Abstract
Background
Malnutrition and sarcopenia are common in inpatient rehabilitation, however individual nutritional therapy (iNT) is often underutilized. This study aimed to assess the effect of iNT on nutrition and muscular health.
Method
Patients with chronic obstructive pulmonary disease (COPD) or post-pneumonia at risk for malnutrition and sarcopenia undergoing inpatient rehabilitation were enrolled. The control group received usual care including enriched food and educational group sessions on nutrition. The intervention group received additional counselling by a dietician twice a week. Both groups received individualized physiotherapy and self-management coaching, endurance exercise sessions on 5 days and strength training sessions on 2–3 days per week as part of the clinical routine. Primary outcomes were changes in energy and protein intake, assessed via menu consumption and macronutrient analysis. Secondary outcomes included handgrip strength, muscle mass by bioimpedance analysis, and physical performance measured by the timed-up-and-go test.
Results
Twenty-six patients per group (median age of 72 years, 60% men, 52% COPD) were included. Energy and protein intake increased significantly more in the intervention group with a 309 kcal and 16 g compared to −53 kcal and −1 g in the control group (p = 0.001 for group differences). Handgrip improved more in the intervention group by a median of 1 kg (p = 0.007), without group differences in muscle mass or physical performance.
Conclusion
INT effectively increased energy and protein intake in patients at risk of malnutrition and sarcopenia undergoing 3 weeks of pulmonary rehabilitation, with a positive impact on prognostic handgrip strength.
Trial registration
The study was registered by the US National Institutes of Health (ClinicalTrials.gov). # NCT05096013.
Keywords
Introduction
Malnutrition and sarcopenia are common health concerns in patients with chronic or severe diseases and related to increased morbidity and mortality, length of hospital stay and treatment costs. 1 According to the European Society of Clinical Nutrition (ESPEN), malnutrition results from an inadequate intake of nutrients, increased nutrient requirement or altered nutrient metabolism with consequences on body composition and reduced physical and mental function. 2 Consequently, malnutrition is a strong predictor for the onset of sarcopenia, which is defined as the presence of low muscle strength and mass according to the European Working Group on Sarcopenia in Older People.3,4 Both diagnoses frequently co-occur in hospitalized patient with sarcopenia, estimated to be present in half of post-acute inpatient rehabilitation patients.5,6 The loss of muscle mass and the reduction in muscle function in sarcopenia results not only in an increased risk of falls and fractures with impairment of activities of daily living but is also associated with respiratory diseases. 4 Research indicates that chronic obstructive pulmonary disease (COPD) and sarcopenia have a mutually reinforcing relationship through systemic inflammation, reduced exercise tolerance, physical inactivity, and an increased risk of exacerbations. This interplay contributes to a vicious cycle that progressively worsens prognosis. 7 In addition, sarcopenia has been recognized as a contributor to swallowing dysfunction (sarcopenic dysphagia), 8 substantially increases the risk of aspiration and subsequent pneumonia.9,10 Therefore, addressing sarcopenia in patients with COPD and in those recovering from pneumonia during pulmonary rehabilitation may be crucial to interrupting this vicious cycle, improving functional outcomes, and reducing the risk of pneumonia recurrence.
A corner stone of the management of sarcopenia is resistance exercise in combination with sufficient protein intake.11–15 A recent meta-analysis found a significant association between increased protein and energy intake during hospital stay with a reduction in mortality and re-admissions in patients who received nutritional support. However, only one third of the 27 included studies involved nutritional support from dieticians. 16 The effect of individualized nutritional therapy (iNT) in achieving protein and caloric goals has been demonstrated in the large EFFORT trial, a randomized controlled study involving more than 2000 hospitalized patients. This trial found improved survival in the intervention group and its findings have been endorsed by the latest ESPEN practical guidelines on nutritional support for polymorbid medical inpatients.17,18 However, data on the effect of iNT in post-acute inpatient rehabilitation setting are sparse and personal resources for dedicated individual nutritional care often lacking. 19
The aim of this study was to evaluate the effect of standardized energy- and protein-enriched diet with additional iNT compared to standardized energy- and protein-enriched diet without iNT during inpatient exercise-based pulmonary rehabilitation on (1) energy and protein intake and (2) muscle strength, mass, and function.
