Abstract
Objectives:
To explore trends in the receipt of commonly prescribed medications (beyond insulin) in people with type 1 diabetes in Australia, including polypharmacy, and to investigate socioeconomic disparities across these trends.
Methods:
A retrospective analysis of 68,287 people with type 1 diabetes registered on the National Diabetes Services Scheme between 2013 and 2019, and linked to the Australian pharmaceutical claims database to capture medication use. The proportion of people with type 1 diabetes dispensed a medication class was assessed in each year, and by levels of Index of Relative Social Disadvantage.
Results:
The study population comprised 37,055 (54.3%) males and 31,232 (45.7%) females captured between 2013 and 2019, with an overall median age of 38.8 (interquartile range [IQR]: 26.1–53.3) years and median diabetes duration of 17.0 (IQR: 7.9–25.7) years. The median number of medications used in the study population was five (IQR: 3–8). Trends in receipt of commonly prescribed medications remained relatively stable between 2013 and 2019, except for the use of noninsulin (adjuvant) glucose-lowering agents, which increased. In 2019, compared with people in the least disadvantaged areas, those in the most disadvantaged areas were more likely to be receiving adjuvant glucose-lowering therapy (crude rates 21.5% vs. 11.6%), antihypertensive therapies (36.1% vs. 28.8%), lipid-lowering therapies (35.7% vs. 29.7%), antithrombotic medication (8.9% vs. 5.6%), proton pump inhibitors (25.2% vs. 17.1%), medications for asthma (13.2% vs. 8.4%), and medications for mental health (27.5% vs. 21.5%). Polypharmacy (defined as ≥5 medications) was more likely in those in the most disadvantaged areas compared with those in the least disadvantaged areas (58.8% vs. 48.5%). Even after adjustment for patient factors, differences in medication use and polypharmacy across socioeconomic strata persisted.
Conclusions:
Receipt of commonly prescribed medications in type 1 diabetes and polypharmacy were associated with increased socioeconomic disadvantage.
Keywords
Introduction
Type 1 diabetes is a chronic disease, characterized by the autoimmune destruction of pancreatic β cells that produce insulin, requiring lifelong insulin replacement. 1 Despite advances in diabetes management, people with type 1 diabetes remain at risk of microvascular and macrovascular complications. 1 Furthermore, people with type 1 diabetes are at higher risk of other autoimmune conditions and depression. 2 As such, people with type 1 diabetes require management of multiple risk factors, complications, and comorbidities over their life course. This typically necessitates the use of multiple medications beyond insulin.
Social determinants of health, such as socioeconomic status (SES), contribute to systematic differences and disparities in health care and outcomes. A recent systematic review identified that inequalities in mortality, morbidity, and use of new technologies for diabetes management exist in adults with type 1 diabetes. 3 Indeed, among adults with type 1 diabetes, associations between lower SES and higher comorbidity risk, as well as lower SES and increased mortality, have been reported.3–5 Numerous studies have demonstrated that access to, and use of, new technology types such as continuous glucose monitoring (CGM) is lower in those in more disadvantaged areas.4,6,7 Moreover, studies have suggested a relationship between lower SES and acute diabetic complications such as ketoacidosis and severe hypoglycemia.6,8
While studies have reported associations between SES and diabetes-related management methods and complications in people with type 1 diabetes, little is known about the relationship between SES and medication use. This study aimed to explore trends in medication use for commonly prescribed therapeutics (beyond insulin) among people with type 1 diabetes in Australia during the period 2013–2019, including polypharmacy, and to identify differences in medication use across levels of socioeconomic disadvantage.
Methods
Data sources
The data for this study were generated by linking the National Diabetes Services Scheme (NDSS) to health administrative datasets. Detailed information on this linkage has been described previously. 9 In brief, the NDSS is an Australian Government initiative, providing subsidized access to diabetes products, as well as information and support services to people with diabetes. The NDSS is a register of people diagnosed with type 1, type 2, gestational, and other forms of diabetes in Australia, with an estimated coverage of 80%–90% of all people with diagnosed diabetes in Australia.10,11 Diabetes type is recorded by a health care professional at the time of registration and modified by informative clinical characteristics. Assignment and reclassification of diabetes type are included in the Supplementary Material. In brief, NDSS registrants were reclassified as having type 1 diabetes based on age at diagnosis, insulin use within a year of diagnosis, or evidence of ongoing use of insulin.
While the NDSS is a national database, the dataset for this analysis included NDSS registrants from the states of Victoria, New South Wales, the Australian Capital Territory, and Queensland (capturing ∼80% of all people on the NDSS). This cohort of registrants was linked to the National Death Index (NDI) to collect mortality status (and determine if they survived the full year) and to the Pharmaceutical Benefits Scheme (PBS) to capture medication dispensation. Linkage was performed by the Australian Institute of Health and Welfare. The NDI records all deaths that occur in Australia. The PBS provides access to subsidized medications for all Australians and collects information on all prescribed and dispensed medications covered by the scheme. When medications are listed on the PBS, the cost is shared between the patient and the government, with the patient paying a copayment. The cost of medications is further reduced for patients who have a concessional status (including those with a pensioner concession card, health care card, or Department of Veterans’ Affairs), or if they meet the PBS safety net. The PBS safety net protects patients and their families requiring a large number of prescription medications by reducing the cost of medications once a spending limit, known as the safety net threshold, is reached. Medications are only subsidized for the indication they are listed on the schedule. Since 2012, the PBS data also include data on medications dispensed whose cost is less than the copayment level. 12 The vast majority of medications for chronic conditions are subsidized through the PBS. While some medications can be accessed via private script, these are likely to be rare for the medications we studied (except for glucagon-like peptide 1 receptor agonist [GLP-1 RA]). Dates of prescription and dispensing, concession status, prescriber specialty, PBS item codes, and Anatomical Therapeutic Chemical (ATC) codes were derived from the PBS data.
