Abstract
Introduction:
The Omnipod® 5 Automated Insulin Delivery (AID) System is the first wearable, on-body, tubeless AID system in the United States. Clinical trials have demonstrated hemoglobin A1C (HbA1c) improvements with Omnipod 5 use. The aim of the study was to evaluate HbA1c changes after initiating Omnipod 5 in a real-world setting.
Methods:
This retrospective study utilized commercial and Medicare Advantage with Part D enrollees’ claims data from the Optum Research Database between August 1, 2021, and December 31, 2023. The study included individuals diagnosed with type 1 diabetes (T1D) or type 2 diabetes (T2D) who initiated Omnipod 5. Participants were required to have continuous enrollment for 12 months pre- and post-initiation and at least two medical claims for diabetes during these periods. Demographic and clinical characteristics were measured at the index date, and HbA1c levels were compared pre- and post-initiation of Omnipod 5.
Results:
The final sample included 2,504 users, with 792 having pre- and post-initiation HbA1c results. The mean age was 54 years; 48% were enrolled in Medicare Advantage, 90% had prior continuous glucose monitoring, and 64% had prior pump therapy. A mean reduction in HbA1c of 0.4% was observed (P < 0.001); mean reductions were 0.4% and 0.5% among those with type 1 and type 2 diabetes, respectively. Among 135 users with pre-initiation HbA1c levels ≥9%, a reduction of 1.4% was noted (P < 0.001). Pre-initiation, 31% achieved the American Diabetes Association (ADA) target of <7% compared to 44% post-initiation (increase of 13%; P < 0.001). For the Healthcare Effectiveness Data and Information Set (HEDIS) target of <8%, 62% of individuals achieved the target pre-initiation compared to 78% post-initiation (increase of 16%; P < 0.001).
Conclusions:
In a real-world setting, Omnipod 5 was associated with reduced HbA1c and an increased proportion of individuals achieving ADA and HEDIS glycemic targets.
Introduction
The 2025 American Diabetes Association Standards of Care in Diabetes support Automated Insulin Delivery (AID) as the preferred insulin delivery method in youth and adults with type 1 diabetes and other types of insulin-deficient diabetes. 1 These guidelines further state that the selection of devices should be individualized based on the person’s preferences and needs. These standards are based on numerous randomized controlled and crossover trials across differing platforms demonstrating the superiority of AID compared to conventional diabetes therapies.2–12
Despite these strong endorsements, real-world adoption of AID remains suboptimal. Data from the T1D Exchange Quality Improvement Collaborative showed that only about one-third of individuals at participating centers were using AID systems as of 2022. 13 This highlights a significant gap between clinical guidelines and actual practice. The under-utilization is particularly concerning given the ADA’s designation of AID as the standard of care, and it underscores the need to address barriers to access and adoption. 14
Several key drivers contribute to this underuse, including insurance-related barriers, perceived difficulties with digital technology, and lack of awareness or support. 1 Addressing these challenges through policy reform, improved awareness about ease of use, and expanded real-world evidence will be essential to increasing equitable access to AID technologies and ensuring that the benefits of these systems are realized across all populations.
The Omnipod® 5 Automated Insulin Delivery System features a wearable, tubeless, waterproof (IP28) insulin pump (the Pod) that contains the dosing algorithm and communicates with a compatible continuous glucose monitor (CGM) to adjust insulin delivery every 5 min based on current and predicted glucose values. It has been cleared by the US Food and Drug Administration and is currently labeled for use in individuals aged 2 years and older with T1D, as well as adults with T2D. Clinical trials have demonstrated improvements in hemoglobin A1C (HbA1c) with the use of Omnipod 5.5,9,15–17 The goal of this study was to evaluate HbA1c changes following the initiation of Omnipod 5 in a real-world setting to provide greater evidence support for expanded access and utilization of AID.
Materials and Methods
Study design
This was a retrospective study of commercial and Medicare Advantage with Part D enrollees using enrollment and claims data from the Optum Research Database (ORD) for the period of August 1, 2021 through December 31, 2023 (study period). Enrollees diagnosed with T1D or T2D who initiated use of the Omnipod 5 were examined for changes in HbA1c pre- and post-initiation. The ORD is Optum’s proprietary administrative claims research database; a large US health care claims database that is de-identified and HIPAA compliant, with fully adjudicated medical and pharmacy claims and linked enrollment information since 1993 on more than 75 million commercial and Medicare Advantage. 18 Outpatient laboratory test results are also available for a subset of individuals in the ORD.
Institutional review board approval or waiver of approval was not required for this study because the study data were secondary and de-identified in accordance with the United States Department of Health and Human Services Privacy Rule’s requirements for de-identification codified at 45 C.F.R. § 164.514(b).
Study population
The study population included individuals having at least one claim specifically for Omnipod 5 during the identification period from August 1, 2022–December 31, 2022. The index date was defined as the date of the first claim within this period. Participants had to be at least 2 years old as of the index year and have 12 months of continuous enrollment prior to the index date (pre-initiation) and 12 months of continuous enrollment starting at the index date (post-initiation). Additionally, consistent with methodologies used in prior claims-based studies of AID systems in T1D and T2D,19,20 participants were required to have at least two medical claims with a diagnosis for T2D, or at least two claims with a diagnosis for T1D, occurring at least 30 days apart at any time from the beginning of the pre-initiation period through the end of the post-initiation period. Patients were classified as having T1D if >50% of the claims with a diabetes diagnosis code indicated T1D; patients were classified as having T2D if ≥50% of the claims with a diabetes diagnosis code indicated T2D.
