Abstract
Background
We aimed to identify the risk factors associated with all-cause mortality in non-cystic fibrosis bronchiectasis (BE) patients in a Finnish cohort.
Methods
In this 4-years follow-up study, the data of non-cystic fibrosis adult BE patients were collected annually from medical records. Finnish translation of the disease-specific quality of life-bronchiectasis (QoL-B) questionnaire, the bronchiectasis severity index (BSI), FACED score, E-FACED score (exacerbations, (E), FEV1 (F), age (A), pulmonary bacterial colonization (C), number of lobes affected by BE (E), and dyspnoea (D), and modified Medical Research Council (mMRC) dyspnoea scale were used. Cox’s regression analysis was used to evaluate factors with mortality.
Results
A total of 95 out of 205 adult non-cystic fibrosis BE patients were included and 79% of them were women with mean age of 69 years (SD ± 13). During the follow-up, eight patients died (8.4%). High scores of FACED (HR 1.9 CI 1.1–3.0), E-FACED (HR 1.5 CI 1.1–2.1) and mMRC (HR 3.2 CI 1.5–6.9) were increased the risk of mortality. The specific aetiology of BE, however, does not affect mortality. Low score of domains in QoL-B, physical (p < 0.01), vitality (p = 0.01), respiration (p = 0.03) and health (p < 0.01), were associated with mortality.
Conclusion
Multifactorial FACED and BSI scores increased the risk of mortality. In addition, mMRC which is a single patient reported variable was predictive for mortality. The simple mMRC scale could provide a valuable tool for clinical use.
Introduction
The main goal in managing bronchiectasis (BE) is to identify its cause, treat it effectively, and prevent exacerbations to limit disease progression. BE mortality is twice as high as in the general population. 1 A key challenge is identifying patients at high risk of frequent exacerbations.
Antimicrobial therapy, both short- and long-term, manages current infections and prevents exacerbations linked to premature death.2–4 Despite advances, severe pneumonia, often caused by Pseudomonas species, remains a leading cause of death in BE. 5 Pseudomonas dominance and reduced airway microbiome diversity are linked to frequent exacerbations, hospitalizations, and mortality. 6
Increased age, number of affected lobes, and chronic obstructive pulmonary disease (COPD) as a comorbidity raise mortality risk in adult patients with BE. 7 Goeminne et al. reported a 20% mortality rate over 5 years, with a median age of 68. 7 Similar studies show higher mortality in adult patients with BE compared to controls, with factors like older age, male sex, asthma, COPD, lung cancer, and cardiovascular disease contributing to risk.8,9 In addition, adult patients with BE have higher all-cause and lung cancer mortality. 8 Airflow limitation and smoking history further increase all-cause mortality risk. 8
In a multicentre study, BSI (bronchiectasis severity index) was stated to be superior to FACED (FACED: FEV1 (forced expiratory volume in 1 minute) (F), age (A), pulmonary bacterial colonization (C), number of lobes affected by BE (E), and dyspnoea (D)) in predicting clinically important disease-related outcomes, including hospital admissions, exacerbations, quality of life (QoL), respiratory symptoms, 6-min walking test and lung function decline in BE. They suggested FACED should be used rather as a mortality risk assessment tool. 10 However, E-FACED (exacerbations, (E), FEV1 (F), age (A), pulmonary bacterial colonization (C), number of lobes affected by BE (E), and dyspnoea (D)) was found to be superior to both FACED and previous exacerbation rate for identifying patients with future exacerbations and hospitalisations. 11 A comprehensive meta-analysis demonstrated that the mMRC predicts all-cause mortality in adult patients with BE. 12 Recent BE studies from Finland characterised aetiology and investigated quality of life of adult patients with BE.13,14 EMBARC (European Multicentre Bronchiectasis Audit and Research Collaboration) is the first international bronchiectasis network, aiming to unite global researchers through a multicentre registry of adult patients with non-CF bronchiectasis. It enables experts to address key questions in the disease’s epidemiology, causes, microbiology, pathophysiology, management, and prognosis.15–19 In this study, as a part of EMBARC, we aimed to analyse predictive factors for mortality from the Finnish BE cohort.
