Abstract
Keywords
Introduction
Lifelong yearly imaging surveillance is recommended for patients who undergo endovascular aneurysm repair (EVAR), since 22% of EVAR patients develop complications that require one or more reinterventions at some time during follow-up. 1 Unfortunately, yearly imaging surveillance may also harm patients and places a burden on the health care system.
The clinical practice guidelines from the Society for Vascular Surgery recommend that all EVAR patients should undergo computed tomography angiography (CTA) 1 month after EVAR. If the first CTA does not show any abnormalities, the next CTA is planned after 1 year. Then, if the 1-year CTA shows no abnormalities, the yearly CTA may be replaced by duplex ultrasonography (DUS). However, CTA should still be performed at least once every 5 years. 2
Studies have shown that yearly CT surveillance increases the attributable lifetime cancer risk due to repetitive radiation exposure, as well as nephropathy due to contrast exposure.3,4 Even if CT surveillance is replaced by DUS, patients still experience the burden of additional hospital visits and anxiety about a potentially negative outcome. 5
Lifelong yearly imaging surveillance also places a burden on health care costs and resources. 6 At present this is even more pertinent since 77% of elective patients with an abdominal aortic aneurysm (AAA) undergo EVAR instead of open repair. 7 In the Netherlands alone, each year 2000 patients undergo EVAR and thus each year 2000 additional patients require annual CT or DUS imaging. 7
Currently, it is unclear whether patients actually benefit from yearly imaging surveillance. Thus, a systematic review was undertaken of the available evidence on the effect of imaging surveillance compliance on reintervention rate and mortality after EVAR. In addition, the percentage of reinterventions that were performed for complications detected through imaging surveillance was compared with the percentage of reinterventions performed for complications detected through patients presenting with symptoms.
Materials and Methods
This systematic review and meta-analysis were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 8 The review protocol was registered in the PROSPERO international prospective register of systematic reviews (CRD42017080494).
Eligibility Criteria
Studies were eligible if the authors reported on patients with an infrarenal AAA who had undergone EVAR. In these studies, authors had to directly compare the reintervention rates or all-cause mortality between patients who were compliant and those who were partially or noncompliant to standard imaging surveillance after EVAR. In addition, studies were included in which authors reported the percentage of reinterventions for complications detected through surveillance imaging vs sequelae detected through patient symptoms. Randomized clinical trials (RCTs) and cohort studies were eligible.
Studies that primarily focused on patients with connective tissue disease were excluded, as these patients may require a different imaging surveillance strategy due to differences in etiology and a higher risk of developing other vascular-related problems. 9 Studies about the replacement of CTA with DUS or magnetic resonance imaging (MRI) were also excluded, since the aim of this review was not to study the best imaging technique for surveillance but the overall effect of compliance to yearly imaging surveillance, regardless of the imaging technique used. Finally, studies were ineligible if EVAR was performed for a diagnosis other than AAA, such as an isolated iliac aneurysm or aortic stenosis.
Information Sources and Search
The search strategy was constructed according to the Patient-Intervention-Comparison-Outcome framework. MeSH terms and synonyms were searched in text words for AAA, endovascular repair, surveillance, imaging techniques, reinterventions, and mortality; the search was not restricted based on publication period or language. The full search strategy is available in the Appendix. The last search was run in the MEDLINE, Embase, and Cochrane Central databases on October 10, 2018. Cross-referencing of systematic reviews and included studies was done to identify additional studies. In addition, the American, European, and Dutch clinical trial registries were consulted to identify recently completed studies.
Study Selection
Two review authors (S.dM, A.G.) independently screened titles and abstracts of the publications identified using the search strategy. Subsequently, they independently confirmed eligibility based on the full-text papers. Disagreements were resolved by discussion and, if necessary, by asking another coauthor to act as an arbiter.
The search identified 1434 articles; an additional 6 articles were found by screening systematic reviews. After removing duplicates, 1020 titles and abstracts were screened, of which 44 publications underwent full-text screening. Twenty-five articles were excluded because, despite reporting on compliance, no comparison was performed between compliant and non- or partially compliant patients. In addition, 8 conference abstracts, guidelines, and commentaries were removed. Eleven articles11–21 were selected for data extraction, of which 8 studies were eligible for comparative meta-analysis (Figure 1).
