Abstract
Objective:
To evaluate trends in insulin delivery and day-to-day variability of insulin requirements over 48 months of hybrid closed-loop use following diagnosis of type 1 diabetes (T1D) in individuals aged 10–16 years.
Methods:
A secondary analysis of the closed-loop arm of an open-label, multicenter, randomized, parallel hybrid closed-loop trial assessing closed-loop insulin delivery in newly diagnosed children and adolescents with T1D was conducted. Mean total daily dose (TDD) over 24 h and during the night, as well as mean total basal and bolus insulin over 24 h, were calculated. Day-to-day variability of insulin requirements was evaluated over 24 h and at night.
Results:
TDD increased from 27.2 ± 16.1 units/d (mean ± standard deviation) at 0–3 months following diagnosis to 65.7 ± 24.9 units/d at 42–48 months. The proportion of total daily insulin delivered as basal insulin rose from 41% to 61% over 48 months. Day-to-day variability of insulin requirements after diagnosis was high (coefficient of variation at 0–3 months: 23.3 ± 0.9%) and remained stable over 48 months. No clinically relevant sex-based differences were observed in insulin requirements.
Conclusions:
During the first 48 months after diagnosis of T1D, insulin requirements in children and adolescents more than double with hybrid closed-loop insulin delivery. Over time, a greater proportion of insulin is administered via the closed-loop algorithm, and the high day-to-day variability in insulin needs underscores the importance of initiating adaptive closed-loop systems from diagnosis.
Keywords
Introduction
Type 1 diabetes (T1D) is caused by immune-mediated destruction of insulin-producing pancreatic β-cells in genetically susceptible individuals, leading to insulin deficiency. 1 The period following T1D diagnosis presents unique management challenges. Many individuals experience a partial remission, during which residual β-cell function temporarily reduces exogenous insulin requirements. However, as β-cell function declines and endogenous insulin production diminishes, insulin requirements typically increase. 2 Limited research has explored the day-to-day variability in insulin requirements after diagnosis.
Hybrid closed-loop insulin delivery systems, which use glucose-responsive insulin delivery to target specific glucose levels, have been shown to improve glycemic outcomes and quality of life in people with T1D.3–7 The CLOuD randomized controlled trial showed that closed-loop insulin delivery improved glycemic outcomes over 48 months after diagnosis compared with standard insulin therapy in children and adolescents with T1D, but this did not appear to confer a protective effect on residual C-peptide secretion. 8
The present post hoc analysis aimed to assess trends in closed-loop insulin delivery and day-to-day variability of insulin requirements in children and adolescents over 48 months following T1D diagnosis.
Materials and Methods
Study design and population
A secondary analysis of the closed-loop arm of an open-label, multicenter, randomized, parallel hybrid closed-loop trial was conducted to assess exogenous insulin requirements in the first 48 months following T1D diagnosis. Participants aged ≥10 and <17 years were recruited within 21 days of diagnosis of T1D and were randomized to either closed-loop or standard insulin therapy for 24 months. Participants were offered a 24-month optional extension phase with the allocated treatment. The present analysis includes only participants who used closed-loop therapy. The study was approved by an independent research ethics committee.
Closed-loop system
The Cambridge model predictive control algorithm (version 0.3.71, CamDiab Ltd., Cambridge, UK) was implemented in two hardware configurations sequentially. The FlorenceM configuration consisted of an unlocked Android smartphone running the algorithm, which communicated with a modified, next-generation sensor-augmented 640G Medtronic insulin pump (Medtronic Minimed, CA, USA) through a proprietary translator, and a Medtronic continuous glucose monitor (CGM) transmitter with Guardian 3 sensor. The CamAPS FX configuration used an unlocked Android smartphone to house the CamAPS FX closed-loop app, a Dana Diabecare RS insulin pump (Sooil Development, Seoul, Korea) or YpsoPump (Ypsomed, Burgdorf, Switzerland), and either the Dexcom G6 (Dexcom, San Diego, CA, USA) or FreeStyle Libre 3 (Abbott Diabetes Care, Alameda, CA, USA) CGM.
Both configurations used an algorithm housed in a mobile app to automatically adjust insulin doses every 8–12 min. The algorithm was initialized by entering the user’s weight and total daily dose (TDD), while insulin sensitivity and active insulin time were automatically calculated and adjusted as necessary. The CamAPS FX algorithm adjusts insulin delivery by modulation of basal infusion rates without any need for autocorrections, while bolus insulin delivery is user-initiated, usually for carbohydrate intake. The algorithm uses adaptive learning with respect to total daily insulin dose, diurnal variation, and insulin delivery around meals.