Our study population was limited to Australians who do not identify as Aboriginal or Torres Strait Islander, with type 1 diabetes registered on the NDSS between January 1, 2013, and December 31, 2019. Aboriginal and Torres Strait Islander people receive diabetes services and medications via separate programs and are consequently not well represented on the NDSS and often do not have all the prescription medications dispensed by the PBS.
This study was approved by the Monash University Human Research Ethics Committee (Project ID 35672) and the Australian Institute of Health and Welfare Ethics Committee (EO2018/5/501).
Medication classes
We categorized commonly used medication classes among people with type 1 diabetes, beyond insulin. This included other (adjuvant) glucose-lowering agents, lipid-modifying and antihypertensive therapies, proton pump inhibitors, antithrombotic medications, as well as medications for asthma, autoimmune conditions, and mental health. Medication classes were derived from ATC codes, 13 based on the chemical subgroup (fifth level). The ATC codes included in each medication class are included in Supplementary Table S1.
Measure of socioeconomic disadvantage
Based on residential postcode, NDSS registrants were assigned a Socio-Economic Index for Areas: Index of Relative Socioeconomic Disadvantage (IRSD) class based on the 2016 Australian census data. The IRSD is produced by the Australian Bureau of Statistics as a measure to rank areas in Australia according to the relative socioeconomic disadvantage (based on various measures such as income, education, employment, occupation, and housing). 14 Registrants were stratified by IRSD quintiles, where the highest quintile represents individuals living in areas of least socioeconomic disadvantage and the lowest quintile represents individuals living in areas of most socioeconomic disadvantage. Of note, while IRSD quintiles are representative of the amount of deprivation in the area in which an individual lives, it may not necessarily reflect the individual’s circumstance or SES.
Data analysis
The proportion of people with type 1 diabetes dispensed a medication class in each year was calculated as the number of people dispensed ≥1 prescription for that year, divided by the number of people with type 1 diabetes registered on the NDSS before the start of that year and survived the full year (i.e., the analysis was only conducted on people with a full year of follow-up). The proportion of people with type 1 diabetes receiving five or more medications concurrently (polypharmacy) was also calculated. Crude proportions were stratified by calendar year, age, and IRSD quintile. We also performed adjusted analyses using logistic regression. In these analyses, the outcome variable was receipt of the medication class in question, with each model adjusted for the linear effect of calendar time, sex, spline effects of current age, and duration of diabetes, 15 a linear effect of the RxRisk comorbidity index (a prescription-based index of comorbidity where a higher score indicates a greater level of comorbidity), 16 and a linear effect of IRSD quintile. These models were fit overall, then separately by IRSD quintile. Each model was used to predict the probability of receipt of a given medication class for a given calendar year at the mean value of all included parameters across the population (thereby enabling comparison).
To estimate the relative change over time in medication use, we calculated the odds ratio (OR) for calendar time for receipt of a given medication class using logistic regression models adjusted for age, sex, duration of diabetes, the RxRisk comorbidity index, and IRSD quintile.
Sensitivity analyses
Two sensitivity analyses were undertaken. The first applied a more conservative definition of type 1 diabetes to the study population by excluding those people with type 1 diabetes who were on any other noninsulin (adjuvant) glucose-lowering therapies. The second applied a definition of medication use that included greater drug dispensation in each study year defined as either ≥3 or ≥9 prescriptions per year.
All statistical analyses were performed in Stata 18.0 statistical software (StataCorp, College Station, TX).
Data Resource and Availability
These data were obtained under a specific license with the AIHW and thus are not publicly available.
Results
Population characteristics
The study population included 68,287 people with type 1 diabetes captured between 2013 and 2019, of which 54.3% were male. The median age of the overall population was 38.8 (IQR: 26.1–53.3) years, with a median diabetes duration of 17.0 (IQR: 7.9–25.7) years. The median number of medications used was five (IQR: 3–8).
Characteristics of the study population and changes over time are included in Supplementary Table S2. Characteristics of the study population by socioeconomic strata are included in Supplementary Table S3.
Trends in medication use
Figure 1 demonstrates trends in medication receipt over time. The most commonly prescribed noninsulin medications in people with type 1 diabetes were antihypertensive and lipid-lowering therapies, and medications for mental health (including antipsychotics, antidepressants, and psychostimulants). Over the period of 2013 to 2019, the proportion of people with type 1 diabetes receiving noninsulin glucose-lowering drugs increased from 12.6% in 2013 to 16.1% in 2019 (adjusted OR per calendar year of 1.08, 95% confidence interval [CI]: 1.08 to 1.09).