Material and methods
Altogether, 95 of 205 adult patients with non-cystic fibrosis BE were included in this cross-sectional study. Patients were recruited from Helsinki University Hospital (HUH) between August 2016 and March 2018, meeting these criteria: (1) age >18, (2) High-resolution computed tomography (HRCT) -confirmed BE, and (3) non-cystic fibrosis BE. A radiologist reviewed HRCT scans, recording the number of affected lobes and the type of bronchial damage (cylindrical, varicose, or cystic). Bronchiectasis was defined as a bronchial airway diameter larger than the diameter of an adjacent blood vessel. Cystic bronchiectases were defined as rounded cysts. 20 Extensive disease was defined as involvement of four or more lobes, with the lingula counted separately. 13 Follow-up ran from October 2017 to April 2021. Data included age, gender, smoking status, body mass index (BMI), lung function, bacterial colonization history, BE aetiology, comorbidities, medication, affected lobes, cystic changes, FEV1%, exacerbation rates, hospital admissions, emergency visits, and novel radiology findings. Data were collected annually from medical records in the EMBARC database. At the end of the study, 10 patients (10.5%) had transitioned to follow-up in primary care. Patients had no role in study design, data collection, or reporting. 13
Quality of life in bronchiectasis questionnaire (QoL-B)
Quality of life was assessed by QoL-B version 3.0.21–23 This version includes 37 questions and eight domains. The domains cover respiratory symptoms, physical activity, role, emotional functioning, social functioning, vitality, health perceptions, and treatment burden. Scoring is from 0 (lowest possible quality of life) to 100 (fewer symptoms and the best possible quality of life). We used the Finnish version of the questionnaire.
Modified medical research council scale (mMRC)
Severity of breathlessness during the daily activities was estimated by using the self-administered mMRC dyspnoea scale which comprises five statements that describe almost the entire range of respiratory disability from none (Grade 0) to almost complete incapacity (Grade 4). 24
Bronchiectasis severity index (BSI), FACED score and E-FACED score
We used BSI, FACED and E-FACED to evaluate the predictive capacity of exacerbations and mortality. The severity of BE according to FACED and E-FACED is mild (0–2 points), moderate (3–4 points), or severe (5–7 points). The results of the BSI are scored as mild (0–4 points), moderate (5–8 points), or severe BE (9 or more points).25,26
Patient and public involvement
Written consents were given by participants. The data were handled in the EMBARC database anonymously. Neither patients nor the public was involved in the design, conduct, or reporting of our research.
Statistical analyses
The chi square test was used for categorical variable comparisons. Continuous and ordinal variables between groups were analysed with a t-test or Mann-Whitney test, depending on variable distributions. To study the association between various symptom domains of QoL, mMRC dyspnoea score and BSI index, Spearman’s correlation coefficient was used. Cox´s regression was used to analyse factors predicting mortality, and Kaplan-Meier analysis was used to estimate the survival function in follow-up. P-values less than 0.05 were considered statistically significant. FACED, E-FACED and BSI were adjusted only for gender, as they already included age as a parameter. All the statistical analyses were conducted with Statistical Package for Social Sciences (SPSS), version 22 (IBM corporation, Armonk, NY)
Ethical approval
The ethics committee of HUH approved the study (registration number 214/13/03/01/2016) and accepted a prolonged permission in February 2023.
Results
Characteristics of the BE Survived Patients and the Non-Survived Patients.

FEV1% = forced expiratory volume in one second, mMRC = modified medical research council dyspnoea scale, BMI = body mass index, BSI = bronchiectasis severity index, QoL-B = Quality of Life Bronchiectasis Questionnaire, QoL = quality of life.
aMWU; median (IRQ).
bX2-test; n (%).