Data analysis
Metrics were calculated daily for each participant and then summarized over 3-month (91-day) periods for the first 24 months and 6-month (182-day) periods for the second 24 months, starting from the day after diagnosis. All metrics were summarized with equal weight given to each participant. For inclusion in the analysis, a participant’s data were only considered if at least 50% of each 24-h period was spent in closed-loop mode. Additionally, data from each participant were only included in a period if at least 30 days of data were available for the 91-day periods and at least 60 days for the 182-day periods.
Metrics included mean TDD over 24 h, during the day (06.00–23.59) and during the night (00.00–5.59), as well as mean basal and bolus insulin doses over 24 h. Insulin variability was assessed by calculating the coefficient of variation (CV) of total insulin dose over 24 h, during the day and during the night. We have previously used CV to evaluate variability of day-to-day insulin delivery in other populations to understand who experiences higher variability of insulin delivery and help inform who would benefit from more advanced therapies. 9 These metrics were calculated for female and male participants, as well as for the overall group.
Data analysis was completed using R Studio (R Foundation for Statistical Computing, Vienna, Austria). Data are presented as mean ± standard deviation (SD).
Results
A total of 48 participants (23 females and 25 males) aged 10–16.9 years were included in the analysis (Table 1). Owing to the data inclusion criteria as outlined in the methods, the number of users in each period varied. Exact numbers for each time period are provided in Supplementary Table S1. One participant was excluded as an outlier due to behavioral (deliberate carbohydrate restriction) factors impacting insulin requirements.
Demographics

Data presented as mean ± standard deviation.
BMI, body mass index.