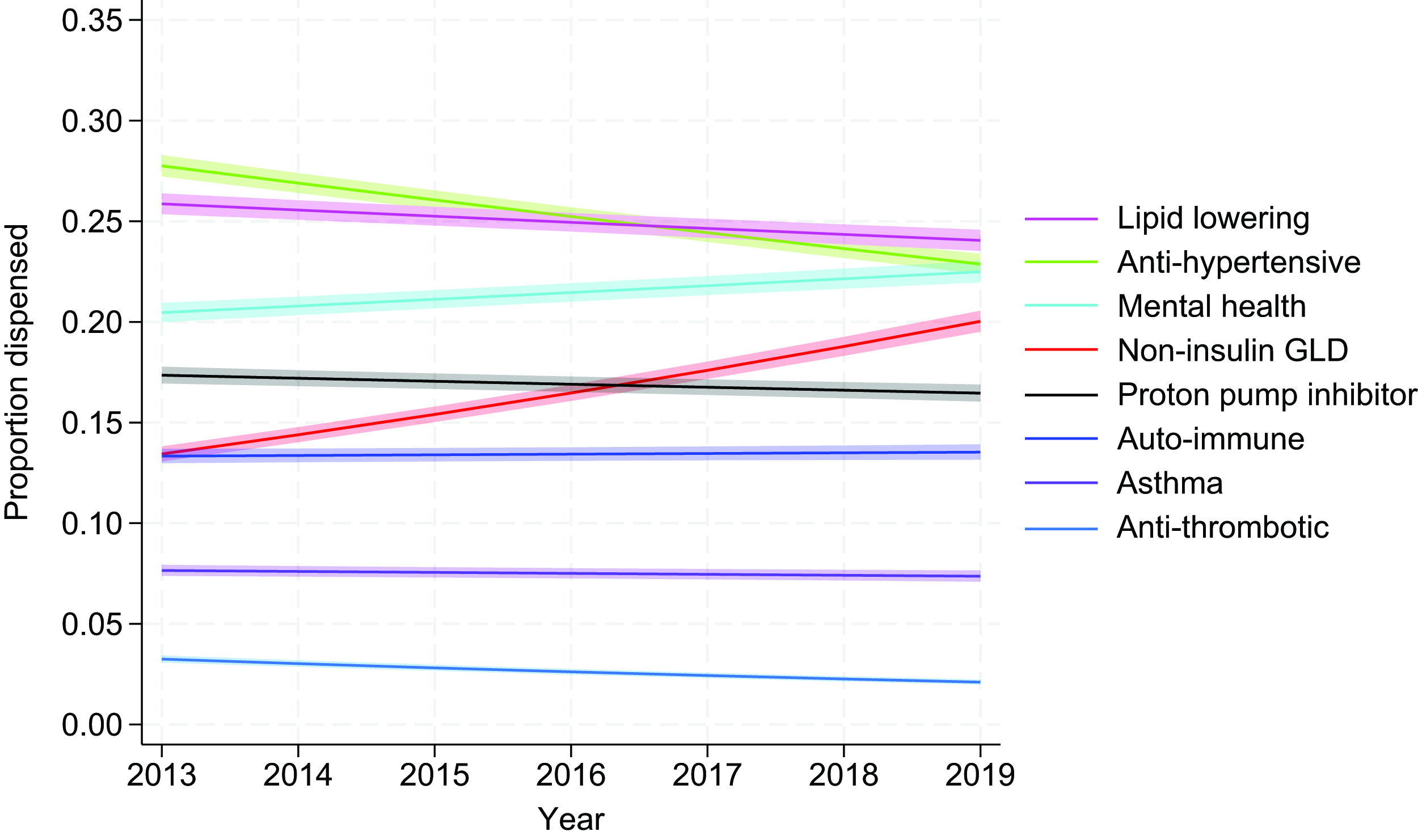
Modeled trends over time in proportion of people receiving each medication class. Models are adjusted for age, sex, duration of diabetes, the RxRisk comorbidity index, and IRSD quintile (socioeconomic status). The solid line represents the mean value and the shaded error clouds represent the 95% confidence interval. GLD, glucose lowering drug; IRSD, Index of Relative Socioeconomic Disadvantage.
Over the same period, receipt of lipid-modifying therapies decreased [crude proportions increased from 31.3% to 32.3% for lipid-modifying therapies, but once adjusted for age, sex, duration of diabetes, comorbidities and SES decreased (adjusted OR: 0.98, 95% CI: 0.98 to 0.99) and receipt of antihypertensive therapies decreased from 33.4% to 32.4% (adjusted OR: 0.96, 95% CI: 0.95 to 0.96)]. Receipt of antithrombotic medications also decreased over this period from 8.5% to 7.3% (adjusted OR: 0.93, 95% CI: 0.92 to 0.93). Receipt of proton-pump inhibitors and medications for asthma and autoimmune conditions remained relatively unchanged. The adjusted ORs were 0.99 (95% CI: 0.99 to 0.99), 0.99 (95% CI: 0.99 to 1.00), and 1.00 (95% CI: 1.00 to 1.01), respectively. The proportion of people receiving medications for mental health increased slightly from 22.8% to 25.0% (OR: 1.02, 95% 1.02 to 1.02). Polypharmacy remained relatively unchanged (54.8% in 2013 compared with 53.8% in 2019).
