Abstract
Objective:
This study was designed to investigate the switch between the open-source automated insulin delivery (OS-AID) system AndroidAPS (AAPS) and commercially available AID systems Control-IQ (CIQ) and MiniMed 780G (780G) conducted in a new extended follow-up study.
Research Design and Methods:
In this prospective open-label single-arm clinical trial, 41 adults with type 1 diabetes (age 35 ± 11 years, glycated hemoglobin [HbA1c] 6.4 ± 2.8% [46 ± 6.8 mmol/mol]) who have voluntarily used AAPS entered a total of three study phases. In the first phase, participants continued with AAPS for 3 months. In the second 3-month study phase, all participants initiated CIQ (n = 25) or 780G (n = 16). Finally, participants were switched back to the AAPS for the last 3 months phase. Results of the treatment with commercially available AID systems were compared with both AAPS phases.
Results:
Commercially available systems were comparable to AAPS in achieving time in range (TIR) (84.2 ± 7.6 vs. 85 ± 6.9%; P = 0.31) and in HbA1c (6.4 ± 3 vs. 6.3 ± 2.7% [46 ± 8.8 vs. 45.7 ± 6.2 mmol/mol]; P = 0.68). In contrast, time in tight range (TITR) was significantly higher in AAPS (66.38 ± 11.84 vs. 63.4 ± 11.77, P = 0.035). However, the time in hypoglycemia <70 mg/dL [<3.9 mmol/L] was significantly lower with commercially available AID systems (2.2 ± 1.2 vs. 3.8 ± 1.9%; P < 0.001). These results were consistent after switching back to AAPS.
Conclusion:
The extension of the Comparison of Different Hybrid Closed-Loop Systems—AndroidAPS and Control-IQ—in adults with Type 1 Diabetes study is the only prospective study to investigate switching between OS and commercially available AID systems. The switch from AAPS to commercially available systems was not associated with a change in TIR. However, the use of AAPS was associated with a higher TITR, but also with a higher risk of hypoglycemia.
Introduction
Automated insulin delivery (AID) systems, also known as hybrid closed-loop or artificial pancreas systems, are regarded as the optimal treatment choice for people living with type 1 diabetes (PwT1D). 1
Many individuals with type 1 diabetes (T1D) utilize open-source AID (OS-AID) systems, otherwise known as do-it-yourself artificial pancreas systems.2–8
Several studies have established that both OS-AID and commercially available AID systems demonstrate greater efficacy than sensor-augmented pumps (SAP).5–7,9–13 However, no prospective studies have compared commercially available AID and OS-AID systems. This lack of evidence challenges health care providers and researchers. They must answer questions of PwT1D regarding whether OS-AID systems deliver equivalent, inferior, or superior results.
We recently reported results from the Comparison of Different Hybrid Closed-Loop Systems—AndroidAPS and Control-IQ—in adults with Type 1 Diabetes (CODIAC) study, which compared the certified Control-IQ (CIQ) technology with Tandem t:slim X2 insulin pump and the OS AndroidAPS (AAPS) with Dana R/RS or Accu-Chek Insight insulin pump in 25 people with T1D. 14
In the current extended follow-up study, we evaluated switching between the OS-AID system AAPS and commercially available AID systems CIQ and MiniMed 780G (780G).
Research Design and Methods
Study design and participants
This nonrandomized, prospective, open-label, single-arm study compared the glycemic outcomes achieved by the commercially available CIQ or 780G technology and the OS AAPS in adults with T1D over 38 weeks. The study consisted of a 12-week AAPS phase, a 2-week run-in phase, a 12-week CIQ or 780G treatment phase, and a final 12-week AAPS phase (Fig. 1).