Total daily dose
Mean ± SD TDD doubled over the first 12 months after diagnosis from 27.2 ± 16.1 units/d at 0–3 months to 54.0 ± 29.5 units/d at 12–15 months (Fig. 1B and Supplementary Table S1). Thereafter, the TDD increased more slowly to 65.7 ± 24.9 units/d at 42–48 months. A similar trend was observed with the daytime (06:00–23:59) TDD, which increased from 24.1 ± 14.3 units/d at 0–3 months to 47.1 ± 26.0 units/d at 12–15 months with a slower increase to 55.5 ± 20.4 units/d at 42–48 months (Supplementary Table S1). Nighttime (00:00–05:59) TDD also doubled from 3.2 ± 1.9 units/night at 0–3 months to 6.9 ± 3.8 units/night at 12–15 months and then increased to 10.2 ± 5.2 units/night at 42–48 months (Supplementary Table S1).
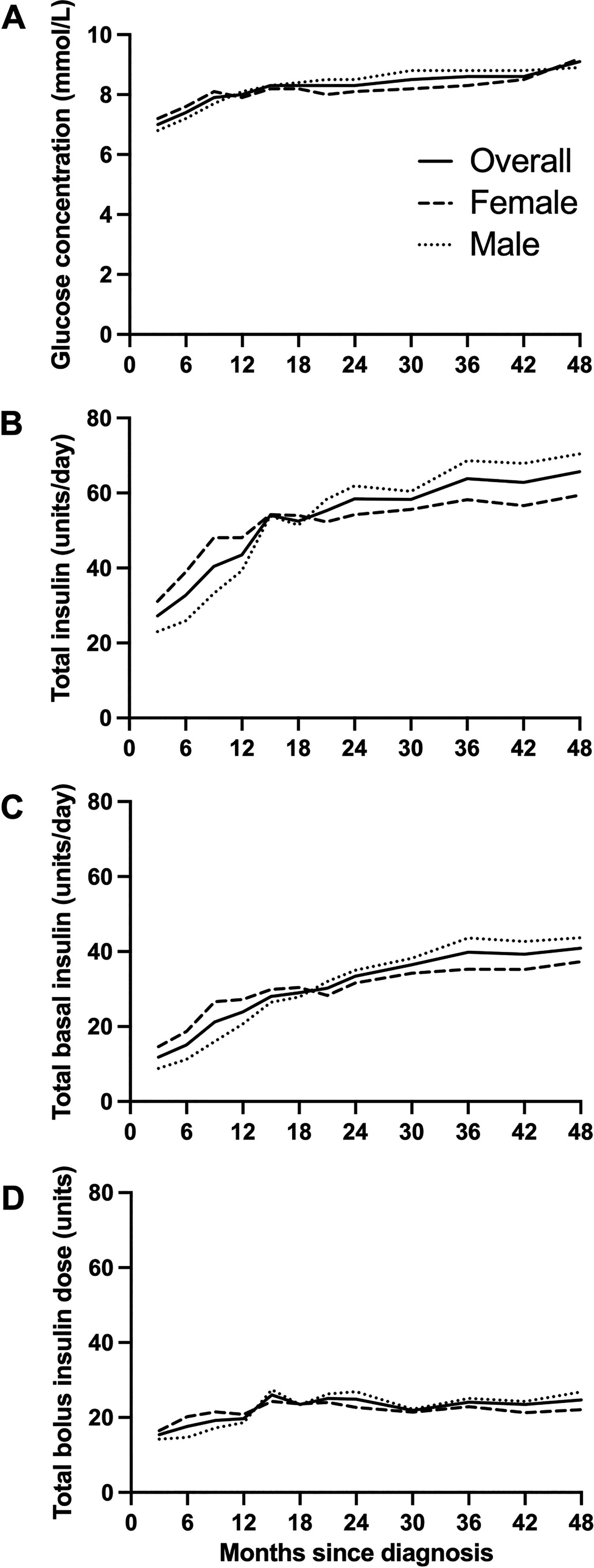
Glucose levels and insulin requirements during hybrid closed-loop insulin delivery over the 48 months following diagnosis of type 1 diabetes:
We observed a trend toward lower mean TDD in females than in males for the first 18 months after diagnosis, whereas from 18 months onwards, mean TDD tended to be higher in females than in males, although this was not tested statistically (Supplementary Table S1, Fig. 1B). This trend occurred in both total daytime and nighttime insulin delivery. A sensitivity analysis of TDD using higher thresholds of time in automode (>70% and >90%) showed a similar trend (data not shown).
Total daily basal insulin increased almost fourfold from 11.8 ± 9.9 units/d at 0–3 months after diagnosis to 40.9 ± 19.8 units/d at 42–48 months, while total daily bolus insulin delivery increased by less than twofold from 15.4 ± 7.0 units/d at 0–3 months to 24.7 ± 9.5 units/d at 42–48 months (Supplementary Table S1). The proportion of TDD delivered as basal insulin increased from 41% to 61% over 48 months (Fig. 2, Supplementary Table S2).
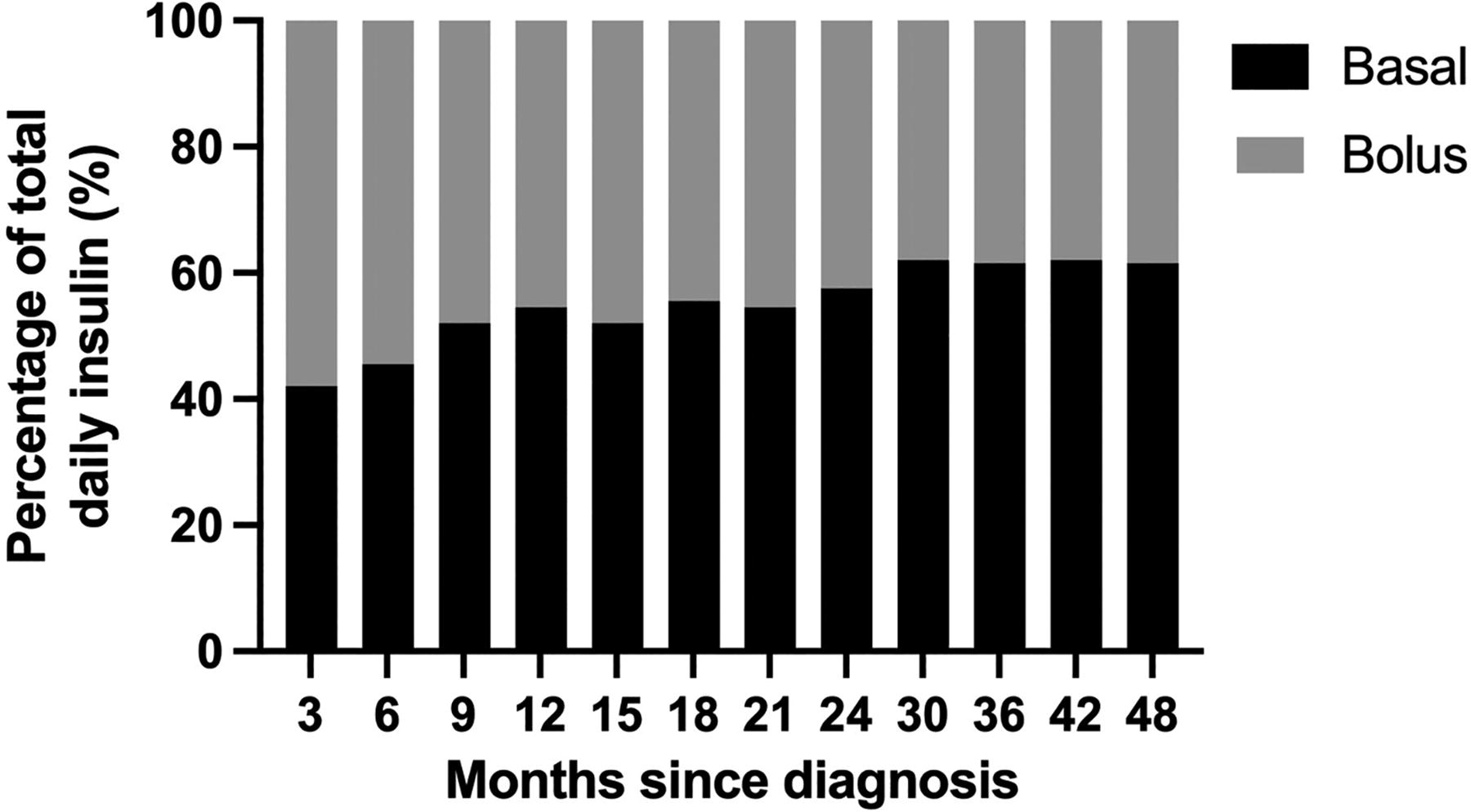
Proportion of total daily insulin as basal and bolus insulin (%) during hybrid closed-loop insulin delivery over the 48 months following diagnosis of type 1 diabetes.