Males were more likely than females to be receiving lipid-modifying and antihypertensive therapies, but less likely to receive all other drug classes (Supplementary Fig. S1). In 2019, 33.9% of males were receiving antihypertensive therapies and 34.2% were receiving lipid-lowering therapies, while the respective values for females were 30.5% and 30.1% (with use increasing with increasing age as described below). Receipt of noninsulin glucose-lowering therapies was 13.8% in males and 18.7% in females. Compared with males, females also had a higher use of autoimmune therapies (24.8% vs. 12.0%), proton pump inhibitors (24.9% vs. 18.0%), and medications for mental health (30.0% vs. 20.8%) and asthma (13.2% vs. 8.7%). Polypharmacy was higher in females compared with males (60.0% vs. 48.5%).
The proportion of people prescribed each medication class increased with increasing age (Supplementary Material). In 2019, receipt of antihypertensive therapies increased from 8.3% in those aged 20–29 years to 55.3% in those aged 50–59 years and 78.4% in those aged 80+ years, and receipt of lipid-lowering therapies increased from 5.0% in those aged 20–29 years to 59.6% in those aged 50–59 years and 63.8% in those aged 80+ years. Concomitant increases in receipt of noninsulin glucose-lowering drugs were 9.1%, 19.9%, and 33.0%, while concomitant increases in mental health therapies were 21.8%, 30.5% and 35.8%, respectively. Polypharmacy increased with increasing age (37.4% in those aged 20–29 years up to 80%–90% in those aged 60+ years). Supplementary Table S4 provides data on receipt of medications in 2019 across all age groups and medication classes.
Socioeconomic disadvantage and medication use
Figure 2 shows trends in the proportion of people prescribed each medication class over time by levels of socioeconomic disadvantage, with the OR for calendar time included in Table 1. Compared with people with type 1 diabetes from the least disadvantaged areas (IRSD quintiles 4 and 5), receipt of medications increased with increasing socioeconomic disadvantage for all medication classes. Moreover, in 2019, 21.5% of people with type 1 diabetes from the most disadvantaged areas received