Abstract
Introduction:
Exercise benefits individuals with type 1 diabetes (T1D) but is often hindered by the risk of hypoglycemia. While guidelines provide strategies to mitigate this risk, adherence to recommendations on exercise remains low. Bolus calculators, such as Hedia Diabetes Assistant (HDA), offer a potential way to address this challenge by integrating exercise-related insulin adjustments into daily routines. This trial evaluates the safety and efficacy of HDA in improving glycemic control during exercise.
Methods:
This randomized, controlled crossover pilot study included 20 adults diagnosed with T1D for >1 year using multiple daily insulin injections and HbA1c between 42 and 72 mmol/mol (6.0–8.6%). Participants were randomized to first use either HDA (arm A) or their habitual diabetes management (arm B) before and after performing a 45-min moderate-intensity session on an exercise bike and subsequently crossover. The primary outcome was change in time in range (TIR) over the two 24 h periods.
Results:
The mean differences (95% confidence interval [CI]) in TIR and time above range (>10.0 mmol/L) were −2.3% (−13.1%, 8.5%) and 3.93% (−5.7%, 13.5%), respectively, and the median difference in time below range (<3.9 mmol/L) was −0.87% (−2.8%, 0.5%). The incidence rate ratio (95% CI) for total hypoglycemic episodes using HDA was −0.56 (−1.41, 0.29). Safety outcomes revealed fewer adverse events when using HDA.
Conclusion:
The HDA insulin bolus calculator with an integrated exercise feature appears safe to use when compared with habitual insulin management in people with T1D during moderate-intensity exercise.
Introduction
Exercise plays an important role in the management of type 1 diabetes (T1D), offering numerous benefits, including improved glycemic control, reduced cardiovascular risk factors, weight management, and improved well-being. 1 Current guidelines recommend that adults with T1D perform at least 150 min of moderate or vigorous intensity aerobic activity with activity occurring at least 3 days per week. 1 However, engaging in exercise can be a challenge for people with T1D due to the risk of hypoglycemia during and after exercise. 1 Traditionally, aerobic exercise has been associated with a lowering of blood glucose levels, whereas anaerobic exercise is thought to increase blood glucose levels. 2 However, recent real-world evidence suggests that blood glucose levels tend to decrease regardless of the exercise modality.3,4 This is especially relevant, as fear of hypoglycemia is the most significant barrier to regular exercise in adults. 5
To mitigate the risks associated with exercise, international guidelines provide evidence-based strategies for managing glycemia around exercise. These guidelines offer recommendations on insulin adjustments, carbohydrate intake, and monitoring strategies to minimize the risk of hypoglycemia. 2 However, adherence to guidelines is often low, as implementing behavioral actions, such as reducing bolus insulin doses, can be complex and difficult to implement in daily routines. 6
One potential solution is integrating the guidelines into a bolus calculator. Hedia Diabetes Assistant (HDA) is one such bolus calculator, designed to adjust insulin bolus doses based on the duration and intensity of the exercise or recommend carbohydrates depending on the blood glucose levels. While bolus calculators could potentially facilitate adherence to exercise-related diabetes management strategies, their safety and efficacy in relation to exercise remain largely unexplored.
This pilot crossover randomized controlled trial aimed to evaluate the safety and efficacy of HDA, assessing its ability to improve glycemic control and prevent hypoglycemic episodes.
Methods
Study design and participants
The study was designed as a prospective, randomized, open-label, blinded-endpoint, pilot crossover controlled study (ClinicalTrials.gov identifier: NCT05458167) comparing the use of HDA with habitual diabetes management. The study was conducted between April 2023 and April 2024.
Participants were recruited from the outpatient clinic at the Department of Endocrinology and Nephrology, Copenhagen University Hospital – North Zealand, Hillerød, Denmark. The participants took part in four visits in total (three onsite visits and one remote visit). At Visit 1, participants were randomized to either arm A or arm B, provided with a blinded Dexcom G6 continuous glucose monitoring (CGM) (Dexcom, San Diego, California, USA) and a smartphone with the HDA app installed, and trained on study procedures. Baseline data were collected and included blood samples, blood pressure measurements, determining maximal heart rate, and self-reported exercise levels using the Saltin–Grimby Physical Activity Level Scale. 7
At Visits 2 and 3, participants performed 45-min bike tests at moderate intensity. Arm A used HDA for insulin dose calculations for 24 h starting from the participant’s breakfast (and insulin dosing) taking place 1 h before Visit 2, whereas arm B used their habitual management, and vice versa for Visit 3. The bike tests were separated by a washout period of at least 48 h.
At Visit 4, participants were contacted by phone to complete the System Usability Scale (SUS) questionnaire, which is a standardized tool for assessing usability through 10 five-point Likert scale questions, generating a score from 0 to 100. 8
Inclusion criteria included age 18–75 years, T1D diagnosis for at least 1 year, basal-bolus insulin regimen, HbA1c between 42 and 72 mmol/mol (6.0–8.6%), and body mass index between 18.5 and 35 kg/m2. Exclusion criteria included pregnancy, breastfeeding, treatment with sodium-glucose transport protein 2 inhibitors, glucagon-like peptide-1 receptor agonists, or glucocorticoids, significant cardiovascular conditions, or expected noncompliance. Full eligibility criteria are detailed in Supplementary Table S1.
