Abstract
Background
Survivors of severe COVID-19 requiring intensive care frequently experience persistent pulmonary and functional impairment consistent with post–critical illness sequelae. The effectiveness of non-pharmacological rehabilitation in this severity-specific subgroup remains uncertain.
Methods
A systematic review was conducted in accordance with PRISMA 2020 guidelines. PubMed, Epistemonikos, LILACS, and Google Scholar were searched for randomized and observational studies evaluating non-pharmacological rehabilitation in adult ICU survivors of COVID-19. Risk of bias was assessed using RoB 2 and ROBINS-I tools. Given substantial clinical and methodological heterogeneity, quantitative meta-analysis was not performed; a structured narrative synthesis was undertaken.
Results
Fourteen studies met inclusion criteria. Five incorporated comparator groups, while nine employed uncontrolled pre–post designs. Interventions ranged from early ICU mobilization to inpatient and outpatient pulmonary rehabilitation. Controlled studies reported variable between-group benefits in dyspnea and functional outcomes, whereas observational studies consistently described within-group improvement over time. However, most studies were at moderate to serious risk of bias, and heterogeneity in intervention timing, dosage, and outcome assessment limited comparability.
Conclusions
Non-pharmacological rehabilitation in ICU survivors of COVID-19 is associated with improvement over time; however, the certainty of causal effectiveness remains low. ICU survivors constitute a distinct recovery population within the broader post-COVID spectrum. Adequately powered, multicenter randomized trials with standardized protocols and harmonized outcomes are required to establish long-term effectiveness.
Keywords
Introduction
The global impact of the COVID-19 pandemic has led to a heterogeneous spectrum of clinical outcomes, ranging from asymptomatic cases to life-threatening respiratory failure. 1 A substantial proportion of patients with severe coronavirus disease 2019 (COVID-19) develop acute hypoxemic respiratory failure requiring admission to an intensive care unit (ICU), frequently necessitating invasive mechanical ventilation, prolonged sedation, neuromuscular blockade, and extended immobilization.2–4 In this context, the clinical trajectory is determined not only by viral pneumonia but also by the pathophysiological consequences of critical illness, including systemic inflammation, multiorgan dysfunction, and catabolic muscle loss. Survivors of ICU-managed COVID-19 therefore represent a clinically distinct population whose long-term outcomes are shaped by both severe acute respiratory distress syndrome (ARDS) and the sequelae of prolonged critical care exposure.
Long-term impairments following critical illness have been conceptualized under the framework of Post–Intensive Care Syndrome (PICS), a multidimensional condition encompassing persistent physical weakness, respiratory dysfunction, cognitive impairment, and psychological morbidity.5,6 Physical limitations are frequently driven by ICU-acquired weakness and critical illness polyneuropathy or myopathy, conditions associated with prolonged mechanical ventilation and extended bed rest. 7 Longitudinal studies conducted prior to the COVID-19 pandemic demonstrated that survivors of ARDS may experience substantial functional disability and reduced quality of life for years following ICU discharge. 8
In the specific context of COVID-19–related ARDS, persistent pulmonary abnormalities—including reduced diffusing capacity, radiographic fibrotic-like changes, and impaired exercise tolerance—have been documented months after hospital discharge. 9 Severe disease and ICU admission have consistently been identified as major risk factors for long-term functional limitation and symptom persistence compared with non-ICU populations.10,11 Accordingly, ICU survivors of COVID-19 differ fundamentally from broader post-COVID cohorts managed in outpatient or non-critical hospital settings, as their recovery trajectory reflects the combined burden of severe viral pneumonia, ARDS-related structural lung injury, neuromuscular deconditioning, and the systemic consequences of prolonged critical illness. 12
Despite the extensive literature on post-COVID rehabilitation in heterogeneous populations, evidence specifically addressing ICU survivors remains limited and methodologically fragmented. Many existing systematic reviews have pooled patients across varying levels of acute severity—including non-hospitalized individuals and general ward admissions—thereby limiting the applicability of pooled estimates to those recovering from critical illness. Given the distinct pathophysiology and recovery trajectory of ICU survivors, rehabilitation strategies evaluated in broader post-COVID cohorts cannot be assumed to have equivalent effectiveness in this high-risk subgroup.13,14 Therefore, this systematic review was designed to focus exclusively on adult ICU survivors of COVID-19 and to critically evaluate the available evidence regarding non-pharmacological rehabilitation interventions targeting pulmonary and physical recovery in this population.15–21
Methods
Reporting standards and protocol registration
This systematic review was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 22 statement to ensure transparency and methodological rigor. The review protocol was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) on March 6, 2023 (CRD42023401148). The final literature search was conducted on May 15, 2024.