Material and methods
Study design
This was a two-arm, parallel-design, open-labelled single-center randomized controlled trial conducted from November 2021 to January 2024 in an inpatient rehabilitation clinic in Switzerland. The study was approved by the Ethics Committee of the Canton of Bern (#2021-01824) and registered with the US National Institutes of Health under ClinicalTrials.gov (#NCT05096013).
Study participants
All patients entering rehabilitation with COPD or a condition following pneumonia as primary indication for PR were defined as the target population. Inclusion criteria were a mild to moderate risk of malnutrition (NRS 2002 score of 3 or 4 points) 20 in combination with an elevated risk of sarcopenia, defined as a SARC-F score ≥ 4 points. 4 Both scores were assessed at the start of rehabilitation.
Patients at high risk of malnutrition with an NRS-2002 of ≥5 in the respective rehabilitation clinic were excluded because they received iNT as part of the clinical routine and could therefore not be randomized. The rationale for adding the SARC-F screening was to further identify among patients with moderate malnutrition risk (NRS 3–4) those who were most likely to benefit from iNT, namely those with concurrent risk of sarcopenia. Further exclusion criteria were cognitive impairment or language barriers limiting patients in following and understanding study procedures, enteral or parenteral nutrition at the start of rehabilitation, status following bariatric surgery and inability to perform a bioelectrical impedance analysis (BIA), e.g., due to implanted cardioverter defibrillators. Subjects who underwent medically prescribed iNT after inclusion in the study were excluded from the study in order to not interfere with clinical routine (n = 2 in the control group). All patients were recruited by a good clinical practice trained study coordinator and dietician and all patient gave written informed consent.
Randomization
Patients were randomly assigned (1:1) to receive either iNT and usual care (intervention group) or usual care alone (control group). Randomization was carried out with the randomization module integrated in the internet-based Clinical Data Management System REDCap® using varying block sizes and stratified according to diagnosis (COPD vs pneumonia), sex (male vs female), age (<70 vs ≥70 years) and energy intake at baseline (<75% vs ≥75% of their individual demand). Investigators involved in patient recruitment (AZ) did not have access to the randomization list at any time and are considered blinded for group allocation.
Blinding
All participants and investigators were aware of group assignment. However, collection of the primary (energy intake) and secondary (protein intake) endpoint was blinded as described in data collection and assessments. Therefore, the study is considered assessor blinded. Blinding was not feasible for sarcopenia related parameters.
Setting
The study was embedded in a 3-weeks exercise-based inpatient pulmonary rehabilitation program. The rehabilitation program included individualized physiotherapy and self-management coaching, group-based exercise training sessions, smoking cessation support if needed, and educational modules on disease and symptom management, oxygen therapy, medication use, and nutrition. Exercise training consisted of endurance and strength components. Endurance sessions were performed five times per week and included activities such as cycling on a bicycle ergometer or walking sessions, typically lasting 15 to 30 min depending on individual capacity. Strength training was conducted two to three times per week and involved resistance exercises targeting major muscle groups. Exercise prescriptions were individualized based on patients’ functional status and rehabilitation goals. The exercise regime did not differ between intervention and control group.
Patients at risk for malnutrition (three or more points on the NRS-2002 screening) are assigned to nutritional educational group sessions about malnutrition and healthy nutrition and receive standardized food fortification. Specifically, soups are enriched with 10 g protein powder, two tablespoons of cream and one tablespoon of rapeseed oil. The dessert component is swapped for a high-energy and high-protein variant if necessary. Patients with a high risk of malnutrition (screened with five or more point on NRS-2002 or according to clinical reasoning) receive iNT from dieticians as described below.