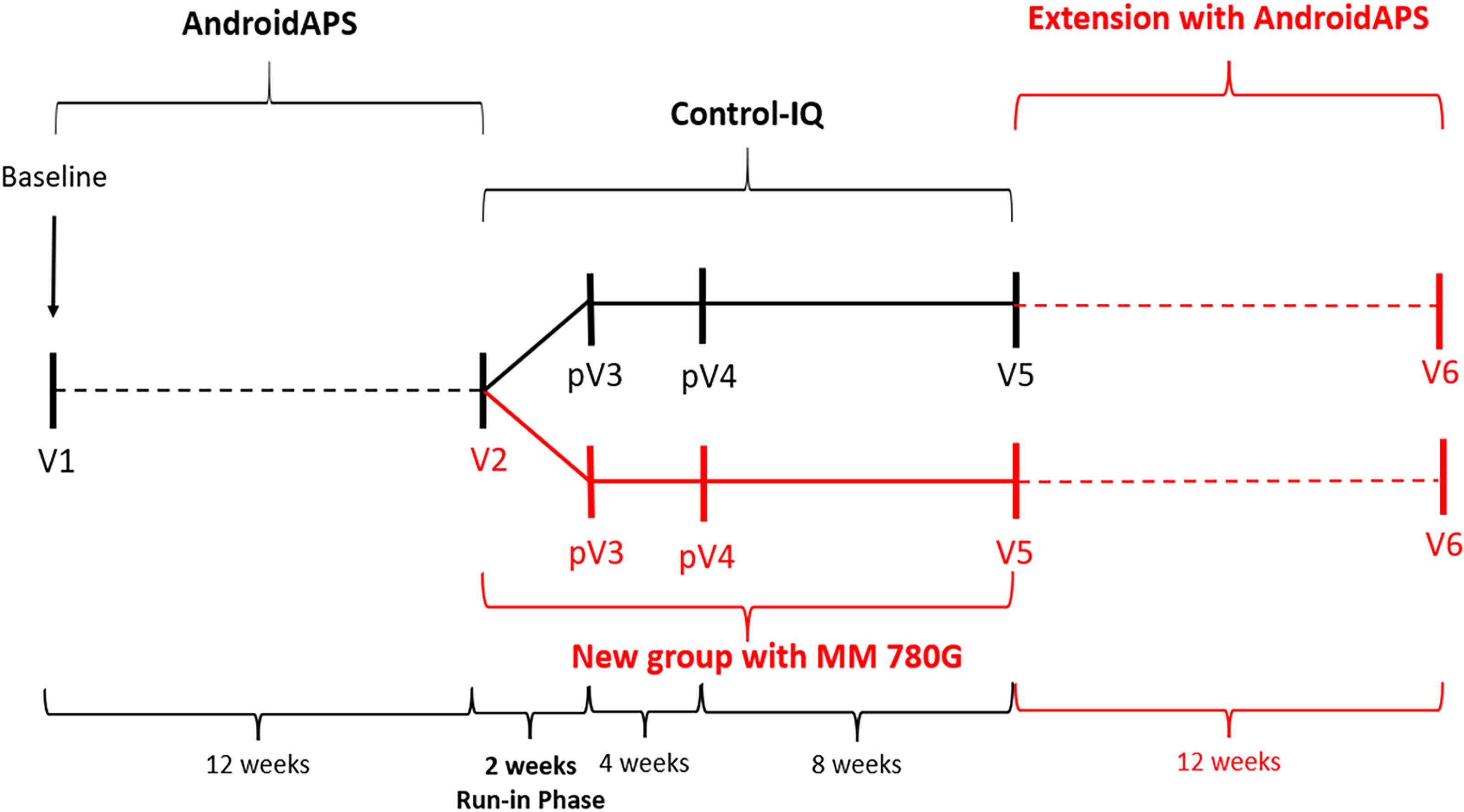
Study flow. V, visit; pV, phone visit.
PwT1D who had previously voluntarily used AAPS were recruited for this study.
The inclusion criteria were as follows: duration of T1D >2 years, age >18 years, and previous use of AAPS for at least 3 months before the start of the study.
Exclusion criteria included concomitant therapies affecting glucose metabolism, lactation, pregnancy, plans to become pregnant during the study, and nonadherence to the current treatment regimen.
The study was approved by an independent ethics review board and was conducted following the Declaration of Helsinki. The trial was registered with ClinicalTrials.gov (NCT 06709729). All participants provided their informed written consent before enrollment.
Procedures
Participants were scheduled for six study visits. At the first visit, participants who had already voluntarily used AAPS were screened for eligibility and informed about the study program. Eligible participants willing to participate in the study signed a written informed consent form. Parameters of glycemic outcomes and relevant medical history were collected. Participants were instructed to photograph and report skin problems at the sensor or cannula insertion sites to the investigators throughout the study.
Participants continued to use the AAPS between visits 1 and 2 (AAPS phase).
At visit 2, investigators obtained blood samples for laboratory analysis (glycated hemoglobin [HbA1c]), and other parameters of glycemic outcomes were collected using a unique uniform resource locator provided by Nightscout (an OS project that provides access to continuous glucose monitoring [CGM] data). Participants were switched to the CIQ or 780G system and trained by the study team on an outpatient basis. The first 25 participants entering the study were switched to CIQ, and the remaining 16 participants initiated 780G.
During the 2-week run-in phase, participants became familiar with the commercially available systems.
All participants were able to change settings between contacts throughout the study. However, staff were instructed to avoid monitoring outside of scheduled visits. This approach was designed to eliminate any effect on outcomes caused by additional planned contact with the study team.
During phone visits (visits 3–4), participants were asked about adverse events and device problems and had the opportunity to consult with the study team.
At visit 5, all data were downloaded from the devices for analysis, and investigators collected blood samples for laboratory analysis (HbA1c). Participants were switched back to the AAPS system.
At visit 6, all data were downloaded from the devices for analysis, and investigators collected blood samples for laboratory analysis (HbA1c).