Eligibility criteria
Eligible studies included randomized or quasi-randomized clinical trials and analytical observational studies enrolling adults (≥18 years) who survived moderate to severe COVID-19 and required admission to an ICU. Studies evaluating non-pharmacological rehabilitation interventions were eligible, including pulmonary rehabilitation, physical exercise programs, respiratory muscle training, or multicomponent multidisciplinary rehabilitation.
Comparators, when present, included usual care, delayed rehabilitation, or alternative physical or respiratory interventions. Studies without a comparator group (uncontrolled before–after designs) were also eligible.
Primary outcomes of interest included pulmonary outcomes (respiratory symptoms, spirometry, lung volumes, diffusing capacity for carbon monoxide, and cardiopulmonary exercise testing), physical performance (validated functional scales and exercise tests), and respiratory or peripheral muscle strength assessed using dynamometry or standardized performance measures.
Studies focusing exclusively on pharmacological interventions, those enrolling non-ICU populations, or those lacking post-intervention outcome data were excluded.
Data sources and search strategy
A comprehensive literature search was performed in Epistemonikos, PubMed, LILACS, and Google Scholar. These databases were selected to ensure broad coverage of biomedical, rehabilitation, and regional literature relevant to post-COVID-19 recovery. No restrictions were applied regarding language or publication status.
Search terms were derived from the components of the PICO framework addressing the review objective: adult ICU survivors of COVID-19 (population), non-pharmacological rehabilitation interventions (intervention), comparator or usual care when applicable (comparison), and pulmonary or physical recovery outcomes (outcomes). Controlled vocabulary terms (MeSH and DeCS) and free-text keywords were combined using Boolean operators. Key terms included “COVID-19,” “SARS-CoV-2,” “Post-Acute COVID-19 Syndrome,” “Rehabilitation,” “Pulmonary Rehabilitation,” “Respiratory Therapy,” “Exercise,” “Muscle Training,” “Physical Activity,” “Dyspnea,” and “Respiratory Function Test.”
Search strategy.

Study selection
Two reviewers independently screened titles and abstracts to identify potentially eligible studies. Full-text articles were subsequently assessed for eligibility based on predefined inclusion and exclusion criteria. Discrepancies were resolved through discussion, with adjudication by a third reviewer when necessary.
Data extraction
Data were independently extracted by two reviewers using a standardized, pre-piloted extraction form. Extracted variables included study design, authorship, year of publication, country, sample size, participant characteristics, ICU-related variables when reported, and detailed descriptions of rehabilitation interventions, including type, components, frequency, intensity, and duration.
Primary outcomes extracted included respiratory symptoms, pulmonary function parameters, respiratory and peripheral muscle strength, exercise capacity, and physical function. When studies reported multiple outcomes or assessment time points, all outcomes relevant to the predefined domains were extracted.
Risk of bias assessment
The risk of bias was assessed by at least two reviewers, and when consensus was not reached, disagreements were resolved by a third reviewer. The selection of risk-of-bias tools was prespecified and based on study design.
Randomized and quasi-randomized trials were assessed using the Cochrane Risk of Bias 2 (RoB 2) tool, which evaluates bias across five domains: bias arising from the randomization process; bias due to deviations from intended interventions; bias due to missing outcome data; bias in measurement of the outcome; and bias in selection of the reported result, leading to an overall judgement of low risk of bias, some concerns, or high risk of bias. 23
Non-randomized intervention studies, including controlled and uncontrolled observational designs, were assessed using the ROBINS-I version 2 (ROBINS-I V2) tool. This tool evaluates risk of bias across seven domains: confounding, classification of interventions, selection into the study, deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported result, and provides an overall judgement ranging from low to critical risk of bias. 24
Risk-of-bias assessments were conducted at the study level and were used to inform interpretation during data synthesis. No studies were excluded based solely on risk-of-bias judgements.