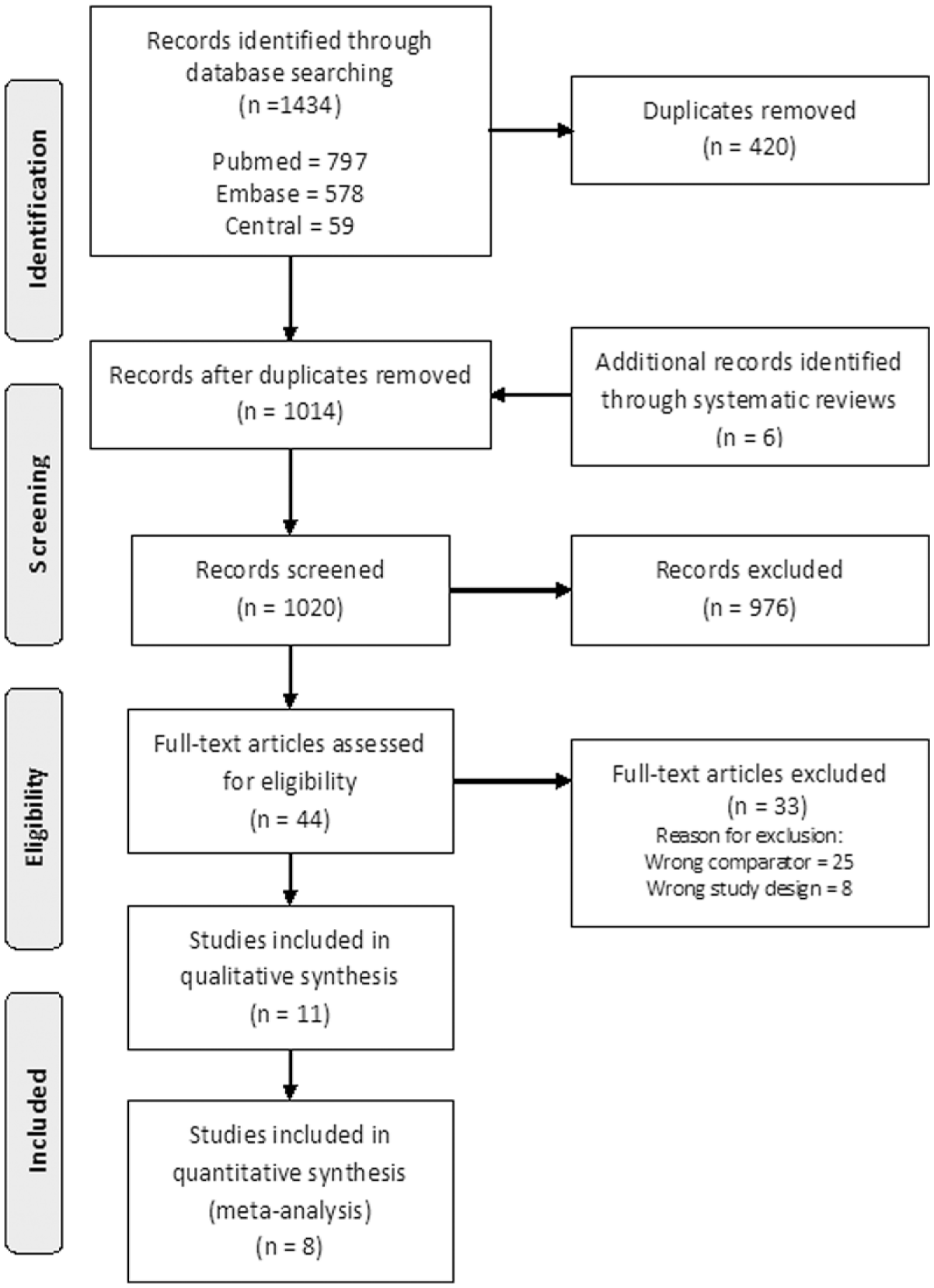
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Data Collection
Using a predefined data extraction form, the review authors independently extracted the following data from text, tables, and/or Kaplan-Meier curves (wherever possible): first author, publication date, study design, patient population, number of patients, standard surveillance protocol, definitions of surveillance subgroups, and follow-up duration. The abstractors also collected the number of patients per surveillance subgroup (compliant, partially compliant, noncompliant, and lost to follow-up); the overall number of reinterventions per subgroup; and the overall number of deceased patients per subgroup (all-cause mortality). As secondary outcomes, aneurysm-related mortality and the number of secondary AAA ruptures per subgroup were recorded. In addition, the number of reinterventions performed for complications detected through imaging surveillance and the number of reinterventions performed for complications detected when patients presented with symptoms were collected. If the articles used for data collection did not present the relevant outcomes in absolute numbers or percentages, the authors of these articles were contacted to obtain these data. Any discrepancies were resolved by discussion and, if necessary, by consulting the arbiter.
For this analysis, 2 subgroups were created: compliant patients and a composite of partially compliant, noncompliant, and lost to follow-up (called the partially/noncompliant subgroup) according to the definitions used in each study. If reinterventions and mortality were reported separately for partially compliant, noncompliant, and lost to follow-up patients, the absolute numbers were summed.
Risk of Bias
Two authors independently evaluated the methodological quality of included studies using risk of bias tools as specified by the Dutch Cochrane collaboration. 10 The appropriate tools for RCTs or for cohort and case-control studies were employed. Risk of bias across studies was examined by evaluating the symmetry of funnel plots.
Statistical Analyses
Summary statistics are given as the median [interquartile range (IQR) Q1, Q3]. The results were synthesized using meta-analytical tools (Review Manager; version 5.3; The Cochrane Collaboration, Copenhagen, Denmark) to compare the rates of reintervention, secondary rupture, all-cause mortality, and aneurysm-related mortality between patients who were compliant and those who were partially/noncompliant to standard imaging surveillance after EVAR. If statistical heterogeneity was limited, ie, if I2 was <50%, a Mantel and Haenszel fixed effect model was used. If statistical heterogeneity was present (I2 >50%), a Dersimonian and Laird random effects model was employed. Results are presented as risk differences (RD) with 95% confidence intervals (CI) and number needed to harm (NNH). Additional analyses were performed to study the percentage of reinterventions performed for complications detected through imaging surveillance vs those performed owing to symptom presentation.
