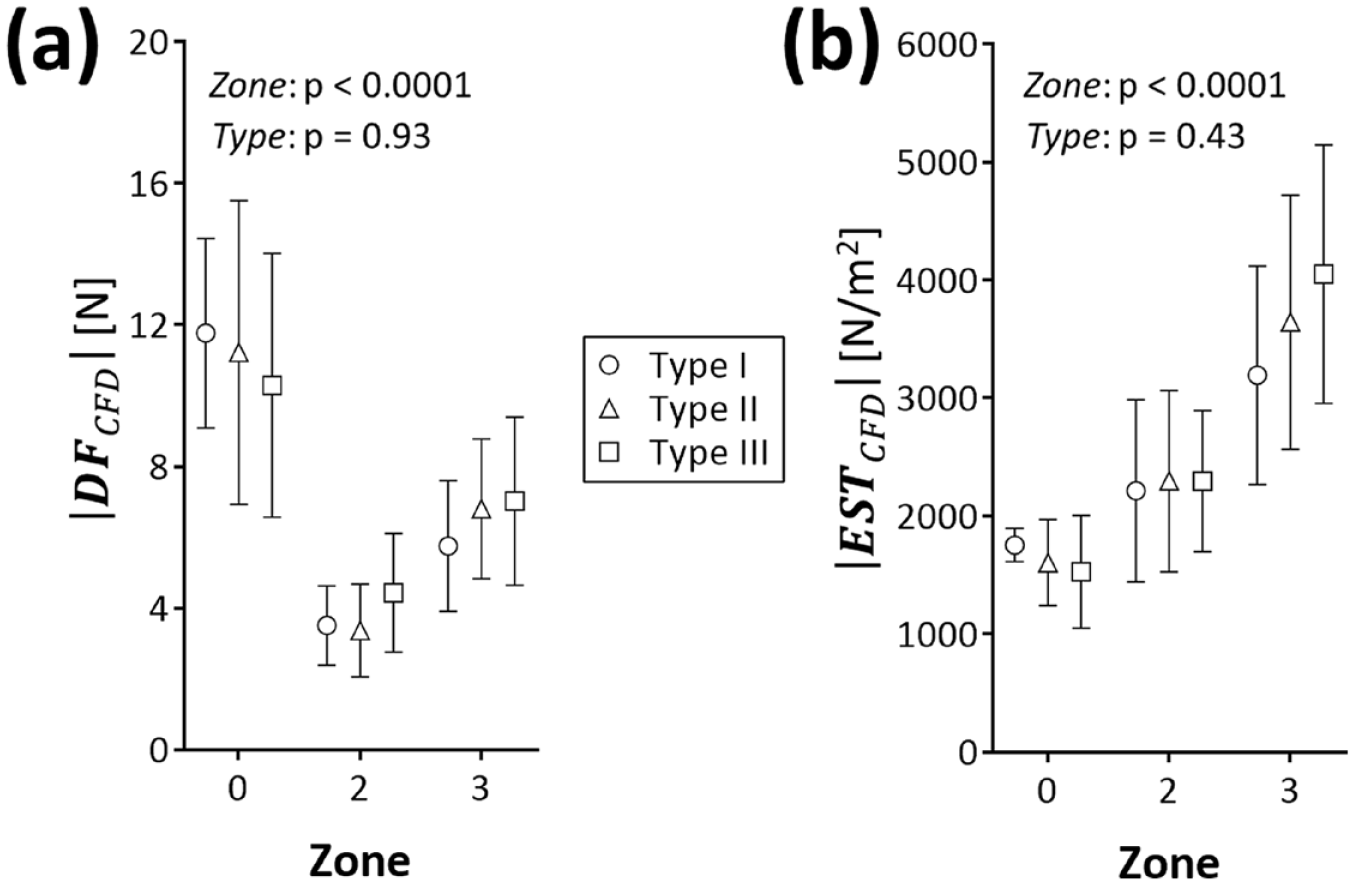
CFD-derived magnitude of (a) displacement force
Confirming the results of the fast-approximate approach, differences remained negligible within each landing zone when comparing both
In Figure 7, pairwise comparison between fast-approximate and CFD computations are reported in terms of
Pairwise comparison in terms of (a)
Specifically, we compared the overall
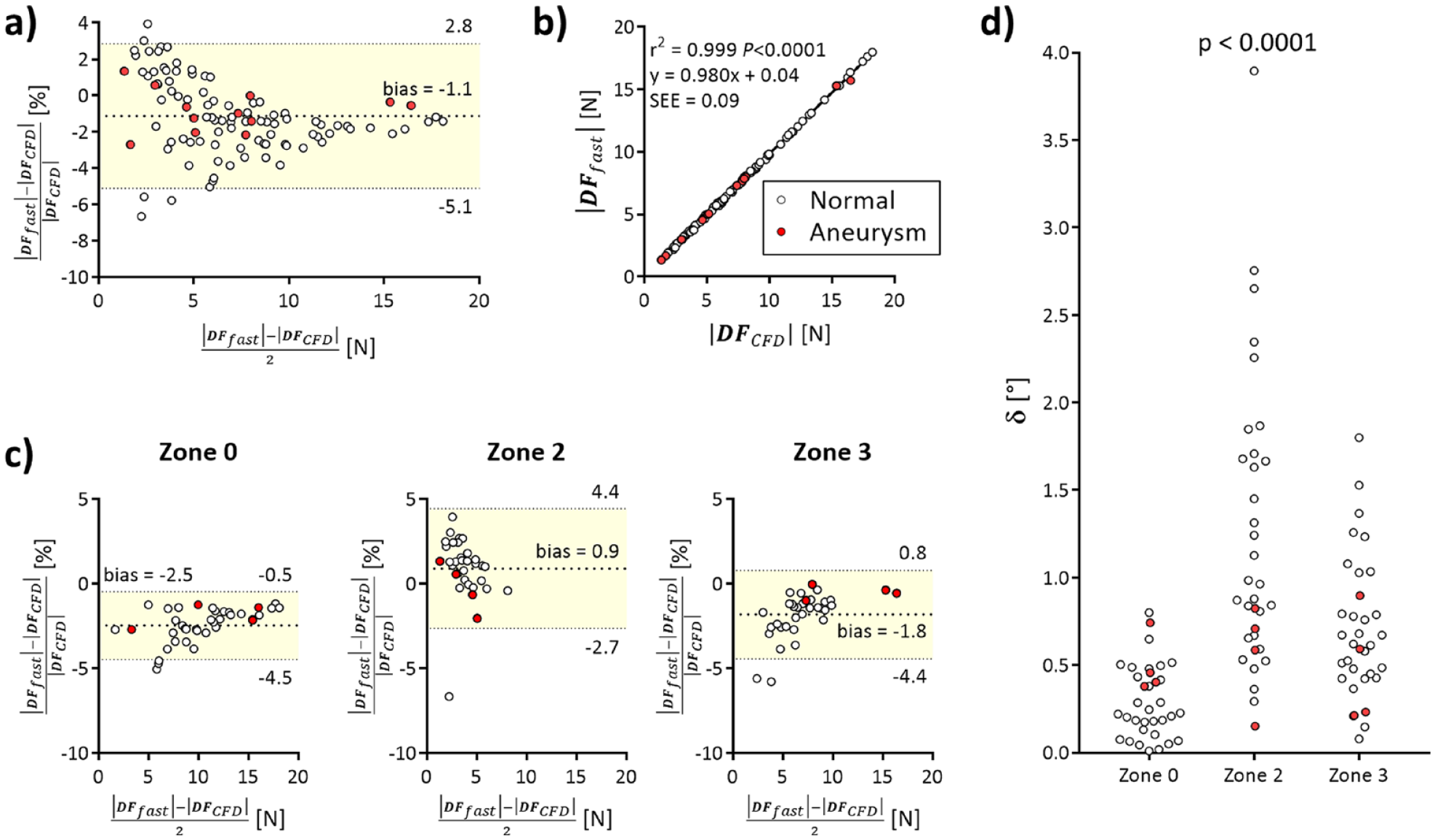
Bland-Altman plot (a) of the differences between
The fast-approximate computation strongly correlated against the CFD gold-standard (Figure 8b), ie, r2=0.99 (p<0.0001, slope regression 95% CI of 0.975 to 0.984, y=0.98x + 0.04). Of note, these differences were not uniformly distributed between the different aortic landing zones (Figure 8c). Specifically,
Furthermore, CFD analysis (Figure 9, Supplementary Table 11) confirmed that aortic blood pressure (
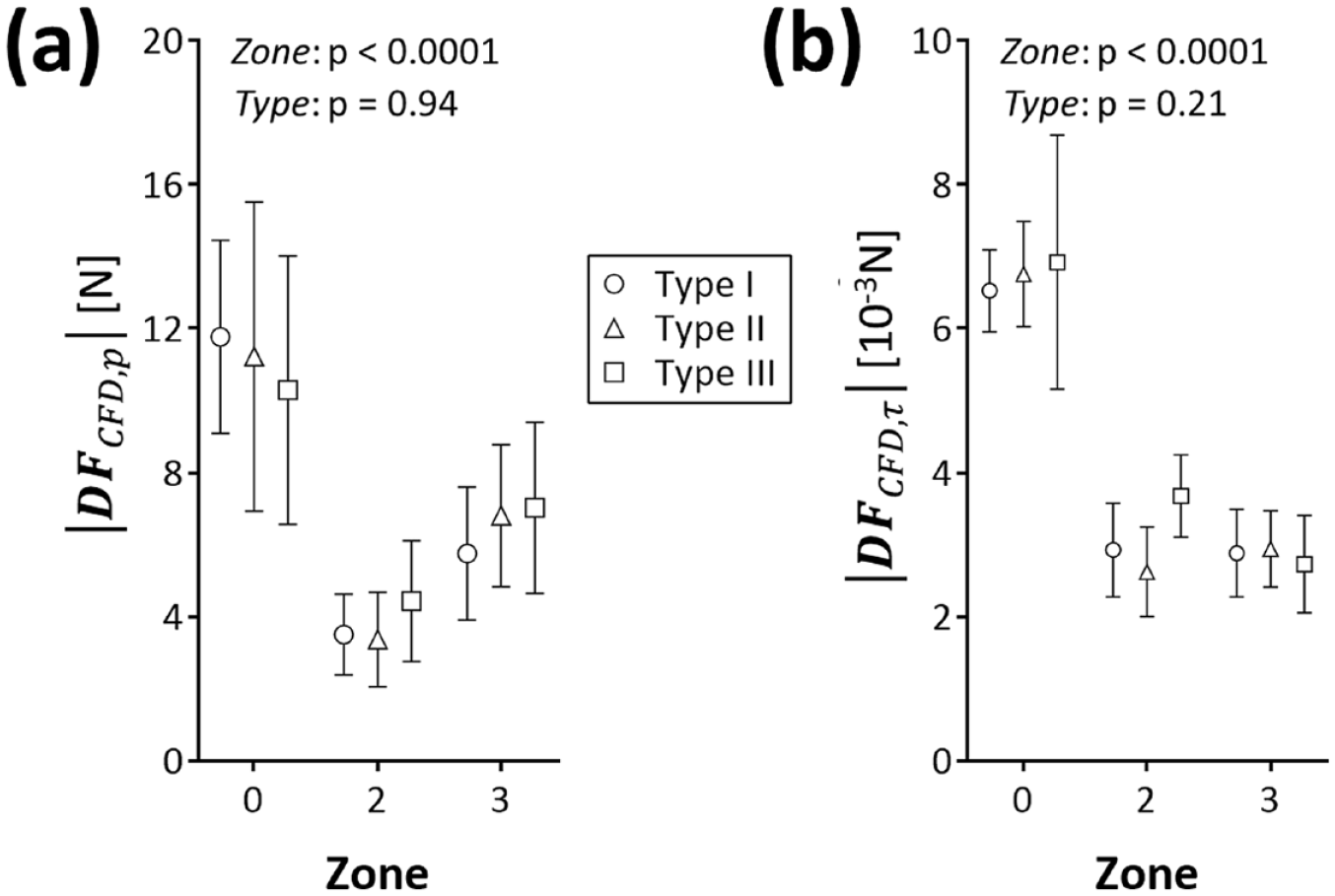
Repartition of displacement force magnitude between
In the small cohort of anatomies with aneurysmatic CILCA arch,
Within each proximal landing zone, we also analyzed the collinearity of each
Discussion
In the present work, we demonstrated that fast-approximate and reliable quantification of displacement forces is feasible in the aortic arch, and this may overcome the need for time-consuming CFD analyses.6,11,12
Our fast-approximate approach reproduced accurately enough the
If compared with time-consuming CFD analysis, which may require several days even exploiting dedicated high-performant computing resources, the computational cost of fast-approximate DF calculation is quantifiable in seconds once the aortic arch geometry is segmented. Hence, the computation of
From a clinical standpoint, the fast-approximate analysis of CILCA arches confirms the presence of a force distribution over the proximal landing zones for TEVAR
