Abstract
African American (AA) churches are valuable partners in implementing health promotion programming (HPP) to combat health disparities. The study purpose was to evaluate AA church characteristics associated with enrollment into the FAITH! (Fostering African American Improvement in Total Health) Trial, a community-based, cluster randomized controlled trial (RCT) of a mobile health intervention for cardiovascular health promotion among AA churches. Churches located in Minneapolis-St. Paul and Rochester, Minnesota were invited to complete an electronic screening survey and follow-up telephone interview including the PREACH (Predicting Readiness to Engage African American Churches in Health) tool to assess church characteristics and infrastructure for HPP. The primary outcome was church enrollment in the FAITH! Trial. Key predictors included overall PREACH scores and its subscales (Personnel, Physical Structure, Faith-based Approach, Funding), congregation size, and mean congregation member age. Of the 26 churches screened, 16 (61.5%) enrolled in the trial. The enrolled churches had higher overall mean PREACH scores (36.1 vs. 30.2) and subscales for Personnel (8.8 vs. 5.6), Faith-based Approach (11.0 vs. 9.6), and Funding (7.3 vs. 4.8) compared with non-enrolled churches; all differences were not statistically significant due to small sample size. Twelve (75.0%) of the enrolled churches had >75 members versus six (60.0%) of the non-enrolled churches. Twelve (80.0%) of the enrolled churches had an average congregation member age ≤54 years versus six (67.0%) of the non-enrolled churches. AA churches enrolling into a community-based RCT reported greater infrastructure for HPP, larger congregations, and members of younger age. These characteristics may be helpful to consider among researchers partnering with AA churches for HPP studies.
Keywords
Assessment of Needs
African Americans (AAs) remain largely underrepresented in clinical trials, perpetuating health disparities (Julian McFarlane et al., 2021). AA churches are trusted organizations within the AA community, and their successful collaborations with academic institutions to conduct health promotion programming (HPP) and increase AA representation in clinical trials are well documented (Julian McFarlane et al., 2021; Maxwell et al., 2019). Fewer studies have investigated church characteristics that predict their readiness and capacity for HPP (Maxwell et al., 2019). Initial findings reveal that larger congregations, funding for HPP, paid staff, and health ministries are associated with HPP delivery (Maxwell et al., 2019).
Although prior studies have evaluated church characteristics for HPP participation and research, none have examined these qualities in the context of the COVID-19 pandemic which exacerbated disparities and limited typical church activities. In addition, no studies have examined church readiness to partner with researchers in implementing a non-face-to-face, mobile health (mHealth) intervention within a randomized controlled trial (RCT). The objective of the current study was to identify church characteristics associated with enrollment into the FAITH! (Fostering African American Improvement in Total Health) Trial, a community-based, cluster RCT of an mHealth intervention for cardiovascular health (CVH) promotion among AA churches in Minnesota (Brewer et al., 2022).
Description of Strategy/Innovation
Study Design/Participants
Employing a community-based participatory research (CBPR) approach, the FAITH! study team, the FAITH! Community Steering Committee, and AA church partners co-designed an innovative mHealth intervention (FAITH! App), which integrates personalized educational content and CVH promotion messaging (Brewer et al., 2022). Participants were recruited to the FAITH! Trial from partnering churches and randomized by church into a two-arm cluster RCT with immediate and delayed intervention groups. The current study incorporated a cross-sectional survey and semi-structured interviews to assess the characteristics of AA churches in Minneapolis-St. Paul (MSP) and Rochester, Minnesota associated with enrollment in the FAITH! Trial (Brewer et al., 2022).