The study was conducted in accordance with the Declaration of Helsinki, ISO 14155:2020, and a Clinical Investigation Plan approved by the Danish Medical Research Ethics Committees (#2210121). Signed informed consent was obtained from all participants as well as a General Data Protection Regulation consent.
Intervention
HDA (
Participants were trained in HDA by the site personnel, who also ensured the settings on insulin-to-carbohydrate ratio and insulin sensitivity factor were appropriate based on a review of the medical record, glucose levels prior to enrollment, and interviewing the patient.
Bike test
Participants were instructed to refrain from exercising 48 h before the bike tests and during the 24 h observation periods following the bike tests. They were also asked to refrain from consuming alcohol and smoking 24 h before the exercise bouts and during the 24 h observation periods. Otherwise, they should live as normally as possible.
On the day of the bike test, participants using HDA (intervention period) would start using HDA in the evening prior to the bike test to become familiar with the app, however, without following the recommendations yet. They used HDA to calculate an insulin dose in the morning, 1 h before the bike test, where they entered 45 min of moderate-intensity exercise and the carbohydrate content of their breakfast into the app. Based on this input, HDA recommends an insulin dose that was reduced by 50%. If glucose levels were low, the app would instead recommend carbohydrates.
The participants needed to follow the advice made by HDA. Participants following their habitual diabetes management (control period) were instructed to manually log their insulin doses from the evening before the bike test on study-specific sheets. Participants were instructed to eat an identical breakfast on the 2 days with the bike tests.
The bike tests were performed on an ergometer bike, continuously adjusted to ensure a moderate intensity (64%–76% of maximal heart rate, Borg scale 12–13). 9
Blood glucose was measured before each bike test using a blood glucose meter. The bike test was postponed if blood glucose was <5 mmol/L or >15 mmol/L or if the participant’s temperature exceeded 38°C. Blood glucose was also measured at 20 and 45 min during the test, and CGM measurements were continuously monitored every fifth minute. If blood glucose fell below 3.9 mmol/L, the test was halted and not resumed, even after the treatment of hypoglycemia.
One hour after exercise, participants consumed a snack containing 50 g of carbohydrates, accompanied by a 50% reduction in the insulin bolus dose during the intervention period and participant-determined bolus insulin dosing during the control period. No other meals were allowed until 4 h postexercise.
Outcomes
Glycemic outcomes were assessed by the blinded CGM.
The primary outcome was the change in time in range (TIR, % of time with glucose 3.9–10.0 mmol/L) between the two 24 h periods at visits 2 and 3 starting 1 h before each bike test. 10
Confirmatory secondary outcomes included hypoglycemic events (glucose <3.9 mmol/L), time above range (TAR, glucose >10.0 mmol/L), and time below range (TBR, glucose <3.9 mmol/L). 10
Supportive secondary outcomes included glycemic variability (coefficient of variation [CV] and standard deviation [SD] of the glucose level distribution), total bolus insulin dose (correction and meal dose), number of correctional events, and SUS. 8
Safety outcomes included adverse events and serious adverse events.
Randomization and blinding
Participants were randomized 1:1 to arm A or arm B using a computer-generated sequence, with allocation determined by scratch-off cards. Participants and site staff could not be blinded to the intervention, but study CGM data were blinded to participants during the entire study. The statistician remained blinded during the initial analysis.
Statistical methods
Statistical analyses followed a predefined statistical analysis plan. No formal sample size calculation was performed; 20 participants were included to provide initial safety and efficacy data and support future studies.
Continuous variables were presented as mean with SD or as median with interquartile range (IQR: Q1–Q3 range), depending on data distribution, and categorical data as proportions. Effect sizes were presented as model-based estimates of treatment effects with 95% confidence intervals (CIs). P values were not reported due to the low sample size.
For the primary and secondary outcomes, the participants were analyzed according to their randomized assignment (intention-to-treat [ITT]). Subgroup analyses evaluated outcomes over 12- and 5 h periods starting 1 h before the bike tests.
Primary and secondary outcomes were analyzed using a linear mixed-effects model, with treatment, period, and sequence as fixed effects and participants as random effects. Hypoglycemic episodes and correctional events were analyzed using negative binomial models. Data transformations or nonparametric methods were applied if model assumptions were violated.
Missing data were handled using the framework proposed by White et al. 11 All available data were used for efficacy endpoints. Sensitivity analyses for the primary outcome included per-protocol analysis, complete case analysis (excluding participants with <70% CGM data), and two imputation methods: conservative imputation (assuming no difference) and multiple imputation (using chained equations under a missing-at-random assumption).
Hypoglycemic events required three consecutive CGM readings below 3.9 mmol/L (totaling 15 min). Hypoglycemic episodes and TBR were classified into level 1 (3.0–3.9 mmol/L) and level 2 (<3.0 mmol/L). Similarly, TAR was divided into level 1 (10.1–13.9 mmol/L) and level 2 (>13.9 mmol/L). 10
Safety outcomes will be presented descriptively without formal statistical testing.
Results
A total of 28 patients were screened, 20 participants were randomized, and 19 participants completed the study. Eight were screen failures, and one withdrew from the study.