Hybrid closed-loop systems
AAPS employs the identical heuristic-based algorithm as OpenAPS, designed to adhere to the mathematical principles utilized by individuals with diabetes in their insulin adjustment decisions. AAPS establishes communication with devices powered by the Android operating system, such as smartphones and tablets.
The connection between the CGM and pump was established through a Bluetooth bridge. In this study, 27 participants utilized the Dana R/RS pump, whereas 14 opted for the Accu-Check Insight pump, all paired with Dexcom G6. The AAPS algorithm fine-tunes the basal rate and incorporates features such as unannounced meals (UAM) and super microbolus (SMB). The SMB feature enables the system to autonomously administer a correction bolus, advancing the peak of insulin activity and implementing a temporary zero basal rate to avert overdosing. The UAM feature equips AAPS to manage incorrect carbohydrate estimations or meals high in protein and fat. The UAM can identify rapid glucose spikes triggered by carbohydrates, adrenaline, and other factors and attempt to compensate with SMB. Conversely, if a rapid glucose decline is detected, SMB delivery can be halted sooner. These functionalities enhance glucose regulation, but do not create an entirely closed-loop system. Every participant in this study utilized both SMB and UAM functionalities on AAPS.
Real-time CGM data flow into AAPS every 5 min, activating the embedded algorithm to forecast a glucose trajectory by taking into account the current sensor glucose value, insulin on board, carbohydrates on board, duration of action, insulin sensitivity factor (ISF), insulin/carbohydrate ratio (CIR), and other pertinent factors. It subsequently provides a temporary basal rate or SMB to the pump based on the discrepancy between the projected glucose value and the established glycemic outcomes target, aiming to maintain glucose levels within that target range. Each new AAPS user is required to undergo training and achieve 10 goals before commencing hybrid closed-loop therapy.9,15–17
The consensus statement for OS-AID systems 18 and the AAPS website 16 provide a detailed overview of AAPS setup options. Likewise, details about the CIQ algorithm can be found on the official Tandem website, whereas information about the 780G algorithm is provided on the official Medtronic website. 19
Outcomes
The primary end point was the difference in percentage of time in range (%TIR, 70–180 mg/dL [3.9–10.0 mmol/L]) between commercially available systems and AAPS. Secondary outcomes were changes in HbA1c (% Diabetes Control and Complications Trial (DCCT) [mmol/mol]), percentage of time spent in hypoglycemia (%TBR, <70 mg/dL [<3.9 mmol/L] and <54 mg/dL [<3.0 mmol/L]), percentage of time spent in hyperglycemia (%TAR, >180 mg/dL [>10.0 mmol/L] and >250 mg/dL [>13.9 mmol/L]), percentage of time in a tight range (%TITR 70–140 mg/dL [3.9–7.8 mmol/L]), glycemic variability expressed as the coefficient of variation (%CV), and mean sensor glucose.
Safety outcomes included the incidence of severe hypoglycemia (requiring second-party assistance to treat), ketoacidosis requiring hospitalization, skin reaction, infection, or hematoma at the sensor insertion site.
Statistical analysis
In addition to the fundamental descriptive attributes, the examination used assessments to ascertain statistical significance for dependent and independent factors. Before utilizing these assessments, the normality of the data was evaluated using the Shapiro–Wilk examination. The null hypothesis of this examination is that the data exhibit a normal distribution. We used the Wilcoxon signed-rank assessment to identify the discrepancies within independent factors, and the paired t assessment was utilized to illustrate potential differences between dependent factors.
In both cases, the null hypothesis was rejected with less than a 5% probability of error, that is, in cases where the value of P was below 0.05.
Results
Baseline characteristics and adherence
Forty-nine adults with T1D who had been previous AAPS users by their own decision were screened for eligibility to participate in this study. Of them, 41 participants were eligible to enter this study. The other eight participants did not meet the inclusion criteria.
Table 1 lists participants’ baseline characteristics. Data at baseline (visit 1) were comparable with those at visit 2 when CIQ or 780G was initiated (Table 2). Data from 30 participants were available for final analysis. Two participants withdrew during the 780G phase (the first participant stated technical issues as the reason, and the second one was not satisfied with the algorithm). Nine participants did not enter the final phase of the study (extension) because they decided to switch from AAPS to commercially available AID systems before the final phase.
Baseline Characteristics

HbA1c, glycated hemoglobin; AAPS, AndroidAPS; BMI, Body Mass Index.
Comparison of Visit 1 (Baseline) and Visit 2 in the AndroidAPS Phase

TIR, time in range; TBR, time spent in hypoglycemia; TAR, time spent in hyperglycemia; %CV, coefficient of variation; CI, Confidence Interval; SD, Standard Deviation.
Primary and secondary end points
The results at the end of the AAPS phase (V2) were compared with those after the 3-month CIQ or 780G phase (V5). In the study extension, the results at the end of the CIQ or 780G phase (V5) were compared with those after the final AAPS phase (V6).