Individuals having at least one claim for Omnipod 5 during the pre-initiation period or missing demographic information such as unknown age or sex were excluded.
Study measures
User characteristics were measured at the index date and included age, age groups (<18, 18–64, 65+), sex, race/ethnicity (Asian, Black, Hispanic, White, Unknown), insurance type (commercial or Medicare Advantage), and geographic region (Northeast, Midwest, South, West), prior CGM status, prior diabetes therapy, and baseline HbA1c level.
Clinical characteristics were measured during the pre-initiation period and included the Diabetes Complication Severity Index, 21 Charlson Comorbidity Score, 22 diabetes type,23,24 CGM utilization, and prior therapy (insulin pump vs. multiple daily injections [MDI]).
The primary outcomes measured included the HbA1c closest to and prior to the index date as the pre-initiation measure, and the latest HbA1c at least 90 days following the index date as the post-initiation HbA1c. The percentage of users who achieved the HbA1c <7% American Diabetes Association (ADA) guideline target and the percentage of users who achieved the HbA1c <8% Healthcare Effectiveness Data and Information Set (HEDIS) guideline target were calculated.19,25 HbA1c levels were compared pre-initiation to post-initiation, and the change in HbA1c from pre-initiation to post-initiation was measured overall and stratified among users with a pre-initiation HbA1c ≥9%, HbA1c ≥8%, and HbA1c ≥7%. These measures were chosen to incorporate endpoints that are relevant to different audiences. The <7% threshold reflects a key clinical target