Ten participants were randomized to arm A, and ten participants were randomized to arm B.
One participant in arm B withdrew consent before the bike test on the intervention day. Otherwise, all participants had some CGM data available for both bike tests and the ITT population included 10 participants in each group (Fig. 1).
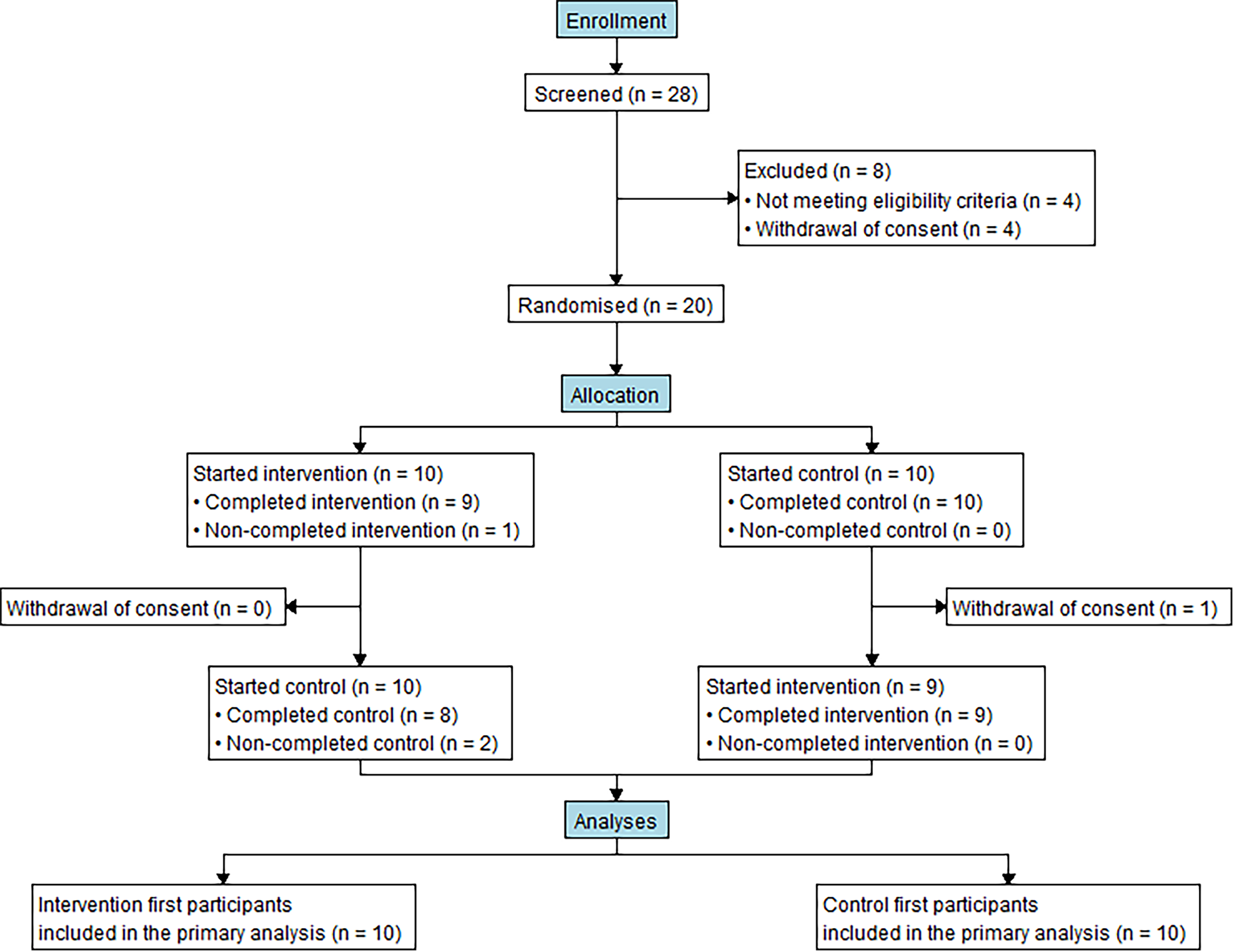
Flowchart of patient inclusion.
Baseline characteristics are shown in Table 1. Adherence with the intervention was high with 96.3% of all recommendations by HDA being followed. In those who deviated from the insulin recommendation, the mean deviation was ±3 units. The maximum number of deviations from the recommendations per participant was 1.