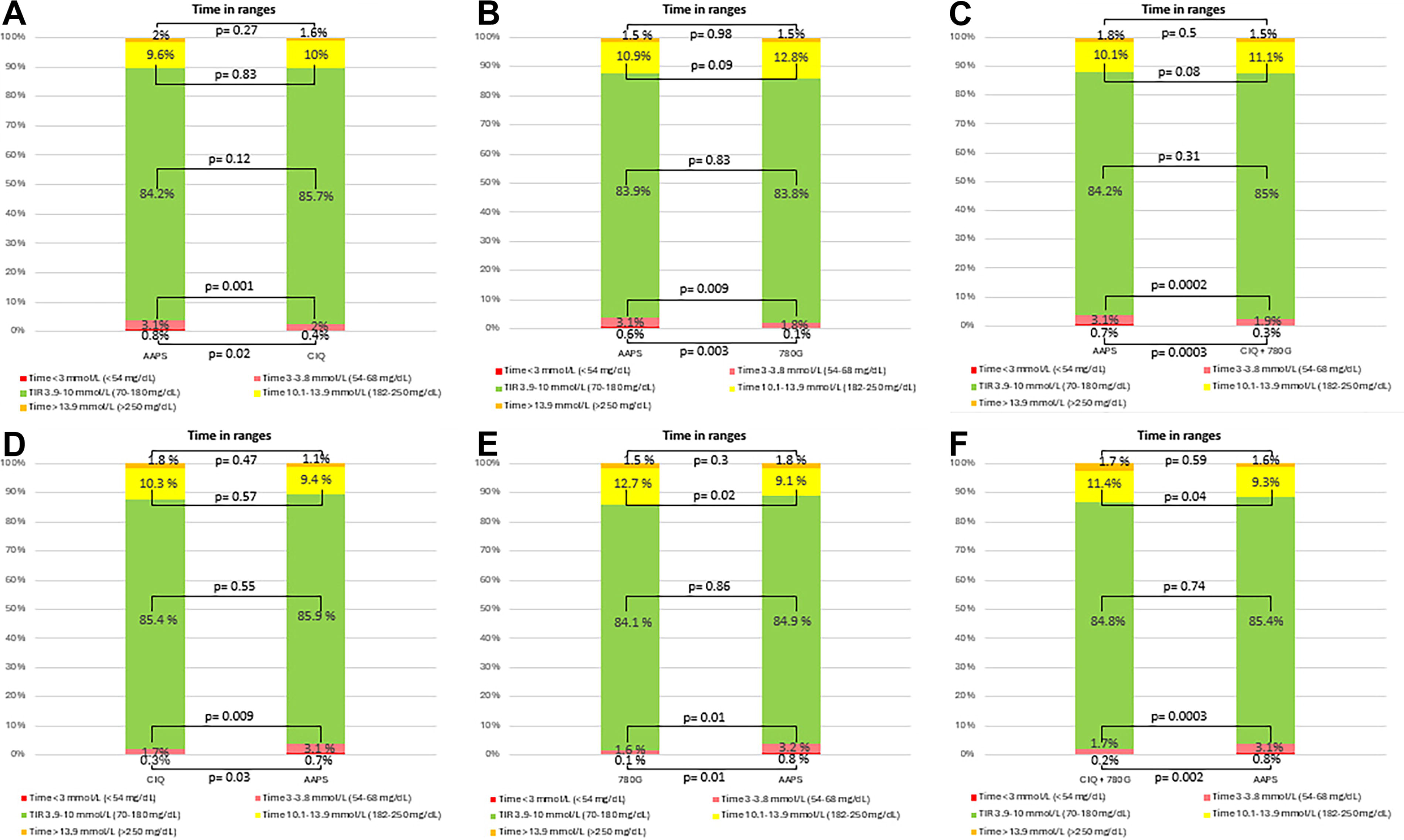
Commercially available systems (CIQ and 780G) were comparable with AAPS in achieving TIR (84.98 ± 6.86 vs. 84.15 ± 7.61%; P = 0.31). No significant difference in TIR was observed in a separate analysis for each AID system—CIQ versus AAPS (85.72 ± 7.64 vs. 84.24 ± 8.46%; P = 0.12) and 780G versus AAPS (83.81 ± 5.54 vs. 83.99 ± 6.38%; P = 0.83).
%TBR (<70 mg/dL [<3.9 mmol/L]) was lower when using commercially available systems than AAPS (2.17 ± 1.17 vs. 3.82 ± 1.92%; P < 0.001), in separate analyses for CIQ versus AAPS (2.4 ± 1.22 vs. 3.88 ± 2.04%; P = 0.001) and 780G versus AAPS (1.81 ± 1.01 vs. 3.74 ± 1.75%; P = 0.003).