noninsulin glucose-lowering drugs compared with 11.6% of people from the least disadvantaged areas. Utilization of noninsulin glucose-lowering drugs by drug class is included in Supplementary Tables S5 and S6. Receipt of antihypertensive therapies was 36.1% in the most disadvantaged areas compared with 28.8% in the least disadvantaged areas, while the respective receipt of lipid-lowering therapies was 35.7% and 29.7%, use of antithrombotics was 8.9% and 5.6%, and use of proton pump inhibitors was 25.2% and 17.1%. Receipt of medications for asthma was 13.2% in the most disadvantaged areas compared with 8.4% in the least disadvantaged areas, while receipt of medications for mental health was 27.5% in the most disadvantaged areas compared with 21.5% in the least disadvantaged areas. By contrast, 19.1% and 17.0%, respectively, were receiving medications for autoimmune conditions. Polypharmacy was common, and increased with increasing socioeconomic disadvantage, with 58.8% of people from the most disadvantaged areas experiencing polypharmacy compared with 48.5% of people from the least disadvantaged areas. Even after adjustment for age, sex, duration of diabetes, and comorbidities, differences in medication use and polypharmacy across SES persisted with increasing proportions of people receiving medications with increasing disadvantage (Figs. 2 and 3). The differences also persisted across all ages (Supplementary Fig. S2 and S3).
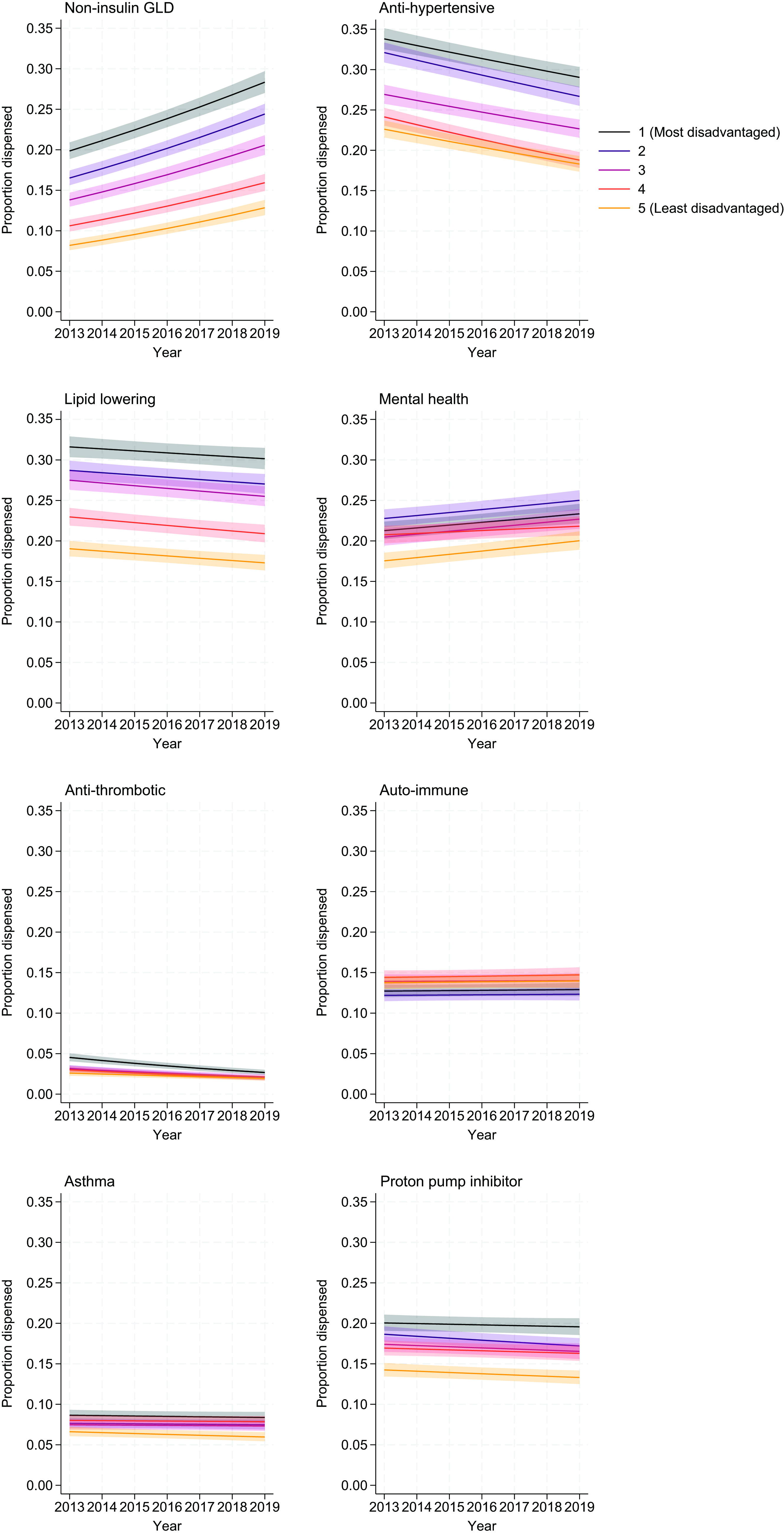
Modeled trends over time in proportion of people receiving each medication class, by socioeconomic disadvantage. Models are adjusted for age, sex, duration of diabetes, and the RxRisk comorbidity index. The solid line represents the mean value and the shaded error clouds represent the 95% confidence interval. GLD, glucose lowering drug.
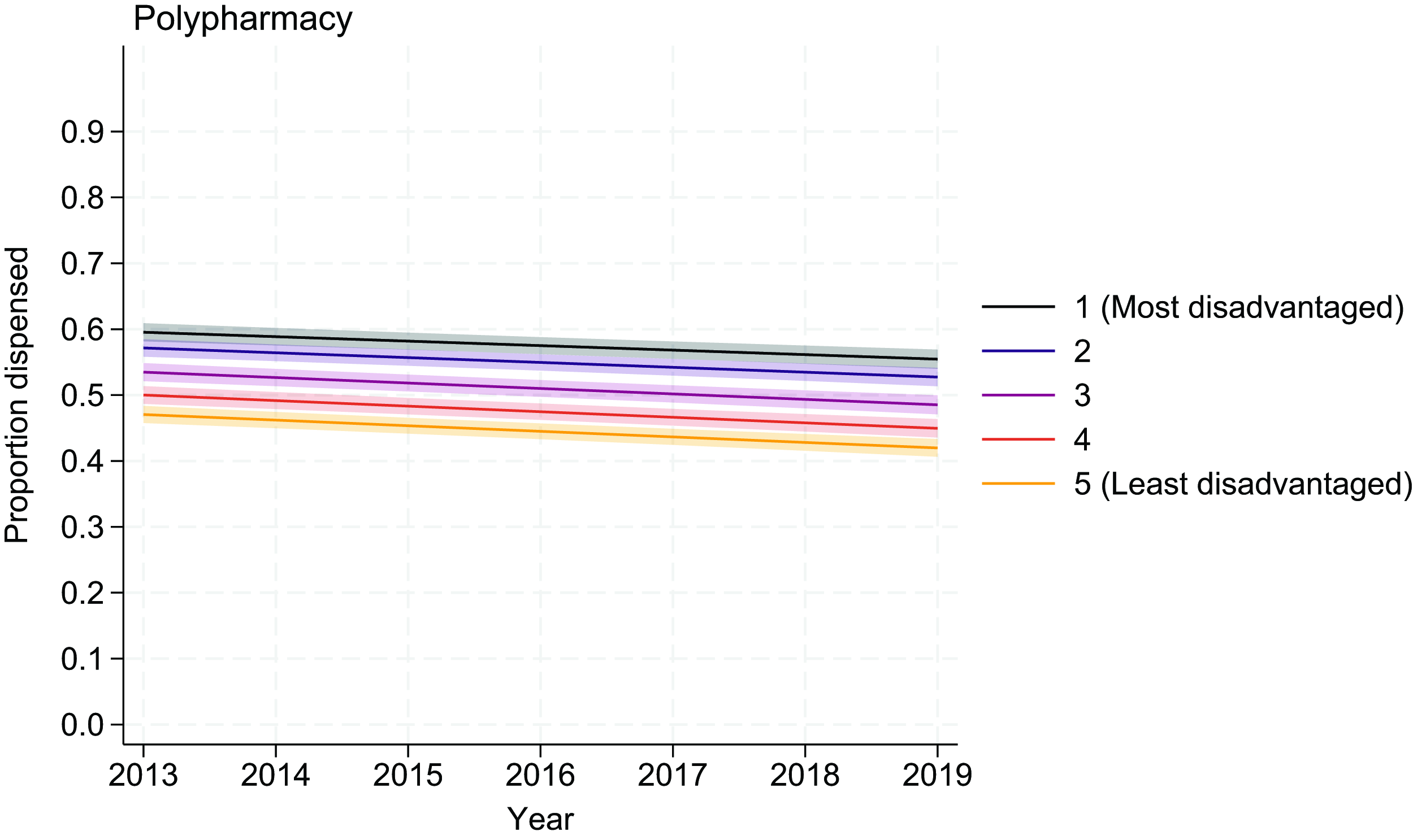
Modeled trends over time in proportion of people receiving ≥5 medications concurrently (polypharmacy), by socioeconomic disadvantage. Models are adjusted for age, sex, duration of diabetes, and the RxRisk comorbidity index. The solid line represents the mean value and the shaded error clouds represent the 95% confidence interval.