Baseline Characteristics
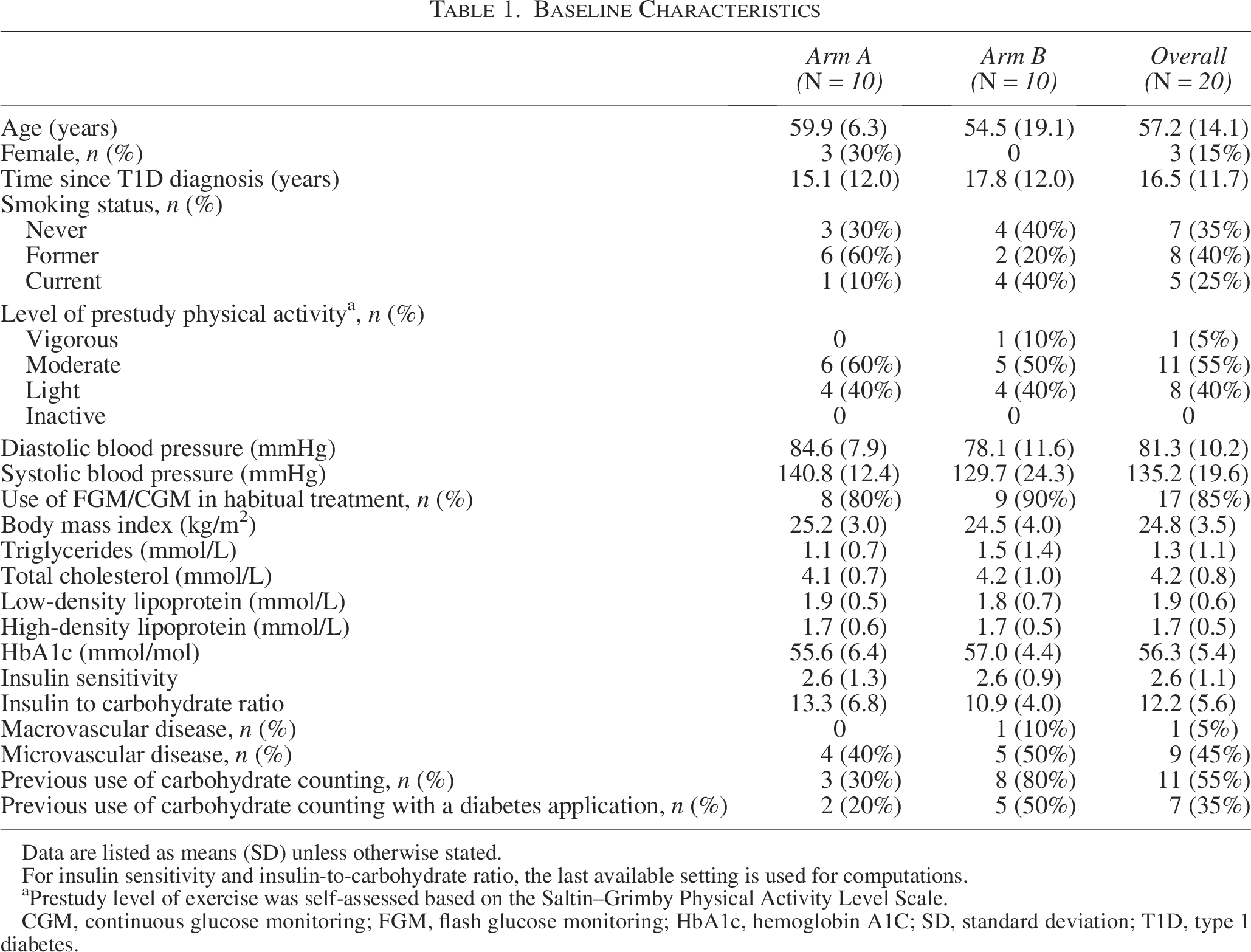
Data are listed as means (SD) unless otherwise stated.
For insulin sensitivity and insulin-to-carbohydrate ratio, the last available setting is used for computations.
Prestudy level of exercise was self-assessed based on the Saltin–Grimby Physical Activity Level Scale.
CGM, continuous glucose monitoring; FGM, flash glucose monitoring; HbA1c, hemoglobin A1C; SD, standard deviation; T1D, type 1 diabetes.
Primary outcome
The mean (SD) TIR (3.9–10.0 mmol/L) during the 24 h study period was 57.6% (21.6%) in the intervention period and 60.1% (17.7%) in the control period. The mean difference (95% CI) was −2.3% (−13.1%, 8.5%) (Table 2, Fig. 2).