In addition, following the transition from AAPS to CIQ, %TBR (<70 mg/dL [<3.9 mmol/L]) decreased in 16 participants, increased in 2 cases, and was unchanged in 7 participants. In the transition from AAPS to 780G, %TBR decreased in 12 participants, increased in 2 participants, and remained unchanged in 2 cases.
%TBR2 (<54 mg/dL [<3.0 mmol/L]) was also lower with commercially available AID systems compared with AAPS (0.27 ± 0.44 vs. 0.69 ± 0.65%; P < 0.001) in separate analyses for CIQ versus AAPS (0.4 ± 0.5 vs. 0.76 ± 0.77%; P = 0.02) and 780G versus AAPS (0.06 ± 0.24 vs. 0.59 ± 0.38%; P = 0.002).
% TITR (70–140 mg/dL [3.9–7.8 mmol/L]) was lower using commercially available systems than AAPS (63.4 ± 11.77 vs. 66.38 ± 11.84; P = 0.13). However, there was no difference in separate analyses for CIQ versus AAPS (65.1 ± 12.97 vs. 67.65 ± 12.44; P = 0.28) and 780G versus AAPS (61.13 ± 9.48 vs. 64.69 ± 10.75; P = 0.13) (Tables 3, 4).
Comparison of AndroidAPS (Visit 2) and Commercially Available Automated Insulin Delivery Systems Control-IQ and MiniMed 780G in a Separate Analysis (Visit 5)
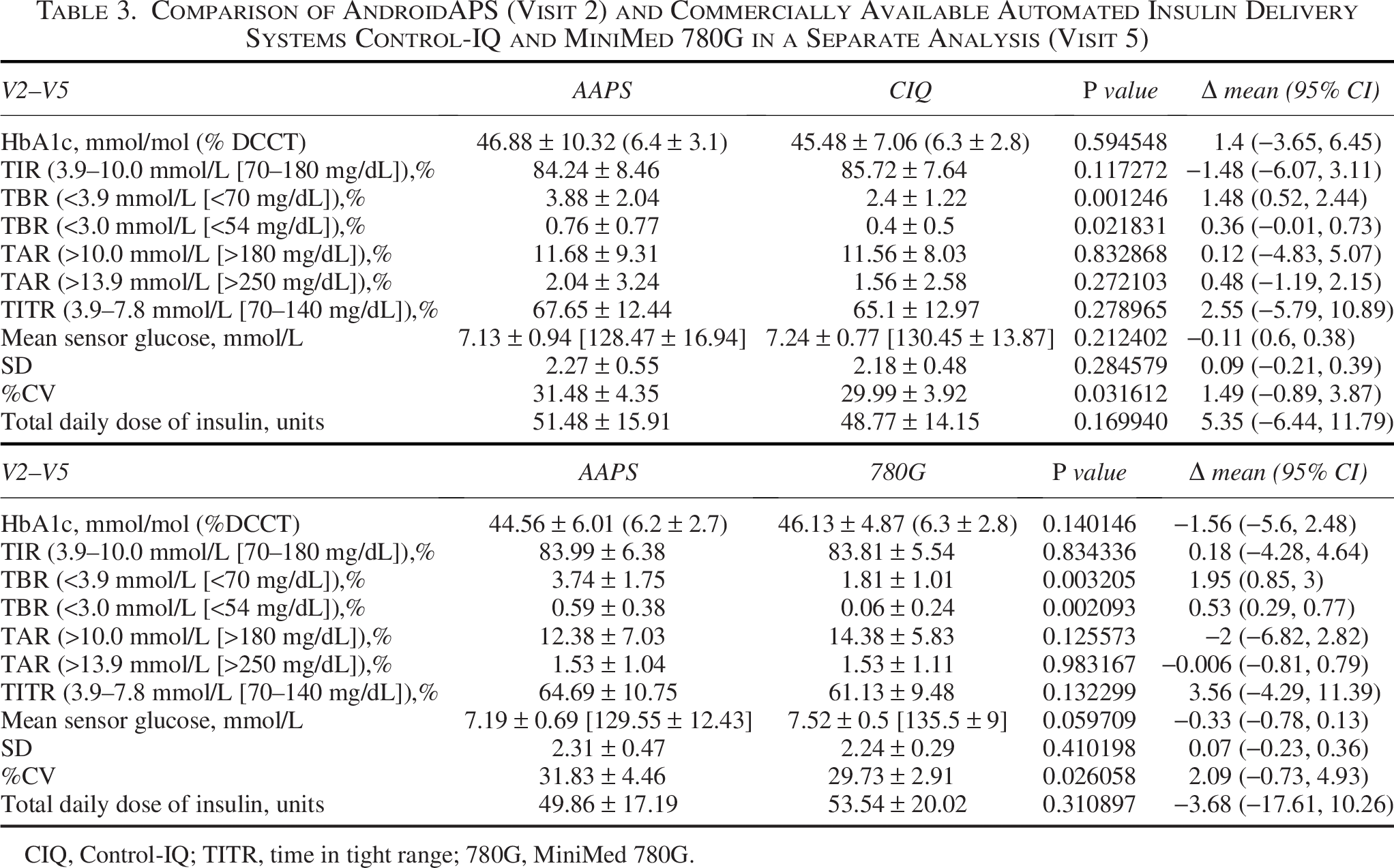

CIQ, Control-IQ; TITR, time in tight range; 780G, MiniMed 780G.