A higher score of BSI (p = 0.03, HR 1.2, Cl 1.0–1.4), FACED (p = 0.01, HR 1.9, Cl 1.2–3.1), E-FACED (p = 0.01, HR 1.6, Cl 1.1–2.2) or mMRC (p = 0.01, HR 3.4, Cl 1.6–7.3) predicted higher mortality. When adjusting FACED (p = 0.01, HR 1.9 CI 1.1–3.0) and E-FACED (p = 0.02, HR 1.5 CI 1.1–2.1) for gender, it retained its statistical power in predicting mortality. When adjusted for age and gender, mMRC (p = 0.03, HR 3.2 CI 1.5–6.9) remained statistically significant. BSI (p = 0.03, HR1.20 CI 1.2–1.4), adjusted for gender only, also retained statistical significance.
Factors (Unadjusted and Adjusted) Predicting Mortality in the Finnish BE Cohort.

BSI = Bronchiectasis severity index, Extended disease = Three or more lobes affected, mMRC = modified medical research council dyspnoea scale, QoL-B = Quality of Life Bronchiectasis Questionnaire, FEV1% = FEV1/FVC = Forced expiratory volume in one second / Forced vital capacity.
*The quantity of cases is insufficient - unanalysed.
Cox’s regression.
aAdjusted with age and gender.
bAdjusted only with Gender.
Considering the domains of FACED score and BSI separately found a higher score of age (p 0.03) correlating with mortality. The higher score of mMRC (p = 0.01) correlates with higher mortality in both tools (Figure 1(a–c)). Lower scores of domains—physical (p < 0.01), vitality (p = 0.01), respiration (p = 0.03) and health (p < 0.01)—were associated with mortality (Table 3). (a) BSI Score Values in Groups Survived and Non-Survived, (b). MMRC