Recruitment/Procedures
To recruit potential churches, in-person events initially occurred at health centers in MSP and Rochester (December 2019) but were transitioned to a virtual platform (Zoom; June 2020) due to the COVID-19 pandemic (SUPPLEMENTAL FILES I-III). Furthermore, in alignment with CBPR, the study team pivoted its activities to address the impact of the pandemic on local AA churches with emergency preparedness support and on AA communities with SARS-CoV-2 testing (Brewer et al., 2022). Recruitment activities were also postponed due to racial/social unrest that ensued in MSP following the death of Mr. George Floyd. Ultimately, churches were invited to complete the PREACH (Predicting Readiness to Engage African American Churches in Health) assessment tool by electronic screening survey and follow-up telephone interview (Brand & Alston, 2018). Churches completing the screening survey received a health ministry starter kit by mail (“Church Health Center. Congregational Health Promoters Leader Guide. Accessed March 26, 2022. https://churchhealth.org/for-congregations/church-health-reader-archive/”). Churches meeting inclusion criteria (SUPPLEMENTAL FILE IV) and enrolling in the FAITH! Trial received a US$250 gratitude incentive.
Measures
The PREACH tool assesses the aspects of church infrastructure that best predict church readiness to engage in HPP (Brand & Alston, 2018). The tool scores churches in four equally weighted subscales: (1) Funding to assess the amount/diversity of church funding available for HPP, (2) Personnel to enumerate the volunteers/staff available to coordinate programming, (3) Faith-based Approach to assess the degree of cultural/social support (e.g., prayer) incorporation into program delivery, and (4) Physical Structure to evaluate facilities (e.g., classrooms) available for HPP (Brand & Alston, 2018). The PREACH score combined subscale scores into an overall score (possible range, 0–64) which was used to further categorize churches into three stages of HPP capacity: Stage 1 (Limited, score 0–22), Stage 2 (Moderate, score 23–40), and Stage 3 (Substantial, score 41–64; Brand & Alston, 2018). The follow-up telephone semi-structured interview included questions to assess church health-related activities and research involvement (SUPPLEMENTAL FILE V).
Outcomes
The primary outcome was church enrollment in the FAITH! Trial. Key predictors of interest were PREACH scores, stages, and subscales. Additional variables included congregation size, mean congregation member age, and prior participation in research conducted by an academic institution.
Results
Of the 30 churches attending recruitment events, 26 (86.7%) completed the screening survey and 24 (80.0%) completed the follow-up interview (Supplemental Material IV). Of the 26 screened churches, six (23.1%) indicated prior research participation and seven (26.9%) were classified as PREACH Stage 3. After eligibility assessment, 16 (61.5%) enrolled in the FAITH! trial.
No statistically significant associations emerged for PREACH scores and trial enrollment, but there were important trends for differences in characteristics between non-enrolled (n = 10) and enrolled churches (n = 16; Table 1). Enrolled churches had higher overall mean PREACH scores (36.1 vs. 30.2) and higher subscale scores for Personnel (8.8 vs. 5.6), Faith-based Approach (11.0 vs. 9.6), and Funding (7.3 vs. 4.8) compared with non-enrolled churches. A higher proportion of enrolled churches had congregations larger than 75 members compared with non-enrolled churches (75.0% vs. 60.0%; Table 1). A higher percentage of churches enrolled in the trial had congregations with an average age <55 years than non-enrolled churches (80.0% vs. 66.7%).