Comparison of AndroidAPS (Visit 2) and Commercially Available Automated Insulin Delivery Systems Control-IQ and MiniMed 780G in Combined Analysis (Visit 5)

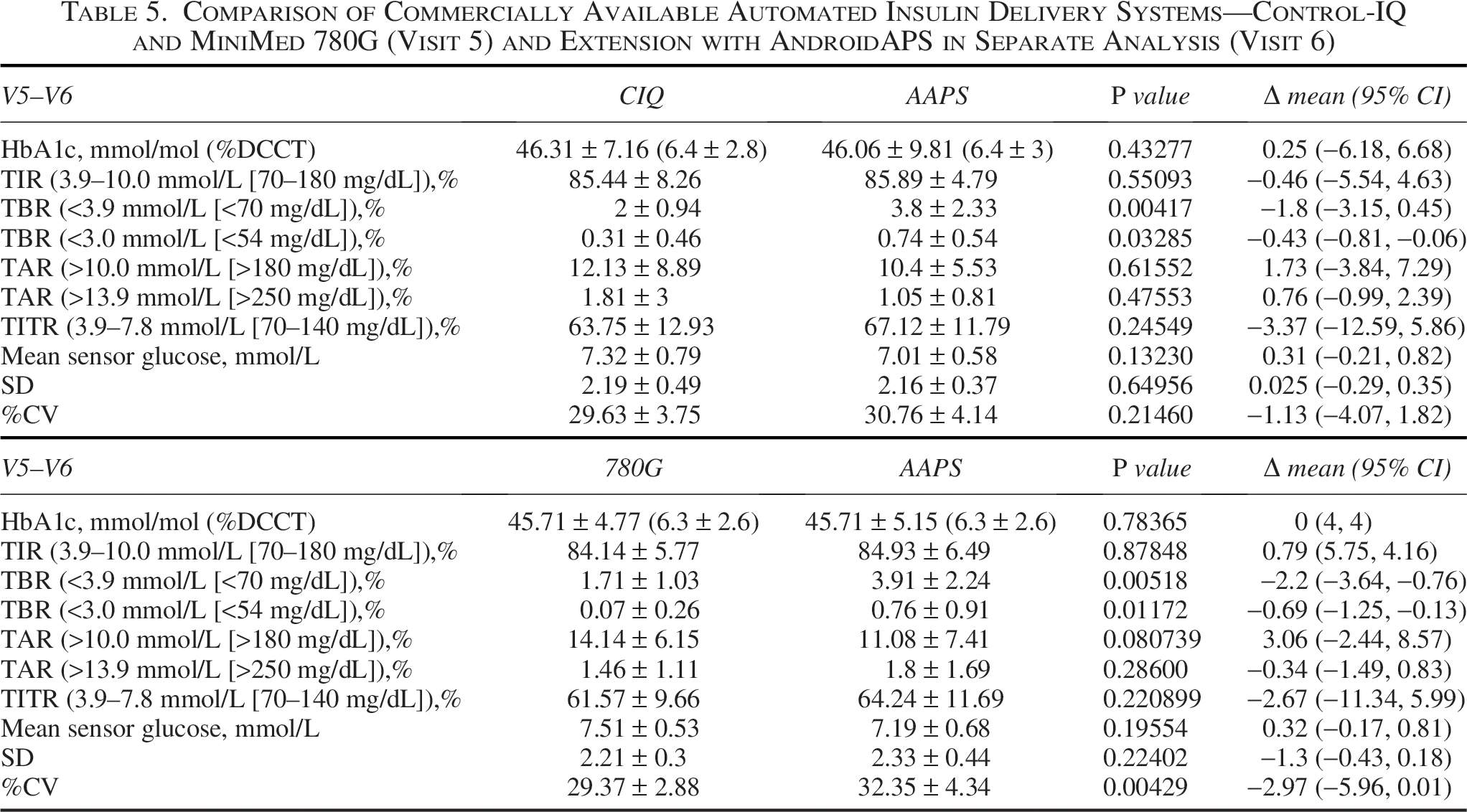
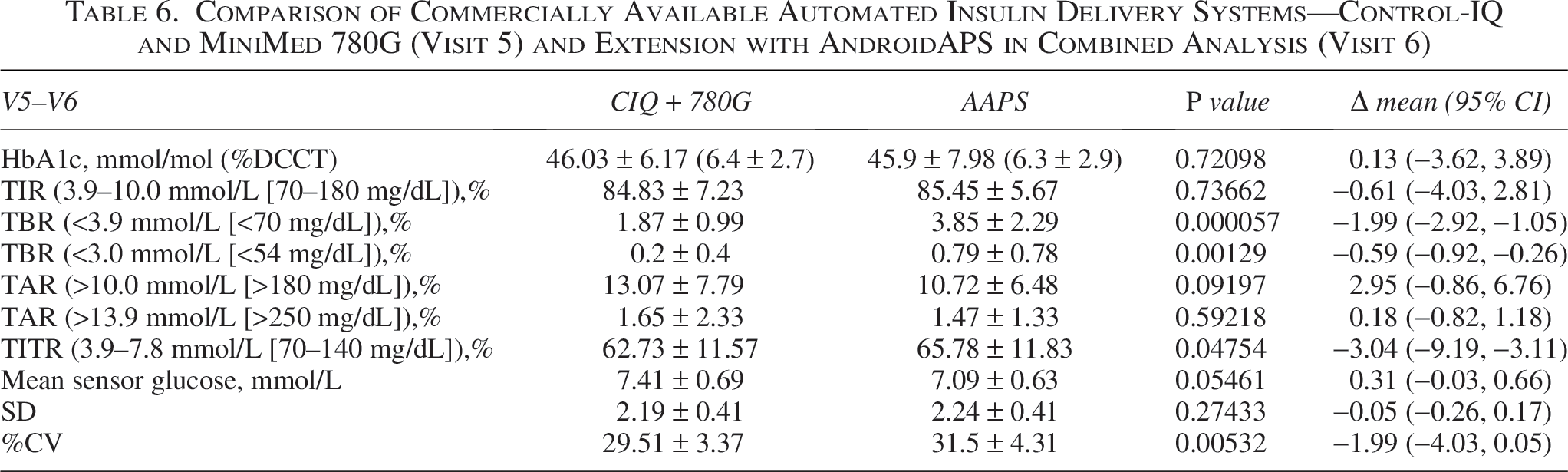
After returning to AAPS in the last study phase, there was no significant difference in TIR, TAR, mean sensor glucose, or HbA1c compared with commercially available systems.
However, time in hypoglycemia was again significantly higher with AAPS (3.9 ± 2.2 vs. 2.2 ± 1.2%; P < 0.001) and in the separate analysis for AAPS versus CIQ (3.8 ± 2.3 vs. 2 ± 0.94, P = 0.004) and for AAPS versus 780G (3.91 ± 2.24 vs. 1.71 ± 1.03, P = 0.005). %CV was significantly lower with commercially available systems (29.51 ± 3.37 vs. 31.5 ± 4.31%; P = 0.005) and in the separate analysis for 780G versus AAPS (29.37 ± 2.88 vs. 32.35 ± 4.34%; P = 0.004), but not for CIQ versus AAPS (29.63 ± 3.75 vs. 30.76 ± 4.14%; P = 0.21).