recommended within the ADA Standards of Care. The <8% threshold is a highly relevant metric for payers, as many health plans in the United States (including all Medicare Advantage plans) are assessed on HEDIS-derived performance measures that include the proportion of their patient population with diabetes who achieve an HbA1c <8%.
Analysis
All study variables were analyzed descriptively. For categorical variables, numbers and percentages (N, %) were presented. For continuous variables, means and standard deviations (SDs) were presented. Pre-initiation and post-initiation comparisons of outcomes used appropriate tests, including McNemar tests for categorical variables and paired t-tests for continuous variables. Statistical significance of P values was based on α = 0.05, two-sided, with no adjustment for multiple comparisons performed.
Results
Demographics and clinical characteristics
A total of 2,504 users were included in the final study sample; of these, 792 had pre-initiation and post-initiation HbA1c results, and of these, 511 users had T1D, and 281 users had T2D (Supplementary Fig. S1). Of the 792 users, the mean (SD) age was 53.5 (16.8) years, 56.1% were female, and 72.9% were White (Table 1). Most users had commercial insurance (52.3%), prior CGM use (90.2%), and prior pump therapy (63.8%). The mean (SD) Charlson comorbidity score was 1.6 (1.8), and the mean (SD) diabetes complication severity score was 2.3 (2.3). Additional characteristics for all users and users with HbA1c at pre-initiation and post-initiation can be found in Table 1.
Demographics and Clinical Characteristics for All Users and Users with HbA1c at Pre-Initiation and Post-Initiation