Results
Study Characteristics
The 11 articles encompassing 21,838 patients were all retrospective cohort studies (Table 1).11–21 Nine studies (n=20,920) compared compliant patients to partially or noncompliant patients.11–19 Two studies (n=832) reported the percentage of complications requiring reintervention detected via imaging surveillance and patients presenting with symptoms.20,21 The imaging surveillance protocols differed among studies (Table 1). Protocols of more recent studies recommended less imaging in the first postoperative year.11,14,17–19 Two studies did not provide detailed information about their imaging surveillance protocol.12,13 All surveillance protocols advised the use of CT.11–21 Eight studies also advised the use of DUS,11–13,15,16,18,19,21 although in 2 of these studies18,19 DUS was allowed only if the first postoperative CT was normal. The median follow-up was 31.7 months (IQR 29.8, 49.3).
Characteristics of Included Retrospective Cohort Studies.
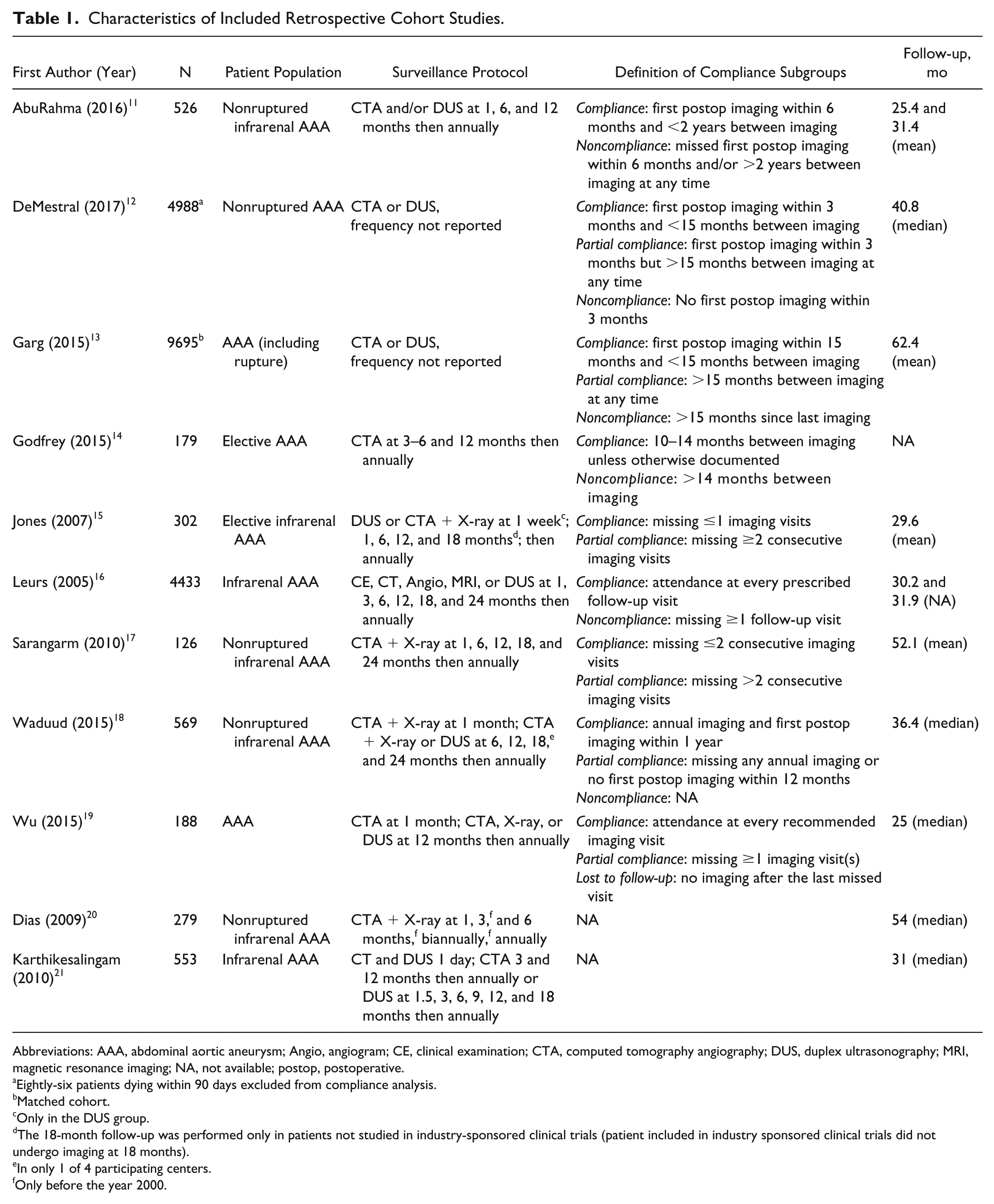
Abbreviations: AAA, abdominal aortic aneurysm; Angio, angiogram; CE, clinical examination; CTA, computed tomography angiography; DUS, duplex ultrasonography; MRI, magnetic resonance imaging; NA, not available; postop, postoperative.
Eightly-six patients dying within 90 days excluded from compliance analysis.
Matched cohort.
Only in the DUS group.
The 18-month follow-up was performed only in patients not studied in industry-sponsored clinical trials (patient included in industry sponsored clinical trials did not undergo imaging at 18 months).
In only 1 of 4 participating centers.
Only before the year 2000.
The definitions of compliance subgroups varied between studies (Table 1). In 5 studies,15–19 the definition of compliance depended on the number of consecutive imaging visits missed (0–3). Four studies defined compliance using a maximum amount of time between imaging visits (10–24 months).11–14 In 4 studies,11–13,18 compliance was also defined using a time restriction regarding the first postoperative CTA or DUS (3–15 months).