%TITR (70–140 mg/dL [3.9–7.8 mmol/L]) was higher using AAPS than commercially available systems (65.78 ± 11.83 vs. 62.73 ± 11.57; P = 0.048). However, there was no difference in separate analyses for CIQ versus AAPS (63.75 ± 12.93 vs. 67.12 ± 11.79; P = 0.25) and 780G versus AAPS (61.57 ± 9.66 vs. 64.24 ± 11.69; P = 0.22) (Tables 5, 6).
Comparison of Commercially Available Automated Insulin Delivery Systems—Control-IQ and MiniMed 780G (Visit 5) and Extension with AndroidAPS in Separate Analysis (Visit 6)

Comparison of Commercially Available Automated Insulin Delivery Systems—Control-IQ and MiniMed 780G (Visit 5) and Extension with AndroidAPS in Combined Analysis (Visit 6)
Safety outcomes
No episodes of severe hypoglycemia or diabetic ketoacidosis requiring hospitalization were reported. Despite 4-step bolus administration in CIQ, one participant mistakenly administered 25 units of bolus insulin. However, this mistake did not result in severe hypoglycemia. No skin reactions, hematomas, or infections at the sensor or cannula insertion sites were reported during this study.
Discussion
The extension of the CODIAC study is the only prospective study investigating the switch between OS and commercially available AID systems.