Score Values in Groups Survived and Non-Survived, (c). FACED Score Values in Groups Survived and Non-Survived. FACED, BSI, QoL and Their Domains Characterised by the BE Survived and Non-Survived Patients. FEV1% = forced expiratory volume in one second, mMRC = modified medical research council dyspnoea scale, BSI = bronchiectasis severity index, BMI = body mass index, QoL-B = Quality of Life Bronchiectasis Questionnaire. aMWU; median (IQR). bX2-test; n (%).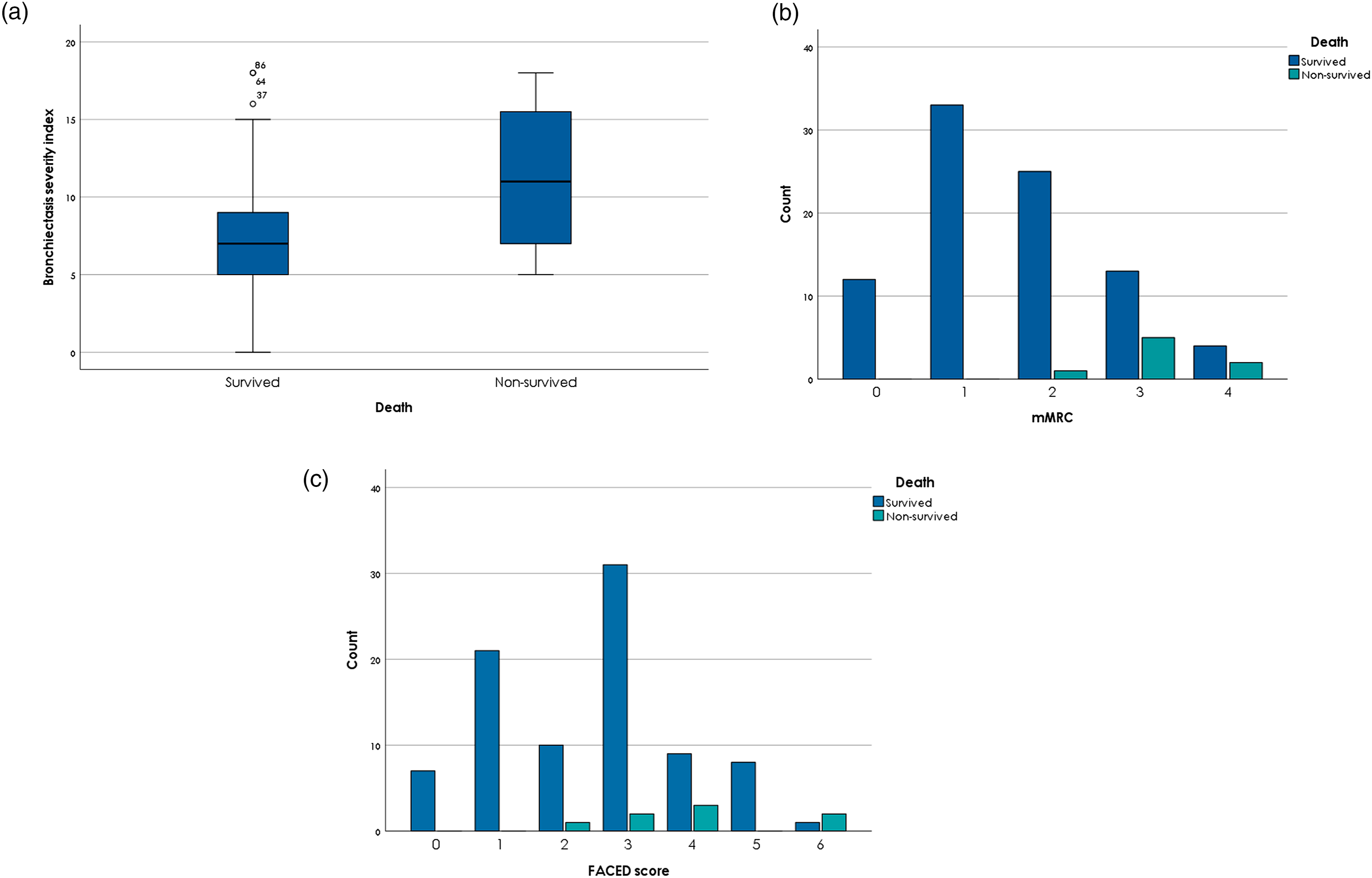

Quality of Life Questionnaire Domains Compared By BE Survived Patients and Non-Survived Patients.

Missing cases n = 3. T-test was used.
Survival in the follow-up is suggested to be higher in adult patients with mild BE compared to those who have moderate or severe BE evaluated by BSI (p = 0.14). FACED-evaluated patients had equal survival curves. Adult patients with moderate or severe BE had suggestive higher mortality compared to patients with mild disease (p = 0.08) (Figure 2(a–b)). Exacerbation mean rate was 2.4/year (CI 1.98–2.86) at start. Annual progress was year two 2.15 (CI 1.73–2.57), year three 1.93 (CI 1.56–2.30) and year four 1.96 (CI 1.52–2.40) (Figure 3 appendix). (a) Kaplan-Maier Survival Curve Compared Mild BE vs. Moderate/Severe BE According to FACED Score, (a) Kaplan-Maier Survival Curve Compared Mild BE vs. Moderate/Severe BE According to BSI Score. Annual Exacerbations (n) In 4 Years of Follow-Up.

Pearson Correlation for Cumulative Number of Exacerbations.