Intervention group
Participants allocated to the intervention group received the usual care program plus the iNT that is otherwise provided only to patients at high risk for malnutrition in clinical routine. In accordance with clinical practice, a detailed initial nutritional assessment was performed, including obtaining a comprehensive nutritional history regarding current intake, requirements, and nutrition-related medications. Information on weight progression, body constitution, chewing and swallowing ability and the gastrointestinal profile and nutrition-relevant laboratory parameters were interpreted and social and individual needs regarding nutrition discussed. Based on this assessment, individualized goal setting and tailored interventions were planned collaboratively with each patient. The interventions included monitoring of intake of macro- and micronutrients, individualized dietary adjustments and enrichment of food components and, in consultation with the treating physicians, the prescription of oral nutritional supplementation and medication if necessary. Nutritional monitoring with individualized counselling was conducted by a registered dietitian twice per week throughout the rehabilitation stay, leading to approximately five to six sessions per patient over the 3-week period. Counselling topics included strategies to optimize energy and protein intake, taking into account individual needs, tolerance, and preferences, as well as approaches to overcome barriers to adequate nutrition.
Control group
Patients in the control group received the usual care rehabilitation program with standardized food fortification as described above. Patients in the control group did not receive iNT or any personal counselling from a dietician, but have participated in nutritional educational group sessions during their rehabilitation stay according to clinical routine.
Primary and secondary outcomes
The primary endpoint was defined as change in energy intake in kilocalories derived from food records at start and before the end of the rehabilitation.
Energy intake was selected as the primary outcome because it directly reflects the intended effect of iNT. Although food provision during inpatient rehabilitation was standardized, patients retained the ability to choose between different menu options, and actual intake varied depending on factors such as appetite, acceptance of protein-enriched meals, gastrointestinal symptoms, and individual preferences. Changes in energy intake were considered clinically relevant and more feasible to detect within the short intervention period compared to functional or structural sarcopenia-related endpoints. Further secondary endpoints were change in protein intake (g) and sarcopenia related parameters, namely change of muscle strength measured by grip strength, change in muscle quantity estimated by lean body mass measured with a body impedance analysis and change in muscle functioning assessed by a timed up-and-go test. 4
Data collection and assessments
Energy and protein intake
Energy and protein intake were averaged over three consecutive days within 5 days after rehabilitation admission and once again over three consecutive days before discharge. Guest service staff documented food intake for each meal in quartiles of the portion size of the provided menus and snacks. Energy and protein intake were calculated based on the consumed menu quantity and analysis of its’ macronutrients as stored in the clinics’ electronic menu selection system. Calculations of energy and protein intake was performed by one investigator (AZ) blinded to group allocation.
Energy and protein requirements
Energy and protein requirements were calculated according to clinical routine and as suggested by the consensus of an ESPEN expert group with the Harris-Benedict formula adjusted for body weight for energy requirement and 1.2 to 1.5 g protein per kg bodyweight depending on clinical status (and 0.8 g/kg for patients with renal insufficiency). 21
Muscle strength
As a surrogate for muscle strength, handgrip strength was measured at the start of rehabilitation and prior to discharge according to the European Working Group on Sarcopenia in Older People (EWGSOP2) criteria. 4 In the present study, the measurements were carried out by a trained dietician (AZ) using a hydraulic Jamar hand dynamometer with two repetitions with the dominant and one repetition with the non-dominant hand, selecting of the strongest measurement thereof. 22
Muscle mass
Bioelectrical impedance analysis (BIA) was performed using the BIA 101 multifrequency device (Akern Srl, Florence, Italy) with the classical tetrapolar technique. Measurements were conducted under standardized conditions: at room temperature, in the morning in a fasting state, after avoidance of prior physical exertion. Patients were positioned supine with legs slightly bent (∼45°) and arms slightly abducted (∼30° from the body). 23
Muscle function
According to EWGSOP2, various physical performance tests are available to measure muscle function. 4 The timed-up-and-go test (TUG) was used as a practical assessment for this study and according to clinics standard operating procedures.
Sample size calculation
Based on a previous study, 24 an increase of 300 kcal (SD 360) in the intervention group and 50 kcal (SD 310) in the control group was expected, which would correspond to a medium to large effect size and a clinical relevant increase. 25 Accordingly, 30 patients per group were deemed as required to achieve a power of 80% with a significance level of 5% and an estimated drop-out rate of 10%.