Day-to-day variability
The CV of 24-h day-to-day insulin delivery remained stable over the 48 months with the lowest CV of 21.8% at 0–3 months and the highest CV of 23.9% at 12–15 months (Supplementary Table S1, Fig. 3A). Daytime (06:00–23:59) CV also appeared to be stable, with the lowest CV of 22.5% at 0–3 months and the highest CV of 25.3% at 12–15 months (Supplementary Table S1, Fig. 3B).
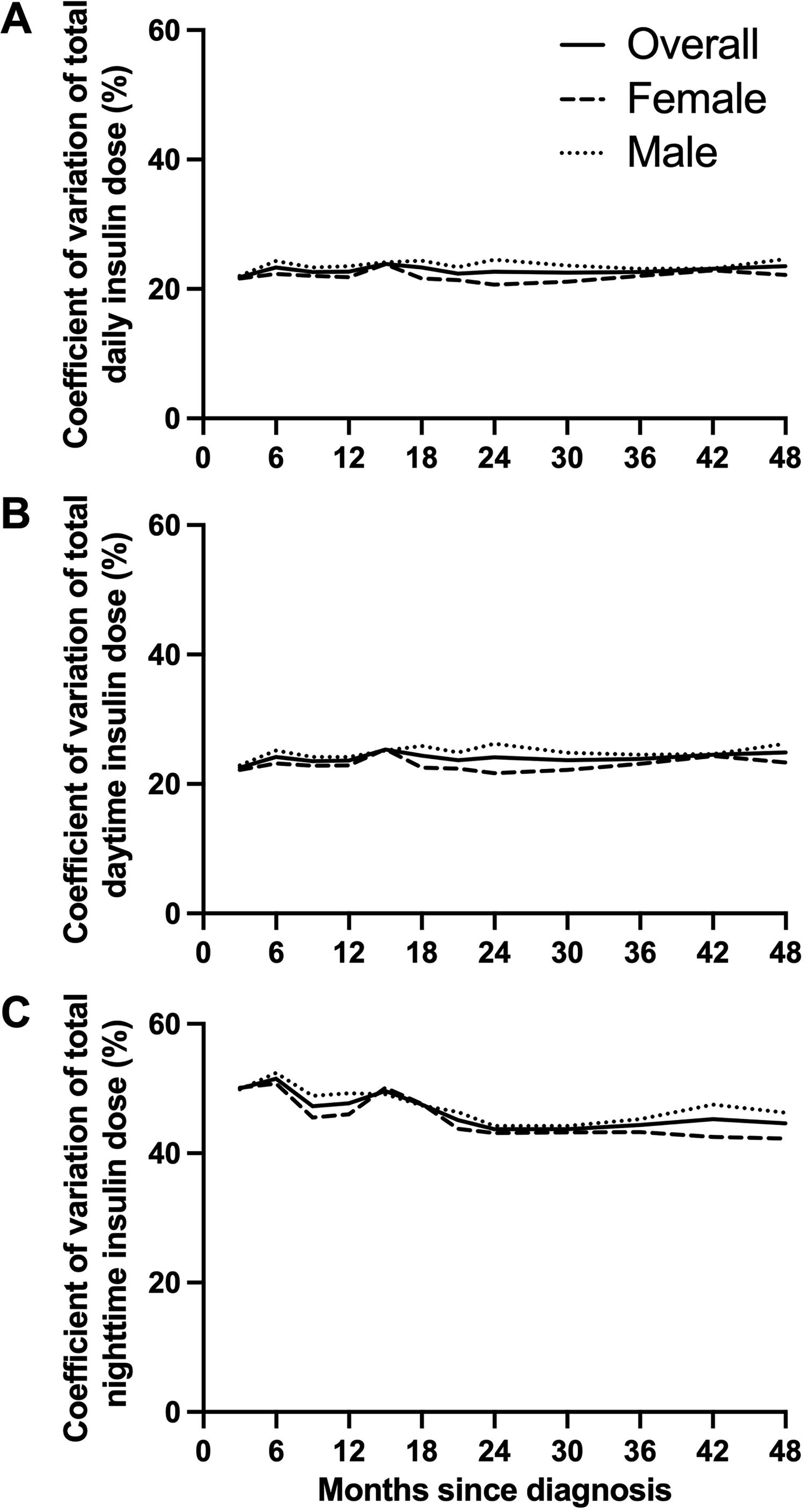
Day-to-day variability in insulin requirements during hybrid closed-loop insulin delivery over the 48 months following diagnosis of type 1 diabetes is depicted as follows:
We observed a trend toward a decrease in nighttime (00:00–05:59) CV from 50.0 ± 2.0% at 0–3 months to 44.6 ± 1.6% at 42–48 months (Supplementary Table S1, Fig. 3C). Nighttime CV ranged from 43.7% to 51.5%, while daytime CV ranged from 22.5% to 25.3% (Supplementary Table S1).
Glucose trends
Over 48 months postdiagnosis, mean glucose increased by 2.1 mmol/L (7.0 ± 0.8 to 9.1 ± 1.6 mmol/L), while time-in-range (TIR, 3.9–10.0 mmol/L) decreased from 82.2 ± 10.9% to 65.3 ± 12.6% (Supplementary Table S3, Fig. 1A).
Discussion
Our analysis demonstrates that total daily insulin requirements more than double with hybrid closed-loop insulin delivery over the first 48 months following a T1D diagnosis. Most of the increase in closed-loop insulin delivery occurred in the first 15 months and may relate to β-cell destruction and decreasing endogenous insulin production. 8
The CamAPS FX algorithm adjusts insulin delivery by modifying basal rates, while bolus insulin is user-initiated, primarily for meals or carbohydrate intake. Therefore, changes in basal insulin delivery directly reflect the algorithm’s adjustments. The increasing proportion of the total daily insulin composed of basal insulin over time suggests that the algorithm adapts to rising insulin requirements, while