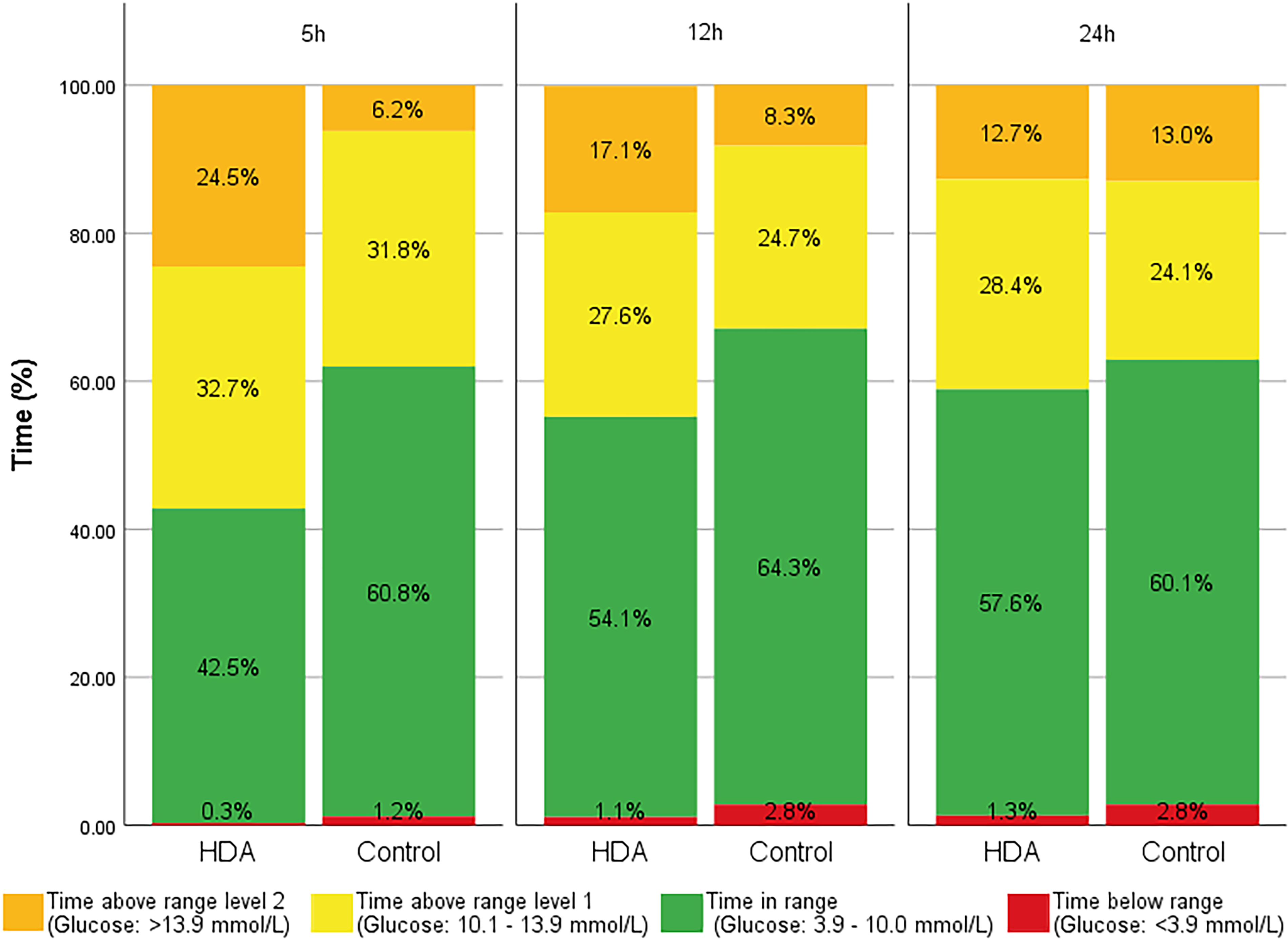
Overview of time in range, time above range, and time below range for the 5, 12, and 24 h periods. 5 h, 12 h, and 24 h denote 5 h, 12 h, and 24 h observation periods each starting 1 h before exercise. Time below range level 1 and level 2 are aggregated due to very low time below range level 2. HDA, Hedia Diabetes Assistant.
Effects of Using Hedia Diabetes Assistant Application or Habitual Diabetes Management During a 24 Hour Period, Including Physical Activity
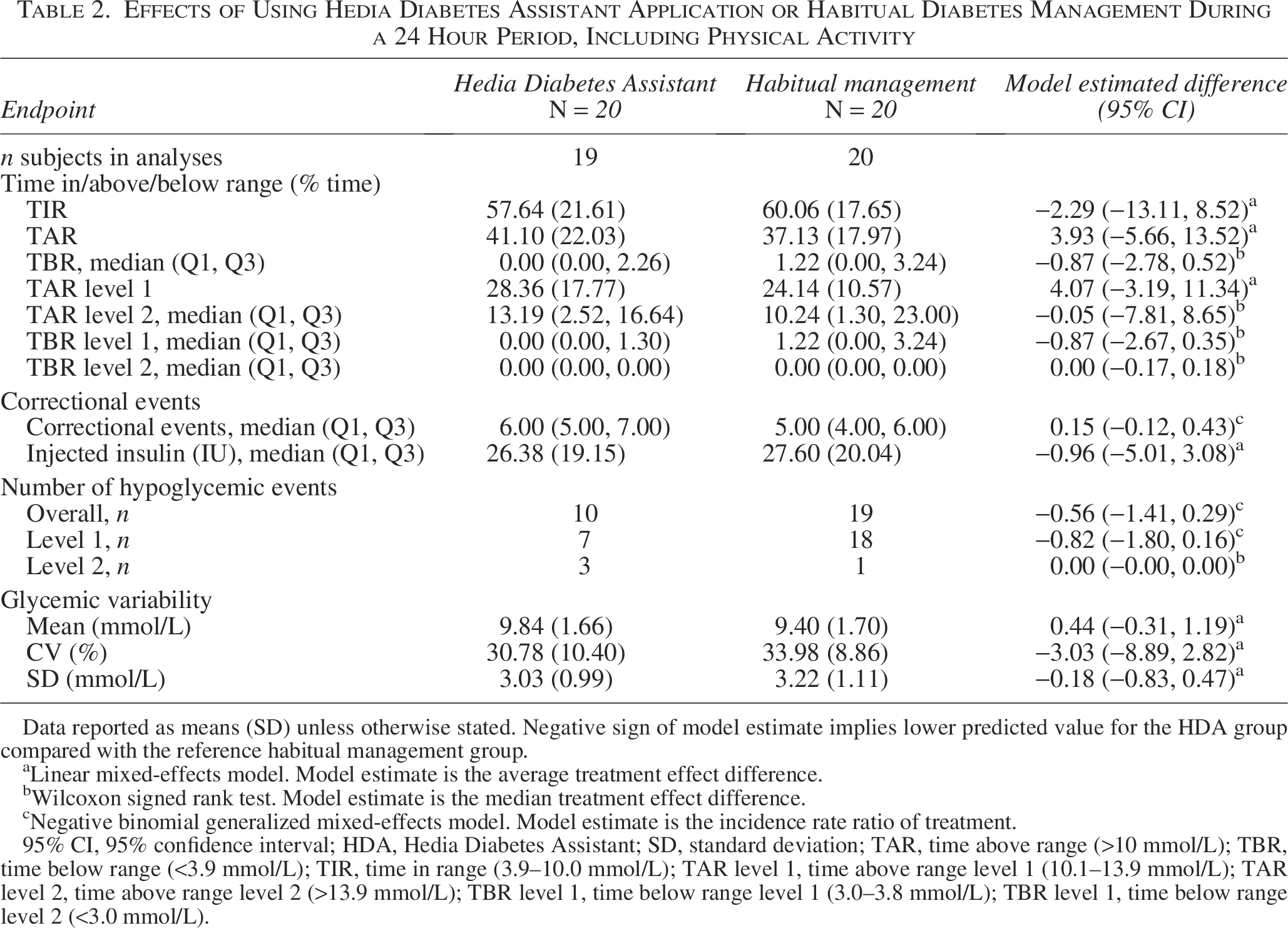
Data reported as means (SD) unless otherwise stated. Negative sign of model estimate implies lower predicted value for the HDA group compared with the reference habitual management group.
Linear mixed-effects model. Model estimate is the average treatment effect difference.
Wilcoxon signed rank test. Model estimate is the median treatment effect difference.
Negative binomial generalized mixed-effects model. Model estimate is the incidence rate ratio of treatment.
95% CI, 95% confidence interval; HDA, Hedia Diabetes Assistant; SD, standard deviation; TAR, time above range (>10 mmol/L); TBR, time below range (<3.9 mmol/L); TIR, time in range (3.9–10.0 mmol/L); TAR level 1, time above range level 1 (10.1–13.9 mmol/L); TAR level 2, time above range level 2 (>13.9 mmol/L); TBR level 1, time below range level 1 (3.0–3.8 mmol/L); TBR level 1, time below range level 2 (<3.0 mmol/L).
Confirmatory secondary outcomes
There were 10 hypoglycemic episodes during the intervention period (7 level 1 episodes and 3 level 2 episodes) and 19 during the control period (18 level 1 episodes and 1 level 2 episodes). The incidence rate ratio (IRR) (95% CI) was −0.56 (−1.41, 0.29) for the total number of hypoglycemic episodes, −0.82 (−1.80, 0.16) for level 1 episodes, and 0.00 (−0.00, 0.00) for level 2 episodes (Table 2). Seven participants had at least one hypoglycemic event during the intervention period compared with 10 during the control period (Table 3).
Number of Subjects Experiencing Hypoglycemic Events