Risk of Bias
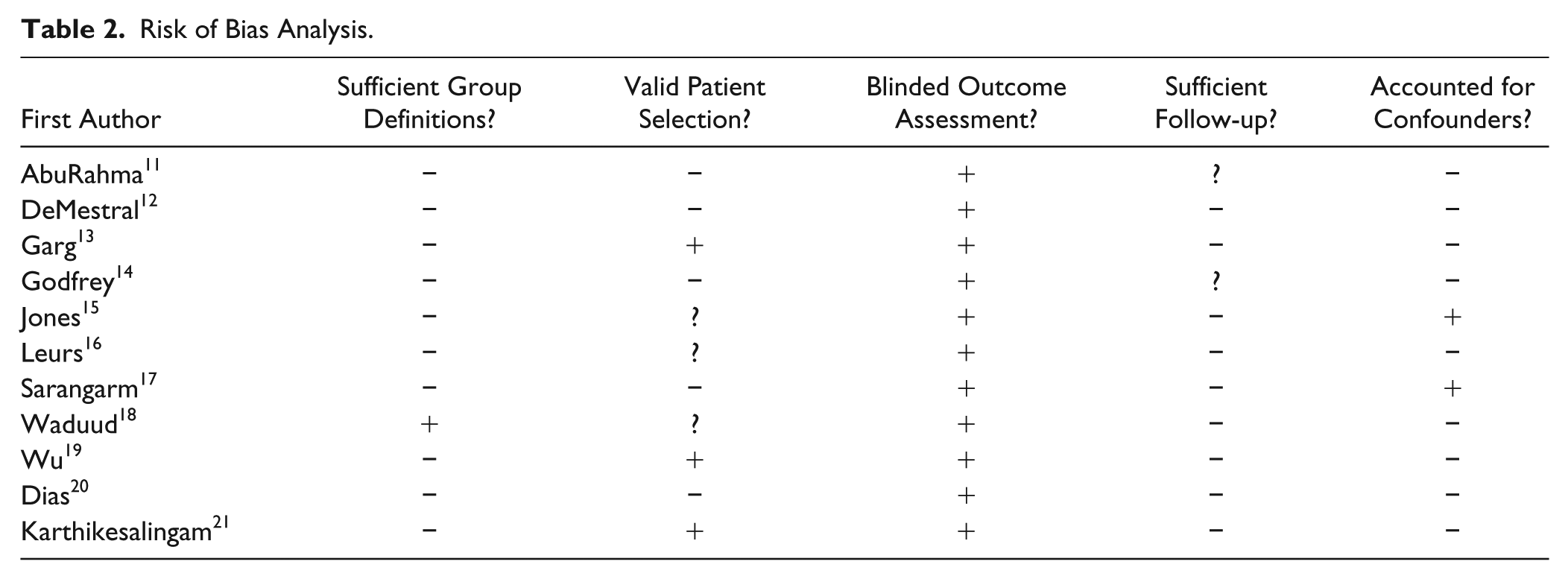
Overall risk of bias was low (Table 2). Ten11–17,19–21 of the 11 studies provided clear definitions of their reported subgroups. Five studies11,12,14,17,20 included only patients with asymptomatic AAA, which was scored as a low risk of bias. Three studies13,19,21 included both asymptomatic and ruptured AAA patients, which was scored as a high risk of bias. In 3 other studies15,16,18 it was unclear if ruptured AAA was included. In addition, 2 studies11,14 did not provide information about the number of excluded patients due to missing data or about the exclusion of more than half of patients for final analysis. Nine studies12,13,15–21 reported a primary outcome, so the adequacy of the duration of follow-up could be assessed.
Risk of Bias Analysis.
Results of Individual Studies
Table 3 shows the number of patients per surveillance subgroup per study and the number of reinterventions and deaths per surveillance subgroup per study. Of the total 20,920 patients in the compliance analysis, 9637 (46%) were compliant with imaging surveillance, 7531 (36%) patients were imperfectly compliant, and the remaining 3752 (18%) were noncompliant or lost to follow-up. The overall reintervention rate of studies presenting data on reintervention11,13–17,19 was 5%. The overall mortality of studies presenting absolute numbers for mortality11,13–19 was 31%. Absolute data were not available from the DeMestral study. 12
Outcomes According to Compliance With Surveillance Imaging Protocol.