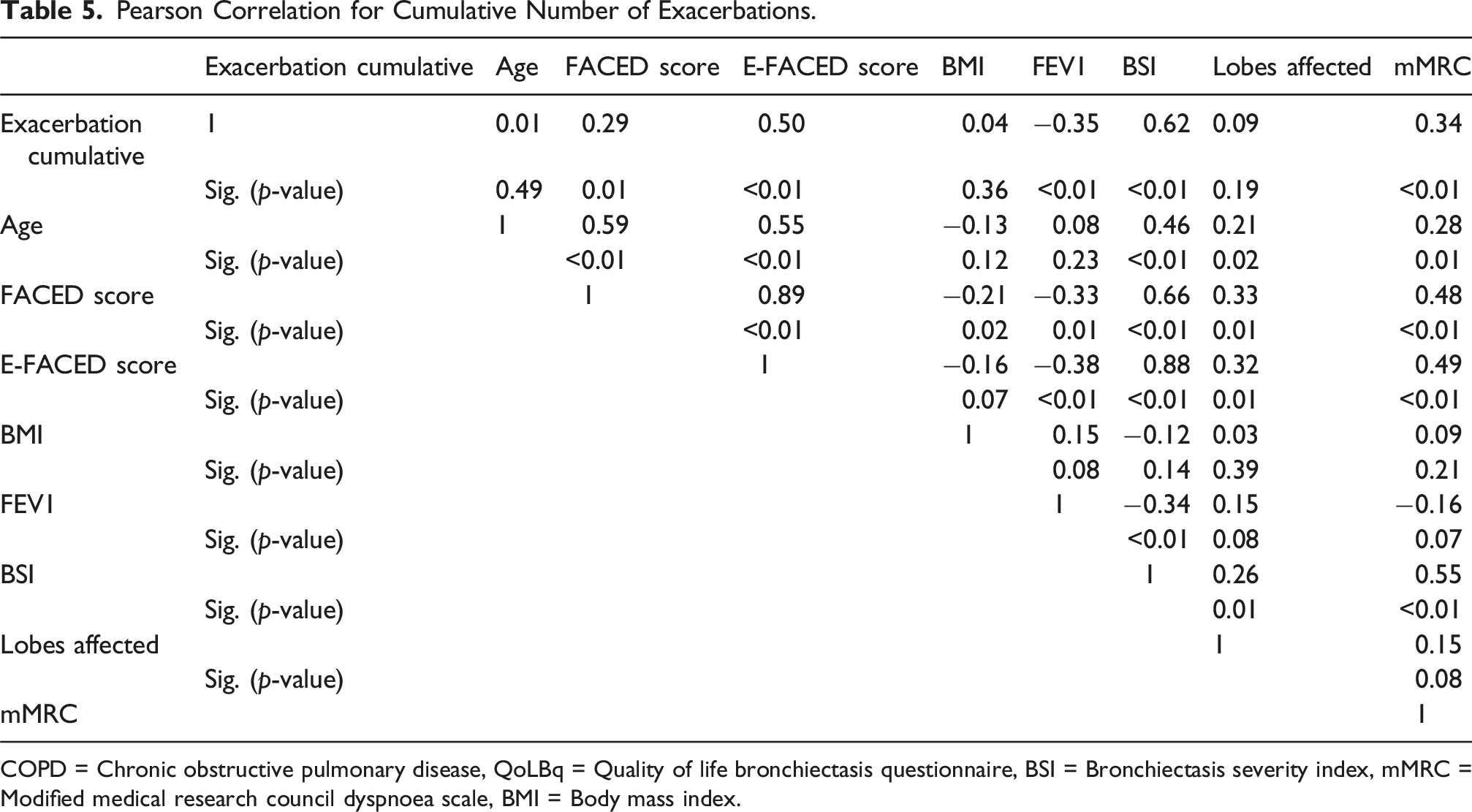
COPD = Chronic obstructive pulmonary disease, QoLBq = Quality of life bronchiectasis questionnaire, BSI = Bronchiectasis severity index, mMRC = Modified medical research council dyspnoea scale, BMI = Body mass index.
Regression Analysis for Cumulative Number of Exacerbations.

COPD = Chronic obstructive pulmonary disease, QoLBq = Quality of life bronchiectasis questionnaire, BSI = Bronchiectasis severity index, mMRC = Modified medical research council dyspnoea scale, BMI = Body mass index.
*Unadjusted with age.