the reducing proportion of bolus insulin could be hypothesized to be due to a lack of adjustment of carbohydrate ratios or reducing bolus frequency over time. This is particularly relevant considering that missed boluses, suboptimal adherence to management, and elevated diabetes distress are frequently reported in adolescent populations.10–12
The variability in day-to-day insulin delivery, measured as CV, was relatively stable over the 48 months after diagnosis. The trend toward decreasing variability of night-to-night insulin delivery over time may reflect declining endogenous insulin secretion. The CLOuD randomized controlled trial showed that C-peptide levels declined most rapidly over the first 24 months and then declined less rapidly over the following 24 months, which would support this observation. 8 The variability of day-to-day insulin delivery we observed in this cohort of newly diagnosed children and adolescents was higher (23%–25%) than is reported in age-matched peers with established T1D (19%) and more comparable to the CV reported in very young children with T1D (25%). 9 Newly diagnosed children and adolescents are therefore likely to derive particular benefit from adaptive closed-loop insulin therapy. This is also supported by the improved glycemic outcomes in those using a hybrid closed-loop system compared to those receiving standard insulin therapy reported in the original study. 8
There is growing interest in the influence of sex on the management and outcomes of T1D, with current evidence conflicting.13–15 No significant differences in glycemic outcomes between sexes have been observed with closed-loop systems.4,16 In the present analysis, we observed a trend toward lower closed-loop insulin delivery in females than in males for the first 18 months after diagnosis and higher closed-loop insulin delivery in females than in males thereafter. However, importantly, pubertal status was not recorded in this study.