The mean (SD) TAR (>10.0 mmol/L) was 41.1% (22.0%) in the intervention period and 37.1% (18.0%) in the control period, with a mean difference (95% CI) of 3.93% (−5.7%, 13.5%).
The median (IQR) TBR (<3.9 mmol/L) was 0% (0–2.3%) in the intervention period and 1.2% (0–3.2%) in the control period, with a median difference (95% CI) of −0.87% (−2.8%, 0.5%) (Table 2 and Fig. 2).
Supportive secondary outcomes
The mean (SD) CV was 30.8% (10.4%) in the intervention period and 34.0% (8.9%) in the control period, with a mean difference (95% CI) of −3.0% (−8.9%, 2.8%). The mean SD of glucose was 3.0 mmol/L (1.0 mmol/L) in the intervention period and 3.2 mmol/L (1.1 mmol/L) in the control period, with a mean difference (95% CI) of −0.2 mmol/L (−0.8, 0.5 mmol/L).
The mean (SD) insulin dose was 26.4 units (19.2 units) in the intervention period and 27.6 units (20.0 units) in the control period, with a mean difference (95% CI) of −1.0 (−5.0, 3.1). Correctional events were 6 (5–7) in the intervention period and 5 (4–6) in the control period, with an IRR (95% CI) of 0.2 (−0.1, 0.4) (Table 2).
The mean (SD) SUS score of HDA was 67.4 (16.0) corresponding to a “C” rating on the Sauro–Lewis Curved Grading Scale indicating average usability. 8
Safety outcomes
No serious adverse events were reported. During the intervention period, three adverse events were recorded, with two attributed to HDA. In contrast, seven adverse events occurred during the control period, six of which were associated with the bike test. In total, seven of the adverse events were related to hypoglycemia, two were related to hyperglycemia, and one was related to dizziness during normoglycemia.
A total of seven participants had their bike tests postponed for safety reasons. Five participants were postponed due to hyperglycemia (>15.0 mmol/L), all during the intervention period, whereas two participants had their tests postponed due to hypoglycemia (<3.9 mmol/L), both during the control period. In addition, one participant had their bike test halted due to hypoglycemia during the habitual treatment period (Table 4, Supplementary Table S5).