Data synthesis
Prior to data synthesis, the feasibility of conducting quantitative meta-analysis was systematically evaluated. Heterogeneity was assessed across three predefined domains: clinical, methodological, and outcome related. Clinical heterogeneity was examined by comparing participant characteristics (restricted to post–ICU COVID-19 survivors), rehabilitation settings, intervention components, timing of initiation, and intervention dose and duration. Methodological heterogeneity was evaluated based on study design, availability and type of comparator groups, risk of bias, and follow-up periods. Outcome heterogeneity was assessed by examining the consistency of outcome definitions, measurement instruments, and timing of outcome assessment across studies. A meta-analysis was planned only if studies demonstrated sufficient homogeneity across these domains and reported comparable quantitative outcome data. In cases where substantial heterogeneity was identified, a structured narrative synthesis was prespecified, conducted in accordance with the Synthesis Without Meta-analysis (SWiM) reporting guideline. 25
Results
Selection of studies
The literature search yielded 2,742 records. After title and abstract screening, 137 full-text articles were assessed for eligibility. Of these, 116 were excluded for not meeting the predefined inclusion criteria. A total of 14 studies fulfilled the eligibility criteria and were included in the final review. The study selection process is presented in Figure 1 (PRISMA flow diagram). PRISMA search flow and inclusion of studies.
Study characteristics and intervention categories
Studies with comparator (n = 5) (Comparator present: randomized, quasi-experimental, or controlled designs).

Abbreviations: ICU = intensive care unit; ADL = activities of daily living; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity; 6-MWT = 6-minute walk test; IMT = inspiratory muscle training; EMG = electromyography; QoL = quality of life.
Characteristics and main findings of studies without comparator (pre–post observational designs) (n = 9).
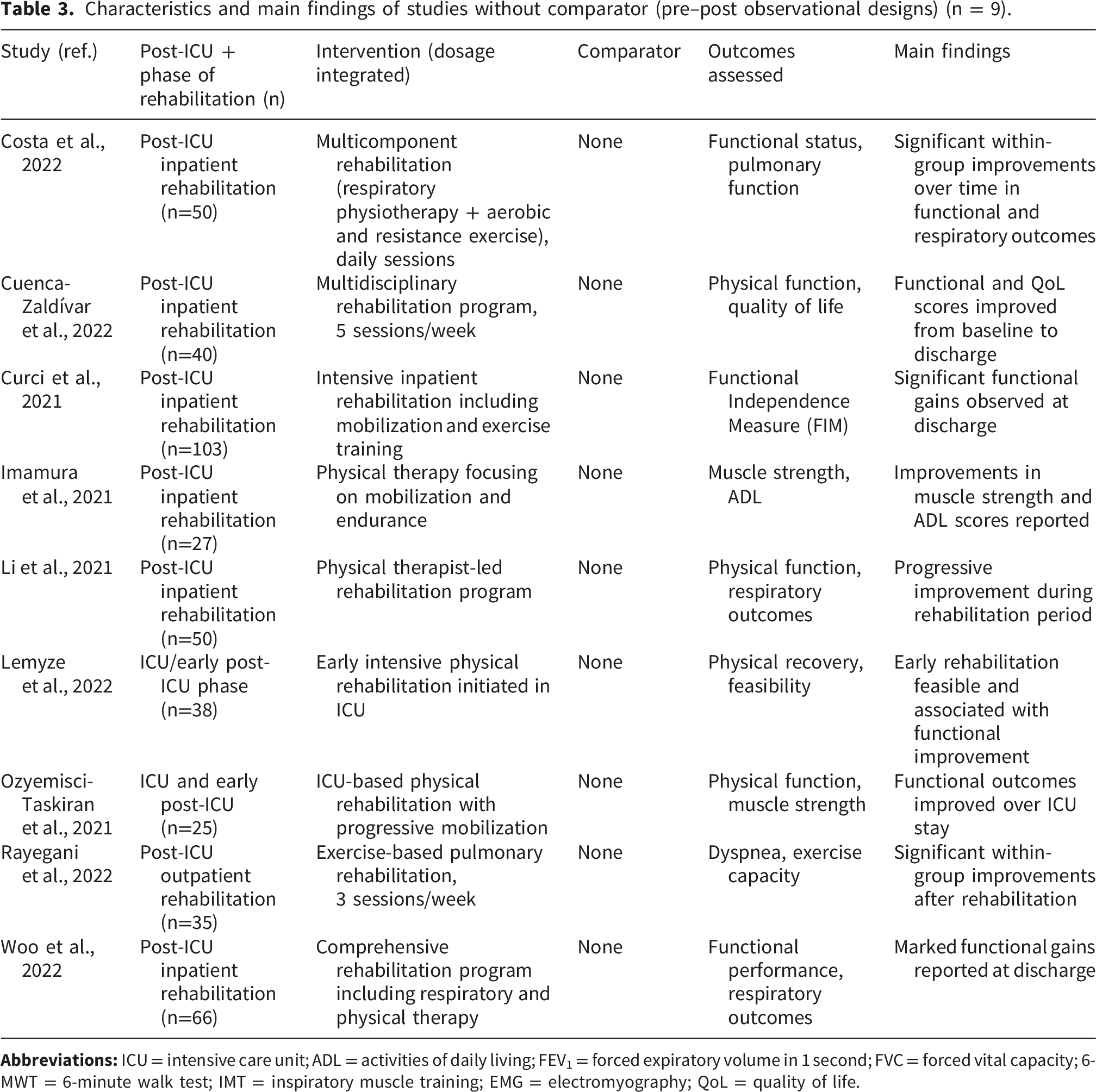
Across both tables, all studies enrolled adult survivors of COVID-19 who required admission to an ICU and evaluated non-pharmacological rehabilitation interventions delivered during different phases of recovery, including early post-ICU care, inpatient rehabilitation, outpatient programs, or home-based settings.
Interventions were predominantly exercise-based or multicomponent rehabilitation programs, integrating respiratory physiotherapy, aerobic and resistance training, and functional exercises. When reported, intervention dosage (frequency, duration, and intensity) is incorporated within the intervention descriptions presented in the tables.
Synthesis of results by study design
Studies with comparator (Table 2)