Odds Ratio for Calendar Year of the Receipt of Medication Class, by Socioeconomic Disadvantage
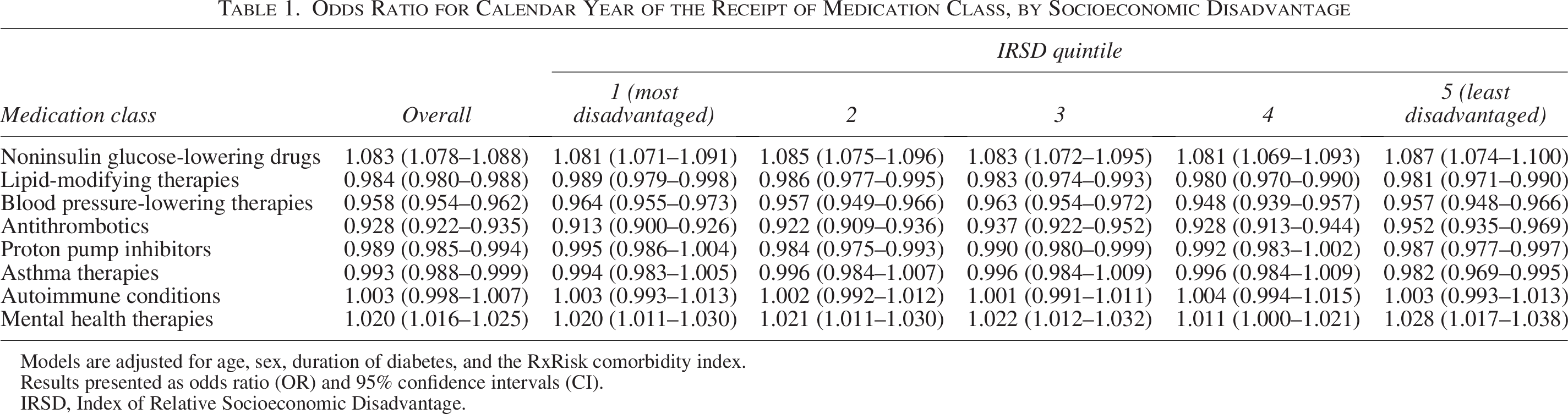
Models are adjusted for age, sex, duration of diabetes, and the RxRisk comorbidity index.
Results presented as odds ratio (OR) and 95% confidence intervals (CI).
IRSD, Index of Relative Socioeconomic Disadvantage.
Our sensitivity analyses demonstrated that even when applying more stringent definitions of type 1 diabetes or level of medication use per year, differences across socioeconomic disadvantage remained (Supplementary Tables S7, S8, S9 and S10).