12
that is preserved in different anatomies, reporting a much greater
From a methodological standpoint, the proposed fast-approximate approach only requires patient-specific aortic geometry and a measure of central aortic pressure, both of which are available during TEVAR planning. The patient-specific anatomy can be accurately extracted from the computed tomography, which is already exploited because of its speed and ease of use to navigate and assess the patient-specific aortic anatomy,29,30 eg, through commercial and clinically oriented semi-automated platforms. Central aortic pressure is recorded in routine clinical practice and generally estimated using noninvasive methods,31,32 eg, through brachial cuff sphygmomanometer. In the present proof-of-concept, a physiological range of blood pressure was imposed and reproduced in the CFD analysis. To mimic the in vivo cuff measure of central blood pressure, this was estimated averaging the overall pressure distribution on the entire aortic arch wall. However, the fact that non-patient-specific pressures were used does not limit the main point of the present work: for a given pressure regime, the fast-approximate method we propose is a very good approximation of the more sophisticated and time-expensive CFD-based method.
From a conceptual standpoint, it is interesting to note that
The proportionality between
Hence, though further and more extensive validation is required, our data suggest that the dimensionless shape vector can effectively describe how pressure-driven blood forces are expected to act and impact on the thoracic aortic wall. This represents a valid biomechanical blueprint of the aortic arch morphology, which can be computed without measuring aortic pressure, and can be interpreted as a scaling factor of the aortic anatomic pattern, see equation (5). As exemplified in Figure 10, two main and complementary factors affecting the shape vector