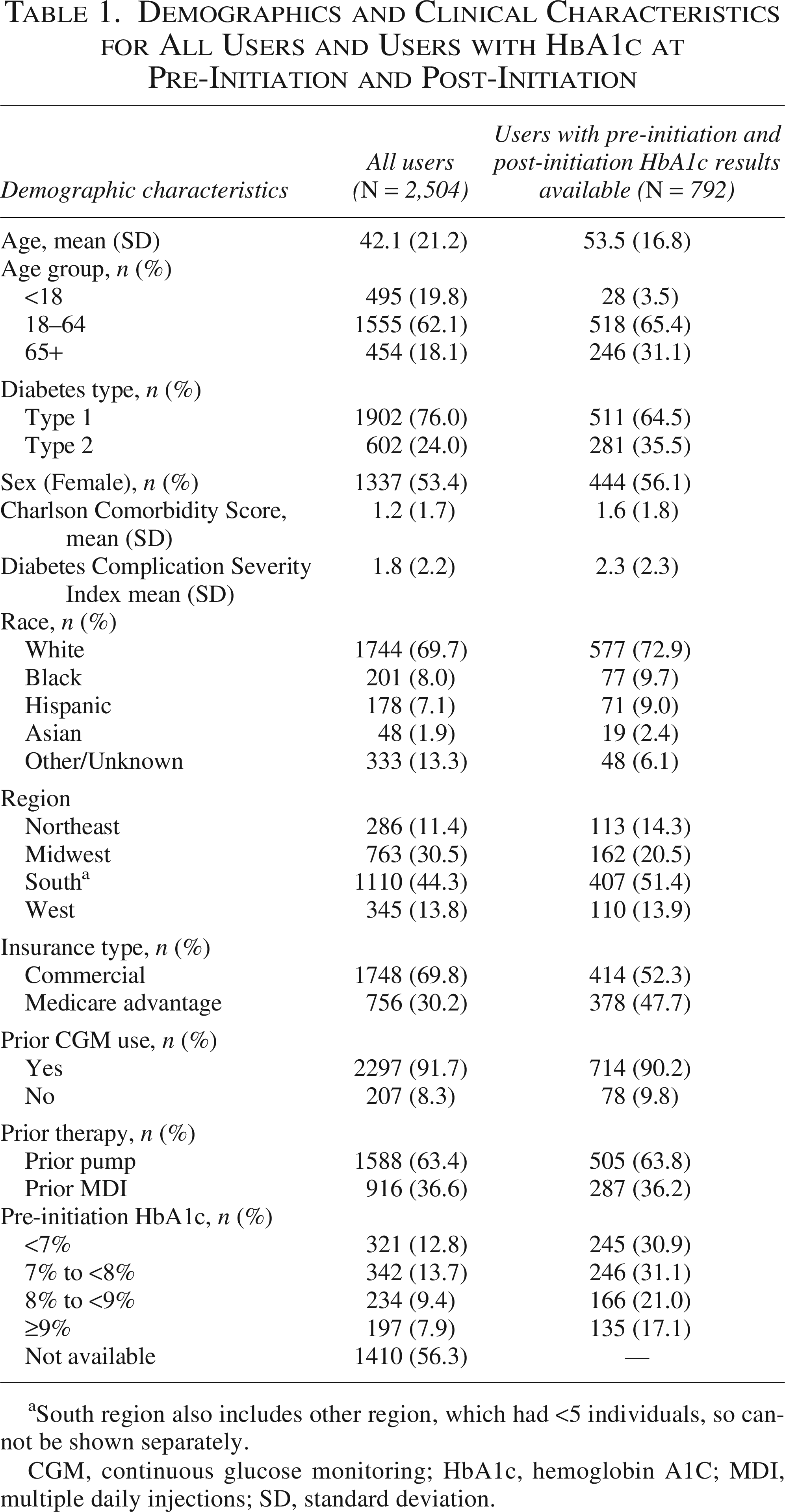
South region also includes other region, which had <5 individuals, so cannot be shown separately.
CGM, continuous glucose monitoring; HbA1c, hemoglobin A1C; MDI, multiple daily injections; SD, standard deviation.
HbA1c results
The HbA1c results are presented for the 792 individuals with pre- and post-initiation HbA1c results.
Mean change in HbA1c for the overall cohort and stratified by pre-initiation HbA1c levels can be found in Figure 1. The mean pre-initiation HbA1c was 7.8% for individuals overall, and a mean reduction of 0.4% was observed (P < 0.001). The mean reduction was statistically significant across the three different HbA1c categories at pre-initiation (P < 0.001), ranging from 1.4% for HbA1c ≥9%–0.7% for HbA1c ≥7%.
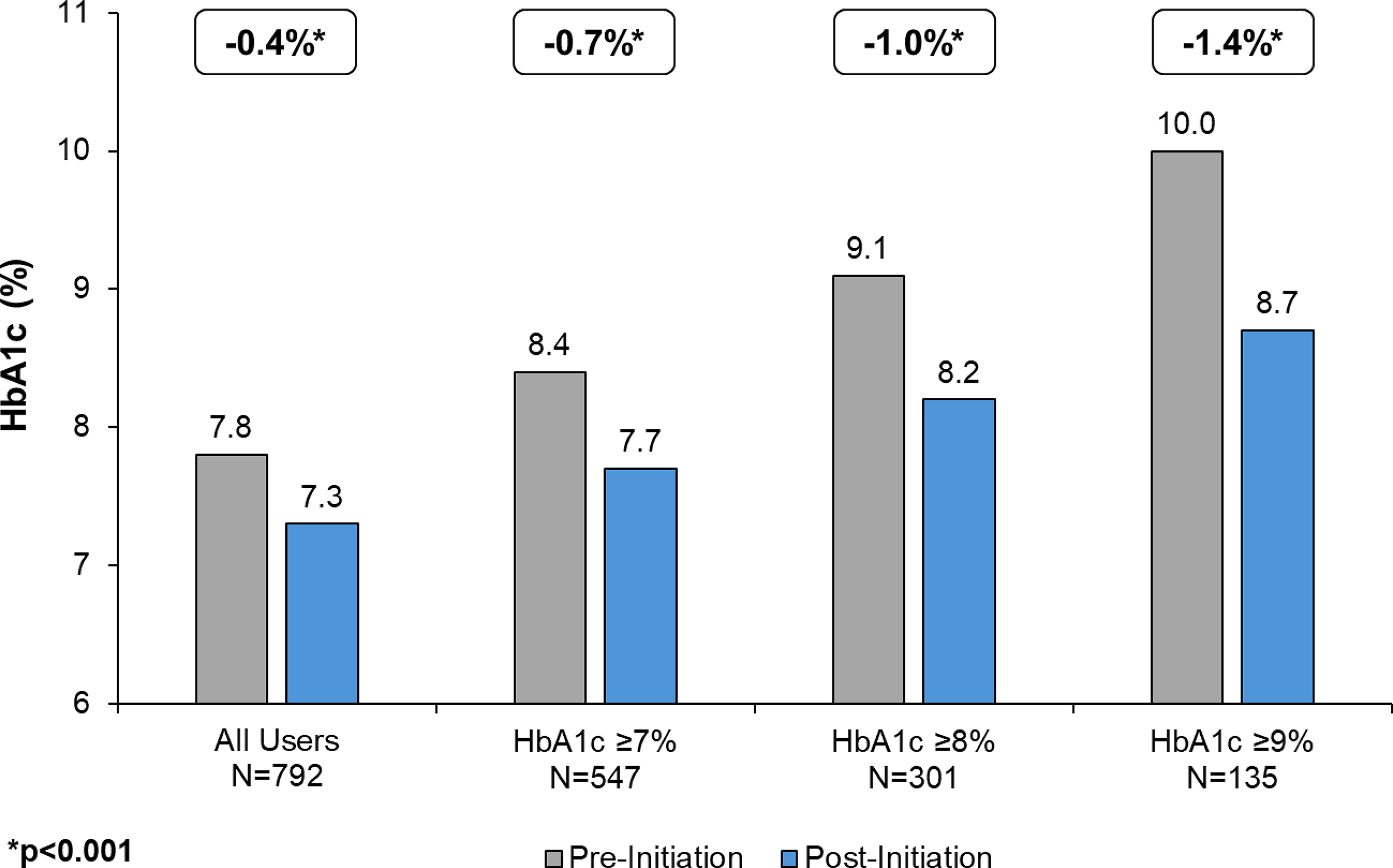
Mean HbA1c pre- and post-initiation with Omnipod 5 overall and stratified by HbA1c at pre-initiation. HbA1c, hemoglobin A1C.
Pre-initiation, 30.9% of individuals achieved the ADA target of <7% compared to 43.7% post-initiation for an absolute increase of 12.8% of individuals (41.3% relative increase) for target achievement (P < 0.001) (Fig. 2). For the HEDIS target of <8%, 62.0% of individuals achieved the target pre-initiation compared to 78.2% post-initiation for an absolute increase of 16.2% of individuals (26.1% relative increase) (P < 0.001).
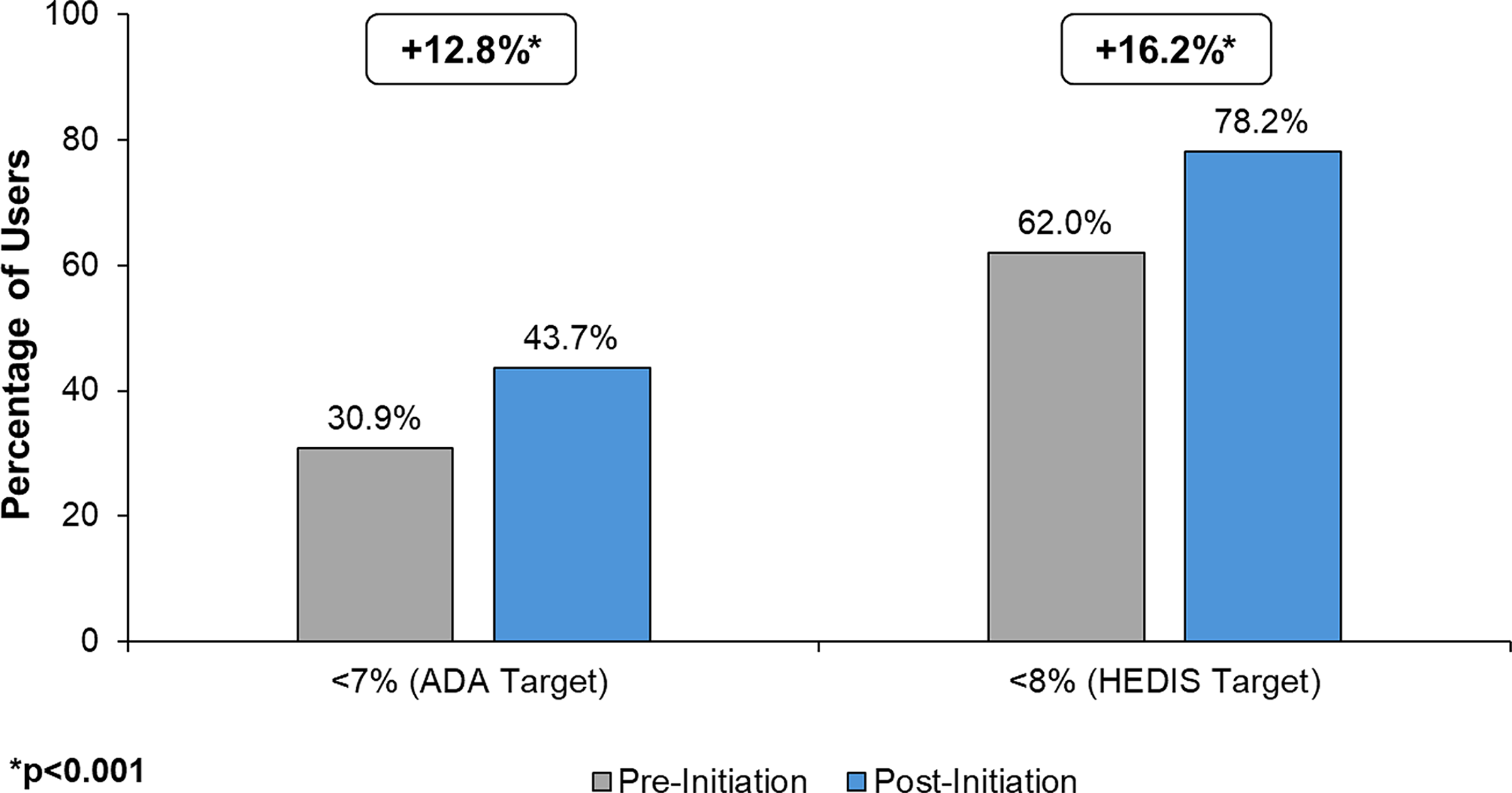
HbA1c target achievement pre- and post-initiation with Omnipod 5. ADA,American Diabetes Association; HEDIS,Healthcare Effectiveness Data and Information Set.