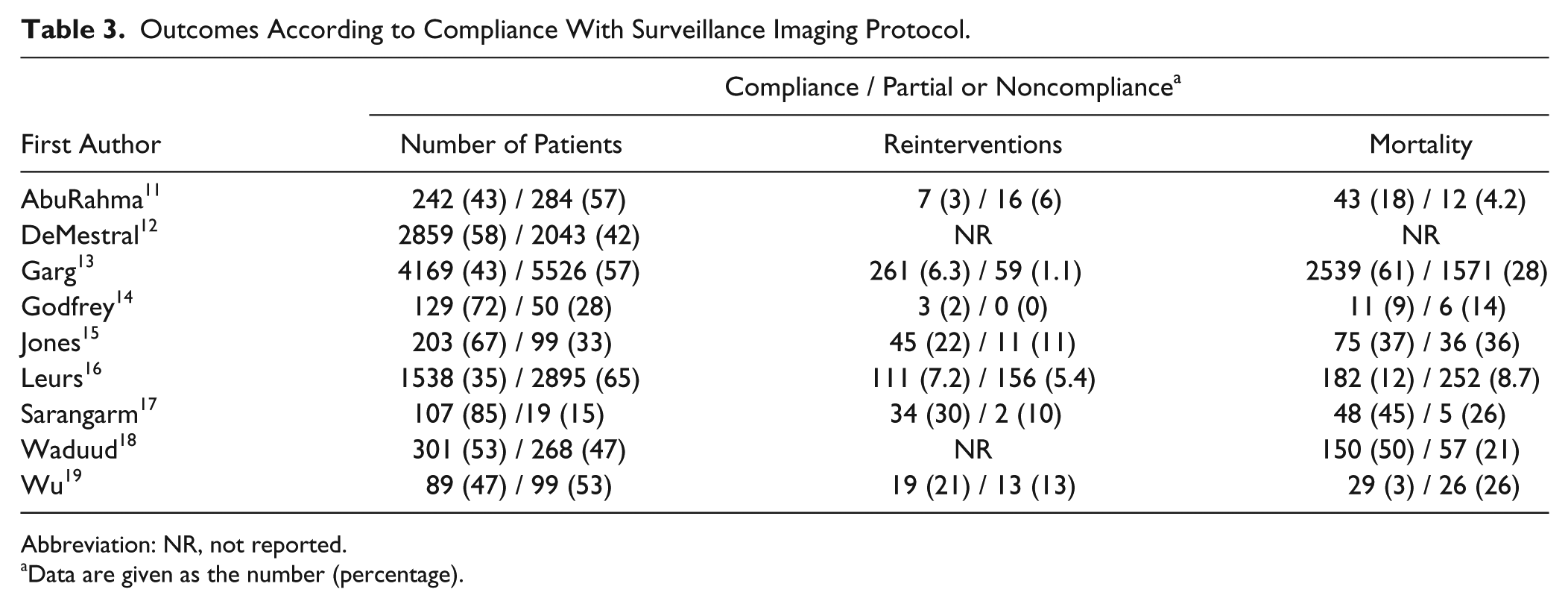
Abbreviation: NR, not reported.
Data are given as the number (percentage).
Synthesis of Results
Reintervention rates from 7 studies11,13–17,19 were pooled, and the compliant subgroup and the combined partially/noncompliant subgroup were compared (Figure 2A). The overall reintervention rate in the compliant subgroup was 480 (7%) of 6477 patients vs 257 (3%) of 8965 patients in the partially/noncompliant subgroup. Meta-analysis showed a statistically significantly higher reintervention rate in the compliant subgroup, with a RD of 4% (95% CI 1% to 7%), which implies an NNH of 25 (95% CI 14 to 100). The risk of bias assessment across studies reporting a reintervention rate produced an asymmetrical funnel plot (Figure 4A).
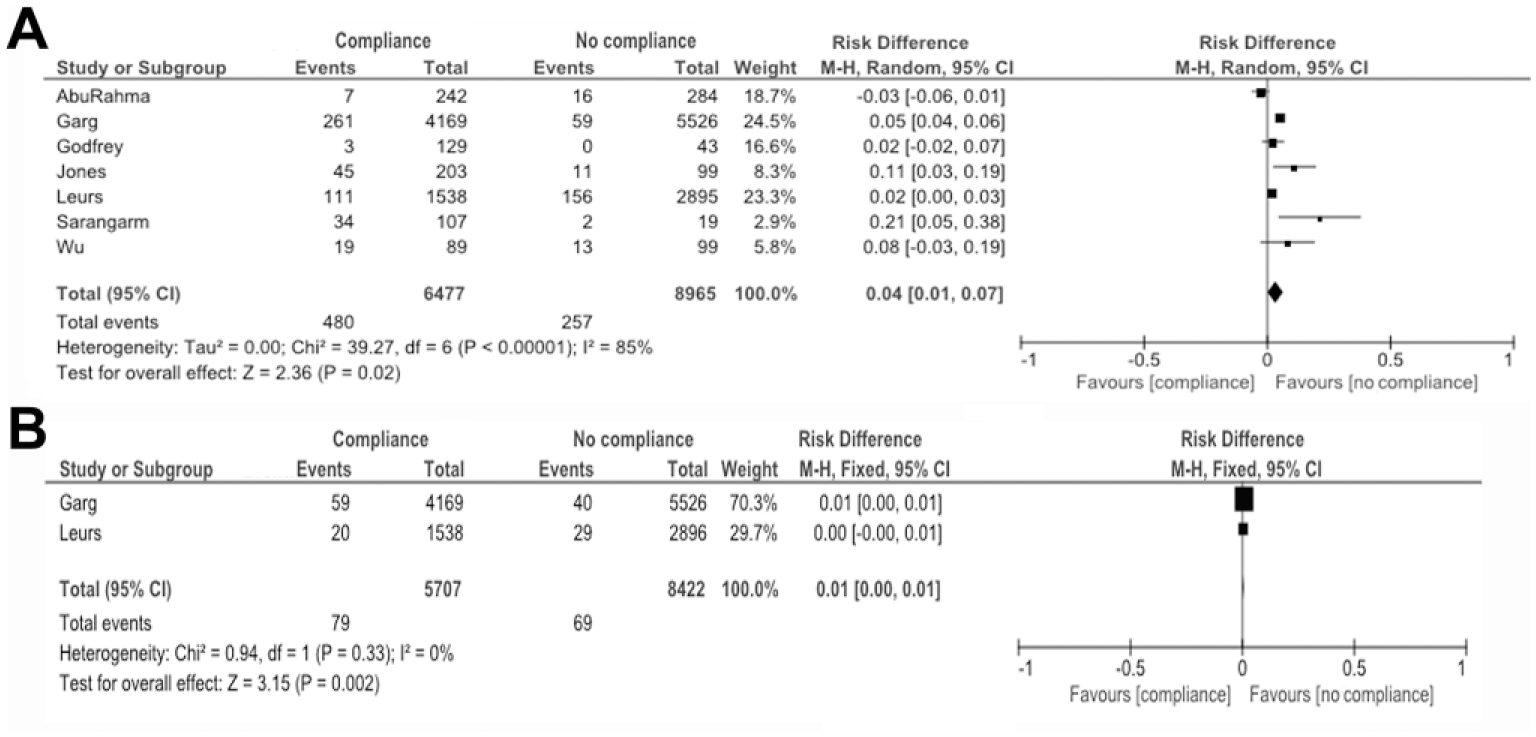
Forest plots of (A) reintervention rates and (B) secondary ruptures in compliant vs partially/noncompliant (“no compliance”) patients.