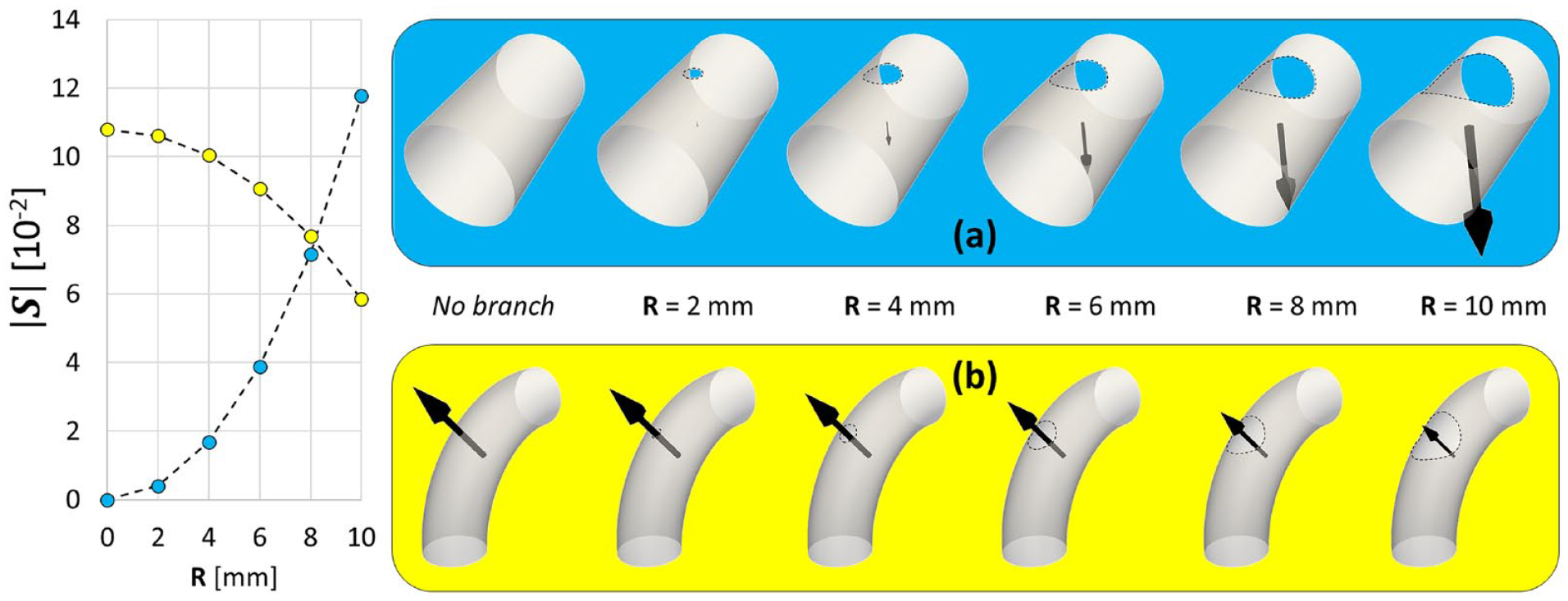
Impact of the dimension (ie, in terms of radius) of vessel branching on the computation of the shape vector
The first factor is determined by the presence of aortic branches and by the size of the respective origins: in a straight cylinder without branches
The second factor, which can be superimposed to the former, is related to the curvature of the vessel: in a curved pipe (Figure 10b), for instance,
Finally, the potential application of the fast-approximate approach may also pave the way for a novel and truly dynamic assessment of aortic
In this respect, the proposed fast-approximate approach could be exploited, combining ECG-gated multiphase CT imaging,35,36 to reproduce the aortic arch geometry over the cardiac cycle, with the patient-specific monitoring of aortic pressure, eg, through continuous noninvasive arterial pressure measurements. 37 To this purpose, the process of aortic segmentation could be fastened by exploiting deep learning-based and fully automatic algorithms.38–40
Limitations
We recognize some limitations of our study.
First, whether the biomechanical environment of the bovine aortic arch variant can effectively entail an increased risk of postoperative complications after TEVAR still remains to be demonstrated. To this purpose, a multicenter ad hoc outcome study is currently in progress.
Second, we adopted the same flow boundary conditions in all the simulated cases, which, however, did not impact on the comparison of both