Among the five studies that included a comparator group, several trials reported statistically significant between-group differences in selected functional or respiratory outcomes following rehabilitation.
In an outpatient setting, Bagherzadeh-Rahmani et al., 2022 26 compared a Pilates-based exercise program with an Aqua-Pilates intervention in post-ICU patients. Although both groups improved over time, the Aqua-Pilates group demonstrated greater improvements in FEV1 and FVC, with statistically significant between-group differences favoring Aqua-Pilates.
Similarly, Romanet et al., 2023 27 evaluated a supervised outpatient pulmonary rehabilitation program combined with inspiratory muscle training. Compared with usual care, the intervention group showed significantly greater reductions in dyspnea scores and improvements in 6-minute walk distance, indicating superior functional and symptomatic outcomes.
In an inpatient setting, Zasadzka et al., 2022 28 reported that EMG-assisted robotic rehabilitation combined with conventional physiotherapy resulted in greater improvements in muscle fatigue indices and functional performance compared with conventional physiotherapy alone.
Early rehabilitation strategies were examined by Bordas-Martínez et al., 2022, 29 who found that intensive physical rehabilitation initiated during ICU stay was associated with faster functional recovery compared with delayed or standard physical therapy.
Finally, Nankaku et al., 2022 30 reported that post-ICU patients receiving a structured inpatient rehabilitation program achieved superior functional independence and mobility outcomes compared with a comparator group defined by lower rehabilitation exposure or intensity.
Despite these positive findings, the magnitude and direction of effects varied across studies, outcome measures were heterogeneous, and effect size estimates were inconsistently reported.
Studies without comparator (Table 3)
The nine observational pre–post studies conducted without a comparator consistently reported within-group improvements over time following rehabilitation.
Multicomponent inpatient rehabilitation programs were associated with improvements in functional status and respiratory outcomes in studies by Costa et al., 2022, 31 Cuenca-Zaldívar et al., 2022, 32 Curci et al., 2021, 33 and Woo et al., 2022 34 , with gains observed from admission to discharge.
Improvements in muscle strength, activities of daily living, and physical performance were also reported in smaller inpatient cohorts by Imamura et al., 2021 35 and Li et al., 2021. 36
Early rehabilitation initiated during ICU or early post-ICU phases was feasible and associated with functional improvement in studies by Lemyze et al., 2022 37 and Ozyemisci-Taskiran et al., 2021. 38
In an outpatient setting, Rayegani et al., 2022 39 reported significant within-group improvements in dyspnea and exercise capacity following an exercise-based pulmonary rehabilitation program.