For users with T1D, a mean reduction in HbA1c of 0.4% was observed (P < 0.001), and for users with T2D, a mean reduction of 0.5% (P < 0.001) was observed (Fig. 3 and Supplementary Table S1). At pre-initiation, 32.3% of users with T1D achieved an ADA target of < 7% compared to 46.2% post-initiation for an absolute increase of 13.9% (43.0% relative increase) (P < 0.001) (Fig. 4). For users with T2D, 28.5% achieved an ADA target of < 7% pre-initiation compared with 39.2% post-initiation for an absolute increase of 10.7% (37.5% relative percentage). At pre-initiation, 66.3% of users with T1D achieved the HEDIS target of <8% compared to 81.6% post-initiation for an absolute increase of 15.3% (23.0% relative increase) (P < 0.001) (Fig. 4). Among users with T2D, there was an absolute increase of 17.8% (32.9% relative increase) (P < 0.001), from 54.1% achieving target pre-initiation compared to 71.9% post-initiation. Also, among the T2D subgroup, there was no statistically significant increase in the percentage of individuals with evidence of GLP-1 use pre-initiation vs. post-initiation (40.9% vs. 43.0%, P = 0.163), and a decrease in the mean number of non-insulin anti-hyperglycemic drug classes (1.23 vs. 1.16, P = 0.20).
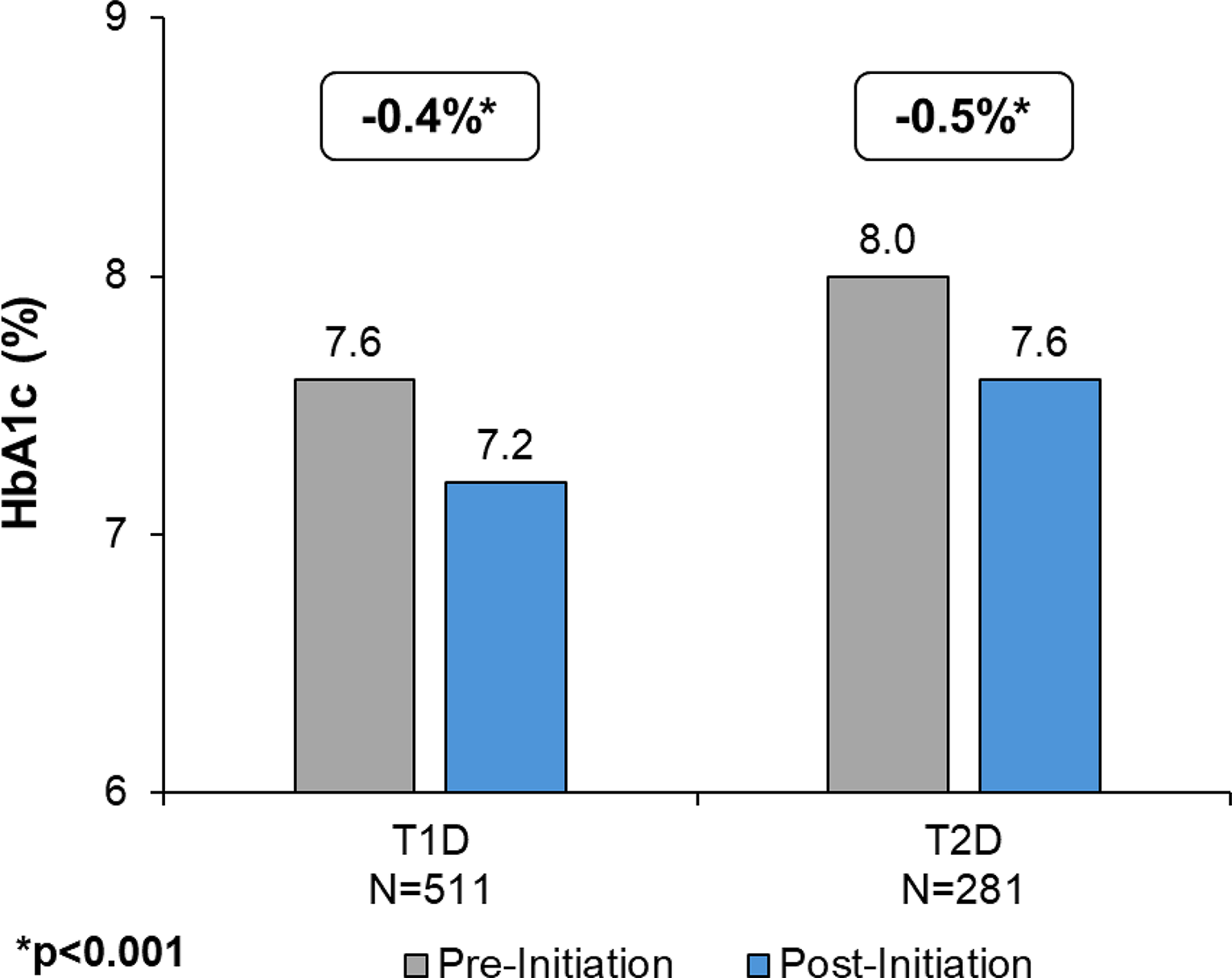
Mean HbA1c Pre- and Post-initiation with Omnipod 5 for Users with T1D and Users with T2D.