Discussion
In adult patients with BE, two of the three used scores (FACED and E-FACED) are better for predicting mortality than any single determinant such as FEV1 or exacerbation rate except for age. In our study, an exception was mMRC, which is a single variable for self-reported breathlessness, predicted mortality at high value (adj. HR 3.2). The mean FACED score showed a higher risk for mortality (HR 1.9) than the mean BSI score (HR 1.2). E-FACED is considered in general as a tool for predicting exacerbation; however, it showed a higher risk for mortality (HR 1.5). For cumulative number of exacerbations, asthma, bacterial colonisation and BSI were risk factors.
Aetiology of BE did not affect mortality in this cohort. Low self-reported QoL-B in the physical capacity, vitality, respiration and health domains were correlated with mortality.
Cystic changes in BE are usually linked to severe disease, but in our study, they weren’t associated with higher mortality. This may be due to our limited sample size. In the study by Pieters et al., computed tomography (CT) findings in BE were analyzed by scoring the CT findings for bronchiectasis, mucus plugging, inflammatory features and total disease findings (Pieters et al.). CT findings were heterogenous and e.g. older age, longer disease history and more severe disease were associated with more findings. We used extensive disease in our analyses, 17 but did not find that as a predictive marker for mortality
During the follow-up, 8.4% of the BE cohort died. The patients with higher scores of FACED were twice as likely to die when compared to the patients with lower FACED scores. Similarly, mortality among the patients with a higher BSI score was 10 % higher than among the patients with a lower BSI score. There was a trend for better survival in the Kaplan-Meier analyses for mild BE compared to the moderate and severe BE estimated either by FACED or BSI. However, the difference was not statistically significant.
Our study demonstrated that the higher rate of mMRC predicted higher mortality (HR 3.2), which came as an interesting result in BE. This self-reported assessment tool for exercise tolerance has been relatively less studied in BE disease. However, there are studies that have shown similar findings. Breathlessness appears in COPD to remain an independent risk factor for both exacerbations and all-cause mortality, even after adjusting for lung function and other relevant risk factors. 27 A large meta-analysis identified a significant relationship between the presence of breathlessness (assessed by mMRC) and increased mortality. The mechanism remained unclear or complex and may reflect breathlessness as a symptom of many diseases. 12
In a large multicentre study, BSI was found to be superior to FACED in predicting many clinically important disease-related outcomes, such as hospital admissions, exacerbations, QoL, respiratory symptoms, 6-min walking test and lung function decline in BE. 28 However, both the FACED and the BSI gave similar results for predicting mortality (area under curve [AUC] 0.76 for both scores) in the same study. 28 Accordingly, Ellis et al. found both scoring systems similar in predicting mortality in adult patients with BE in a 5-years follow-up with an AUC 0.79 for BSI and 0.8 for FACED. 29 When comparing these estimates, a FACED score with a cut-off value of >5 and a BSI with a cut-off value of >9 have been reported to predict mortality and hospitalisations, respectively. 30 In our study, the FACED score for the non-survived group had a median value of four and BSI a median value of 11. In a Korean BE patient population, low BMI predicted mortality and current smoking increased the risk for mortality by 11-fold. 31 Smoking is mostly associated with increased risk of mortality, both for overall mortality as well as cardiovascular, cancer and respiratory mortality. 32 BMI is associated with survival with a U-shaped curve with increased mortality in both underweight and overweight patients. 33 Compared to our study, smoking was not associated with mortality, and the non-survived group had lower BMI than the survived group. However, the difference was small and did not reach statistical significance. We assume that the relatively small sample size explains the lack of association.