African American Church Characteristics (N = 26) a
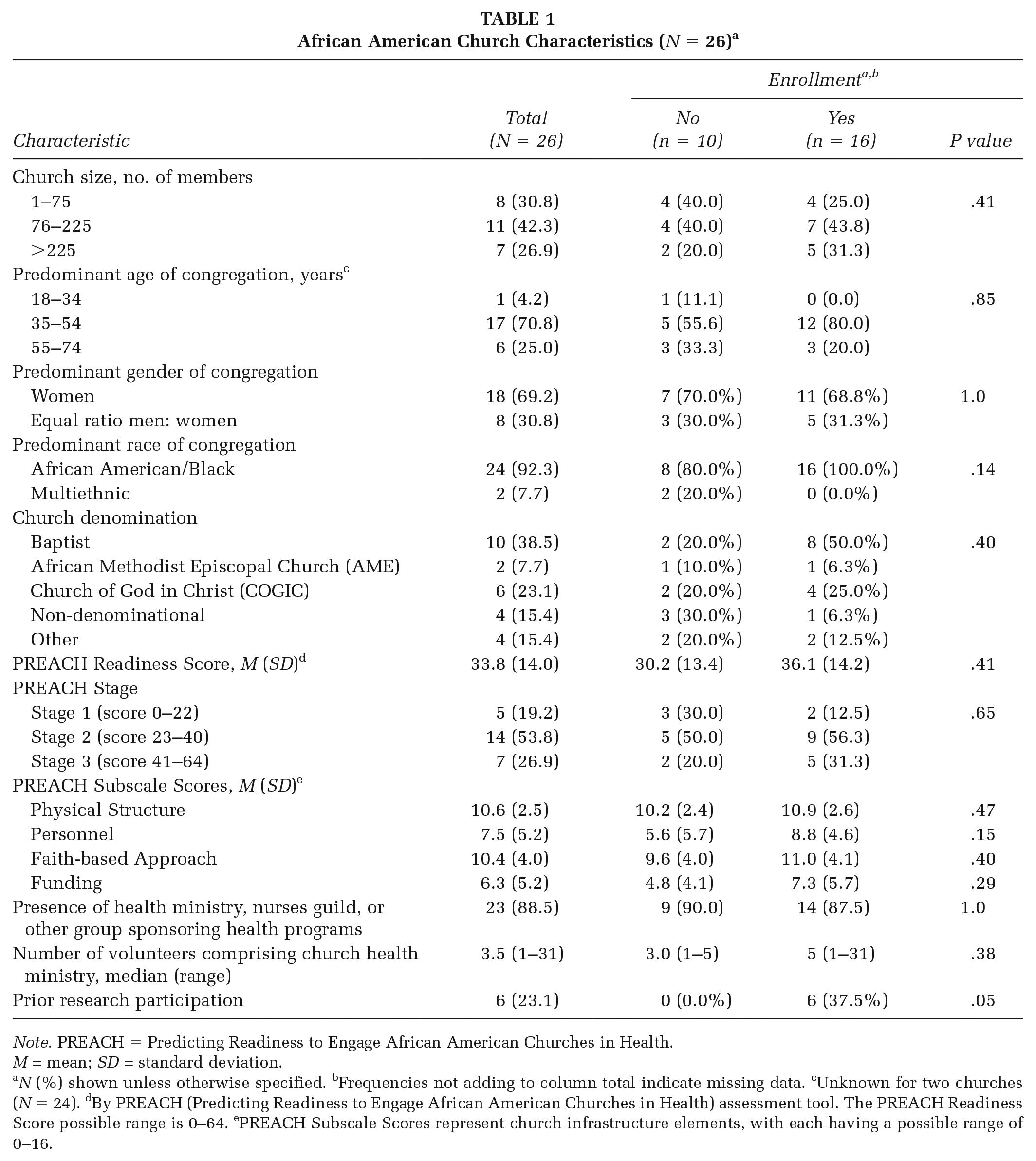
Note. PREACH = Predicting Readiness to Engage African American Churches in Health. M = mean; SD = standard deviation.
N (%) shown unless otherwise specified. bFrequencies not adding to column total indicate missing data. cUnknown for two churches (N = 24). dBy PREACH (Predicting Readiness to Engage African American Churches in Health) assessment tool. The PREACH Readiness Score possible range is 0–64. ePREACH Subscale Scores represent church infrastructure elements, with each having a possible range of 0–16.
Implications for Practice
Our findings suggest that facilitators to church enrollment in a culturally tailored, community-based RCT were greater overall infrastructure, personnel, faith-based approaches, funding for HPP, and having members of younger age. These findings align with existing literature and suggest that researchers seeking to partner with AA churches should provide support to these organizations through adequate personnel and funding, especially to small-sized congregations with older members. Although nominal, our study team provided churches with health ministry starter kits and a monetary incentive to assist them in developing a health ministry. Furthermore, using virtual recruitment strategies and an mHealth intervention instead of in-person programming may have imparted fewer infrastructural barriers for churches. Further study should explore the benefits of mHealth interventions in facilitating CBPR partnerships in limited-resource environments.