Given the absence of concurrent control groups, improvements observed in these studies cannot be distinguished from natural recovery following critical illness and should therefore be interpreted descriptively rather than causally.
Risk of bias results
Randomized and quasi-randomized trials (RoB 2)
The three comparator trials judged as randomized or quasi-randomized were assessed using the Cochrane Risk of Bias 2 (RoB 2) tool: Bagherzadeh-Rahmani et al., 2022
26
; Romanet et al., 2023
27
; Zasadzka et al., 2022
28
. Overall judgements ranged from some concerns to high risk of bias, mainly due to incomplete reporting of key features of the randomization process (e.g., allocation concealment) and the inherent inability to blind participants and personnel in rehabilitation interventions. Additional concerns related to outcome measurement for subjective endpoints and incomplete outcome data were identified in some trials. A summary of RoB 2 domain-level judgements is presented in Figure 2(a). (a) Synthesis of the risk of bias assessed with the ROBINS-2 tool for quasi-randomized trials. (b) Synthesis of the risk of bias assessed with the ROBINS-I V2 tool for quasi-randomized trials.
Non-randomized studies (ROBINS-I V2)
The remaining 11 non-randomized studies were assessed using the ROBINS-I V2 tool 40 : Bordas-Martínez et al., 2022 29 ; Nankaku et al., 2022 (30); Costa et al., 2022 31 ; Cuenca-Zaldívar et al., 2022 32 ; Curci et al., 2021 33 ; Imamura et al., 2021 35 ; Li et al., 2021 36 ; Lemyze et al., 2022 37 ; Ozyemisci-Taskiran et al., 2021 38 ; Rayegani et al., 2022 39 ; Woo et al., 2022. 34 Most studies were judged to be at serious overall risk of bias, primarily driven by confounding, reflecting the predominance of uncontrolled pre–post designs and limited ability to separate intervention effects from natural recovery after critical illness. Additional concerns commonly included selection into rehabilitation programs and outcome measurement for symptom-based or patient-reported outcomes, where blinding was not feasible or not reported. A summary of ROBINS-I V2 domain-level judgements is presented in Figure 2(b).
Data synthesis and assessment of heterogeneity
Given the substantial clinical, methodological, and outcome heterogeneity across the included studies, quantitative meta-analysis was not undertaken. Clinical heterogeneity reflected variability in patient characteristics restricted to post-ICU COVID-19 survivors, rehabilitation settings, intervention components, timing of initiation, and dosage. Methodological heterogeneity arose from differences in study design, including a predominance of uncontrolled before–after cohorts, limited randomized evidence, variability in comparator groups, and heterogeneous follow-up durations. Outcome heterogeneity resulted from inconsistent outcome definitions, measurement instruments, and assessment time points.
Accordingly, a structured narrative synthesis was conducted in line with the Synthesis Without Meta-analysis (SWiM) reporting guideline. 25 Studies were grouped by study design and type of rehabilitation intervention. Outcomes were summarized descriptively within each group, with quantitative effect estimates reported when available and interpreted in the context of study design and risk of bias. When effect size estimates were not reported, findings were summarized qualitatively to avoid inappropriate imputation or standardization. 41
Discussion
Interpretation of findings
This review synthesizes the available evidence on non-pharmacological rehabilitation specifically in adults who survived severe COVID-19 requiring intensive care. The synthesis of the 14 included studies demonstrates that while pulmonary and physical improvements are achievable, the recovery trajectory of this specific phenotype is uniquely arduous. Unlike broader meta-analyses that dilute their findings by pooling mild cases with critical ones, our focus on the post-ICU population highlights a recovery process dictated by the “double hit” of severe viral pneumonia and the systemic sequelae of critical care, such as ICU-acquired weakness. 42 When interpreted within the broader framework of critical illness recovery, the improvements reported across functional and respiratory outcomes must be understood considering the established natural trajectory following ARDS and prolonged ICU exposure. Survivors of critical illness frequently develop PICS, a multidimensional condition characterized by persistent physical weakness, pulmonary dysfunction, cognitive impairment, and psychological morbidity. 43 Long-term follow-up studies conducted prior to the COVID-19 pandemic demonstrated that ARDS survivors may experience substantial functional disability for years after discharge, with gradual, and often incomplete, recovery. 8
In patients with COVID-19–related ARDS, persistent pulmonary abnormalities—including reduced diffusing capacity and radiographic fibrotic-like changes—have been documented months after hospital discharge. 9 In addition, ICU-acquired weakness and critical illness polyneuropathy or myopathy remain major contributors to prolonged functional impairment in mechanically ventilated patients. 7 Against this background, the within-group improvements consistently reported in uncontrolled rehabilitation studies among ICU survivors31–34,37,39 cannot be interpreted as definitive evidence of treatment efficacy, as spontaneous recovery following critical illness is both expected and well documented.