Statistical analyses
Patient characteristics were compared between groups using descriptive statistics with counts and frequencies for categorical and median and quartiles for continuous data. Change in primary and secondary endpoints were analysed using Wilcoxon rank-sum tests for non-normally distributed data. As additional sensitivity analyses,, multi-variate linear models for change in energy and protein intake as well as change in handgrip strength were performed to adjust for age, sex, primary pulmonary rehabilitation indication and baseline value. Frequencies of patients reaching energy and protein-requirements at discharge were compared between groups using a X2 test.
In explorative post-hoc analyses, change in handgrip strength was correlated with changes in energy and protein intake respectively using Pearson statistics.
The significance level was set at 0.05 for all tests with no adjustment for multiple testing. All analyses were done in the intention-to-treat population, which included all randomized patients that did not withdrew consent, were lost-to follow-up or excluded from the study. All analyses were performed using the statistical software R, version 4.0.3.
Results
Overall 146 patients admitted to pulmonary rehabilitation after pneumonia or COPD exacerbation were assessed for eligibility, 59 randomly allocated to the intervention (n = 30) or control group (n = 29). Due to drop-outs, 26 patients per group were included in the intention-to-treat analysis (Figure 1). Baseline characteristics were comparable in both study groups (Table 1). Overall, patients had a median age of 72 (interquartile range 61 to 77) and 31 (60%) were men. At baseline, 10 (19%) and 6 (12%) of the patients have not reached at least 75% of their recommended energy or protein intake, respectively. Across both groups, 26 (50%) of the patients fulfilled the GLIM criteria
26
for a diagnosed malnutrition. Flow chart. Patient characteristics. Median (quartiles) for continuous and counts (frequencies) for categorical data.

Energy & protein intake
Outcomes.

Median (quartiles) for continuous data, Δ Change from entry to discharge assessement.
p-value for group difference from unpaired Wilcoxon rank sum test.
***p < 0.001 for within-group differences between entry and discharge assessments from paired Wilcoxon-test

Energy intake, protein intake and handgrip strength by group and timepoint.

Energy and protein intake of each study participant at the end or rehabilitation in percentage of the individual requirement.
Multivariate linear models for changes in energy and protein intake and change in handgrip strength.
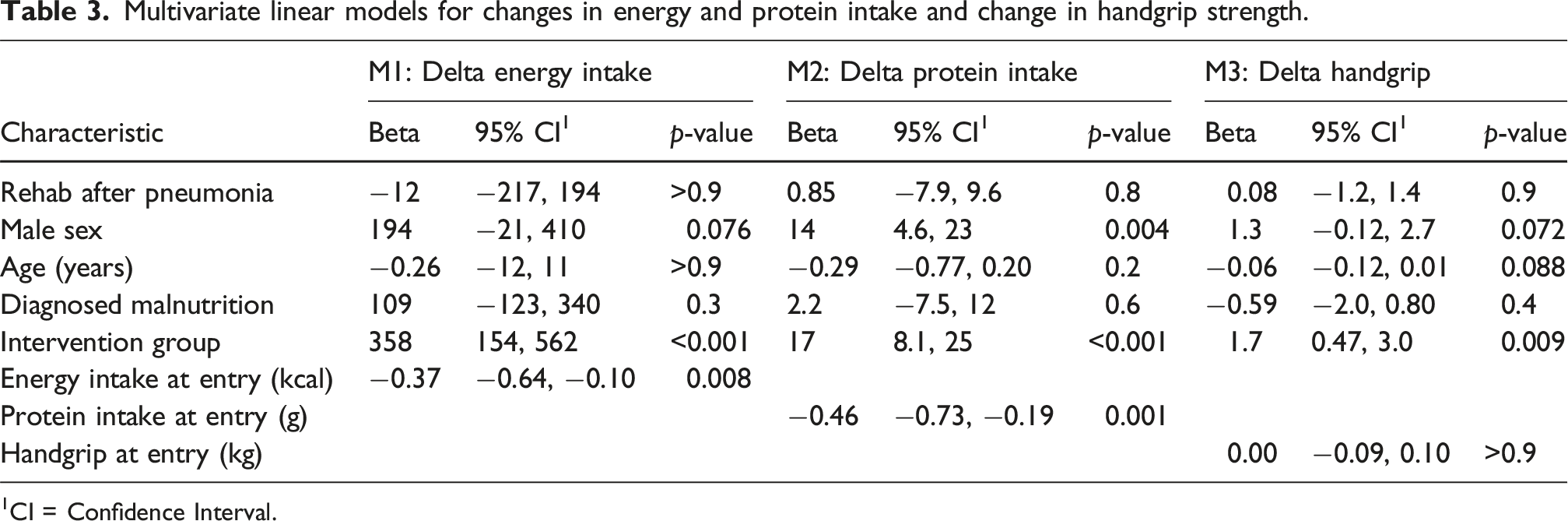
1CI = Confidence Interval.