Discussion
This study provides comprehensive insights on medication use among people with type 1 diabetes. Our study provides several important findings. First, the proportion of people with type 1 diabetes receiving commonly prescribed medications remained relatively unchanged between 2013 and 2019. The exception was receipt of noninsulin (adjuvant) glucose-lowering drugs, which increased and may reflect the increasing availability of glucose-lowering drugs with cardiorenal benefits such as sodium glucose transport protein 2 (SGLT2) inhibitors, which increased from 0.3% in 2014 to 5.1% in 2019 and GLP-1RA, which increased from 0.5% in 2013 to 1.8% in 2019. The increased availability of these newer glucose-lowering drugs with cardiorenal benefits may also partly explain the small decreases in receipt of antihypertensive and lipid-lowering therapies, as well as antithrombotic medications observed. This finding is in line with national trends that also show a small decrease in the dispensation of statins (the most commonly prescribed lipid-lowering therapy) and antihypertensive polytherapy over the period of 2013–2018/19.17,18
Second, compared with females, males were more likely to receive antihypertensive and lipid-lowering therapies but less likely to receive all other medication classes. Although the impact of diabetes on cardiovascular disease risk is greater in females than in males, the incidence and prevalence of cardiovascular disease (as well as coronary artery calcification, a marker of atherosclerosis and strong indicator of cardiovascular disease) remain higher in males with diabetes,9,19–23 likely explaining the greater use of antihypertensive and lipid-lowering therapies in males. The higher rates of autoimmune disease and treated mental health conditions among females likely contribute to the sex differences in other medication classes.24,25 Our findings on antihypertensive and lipid-lowering therapies are consistent with prior research indicating lower use of aspirin, statins, angiotensin converting enzyme inhibitors, or angiotensin II receptor blockers in females compared with males.22,23 Emerging evidence suggests that the cardioprotective advantage typically observed in females in the general population is attenuated with the onset of diabetes.21,23 The exact mechanisms underlying this shift remain unclear, but contributing factors may include challenges with glycemic control in females, sex-specific differences in cardiovascular risk factor profiles, as well as female-specific changes such as premature menopause and polycystic ovary syndrome. 26
Third, receipt of all medication classes, as well as polypharmacy, increased with increasing age. Fourth, disparities in receipt of commonly prescribed medications in people with type 1 diabetes across SES exist, with people from the most disadvantaged areas more likely to receive medications for cardiovascular risk management, other autoimmune conditions, and mental health compared with people from the least disadvantaged areas. Moreover, polypharmacy was more common in people from the most disadvantaged areas. These differences persisted after adjustment for age, sex, duration of diabetes, and comorbidities. Previous studies have similarly highlighted health disparities among people with type 1 diabetes. However, most have focused on diabetes-related management technologies such as CGM and/or pumps and complications rather than on medication use, making direct comparisons difficult. Some studies have suggested reduced use of CGM and/or pumps among people with type 1 diabetes from more disadvantaged areas compared with those from less disadvantaged areas. 7 Moreover, glycemic management (in terms of HbA1c level) has been shown to be associated with SES, with people from more disadvantaged areas having poorer glycemic management. An analysis of 21,253 people with type 1 diabetes captured in the U.S. T1D Exchange Clinic Registry reported that lower SES (defined by income and lack of insurance or state/federal insurance) was associated with higher average HbA1c levels across the lifespan of type 1 diabetes. 27 Similarly, a study of 1060 adults with type 1 diabetes in Spain demonstrated that time in range was lower among people from the lowest SES quartile compared with the highest SES quartile, with use of flash/CGM helping to alleviate the impact of social inequalities on glycemic management by narrowing the gap in glycemic management across SES, although differences persisted. 8 Comparable results have also been demonstrated in children and youth with type 1 diabetes.28,29
Glycemic management is directly correlated with the development of both acute glycemic and chronic diabetes-related complications. This may explain the higher reported rates of complications seen in those from the lower SES. For example, presentation for diabetic ketoacidosis (DKA) at diagnosis and/or hospitalization for DKA in people with type 1 diabetes were associated with socioeconomic disadvantage.30–32 Moreover, an analysis of the Scottish National diabetes register (SCI-Diabetes) showed that those with type 1 diabetes in the most disadvantaged areas experience severe hypoglycemia requiring hospitalization 2.58 times more than those in the least disadvantaged areas. 33 Studies have also reported higher rates of more chronic diabetes-related complications such as diabetic retinopathy 34 and cardiovascular disease in people with type 1 diabetes from lower SES with socioeconomic deprivation reported to increase the risk of cardiovascular disease and death by a factor of 2–3 in type 1 diabetes.35,36 Diabetes distress has also been shown to be correlated with lower SES, potentially explaining the increased use of therapies for mental health that we observed in the most disadvantaged areas. 37 The higher rates of complications and comorbidities seen in people with type 1 diabetes from lower SES in other studies may explain the higher proportion of people receiving medications from the most disadvantaged areas in our study. While a partially publicly funded health system provides improved access to health care over a completely private system, we hypothesize that greater medication use may be more common among people from more disadvantaged areas due to an increased prevalence of diabetes-related complications, as well as higher long-term average glycemia. In line with our study findings, a study of the Scottish population with type 1 diabetes (based on SCI-Diabetes data), who had access to a publicly funded health system, demonstrated that the prevalence of polypharmacy (defined as individuals prescribed five or more drugs) increased with increasing deprivation. 38 Moreover, the authors found that polypharmacy was associated with increased risk of adverse health outcomes, including acute diabetes-related complications, which may further exacerbate the need for medications. This contrasts with a study from the United States, where no such system is available, which demonstrated that there may be under-prescribing of multiple medications in people from lower SES, 39 as well as reduced medication adherence. 40 Moreover, among U.S. adults taking prescription medications, those who purchased medication outside of the United States were more likely to be older, Hispanic, or an immigrant, have higher educational attainment, have lower family income, and be uninsured. 41 Cost-related medication adherence was also a factor associated with purchasing medications outside the United States to reduce the cost burden. 41
Our study has demonstrated an association between SES deprivation and medication use, including polypharmacy. However, due to the nature of the dataset used in this study, we could not ascertain if these medications are being prescribed and used appropriately to reduce the risk of adverse outcomes (e.g., Are they being prescribed according to treatment guidelines and for more intensive treatment in people at high risk of cardiorenal disease?). We hypothesize that the increased use of medications is a result of increased risk factors and complications as proposed by other studies. However, we may have underestimated medication needs, particularly in people from more disadvantaged areas, as SES may affect the ability to fill scripts due to the cost of copayments and travel-related costs to attend pharmacies, and our analysis explores dispensation of medications only (not prescribing). In addition, people from lower SES may be less engaged with health care due to cost, once again, underestimating medication needs. Further studies are needed to investigate treatment and prescribing gaps in people with type 1 diabetes and across SES, as well as the impact of access to best diabetes-management technology on medication use.