The number of secondary ruptures per compliant and partially/noncompliant subgroups were reported in 2 studies.13,16 Meta-analysis of these 2 studies showed a statistically significant higher secondary rupture rate in the compliant subgroup (Figure 2B), with a RD of 1% (95% CI 0% to 1%).
Figure 3A shows the forest plot of the 8 studies11,13–19 reporting overall mortality, comparing the compliant subgroup and the partially/noncompliant subgroup. The overall mortality in the compliant subgroup was 45% (3077/6778) vs 21% (1965/9233) in the partially/noncompliant subgroup. Meta-analysis did not show a statistically significant RD between these subgroups [12% (95% CI −2% to 26%)]. The risk of bias assessment across studies reporting mortality produced a fairly symmetrical funnel plot (Figure 4B).
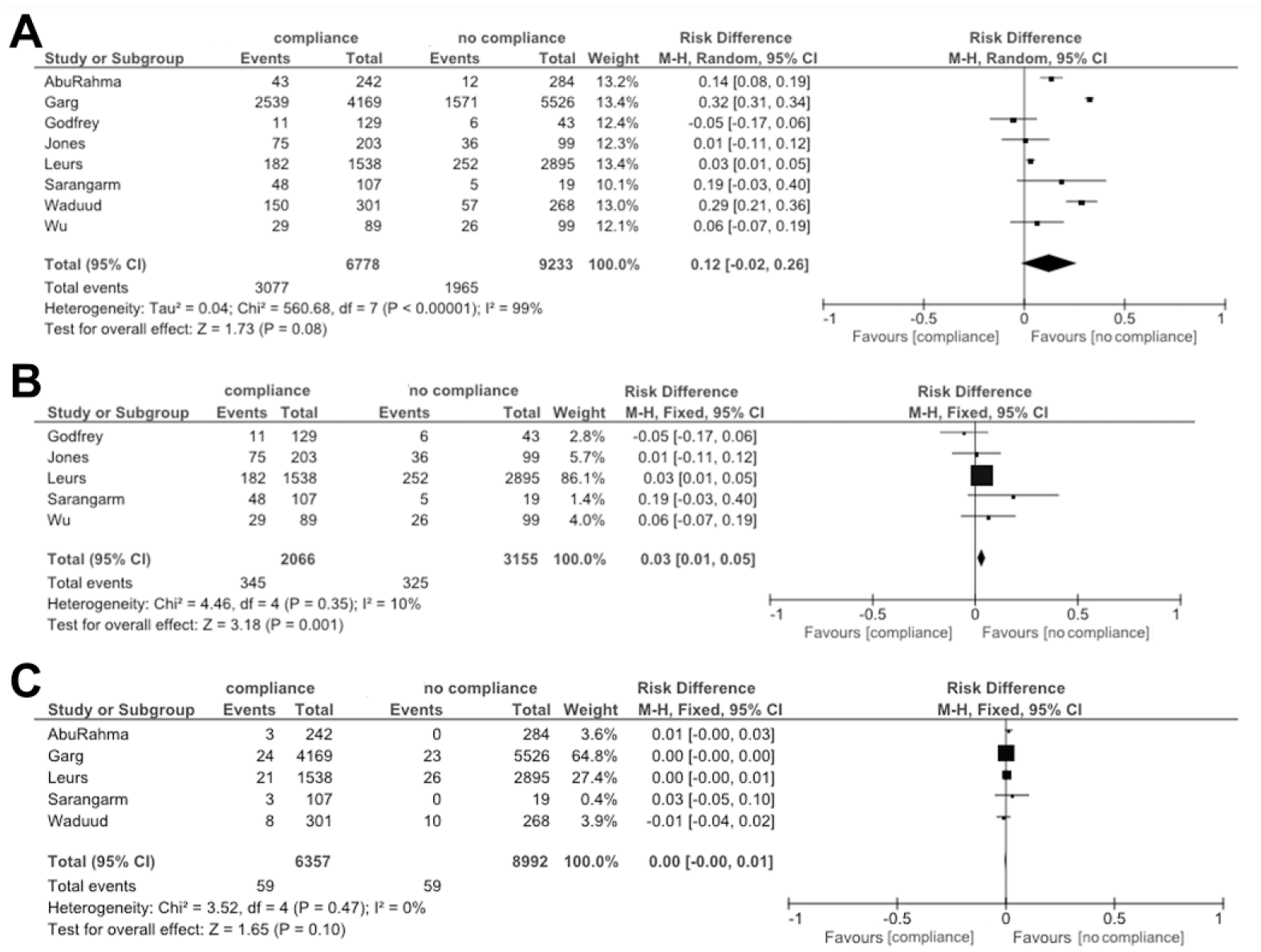
Forest plots of (A) all-cause mortality (all studies), (B) all-cause mortality (studies based on the compliance definition), and (C) aneurysm-related mortality in compliant vs partially/noncompliant (“no compliance”) patients.