Our CBPR-enhanced research strategy also significantly contributed to church recruitment. Few of the churches surveyed had previously participated in research before their involvement in the FAITH! Trial, potentially reflecting hesitancy of AA communities to engage in clinical trials (Julian McFarlane et al., 2021). However, in alignment with our CBPR approach, we prioritized the interests of AA churches in study development/implementation, received guidance from community partners, and provided pandemic-specific support to local AA churches and communities. These steps likely bolstered enrollment and enhanced innovation to overcome pandemic-related barriers. Researchers should consider adoption of community-engaged approaches to facilitate greater inclusion of racial/ethnic minorities in clinical trials.
Supplemental Material
sj-pptx-1-hpp-10.1177_15248399221118394 – Supplemental material for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic
Supplemental material, sj-pptx-1-hpp-10.1177_15248399221118394 for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic by Kristene Tadese, Sarah Jenkins, Dawn Aycock, Clarence Jones, Sharonne N. Hayes, Lora E. Burke, Lisa A. Cooper, Christi A. Patten and LaPrincess C. Brewer in Health Promotion Practice
Supplemental Material
sj-pptx-2-hpp-10.1177_15248399221118394 – Supplemental material for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic
Supplemental material, sj-pptx-2-hpp-10.1177_15248399221118394 for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic by Kristene Tadese, Sarah Jenkins, Dawn Aycock, Clarence Jones, Sharonne N. Hayes, Lora E. Burke, Lisa A. Cooper, Christi A. Patten and LaPrincess C. Brewer in Health Promotion Practice
Supplemental Material
sj-pptx-3-hpp-10.1177_15248399221118394 – Supplemental material for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic
Supplemental material, sj-pptx-3-hpp-10.1177_15248399221118394 for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic by Kristene Tadese, Sarah Jenkins, Dawn Aycock, Clarence Jones, Sharonne N. Hayes, Lora E. Burke, Lisa A. Cooper, Christi A. Patten and LaPrincess C. Brewer in Health Promotion Practice
Supplemental Material
sj-pptx-4-hpp-10.1177_15248399221118394 – Supplemental material for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic
Supplemental material, sj-pptx-4-hpp-10.1177_15248399221118394 for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic by Kristene Tadese, Sarah Jenkins, Dawn Aycock, Clarence Jones, Sharonne N. Hayes, Lora E. Burke, Lisa A. Cooper, Christi A. Patten and LaPrincess C. Brewer in Health Promotion Practice
Supplemental Material
sj-pptx-5-hpp-10.1177_15248399221118394 – Supplemental material for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic
Supplemental material, sj-pptx-5-hpp-10.1177_15248399221118394 for Factors Facilitating Academic–Community Research Partnerships With African American Churches: Recruitment Process for a Community-Based, Cluster Randomized Controlled Trial During the COVID-19 Pandemic by Kristene Tadese, Sarah Jenkins, Dawn Aycock, Clarence Jones, Sharonne N. Hayes, Lora E. Burke, Lisa A. Cooper, Christi A. Patten and LaPrincess C. Brewer in Health Promotion Practice
Footnotes
Acknowledgements
The FAITH! study team would like to show appreciation to all past and current FAITH! Community Steering Committee members for their unwavering support. Their input has enhanced the impact of the FAITH! Program by virtue of remaining steadfast in our mutual goal to achieve cardiovascular health equity in Minnesota and beyond through community outreach and interventions. A special thanks is extended to Martha Bock for her assistance with organizing our recruitment events and conducting the church interviews.
The research reported herein was supported by the National Institutes of Health (NIH)/National Institute on Minority Health and Health Disparities (NIMHD; Grant No. 1 R21 MD013490-01) and the Clinical and Translational Science Awards (CTSA; Grant No. UL1 TR000135) from the National Center for Advancing Translational Sciences (NCATS) to Mayo Clinic and the Mayo Clinic Center for Health Equity and Community Engagement in Research. Dr. Brewer was supported by the American Heart Association-Amos Medical Faculty Development Program (Grant No. 19AMFDP35040005), NCATS (CTSA Grant No. KL2 TR002379), and the Centers for Disease Control and Prevention (CDC; Grant No. CDC-DP18-1817) during the implementation of this work. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NCATS, NIH, or CDC. The funding bodies had no role in study design; in the collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