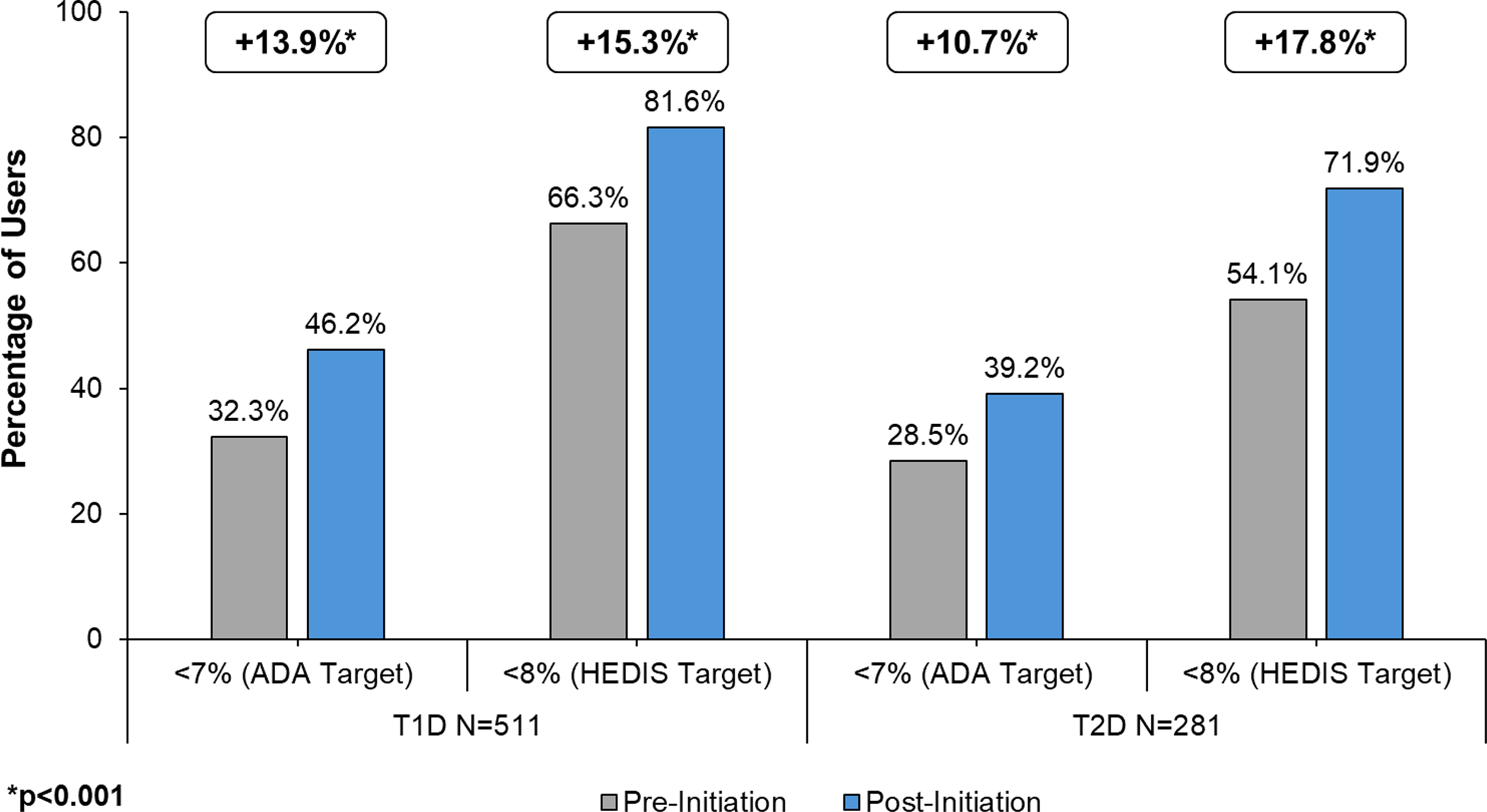
HbA1c target achievement pre- and post-initiation with Omnipod 5 for users with T1D and users with T2D.