Strengths and limitations
The strength of the study lies in the use of a large, national population-based dataset with a sample of ∼70,000 people. Moreover, given that the NDSS subsidizes diabetes management products, including insulin needles, blood glucose test strips, urine test strips, and insulin pump consumables, it is likely that the NDSS captures the overwhelming majority of people living with type 1 diabetes. 42
There are, however, several limitations that warrant mention. First, the nature of register data is that there will inevitably be some misclassification of diabetes type, particularly at older ages. However, due to the similar median age and median duration of diabetes across socioeconomic strata, we believe that the level of misclassification is low and unlikely to affect our conclusions. Moreover, differences across SES persisted even we used a stricter definition of type 1 diabetes (by excluding those on noninsulin (adjuvant) glucose-lowering drugs, including those on SGLT2 inhibitors and GLP-1RA, which are not currently reimbursed for people with type 1 diabetes). Second, there may be some misclassification of deprivation based on residential postcode as individuals may move and/or the IRSD quintile assigned to a postcode may not be reflective of the individual’s SES, with some postcodes reflecting a diverse SES mix. Third, in Australia, newer glucose-lowering medications such as GLP-1 RAs are currently only subsidized for people with type 2 diabetes and as such, the PBS data may significantly underestimate use of these drugs by people with type 1 diabetes, as there is no equivalent dataset that comprehensively captures information on all privately dispensed medications. Although some privately prescribed and/or dispensed medications may be recorded in other datasets drawn from general practice electronic medical record data or pharmacy data, these sources have notable limitations.43,44 Fourth, because medication use was drawn from administrative data, we do not have information on anthropometrics, laboratory data, or complications and comorbidity data to understand if the medications are being used and/or prescribed appropriately. Furthermore, as the focus of this study was trends in medication use, we did not explore adherence and persistence to medications. We also focused on medication classes as a whole, and did not explore trends in the use of each specific medication within the relevant class.
In conclusion, this study identified that socioeconomic disparities in medication use and polypharmacy among people with type 1 diabetes exist. Future work should focus on understanding if the differences observed in our study are due to systematic differences in risk factors, clinical care provision, or health outcomes across socioeconomic strata, as any differences may reflect higher prevalence of risk factors (including HbA1c) and complications in people from more disadvantaged areas. This is pivotal to informing strategies to reduce socioeconomic disparities and improve the health and well-being of all people with type 1 diabetes.
Authors’ Contributions
E.Z.: Conceptualization (lead), methodology (equal), writing—original draft (lead), and writing—review and editing (equal). J.I.M.: Formal analysis (lead), methodology (equal), and writing—reviewing and editing (equal). L.C.: Writing—reviewing and editing (equal). S.T.: Writing—reviewing and editing (equal). S.A.M.: Writing—reviewing and editing (equal). E.A.D.: Writing—reviewing and editing (equal). T.W.J.: Writing—reviewing and editing (equal). K.E.L.: Writing—reviewing and editing (equal). D.J.M.: Methodology (equal) and writing—reviewing and editing (equal). J.E.S.: Methodology (equal) and writing—reviewing and editing (equal). S.Z.: Methodology (equal) and writing—reviewing and editing (equal). All the authors have read and approved the final version of this article. J.I.M. is the guarantor of this work, and as such had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Author Disclosure Statement
S.T. is a regular discussant and advisor at the Drug Utilization Sub-Committee (DUSC) of the Pharmaceutical Benefits Advisory Committee (PBAC). This work is independent of DUSC and does not reflect the views of DUSC. S.T. reports grant support from MRFF CVD Mission, paid to Monash University, independent of this work; travel and meeting support from Monash University CCRET independent of this work, grant from Bethlehem Griffiths Research Foundation, independent of this work. S.A.M. reports support for research from Medtronic to her institution; receiving speaker honoraria from Eli Lilly Australia, Roche Diabetes Care Australia, and Sanofi-Aventis Australia; serving on advisory boards for Medtronic and Ypsomed; and facilitating workshops for the Australian Diabetes Society. E.A.D. has received payment to the institution (Kids Research Institute Australia) from Eli Lilly, and Sanofi for consultancy work outside the submitted work. J.E.S. has received honoraria for scientific advisory, lectures, and clinical research from Zuellig Pharma, Astra Zeneca, Sanofi, Novo Nordisk, Eli Lilly, Abbott, Mylan, Boehringer Ingelheim, Roche, Pfizer, and GSK. S.Z. has received payment to the institution (Monash University) from Eli Lilly Australia, Boehringer-Ingelheim, Merck Sharp & Dohme Australia, AstraZeneca, Novo Nordisk, Sanofi, and Servier for consultancy work outside the submitted work. All other authors have no conflicts of interest to disclose.
Funding Information
E.Z. was supported by funding from Breakthrough T1D Australia (grant number 5-SRA-2021-1088-M-X). J.I.M. was supported by a Heart Foundation Postdoctoral Fellowship (ID: 108269-2024) from the National Heart Foundation of Australia.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