Muscle strength, mass and function
Both groups significantly improved in handgrip (muscle strength) and muscle function, but not in body composition. The intervention group improved significantly (p = 0.007) more in handgrip strength with a median of 2 kg versus 1 kg in the control group. The greater improvement in handgrip observed in the intervention group compared to the control group remained (β = 1.7 kg, 95% confidence interval 0.47; 3.0) in the multivariate adjusted model (Table 3). Male patients tended to improve greater in energy and protein intake as well as handgrip strength (Table 3). However, only weak associations were observed between change in handgrip strength with change in energy intake (R = 0.23, p = 0.1) and with change in protein intake (R = 0.3, p = 0.03) (Supplemental Figure 1).
Discussion
In this single-center randomized controlled trial, patients at risk of malnutrition and sarcopenia significantly increased their energy and protein intake as well as handgrip during a short 3-week exercise based pulmonary inpatient rehabilitation when accompanied by iNT.
Energy and protein intake increased by a median of 309 kcal and 16 g respectively with PR in the intervention group versus −53 kcal and −1 g in the control group, respectively. While a significant proportion of patients did not meet their individual required energy and protein intake at end of PR, the intervention nearly doubled this percentage (58% vs 23%). This underscores the necessity of individualized nutritional counselling to help patients better meet their dietary requirements during PR. We also observed a statistically significant higher improvement in handgrip strength (+1 kg) in the intervention group compared to the control group. Muscle mass and functional mobility however, did not differ between groups.
Our results are in line with a meta-analysis of malnourished non-critically medical inpatients, which found higher energy (+365 kcal) and protein (+17.7 g) with nutritional support compared to usual care. 16 In this meta-analysis the higher energy and protein intake achieved with nutritional support was associated with a reduction in mortality and nonelective hospital readmissions, suggesting that our observed effects are of clinical relevance. Noteworthy, the effects of nutritional support on mortality were particularly strong in the most recent trials included in the meta-analysis, where patients with malnutrition were followed individually by a nutritionist.
Half of the patients included in our study were already close to or above their recommended energy and protein intake at start of PR before or without seeing any dietician. On the other hand, 50% of the included patients met the GLIM criteria for diagnosed malnutrition, 12% with severe malnutrition. These patients have a clear indication for iNT, but are not receiving optimal nutritional care under the clinics’ current nutritional regimen with iNT only prescribed to patients with NRS-2002 of 5 points or higher. Correspondingly, patients with diagnosed malnutrition according to the GLIM criteria but only low to moderate risk of malnutrition according to the clinical screening tool NRS-2002, do not receive iNT.
A recent multi-center prevalence study across various rehabilitation disciplines found that 35% of all rehabilitation inpatients have malnutrition and 33% sarcopenia with a co-occurrence of 22% and highest prevalence in geriatric and pulmonary patients. 27 Given the high prevalence and limited personnel resources in rehabilitation care, it is critical to identify patients most in need for iNT in clinical routine.