Strengths of the present analysis include the long duration of closed-loop insulin delivery following diagnosis and the use of a highly adaptive closed-loop algorithm. Limitations include the small sample size (N = 48), low ethnic diversity (85% White), lack of data on pubertal status, limited participant data for some periods, and the presentation of insulin as TDD instead of by weight, as weight data were not available for all the time periods. It was also not possible to calculate how much of the total daily insulin is directly due to use of the Boost function; however, the median (interquartile range) frequency of Boost use was 0 (0, 1) per day (data not shown).
Conclusions
In conclusion, over the first 48 months after diagnosis of T1D, insulin requirements in children and adolescents more than double with closed-loop insulin delivery. Over time, a greater proportion of insulin is delivered by the closed-loop algorithm. The day-to-day variability of insulin requirements is high from diagnosis, supporting early use of adaptive closed-loop systems after diagnosis of T1D.
Authors’ Contributions
C.R., C.K.B., and R.H. designed the analysis. C.K.B., J.W., J.M.A., M.E.W., S.H., A.T., T.R., A.G., R.E.J.B., D.E., N.T., and F.M.C. were responsible for the enrollment of participants, arranged informed consent from the participants, and provided participant care throughout the trial. R.H. designed the control algorithm. C.R. carried out data analysis. C.R., C.K.B., and R.H. contributed to the interpretation of the results. All authors critically reviewed the article. C.R., C.K.B., and R.H. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Author Disclosure Statement
C.R. reports no conflicts of interest related to the present article. J.W. has received speaker honoraria from Ypsomed and Novo Nordisk. M.E.W. reports patents related to closed-loop and being a consultant at CamDiab. S.H. serves as a member of the Medtronic advisory board, is a director of Ask Diabetes Ltd., providing training and research support in health care settings, and reports having received training honoraria from Medtronic and Sanofi and consulting fees for CamDiab. T.R. receives consultancy fees from Abbott Diabetes Care, has received honoraria from Novo Nordisk for delivering educational meetings, and has received honoraria from Sanofi for delivering educational meetings and participating in advisory boards. R.E.J.B. reports receiving speaker honoraria from Eli Lilly and Springer Healthcare, reports sitting on the Novo Nordisk UK Foundation Research Selection Committee on a voluntary basis, acted as an independent advisor for Provent Bio, and received speaking honoraria from Sanofi and Medscape, which were donated to an education research fund. F.M.C. has received honoraria for speakers’ fees and advisory board attendance from Abbott, Lily, Sanofi, Medtronic, and Insulet. C.K.B. is a consultant at CamDiab and has received speaker honoraria from Ypsomed. R.H. reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk; receiving license fees from BBraun; having patents related to closed-loop; being a consultant to Abbott Diabetes Care; and being a director at CamDiab. No other potential conflicts of interest relevant to this article were reported.
Funding Information
This project was funded by the National Institute for Health Research (NIHR) Efficacy and Mechanism Evaluation program (14/23/09), the Helmsley Trust (2016PG-T1D045 and 2016PG-T1D046), and JDRF (22-2013-266 and 2-RSC-2019-828-M-N). Additional support for the artificial pancreas work from the NIHR Cambridge Biomedical Research Centre and the NIHR Oxford Biomedical Research Centre. Abbott Diabetes Care supplied free glucose monitoring devices, and Dexcom and Medtronic supplied discounted continuous glucose monitoring devices. Medtronic supplied discounted insulin pumps, phone enclosures, continuous glucose monitoring devices, and pump consumables. C.R. is funded by a Diabetes UK PhD Studentship. C.K.B. is a recipient of a EFSD/Novo Nordisk Foundation Future Leaders Award. R.H. is a recipient of the EASD-Novo Nordisk Foundation Diabetes Prize for Excellence. The views expressed are those of the author(s) and not necessarily those of the NIHR, the Department of Health and Social Care, or other funders.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