Among the five studies incorporating a comparator,26–30 some reported statistically significant between-group improvements in dyspnea, exercise capacity, or functional independence. However, limitations including small sample sizes, incomplete reporting of allocation procedures, and the inherent difficulty of blinding rehabilitation interventions constrain causal interpretation. In ICU populations—where progressive improvement in muscle strength and functional capacity may occur during the first year after discharge even in the absence of structured programs. 44
Differentiating critical care recovery from general Post-COVID cohorts
Many prior systematic reviews have pooled heterogeneous post-COVID populations across varying levels of acute severity, including non-hospitalized individuals, general ward admissions, and mixed cohorts.13,14 While such pooling increases statistical power and facilitates meta-analysis, it reduces applicability to ICU survivors, whose recovery trajectory is shaped not only by viral infection but also by prolonged mechanical ventilation, sedation exposure, systemic inflammation, and immobility. These factors are independently associated with long-term disability in survivors of critical illness.7,43
To our knowledge, relatively few systematic reviews have restricted inclusion exclusively to ICU survivors of COVID-19, despite the distinct pathophysiological profile and recovery dynamics documented in critical care literature. Even recent syntheses focused on critically ill COVID-19 patients have highlighted marked clinical and methodological heterogeneity, limiting quantitative pooling and definitive conclusions. 45 The principal contribution of the present review, therefore, lies in recognizing ICU survivors as a severity-specific subgroup and evaluating rehabilitation evidence within that defined context rather than extrapolating from broader post-COVID cohorts.
From conventional critical care to the COVID-19 context
Rehabilitation in critically ill patients has been investigated extensively prior to the COVID-19 pandemic. Randomized trials demonstrated that early physical and occupational therapy during mechanical ventilation is feasible and may improve functional outcomes at hospital discharge. 46 Early mobilization strategies have also been associated with improved functional independence in selected ICU populations. 47
However, even in pre-COVID ICU trials, heterogeneity in illness severity, timing of intervention, and rehabilitation dose limited the ability to draw definitive long-term conclusions. The same methodological challenges are evident in COVID-19 ICU cohorts, where variability in ventilation duration, ARDS severity, and post-ICU recovery phase complicates interpretation. Framing post-COVID rehabilitation within this established body of critical care literature underscores that the current uncertainty reflects broader challenges inherent to ICU survivorship research rather than a uniquely COVID-specific phenomenon. 48
Methodological limitations and the challenge of attributing functional recovery
The predominance of uncontrolled pre–post designs in the available literature31–35 highlights the difficulty of conducting adequately powered randomized controlled trials in this population. Regression toward baseline and spontaneous physiological recovery are expected during convalescence from critical illness, particularly in the first months after discharge. 8 Without well-defined comparator groups and standardized intervention protocols, attribution of observed improvements to rehabilitation interventions remains uncertain.
Furthermore, heterogeneity in intervention timing—from ICU-based mobilization29,37,38 to inpatient and outpatient rehabilitation programs26–28,31–34,39 —reflects distinct phases of recovery characterized by differing physiological constraints. Harmonization of outcome measures, detailed reporting of intervention dose, and severity-stratified analyses will be essential for future research.