In an earlier study, BSI and FACED were correlated with acute exacerbations, and FACED score results were correlated with emergency department visits. 34 Moreover, a parameter for 6 min walking distance together with the lowest saturation measure was equal to FACED and BSI in predicting mortality but probably easier to calculate or to measure in clinical practice. 34
In a Spanish study (185 patients), 12.4% died in 1 year’s follow-up, and the risk was increased in males. The mortality causes were respiratory related (68%) and other common causes were cardiovascular (18%) and septic shock (14%). 35 In Goeminne et al.'s 2012 study, the 41-months follow-up revealed a death rate of 10.6% among a cohort of 539 patients, with thoracic cancer and COPD showing the highest mortality rates (30% and 26%, respectively). 36 Additionally, in Goeminne’s 2014 study, the 5-years follow-up recorded a 20.5% death rate in a 245-patient cohort, with respiratory causes accounting for the majority (58%) of deaths. 7 In a 2022 study by Mateus et al., involving 120 BE patients followed for 3 years, the all-cause mortality rate was 10.8%, with cardiovascular disease contributing to 30.8% of the deaths observed in the cohort. 37 In our follow-up, the causes of death were heterogeneous, with three out of eight patients having a respiratory infection as the cause of mortality. In an Indian study, frequent exacerbations, COPD, cardiovascular disease, current smoking, bronchial colonization with Enterobacterales or Klebsiella pneumoniae increased the risk of mortality. 38 As well, per 1-unit increase in mMRC dyspnoea score increased mortality risk. In addition, in the study by Aliberti et al., increasing purulence of sputum increased the risk of exacerbations, hospitalizations and finally also mortality. 39
Exacerbation rate had a decreasing trend in the follow-up (2.4/year [CI 1.98–2.86] at start and in the endpoint 2/year [CI 1.52–2.40, p = 0.09]), suggesting that in some patients, exacerbation rate can be decreased by treatment. The size of the cohort decreased during the follow-up due to deaths and patients who were transferred to observation in primary care. The change to a lower exacerbation rate, however, did not reach statistical significance.
In a meta-analysis, macrolides significantly reduced exacerbations and prolonged time between them in adult patients with BE across all subgroups. Further, QoL was improved, but the therapy had no effect on lung functions. 40 In our study, lung function alone was not predictive for mortality, in contrast to a relatively small Brazilian cohort in which age and FEV1 % predicted mortality. 41
Strengths and limitations
Our study population of adult patients with BE is an independent part of the EMBARC study and had a limited size. BE diagnosis was confirmed by HRCT. The follow-up time of the cohort was relatively short, and 10 patients were lost from the follow-up because the treatment was transferred into primary health care. The small study population limit our ability to assess mortality accurately. Mortality rarity was unknown before analysis, and no prior sample size calculation was done, but this would not have changed the limited cohort size due to the small population.
Conclusions
According to our evaluation, using multidimensional tools is preferable to using a single value for assessing mortality. An exception turned out to be the mMRC, which at high value predicted mortality 3-fold. A higher FACED score indicates twice as high a risk for mortality compared to lower FACED scores. FACED and mMRC scores as tools can be used both in research and in clinical practice when estimating adult BE patients’ survival possibilities. However, even in this rather small study population with a follow-up time of 4 years, mortality was 8.4%. This highlights the severity of BE as a chronic pulmonary disease and the need for better treatment options.
Footnotes
Acknowledgments
The authors would like to acknowledge all the patients who participated in the study. We thank Statistician Ville Kinnula at Helsinki University for statistical advice.
Contributorship statement
PK was responsible for the design, analysis, interpretation, drafting and finalizing of the manuscript. WM was involved in the analysis, interpretation, drafting and finalizing of the manuscript. JM was responsible for the data collection, analysis, interpretation, drafting and finalizing of the manuscript. TT was involved in the data collection, drafting and finalizing of the manuscript. PB provided statistical assistance and was involved in finalizing of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding of the Foundation of the Finnish Anti-Tuberculosis Association, The Finnish Research Foundation of the Pulmonary Diseases and University of Helsinki and Helsinki University Hospital, Heart and Lung Center, Department of Respiratory Diseases, Helsinki, Finland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This is part of the EMBARC study and data can be obtained from EMBARC register or from the authors by an approved application.