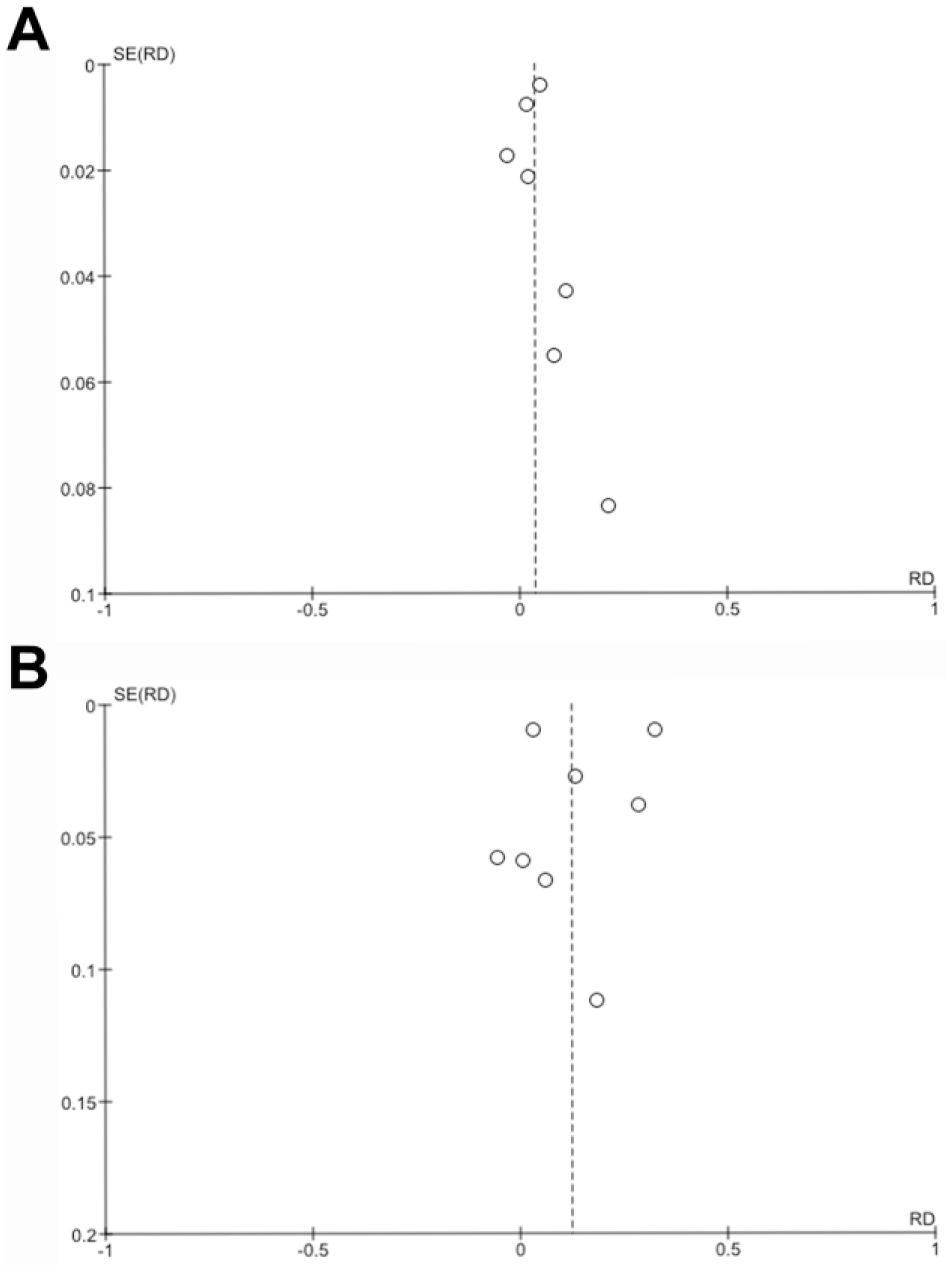
Funnel plots of (A) reintervention rates and (B) mortality in compliant vs partially/noncompliant patients. RD, risk difference, SE, standard error.
In an attempt to reduce heterogeneity among studies based on definitions of compliance, 3 studies11,13,18 were excluded that incorporated patients who missed their initial postoperative imaging as part of their definition of partial/noncompliance. Meta-analysis of the 5 remaining studies14–17,19 showed a statistically significant difference between the groups [RD 3% (95% CI 1% to 5%)] in favor of the partially/noncompliant group (Figure 3B).
Five studies11,13,16–18 reported the number of aneurysm-related deaths per compliant and partially/noncompliant subgroups. The aneurysm-related mortality in the compliant subgroup was 0.9% (59/6357) vs 0.6% (59/8992) in the partially/noncompliant subgroup. Meta-analysis of these 5 studies showed no difference between the subgroups (Figure 3C), with a RD of 0% (95% CI −0% to 1%).
Additional Analysis
Dias et al 20 reported that 41 (53%) of 78 reinterventions performed in 57 patients were for complications detected through imaging surveillance, 23 (29%) via patients presenting with symptoms, and the remaining 14 (18%) reinterventions were performed within 30 days after EVAR, but whether they were due to patient symptoms or were planned based on the primary repair was not noted. Karthikesalingam et al 21 reported that 28 (41%) of 69 patients required one or more reinterventions for complications detected via imaging surveillance; sequelae requiring reintervention because of symptoms were detected in 41 (59%) patients.