Low handgrip strength is considered as indicator of sarcopenia 4 and associated with increased mortality, morbidity and poorer health related quality of life in patients with COPD. 28 If the 1 kg greater improvement in handgrip strength in the intervention group is of clinical relevance remains unclear as there is not sufficient evidence to specific minimal important clinical differences. 29 Nevertheless, our findings are noteworthy given the short observation time of 2–3 weeks. Body composition did not change during the course of rehabilitation. This seems plausible given the short observation time in our study. A recent meta-analysis found that exercise and nutritional supplementation interventions in patients with COPD were only effective in improving fat-free mass when combined. However, the three trials that reported significant improvements in fat-free mass ranged from 8 to 12 weeks, suggesting that pulmonary rehabilitation of 3 weeks (with or without iNT) may be too short to initiate changes in body composition. 30 In contrast, TUG-time significantly improved over the course of PR with no differences between groups (−4 s). Both groups followed the same exercise regime of the clinical routine. As the exercise intervention did not differ between groups, this could be expected. The overall substantial regain in function capacity during inpatient rehabilitation is noteworthy though. In comparison, a previous study in patients with COPD identified already a change of 0.9–1.4 s over 8–12 weeks of PR as clinically relevant. 31
In our explorative sub-analyses, we have observed that male patients tend to improve more in energy and protein intake as well as handgrip when compared to female patients. However, whether sex differences in PR outcomes exists requires further investigation. Existing evidence does not support such a hypothesis.32,33
Limitations
This study has several limitations. First, it was a single-center study, which limits its external validity. We also did not include patients with high risk of malnutrition (NRS ≥ 5), since they receive iNT as part of the clinical routine and therefore could not be ethically randomized. Nonetheless, it could be hypothesized that its effectiveness would be comparable—or potentially even greater— in patients at higher risk, who typically have a stronger indication and potentially greater benefit from nutritional interventions. Additionally, the study was conducted in an inpatient rehabilitation setting where diet can be well controlled, which is less feasible in outpatient PR programs.
Second, blinding of study personnel and patients towards group allocation was not feasible. However, food intake documentation was performed by clinical staff not otherwise involved in the study and calculations of energy and protein intakes for follow-up assessments were performed in a blinded manner. Bliding of sarcopenia related assessements (handgrip, body composition and physical functioning) was not feasible for this study. Third, our approach of estimating energy and protein intake may not be very precise and some snacks consumed by patients may have been missed. However, this procedure is close to the clinical routine where dietician base their counselling on such documentations and calculations. Lastly, there was no long-term follow-up after rehabilitation discharge. It remains to be investigated whether the improvements in energy, protein and handgrip strength are sustainable after PR discharge and whether they translate into differences in body composition and muscle health or hard clinical endpoints such as mortality or re-hospitalization.
Conclusion
Individual nutritional counselling during pulmonary rehabilitation leads to an increased energy and protein intake as well as greater improvement of handgrip strength in patients with COPD or after pneumonia at low to moderate risk of malnutrition and sarcopenia. Half of the patients identified at low to moderate risk of malnutrition with the clinical routine screening have finally met the criteria for the diagnosis of (severe) malnutrition. Given the potential positive effect of iNT during PR, it is crucial to further assess patients at low to moderate risk of malnutrition and sarcopenia to identify those most in need of iNT and to strengthen nutritional management during rehabilitation. Further investigations are needed to assess and maintain improvements after rehabilitation discharge.
Supplemental Material
Supplemental Material - Effect of individual nutritional therapy during inpatient pulmonary rehabilitation in patients at risk for malnutrition and sarcopenia - a randomized controlled trial
Supplemental Material for Effect of individual nutritional therapy during inpatient pulmonary rehabilitation in patients at risk for malnutrition and sarcopenia - a randomized controlled trial by Marco Coiro, Andrea Zurfluh, Undine Lehmann, Patrick Brun, Anke Scheel-Sailer, Hansueli Tschanz, Ann van Hoof, Matthias Wilhelm and Thimo Marcin in Chronic Respiratory Disease
Footnotes
Consent to participate
All patients were recruited by a good clinical practice trained study coordinator and dietician and all patient gave written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was initiated by the investigators. AZ received financial support from the Stiftung zur Förderung der Ernährungsforschung Schweiz. The institution of UL recieved an investigator-initiated trial grant unrelated to this project from Fresenius Kabi AG.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