Strengths and limitations
A major strength of this review is its exclusive focus on ICU survivors of COVID-19, a subgroup consistently characterized by greater baseline impairment and more complex recovery trajectories than non-critical cohorts.9,11 By anchoring the analysis within established frameworks of Post–Intensive Care Syndrome and ARDS survivorship, the review situates post-COVID rehabilitation within the broader continuum of critical illness recovery rather than within heterogeneous post-COVID populations. In addition, studies were explicitly categorized according to the presence or absence of comparator groups, allowing clearer distinction between controlled evidence and uncontrolled observational data. When heterogeneity precluded meta-analysis, synthesis was conducted transparently in accordance with SWiM principles, enhancing methodological clarity and reproducibility. 25
Several methodological limitations must be acknowledged. The current evidence base is dominated by small cohorts and predominantly non-randomized designs, many of which were judged to be at serious risk of bias.31–39 The absence of robust comparator groups substantially limits the ability to distinguish true intervention effects from spontaneous recovery or residual confounding. 24 In survivors of critical illness, functional improvement during the first months after discharge is well documented, reflecting natural physiological recovery and, in some cases, regression toward the mean. 8 In this context, isolating the independent contribution of rehabilitation interventions remains challenging, and the overall certainty of causal inference is therefore limited. Substantial clinical and methodological heterogeneity—across rehabilitation settings, intervention timing and dosage, and outcome assessment—further constrained quantitative synthesis and comparability. 49 Measurement bias was also a concern, as several studies relied on subjective, patient-reported outcomes without blinded assessment, 50 and key intervention parameters such as adherence and treatment fidelity were frequently underreported. The predominance of non-randomized designs introduces a substantial risk of confounding,. 23 Finally, although restricting inclusion to ICU survivors strengthens clinical specificity, variability in post-ICU care pathways and healthcare systems may limit generalizability. Taken together, these limitations underscore the need for adequately powered, multicenter randomized trials specifically designed for ICU-managed COVID-19 populations before definitive conclusions regarding long-term effectiveness can be drawn. 51
Implications for clinical practice and future research
Given the low certainty of the current evidence and the predominance of uncontrolled designs, rehabilitation for ICU survivors of COVID-19 should be viewed as a supportive component of post–critical illness recovery rather than a treatment with established long-term efficacy. Clinical application should emphasize individualized, severity-informed approaches tailored to comorbidities and functional capacity, while avoiding assumptions of causal benefit based on observational data. 52
Substantial heterogeneity in rehabilitation timing, setting, and intensity underscores the absence of standardized care pathways. Future research should prioritize adequately powered, multicenter randomized trials with clearly defined rehabilitation phases, robust comparator groups, and detailed reporting of intervention parameters. Harmonized outcome measures and longitudinal follow-up will be essential to distinguish natural recovery from true intervention effects and to clarify how illness severity modifies rehabilitation outcomes. 53
Conclusions
This systematic review synthesizes the available evidence on non-pharmacological rehabilitation for pulmonary and physical recovery in adults who survived COVID-19 requiring intensive care. Although improvements in respiratory and functional outcomes are frequently reported, the certainty of the evidence remains low, reflecting the predominance of uncontrolled study designs, methodological limitations, and substantial heterogeneity in intervention delivery and outcome assessment.
ICU survivors represent a severity-specific subgroup whose recovery trajectory is shaped by ARDS-related pulmonary injury, ICU-acquired weakness, and the systemic consequences of prolonged critical illness. Within this context, improvements observed over time cannot be confidently attributed to rehabilitation interventions alone, as spontaneous physiological recovery is an expected component of post–critical illness convalescence. A central contribution of this review is the explicit positioning of rehabilitation evidence within this ICU-focused framework, underscoring the limitations of extrapolating findings from heterogeneous post-COVID populations.
Until adequately powered, multicenter randomized controlled trials with standardized protocols and harmonized outcomes are conducted, rehabilitation for ICU-managed COVID-19 survivors should be delivered as an individualized, supportive component of comprehensive post–critical illness care rather than as an intervention with established long-term efficacy.
Supplemental material
Supplemental material -Non-pharmacological rehabilitation strategies for pulmonary and physical recovery in ICU survivors after COVID-19: A systematic review
Supplemental material for Non-pharmacological rehabilitation strategies for pulmonary and physical recovery in ICU survivors after COVID-19: A systematic review by Yimy F. Medina, Eliana Isabel Rodríguez Grande, Javier Leonardo Galindo, Olga Cecilia Vargas Pinilla, Franklin Soler and Gleidy Vanessa Espitia in Chronic Respiratory Disease.
Footnotes
Ethical considerations
This study is a systematic review of previously published literature and does not involve any new data collection from human participants or animals. Therefore, ethical approval and informed consent were not required.
Author contributions
All authors contributed equally to the conception, design, data collection, analysis, and writing of this manuscript. Each author has reviewed and approved the final version and agrees to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data analyzed in this study are from previously published articles included in the systematic review. No new data was generated or analyzed by the authors. The data supporting the findings of this study are available within the cited literature.
Supplemental material
Supplemental material for this article available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