HbA1c outcomes across additional subgroups
Additional subgroup results can be found in Supplementary Table S1. Hypothesis tests and corresponding p-values compared post-initiation versus pre-initiation values.
Insurance subgroups
Individuals with commercial insurance experienced a mean reduction in HbA1c of 0.5% (P < 0.001), while those with Medicare Advantage insurance experienced a mean reduction of 0.4% (P < 0.001).
Age subgroups
The aged 18–64 and 65 years and older subgroups demonstrated statistically significant reductions in HbA1c following initiation of Omnipod 5 (P < 0.001 for both). Adults aged 18–64 years experienced a mean reduction of 0.5%, from 7.8%–7.3%. Similarly, individuals aged 65 years and older showed a mean reduction of 0.3%, from 7.6%–7.3%. Individuals under 18 years of age had a mean HbA1c reduction of 0.4%, decreasing from 8.0%–7.6%; however, this reduction did not achieve statistical significance (P = 0.126), potentially due to the small subgroup sample size (N = 28).
CGM subgroups
Individuals with prior CGM had a mean HbA1c reduction of 0.3% (P < 0.001); those without prior CGM had a mean reduction of 1.2% (P < 0.001).
Insulin delivery subgroups
Individuals using MDI had a mean HbA1c reduction of 0.7% (P < 0.001); those with prior pump therapy had a mean reduction of 0.3% (P < 0.001). Prior pump therapy included both AID and non-AID pumps.
Discussion
Omnipod 5 was associated with reduced HbA1c and an increased percentage of individuals achieving ADA and HEDIS glycemic targets in a real-world claims database setting. The sample was fairly well-controlled at index, with a mean HbA1c under 8% pre-initiation. These analyses demonstrate the observed benefit of Omnipod 5 in those with HbA1c near target as well as those with suboptimal HbA1c levels. Prior clinical trials have demonstrated the HbA1c impact of Omnipod 5 across a wide range of HbA1c levels and prior treatment.5,9,15–17
For clinicians making decisions on therapies, it is particularly important to compare prospective research study data with real-world evidence. The pivotal trials for Omnipod 5 were published as users 6–70 years of age 9 and 2.0–5.9 years of age.5,15 These studies demonstrated an HbA1c reduction with the use of Omnipod 5 compared to conventional therapy of 0.4%, 0.7%, and 0.6% for adults, children, and young children, respectively. The present analysis of primarily adults demonstrated a similar reduction of 0.4% with the use of Omnipod 5, demonstrating the robustness of the results to trial and real-world settings. Findings of this study are consistent with those of Carlson et al. (2021) and Forlenza et al. (2024). 26,27 Carlson et al. found a larger decrease in HbA1c among individuals with Type 2 diabetes using similar methods to those of this analysis, though their sample and data collection are derived from direct training sessions for Omnipod and patient-reported HbA1c levels as opposed to a health insurance claims database. Forlenza et al. (2024) found similar changes in glycemic control to those in the present study, though they used glucose management indicator (GMI), a CGM-derived approximation of HbA1c, as a proxy rather than directly observed, lab-tested HbA1c. Findings of this analysis are also similar to results of real-world analyses of other AID systems on HbA1c, though head-to-head comparisons are difficult due to non-comparable patient populations and differing timelines.28–31
The Omnipod 5 clinical trials and real-world studies also showed that users with higher pre-initiation HbA1c values had larger relative reductions in HbA1c, as demonstrated by the child cohort with a baseline HbA1c of 7.7% seeing a reduction of 0.7%. The subgroup analyses from the current study further support these findings. Individuals under 18 years of age experienced a mean HbA1c reduction of 0.4% which was not statistically significant (P = 0.126); however, there were only 28 patients, while adults aged 18–64 years and those aged 65 years and older each demonstrated a reduction of 0.5% and 0.3%, respectively (P < 0.001). These consistent improvements across age groups reinforce the real-world effectiveness of Omnipod 5, aligning with outcomes observed in controlled clinical trials. The HbA1c reductions in all other subgroups were statistically significant.
At pre-initiation, 66.3% of users with T1D achieved the HEDIS target of <8% compared with 81.6% post-initiation for a 23.0% relative increase. Among users with T2D, there was a 32.9% relative increase from 54.1% achieving target pre-initiation compared to 71.9% post-initiation. From a payer standpoint, these improvements have significant implications. HEDIS measures are a core component of Medicare Advantage STAR Ratings, which are used by the Centers for Medicare & Medicaid Services to evaluate plan quality and determine associated bonus payments and rebate structures.32,33 Higher STAR Ratings are linked to increased revenue potential, enhanced market competitiveness, and improved member retention. Additionally, in this context, the subgroup analysis by insurance type revealed that individuals with commercial insurance and individuals with Medicare Advantage both experienced a mean reduction in HbA1c. As such, interventions like Omnipod 5 that improve glycemic control not only support better clinical outcomes but also align with payer objectives related to quality performance and value-based care strategies.