Discussion
This systematic review showed that compliance to post-EVAR imaging surveillance appears not to protect against secondary rupture or mortality compared to partial/noncompliance during a median of 31.7 months’ follow-up even though more reinterventions were performed in the compliance subgroup. The conclusions of this meta-analysis are in agreement with the publication by Grima et al. 22
A recently published observational studies–based systematic review by Zaiem et al 23 stated that for surveillance to be justified there needs to be diagnostic accuracy, detection of important prevalent findings, and an effective intervention. Since complications requiring reintervention were observed at all imaging surveillance visits (1, 6, 12, 24, 36, 48, and 60 months) after EVAR, there appeared to be a rationale for standard imaging surveillance. However, when interpreting our findings, perhaps not all complications detected by imaging surveillance are important. There appears to be overtreatment of complications detected by imaging surveillance, since the additional reinterventions did not increase survival. These reinterventions per se may even cause new complications that will reduce the patients’ quality of life or result in death. In addition, 29% to 59% of the complications requiring reintervention were detected due to the patient presenting with symptoms in between imaging surveillance visits. Perhaps survival may improve by treating symptomatic complications only. Unfortunately, this outcome measure was reported in only 2 studies.20,21
Another important reason that may prevent compliance to standard imaging surveillance to protect against mortality is something learned from the UK EVAR trials. In these trials, over 90% of EVAR patients died from non-aneurysm-related causes, such as coronary artery disease and cancer. 24 This small percentage of patients who die from aneurysm-related deaths was also noticeable in this systematic review, where aneurysm-related deaths occurred in <1% of the patients, without any differences between subgroups.
The long-term outcome of the Dutch Randomised Endovascular Aneurysm Repair (DREAM) trial showed that most reinterventions were performed in the first few months after EVAR and again at 4 years after EVAR. 25 In addition, some studies suggest that a normal first postoperative CTA or DUS may be predictive of a low reintervention risk within the first 3 years of follow-up.5,26,27 Other studies, however, highlight the importance of preoperative anatomy in predicting the reintervention risk, for instance by applying the St George’s Vascular Institute Score, or by treating patients according to instructions for use (IFU).28,29
Based on this information, it may be feasible to reduce the frequency of imaging surveillance in patients undergoing EVAR for an asymptomatic infrarenal AAA, provided that they are treated inside the IFU and the first postoperative imaging shows no abnormalities. Larger cohorts of retrospective studies with prolonged follow-up should be designed to evaluate the effect of the normal first postoperative imaging on reintervention rate and mortality to see if secondary imaging can safely be postponed. A prospective study to generate more evidence is likely to take too long, considering the accelerating strain on the health care system.
Future studies should also focus on the psychological burden vs the benefit of imaging surveillance in patients. Anxiety surrounding imaging surveillance, also known as “scanxiety,” has been found to be present in cancer patients. 30 However, in patients with AAA this has not yet been studied. The presence of anxiety or, perhaps in contrast, the secure feeling that no abnormalities were found, may play an important part in the discussion about imaging surveillance frequency.
Limitations
Limitations of this study are the heterogeneity of the available studies regarding imaging surveillance protocols and the definitions of compliance and follow-up duration. This heterogeneity may have also caused the asymmetry of the funnel plot for mortality, rendering it difficult to evaluate the reporting bias. However, when looking at individual studies, all studies, except the third smallest study of Godfrey et al, 14 showed a trend toward decreased mortality in the partially/noncompliant subgroup. In addition, there was a statistically significant difference in favor of partial/noncompliance when attempting to reduce heterogeneity between study designs based on definitions of compliance.
Another limitation may be the presence of bias in patients who were compliant to imaging follow-up and patients who were partially or noncompliant. On the one hand, noncompliant patients may have been patients who would have had multiple consecutive normal imaging results, making them less concerned about returning for follow-up. Patients with abnormal imaging results may have been more inclined to return for a follow-up visit. This may also be the reason why secondary ruptures occur more frequently in the compliant subgroup. Unfortunately, the included studies did not provide information about the imaging results prior to secondary rupture in the compliant subgroup. In addition, patients with multiple comorbidities may be more compliant, since they undergo follow-up for other diseases, but they do have an increased mortality risk. On the other hand, patients with multiple comorbidities may also lack the mobility to attend multiple follow-up visits. Unfortunately, this information was not mentioned in the included studies. Likewise, it was not possible to study whether the presence of hostile neck anatomy in the compliant vs the partially/noncompliant subgroups directly affected reintervention rates and mortality. More important, if we look closely at the definitions used for partial or noncompliance, patients who miss one imaging visit are forever judged as partially/noncompliant, even if this was the only visit they missed. This was also the main concern of DeMestral et al, 12 which is why they refrained from reporting crude mortality per subgroup. Consensus on a definition for compliance and noncompliance to imaging surveillance seems a necessary requirement for comparisons of these subgroups in future studies.
Conclusion
Compliance to imaging surveillance after EVAR appears not to protect against mortality. Imaging surveillance may even lead to unnecessary reinterventions, which can cause complications. In some cases reintervention would have taken place irrespective of compliance to surveillance, since imaging was performed because the patient presented with symptoms. Apparently, yearly imaging surveillance is not beneficial to all EVAR patients.
Footnotes
Appendix
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the AMC Foundation, which was not involved in the study design, data analysis, or interpretation of results.