Safety of Using HDA Application or Habitual Diabetes Management with Physical Activity
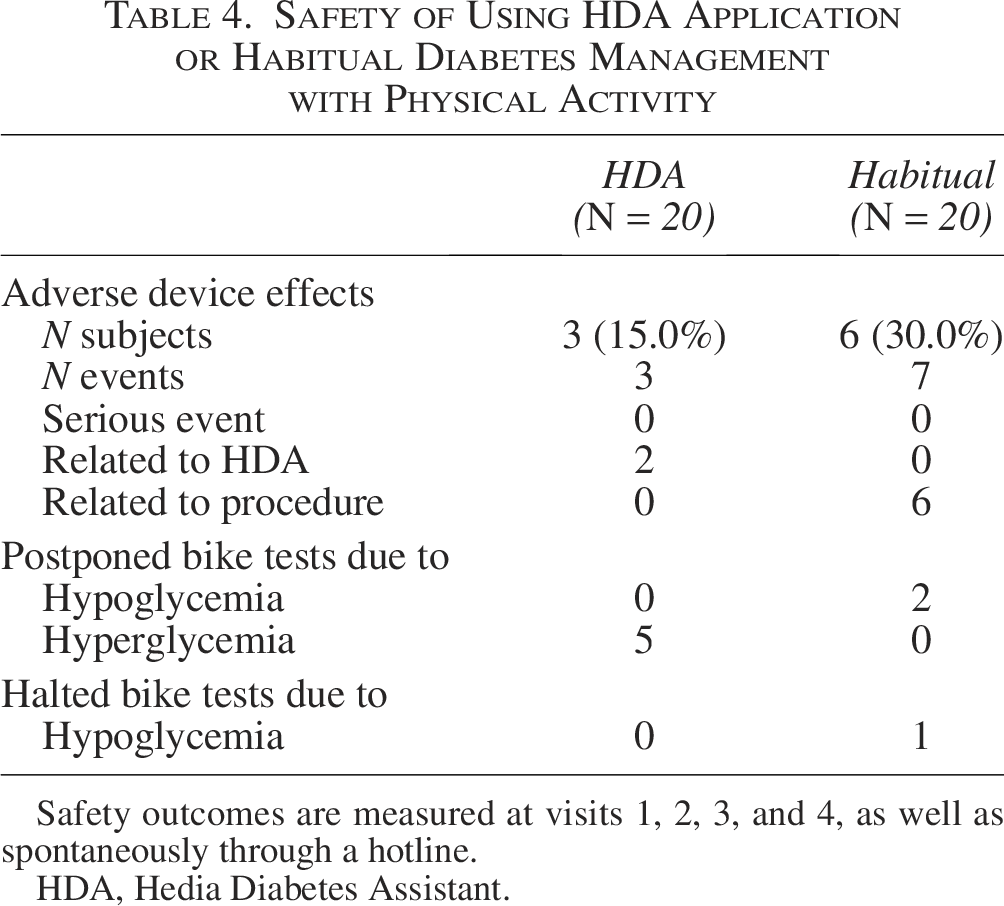
Safety outcomes are measured at visits 1, 2, 3, and 4, as well as spontaneously through a hotline.
HDA, Hedia Diabetes Assistant.
Subgroups
Results for the 12- and 5-hour periods were largely consistent with the 24 h period (Supplementary Tables S2 and S3 and Supplementary Fig. S1). During the 5 h period, the mean difference (95% CI) in mean glucose was 2.0 mmol/L (0.2, 3.7 mmol/L).
Sensitivity analyses
Four different sensitivity analyses were performed, and all supported the main analysis. Results from the per-protocol analysis are listed in Supplementary Table S4.
The difference in TIR from the main analysis was −2.64% (−13.66, 8.37), −3.21% (−13.65, 7.23), and −1.98% (−10.50, 6.55) for complete case analysis, conservative imputation of missing data, and multiple imputation, respectively.
Period effect
A period effect was observed for TIR, TAR, and mean sensor glucose. Specifically, the mean (95% CI) TIR was 14.2% (3.4, 25.1%) higher following the second bike test for both arms, regardless of which arm participants were randomized to. Similarly, TAR and mean sensor glucose were lower after the second bike test, with reductions of −15.6% (−25.2, −6.0%) and −1.5 mmol/L (−2.2, −0.7 mmol/L), respectively.
Discussion
This randomized crossover pilot trial evaluated the effects of using HDA compared with habitual insulin management during and after exercise in individuals with T1D on multiple daily injection insulin therapy. The participants had an average age of 57 years, a low burden of diabetes complications, and had on average lived with T1D for 16 years. Subjects engaged in two bouts of 45 min of moderate-intensity exercise and were randomized to use either HDA (and receive an insulin reduction in relation to the exercise) or their habitual diabetes management for the first bout and then crossed over for the second bout. The primary findings revealed no clear differences in glucose levels between use of HDA and habitual diabetes management during and after exercise, suggesting that HDA is safe to use in this context. Analyses of secondary outcomes and subgroup analyses showed a tendency toward fewer hypoglycemic episodes at the cost of transient hyperglycemia using HDA, especially when considering the number of bike tests that were postponed or halted due to hypoglycemia in the control period and the adverse events related to the bike exercise in the control period. Further, the analysis showed that the effect of HDA on TIR and TAR, respectively, seemed more pronounced shortly (5 and 12 h) after the bike test (in a dose-dependent way), whereas the effect on TBR (level 1) and hypoglycemic events was minimal shortly after the bike test and appeared more pronounced over time (24 h).