This study represents the first published claims-based real-world data reporting HbA1c reduction following initiation of the Omnipod 5 for both T1D and T2D. Of note, HbA1c reductions and targets were demonstrated in both users with T1D and T2D. It is also noteworthy that among the T2D subgroup, there was no statistically significant increase in the percentage of the individuals with evidence of GLP-1 use pre-initiation versus post-initiation, suggesting concomitant anti-hyperglycemic medication use was unlikely to have contributed to the HbA1c improvements demonstrated within the T2D subgroup. These results align with the findings of the SECURE T2D trial, which demonstrated similar glycemic benefit across GLP-1 users and non-users. 15 These findings underscore the clinical utility of AID systems such as Omnipod 5 across a broader spectrum of individuals with diabetes, including those with T2D who face challenges in achieving glycemic targets. By demonstrating meaningful HbA1c reductions, particularly among those with elevated baseline levels, this study supports the integration of AID technology into routine diabetes care, with the potential to enhance glycemic control, reduce the risk of complications, and improve long-term health outcomes in real-world settings.
One limitation of the study was that approximately half of the users had claims with a diagnosis of both T1D and T2D; however, rules were implemented to classify users into two mutually exclusive groups using the percentage of claims with T1D and T2D based on an approach that has been shown to be a good differentiator.23,24 We acknowledge the potential for misclassification, particularly given the increasing similarities in care between individuals with T1D and insulin-requiring T2D. Another limitation is that the study used a pre-post design. Without a control group, it is challenging to determine whether the changes observed from pre-initiation to post-initiation are better or worse than users who did not start Omnipod 5. The availability of HbA1c data, both pre- and post-initiation, was only available for 32% of the original sample. This study may suffer from selection bias arising from the limited availability of both pre-initiation and post-initiation HbA1c values. However, we note that the mean HbA1c at baseline among those with baseline values (7.8%) and the mean HbA1c at follow up among those with follow up values (7.3%) are extremely similar to those with both baseline and follow up values (7.8% and 7.3%, respectively), indicating selection bias may not be a significant concern in this context. The percentage of individuals <18 years was 4% in the subsample compared to 20% in the full sample. It can be theorized that many pediatric individuals have their HbA1c tests performed at the point-of-care or at hospital-based laboratories affiliated with their practice, rather than at reference laboratories, and therefore these results are not captured in the Optum database. Lastly, the study window includes a period prior to FDA approval of the Type 2 diabetes indication for Omnipod 5; it is unclear whether this could have influenced usage dynamics among those using the product off-label.
There are also several strengths of this study. The patient sample had a large proportion of individuals who used CGM prior to initiating Omnipod 5—this limits the extent to which the CGM component of the AID system would be indirectly influencing glycemic changes. Additionally, the subgroups and stratifications demonstrate robust benefit across several key clinical and demographic strata. Lastly, the diabetes type classification approach offers better accuracy and greater consistency than using other methods in claims data, such as the earliest or latest diagnostic code available.
In conclusion, this claims analysis demonstrates the effectiveness of Omnipod 5 in reducing HbA1c and achieving glycemic targets in a real-world setting and across a variety of different user subpopulations. Future real-world studies should examine glycemic changes associated with initiation of diabetes technology within the context of the usage of non-insulin diabetes medications, including GLP-1 RA and SGLT-2 medications.
Authors’ Contributions
Study design was conducted by D.C., D.S., R.R.-S., N.N.G., and V.W. Analysis of data was conducted by E.K.B., N.N.G., and V.W. C.J.L., G.F., D.C., D.S., R.R.-S., N.N.G., E.K.B., and V.W. interpreted the findings. All the authors read and approved the final article and reviewed and commented on all previous versions.
Footnotes
Acknowledgments
The authors would also like to thank Insulet Corporation employee Joshua Weinstein, PhD for significant contributions to article development and Optum employees Daryl Truong, PharmD for medical writing (for which Optum was compensated) and Sarah Hague for project management support.
Author Disclosure Statement
E.K.B., N.N.G., and V.W. are employees of Optum. C.J.L. has received research support: NIH, Helmsley Foundation, Abbott, Dexcom, Insulet, Tandem, Mannkind, and DEKA/Sequel. C.J.L. has served as a consultant for Dexcom, Eli Lilly and Tandem Diabetes and has received travel support from Insulet. G.F. has received research support from NIH, Breakthrough T1D, Helmsley Charitable Trust, Medtronic, Dexcom, Abbott, Insulet, Tandem, Beta Bionics, Lilly, and Mannkind and has been a speaker/consultant/ad board member for Medtronic, Dexcom, Abbott, Insulet, Tandem, Beta Bionics, Lilly, Sequel, and Mannkind. D.C., D.S., and R.R.-S. are employed by and own stock in Insulet Corporation.
Funding Information
This work was funded by Insulet, which participated in the study design, interpretation of the data, writing and revision of the article, and the decision to submit the article for publication. Insulet was given the opportunity to review the article for medical and scientific accuracy as well as intellectual property considerations. The author(s) met criteria for authorship as recommended by the International Committee of Medical Journal Editors. The authors received no direct compensation related to the development of the article. The study was performed by Optum under contract with Insulet.
Availability of Data and Materials
The data contained in the database used for the study contains proprietary elements owned by Optum and, therefore, cannot be broadly disclosed or made publicly available at this time. The disclosure of these data to third parties assumes certain data security and privacy protocols are in place and that the third party has executed a standard license agreement which includes restrictive covenants governing the use of the data.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