Third, we approximated the spatial blood pressure distribution within the aortic arch with a single, noninvasive and clinically pertinent measure. Although only minor changes in blood pressure are expected along the aortic arch,14–16 the validity of this assumption should be further tested against in vivo evidence. This could be obtained either invasively or noninvasively: in the first case, measurements available in the cardiac catheterization laboratory, 42 eg, in patients undergoing diagnostic catheterizations or percutaneous procedures, could be exploited. In the second case, the relative pressure field, and hence pressure space-dependency, within the aortic arch could be extracted from time-resolved velocity-encoded magnetic resonance imaging (4D Flow).43,44
Conclusion
The present work provides a reliable and simplified method enabling a preoperative decision-making process for TEVAR through quantitative and physics-based parameters measured in aortic arch proximal landing zones, which allows further studies on the definition of clinically relevant threshold values of such parameters.
Supplemental Material
sj-docx-1-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-1-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-10-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-10-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-11-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-11-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-12-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-12-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-2-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-2-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-3-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-3-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-4-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-4-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-5-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-5-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-6-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-6-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-7-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-7-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-8-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-8-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-9-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-9-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Supplemental Material
sj-docx-13-jet-10.1177_15266028221095403 – Supplemental material for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant
Supplemental material, sj-docx-13-jet-10.1177_15266028221095403 for Fast Approximate Quantification of Endovascular Stent Graft Displacement Forces in the Bovine Aortic Arch Variant by Francesco Sturla, Alessandro Caimi, Rodrigo M. Romarowski, Giovanni Nano, Mattia Glauber, Alberto Redaelli, Emiliano Votta and Massimiliano M. Marrocco-Trischitta in Journal of Endovascular Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support of the Italian Ministry of Health (Ricerca Corrente and 5xmille) is gratefully acknowledged.
Ethics
The study was approved by the Ethics Committee of reference, i.e., Ethics Committee of IRCCS San Raffaele (Milan, Italy); protocol code “CardioRetro” number 122/int/2017 approved on September 14, 2017 and amended on July 18, 2019. Informed consent was waived by the Ethics Committee because of the retrospective nature of the study and the analysis of anonymized images. The study was conducted according to the principles outlined in the Declaration of Helsinki.
Data Availability
Data and support material are available as part of the electronic Supplementary Material.
Supplementary Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.