Commercially available AID systems and AAPS were comparable in TIR (70–180 mg/dL [3.9–10.0 mmol/L]). In addition to TIR, we did not observe any significant difference in secondary outcomes such as HbA1c and TAR (both >180 mg/dL [>10.0 mmol/L] and >250 mg/dL [>13.9 mmol/L]). However, the use of AAPS was associated with higher TITR, but also with risk of both levels of hypoglycemia (<70 mg/dL [<3.9 mmol/L] and <54 mg/dL [<3.0 mmol/L]). Although TBR was higher in AAPS, all participants using AAPS or commercial systems met consensus guidelines for TBR (<4%). 20
Consistent with hypoglycemia, glycemic variability (%CV) was also lower when using commercially available AID systems. The observed differences can probably be linked to the diverse algorithms utilized by these systems. AAPS, specifically, allows a high level of customization, which includes the option to set a target glycemia as low as 4.5 mmol/L. These features may, in turn, be associated with a higher incidence of hypoglycemia compared with commercially available systems. However, AAPS has more settings that can be adjusted and this makes it challenging to directly compare with commercially available AID systems, which have fewer settings other than basal rate, ISF, and CIR that a user can adjust.
In this study, we also assessed the TITR as a new, emerging metric for the assessment of normoglycemia.20,21
When using AAPS, the average glycemia and TITR appeared to be significantly better compared with a combined set of commercial systems. This difference might be explained by the numerically higher, although not statistically significant, mean sensor glucose and lower TITR observed with the 780G system. In contrast, the 780G system was associated with the lowest risk of hypoglycemia. Similar trends were noted in the extension study, suggesting a consistent pattern in the outcomes.
Although the occurrence of hypoglycemia in all systems aligns with expert consensus, 22 it is essential to recognize that the evaluation of hypoglycemia can differ in specific clinical situations, particularly in individuals who experience hypoglycemia unawareness or in elderly PwT1D with diabetes-related complications. Moreover, meeting the existing recommendations does not imply that we should not strive for even more optimal outcomes.
Clinicians frequently encounter the use of OS-AID in their daily practice. However, there is yet to be any established structured training for clinicians regarding OS-AID, and significant gaps remain in the evidence from clinical studies.
The majority of studies rely on self-reported data, which present a considerable risk of bias,23–26 with a few exceptions.5–8,27 A retrospective observational trial conducted by Jeyaventhan et al. 28 compared commercially available AID Medtronic MiniMed 670G with OS-AID. However, the specific type of OS-AID utilized in that study was not detailed.
In contrast to our study, we included only participants utilizing the single type of OS-AID system, which minimizes the potential for bias.
Moreover, 670G was the first generation of commercially available AID with many limitations.29,30 Burnside et al. conducted a clinical trial with AAPS. However, in contrast with our study, their trial evaluated the effectiveness of AAPS compared with SAP.6,7 In addition, a similar design was also used in other studies.5–8 However, we believe that comparing the AID system with SAP will not provide many new insights, and there is a need to concentrate on research involving diverse AID systems, as these are recognized as the standard treatment for type 1 diabetes.31,32 While significant variations among AID systems are typically not anticipated, 32 our study indicates that there might be differences that can be considered clinically relevant. It is also important to note that our trial involved adults with already optimal glycemic outcomes experienced in using AID, in contrast to other studies that included participants lacking AID experience. These aspects could have affected the inferior average outcomes observed in AAPS users compared with our findings.5–7 Our study is not without its limitations.
Participants were trained on commercial systems as part of the study design. No training by the study team or evaluation of settings or skills was available for the AAPS, this may have influenced the resulting rates of hypoglycemia in AAPS. However, AAPS training requires several weeks of self-training, and users must complete a series of 10 objectives before initiating AAPS. In contrast, the study team provided participants with standard training for CIQ or 780G, which lasted around 60 min.
Because this was a nonrandomized study, there is a possibility that certain unmeasured variables could influence our results. However, numerous countries mandate national regulatory approval for each device and software used in Randomized Control Trials (RCTs) and additional regulatory clearance for every version of those devices and software. These constraints make RCTs with OS-AID systems challenging to conduct in many countries, with a few exceptions.5–8 In the Czech Republic, where this trial was conducted, national regulatory authorities approved RCTs solely for studies conducted within hospital environments. 27 However, they did not allow RCTs to be conducted in home settings. The design of our trial represents the most that we could achieve within the existing regulatory landscape while still keeping the system OS.
Conclusions
The results showed that switching from AAPS to commercially available systems was not associated with a change in TIR and HbA1c. However, this study suggests that TITR may be higher when using AAPS, but also with a higher risk of hypoglycemia compared with both commercially available AID systems (Fig. 2).
Authors’ Contributions
J.Š. was responsible for the protocol design. J.Š., L.R., A.H., J.K., Q.D.D., E.H., G.G., C.G.P., M.F., and M.P. wrote and revised the article. Q.D.D., L.R., J.Š., A.H., J.K., M.F., E.H., and M.P. were responsible for study implementation and administration. Q.D.D., L.R., J.Š., A.H., J.K., C.G.P., and E.H. reviewed the data. J.Š. is the guarantor of this work and, as such, had full access to all the data in the study and had final responsibility for the decision to submit for publication.