The HDA decision support algorithm is based on international guidelines, directing a 50% reduction of mealtime bolus insulin prior to moderate-intensity exercise lasting 45 min. 2 Our results indicate that the suggested bolus insulin reduction on average was too large, or the exercise done in the bike test was not as intensive as the guidelines describe. However, trading hypoglycemic events for transient hyperglycemia seems like a low cost. This does demonstrate that the most optimal insulin dose adjustment in relation to exercise is difficult to calculate and should probably be based—in an optimal setting—on more personalized variables than exercise intensity and duration (this could include machine learning algorithms). While duration can be determined very precisely (when the exercise has been done, but not before), the intensity of exercise can be difficult to foresee and assess for most people in daily life (however pulse watches could simplify this task). This is a challenge for all current and future implementations of the international guidelines. Furthermore, the timing of insulin dose reduction may play a crucial role; if patients reduce their insulin too early before exercising, they risk developing hyperglycemia before starting the activity. In addition, dose adjustments of basal insulin are important. HDA only provides guidance on dose adjustments to rapid-acting insulin and recommends carbs in case glucose level is below the target. This is highly relevant because research shows that many people with T1D initiate exercise with a too-low glucose level. 12 Strategies for basal insulin dosing in relation to exercise exist, 13 but the evidence is limited and therefore, this has not been built into HDA or other bolus calculators.
Our findings highlight the practical challenges of implementing the international guidelines and balancing pre-exercise insulin reductions with postexercise glycemic control into bolus calculations. Moreover, given that fear of hypoglycemia is a known barrier to exercise, the findings suggest that decision-support tools such as HDA help break down this barrier and thus help individuals with T1D achieve the health benefits associated with exercise.1,14
Notably, this is the first randomized controlled trial to assess a marketed insulin bolus calculator with an integrated exercise module aimed at preventing postexercise hypoglycemia. A key strength of the study is its real-world clinical relevance, as the exercise duration and intensity reflect common patterns among individuals using a bolus calculator, as supported by data from a study of HDA users. 12 Furthermore, the duration and intensity of the exercise were precisely controlled, and the use of blinded CGMs ensured that participants were blinded toward the outcomes.
The limitations of this study must be acknowledged. First, the small sample size restricts the generalizability of the findings, as it limits the ability to detect smaller effects and increases the potential for selection bias, demonstrated by the inclusion of participants who were generally very physically active at baseline. Second, the short duration of the two study periods (24 h) tends to give results that are impacted by large intra- and interparticipant variability of glucose measurements. Third, a period effect was observed, with increased TIR, decreased TAR, and lower mean glucose following the second bike test, regardless of the order of the intervention or control period. This suggests that participants may have adapted to the exercise protocol after the first period, (subconsciously) influencing their behavior and glycemic responses. Fourth, variability in carb-counting experience may have influenced the use of HDA, as only 55% of participants had previous carb-counting experience. Moreover, the short run-in period, during which participants used HDA only the evening before the bike test, may have limited their familiarity with the device. Lastly, the gender imbalance with relatively few female participants limits the generalizability. This is a problem because female gender is associated with an increased risk of being inactive 15 ; hence, women could potentially benefit most from using bolus calculators with an exercise module.
Future studies should aim for larger and more diverse populations, potentially with additional exercise events and longer CGM monitoring periods for more comprehensive data. As HDA is based on consensus guidelines with rough classification of the type of exercise intensity and duration, studies exploring and comparing alternative methods are warranted, including comparisons with automated insulin delivery systems.
Conclusions
In conclusion, the findings of this randomized crossover pilot trial suggest that HDA is safe to use during moderate-intensity exercise, offering potential benefits in reducing postexercise hypoglycemia. However, this comes with a trade-off, as pre-exercise insulin reductions may lead to transient hyperglycemia.
Authors’ Contributions
K.F.P.: Conceptualization, methodology, visualization, and writing—original draft. P.R.M.: Conceptualization, methodology, and writing—review and editing. M.T.O.: Investigation and writing—review and editing. G.L.: Formal analysis, visualization, and writing—review and editing. M.A.K.: Project administration and writing—review and editing. D.H.L.: Supervision and writing—review and editing. P.L.K.: Conceptualization, investigation, methodology, supervision, and writing—review and editing.
Footnotes
Acknowledgments
The authors would like to thank study nurses Bonnie Britt Myhre, Pernille Banck-Petersen, and Rikke Charlotte Carstensen for their invaluable assistance in including participants in the study and conducting the bike tests.
Author Disclosure Statement
Funding Information
The study was funded by Hedia ApS. The last author held the contractual right to have the final say regarding the content of the article.
Approvals
Approval was obtained from the Danish Medical Research Ethics Committees (#2210121).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
