Abstract
Background
The paradigm of bronchiectasis is shifting away from its exclusive characterization as a neutrophilic condition. Patients with bronchiectasis and high eosinophil levels have been found to have a specific phenotype, but the clinical effect of eosinopenia remains unclear.
Method
A retrospective, single-center, observational study was conducted at a tertiary medical center. Adult patients under follow-up for bronchiectasis from January 2007 to August 2020 were categorized by blood eosinophil count (BEC) as follows: eosinopenia (<100 cells/µL), normal (100-299 cells/µL), and eosinophilia (≥300 cells/µL). Data on the first hospitalization due to exacerbation and the community exacerbation rate in the first year of follow-up were analyzed. Mortality rates were assessed up to the end of follow-up on September 1, 2023.
Results
The cohort included 724 patients (100%), 61% female (n = 442), of mean age 61 ± 16 years. The median follow-up period was 7.5 years (IQR: 5.1–10.8). Eosinopenia was found in 14.7% (n = 107), normal BEC in 56.6% (n = 417), and eosinophilia in 28.7% (n = 200). Patients with eosinopenia had a higher hazard ratio for first hospitalization than the normal-count group (1.71, 95% CI 1.11–2.64, p = .01) and the highest mean exacerbation rate (p = .04). On multivariate analysis, eosinopenia was significantly associated with higher mortality (HR 2.15, 95% CI 1.42–3.24, p < .001) after adjusting for age and sex.
Conclusion
Eosinopenia in bronchiectasis emerged as a potential biomarker for adverse outcomes. Further study of its role in disease behavior may provide insights for the development of therapeutic strategies.
Introduction
Bronchiectasis is defined by the presence of irreversible bronchial dilation associated with symptoms such as a productive cough with a purulent component and exacerbations. 1 It is the most frequent chronic airway inflammatory disease after chronic obstructive pulmonary disease and asthma. 2 The incidence, prevalence, and disease burden of bronchiectasis are increasing worldwide. 3
Although bronchiectasis is historically characterized by neutrophil-predominant airway inflammation, recent studies suggest that the inflammation is heterogeneous. There is growing evidence of a subpopulation of patients with bronchiectasis with a high blood eosinophil count (BEC) (high type 2 inflammation) who have a different phenotype in terms of clinical manifestations, lung function, exacerbation frequency, and mortality.4,5
Conversely, eosinopenia, characterized by a low BEC, represents a less well-explored aspect of bronchiectasis. Eosinopenia reflects low type 2 inflammation and may signify a different inflammatory pathway or disease mechanism compared to those with high eosinophil counts. Understanding eosinopenia is crucial as it may influence disease severity, response to treatment, and overall patient outcomes. Despite its potential impact, the clinical significance of eosinopenia in bronchiectasis has not been thoroughly investigated.6,7
This study aims to investigate the impact of eosinopenia on clinical outcomes in patients with bronchiectasis. Specifically, we compare patients with eosinopenia to those with normal BEC and eosinophilia in terms of exacerbation rates, hospitalization rates, and mortality outcomes. By examining these comparisons, we hope to clarify the role of eosinopenia in the broader context of bronchiectasis management and outcomes.
Methods
Study population and design
A retrospective, single-center, observational study design was used. The eligible population consisted of adult patients with bronchiectasis who were followed between January 2007 and August 2020 at a tertiary university medical center in Israel. The diagnosis of bronchiectasis was made by high-resolution computed tomography (CT) scan in all cases. 8 Patients were recruited as outpatients during their regular follow-up visits at the Pulmonary Institute.
The study was carried out in accordance with the Declaration of Helsinki and was approved by the institutional Ethics Committee (certification number: RMC-0373-24). Due to the retrospective nature of the study, informed consent was not required.
Data collection
Data were obtained from the electronic medical records database which integrates medical information from all hospitals in the country. Baseline clinical variables were recorded, namely, age, sex, body mass index (BMI), comorbid conditions, and long-term medication, in addition to basic laboratory variables of complete blood count, levels of albumin and creatinine, positive bacterial and fungal cultures from bronchoalveolar lavage (BAL), results of pulmonary function tests, and features of high-resolution CT scan. Data on the rate of bronchiectasis exacerbations leading to community healthcare visits, emergency room visits, and hospitalizations were recorded for the first year of follow-up. The number of exacerbations in the year prior to follow-up was also captured using the same electronic medical records database and applying the same methodology and criteria used during the follow-up period. Mortality rates were assessed until the end of follow-up on September 1, 2023.
Groups allocation
Patients were categorized into three groups based on their baseline blood eosinophil count (BEC) at the start of the follow-up period: (1) eosinopenic bronchiectasis (<100 cells/µL), (2) normal eosinophil count (100–299 cells/µL), and (3) eosinophilic bronchiectasis (≥300 cells/µL). The follow-up period began from the date of the initial assessment at our pulmonary institute. The cut-offs used are widely accepted in the COPD and bronchiectasis literature (4,7). All blood tests for categorization were conducted during a stable period, at least 4 weeks prior to any exacerbation events.
Definitions
The diagnosis of bronchiectasis was established based on clinical criteria, including presence of chronic cough, sputum production, and history of exacerbations, along with evidence on chest CT scan. 8 An exacerbation was defined when there is worsening in any three of the key symptoms of bronchiectasis (cough, sputum volume/consistency, sputum purulence, breathlessness, fatigue/malaise, and hemoptysis) for at least 48 h and which necessitates a change in therapy. 9
Statistical analysis
Descriptive data were summarized as mean and standard deviation or as number (percentage) of reported cases. Chi-square test was used to compare categorical variables, and Student’s t-test, to compare continuous variables. Statistical comparisons were conducted among BEC groups (<100, 100–300, or ≥300 cells/µl). A p value ≤.05 was considered significant.
Cox proportional hazard regression was applied to compare the time to first hospitalization due to bronchiectasis exacerbation among the three BEC groups, and Kruskal-Wallis test was used to compare the rate of exacerbations (including total hospitalizations, community healthcare visits, and emergency room visits because of bronchiectasis exacerbation) per patients during the first year of follow-up.
Kaplan-Meier survival analysis was conducted for three BEC categories. Univariate and multivariate analyses, including a logistic regression model, were performed to identify the variables most significantly associated with mortality.
All statistical analyses were carried out using SAS software, version 9.4 (SAS Institute Inc.).
Results
Patient characteristics
Demographic and clinical characteristics of patients with bronchiectasis by eosinophil count.

Data are presented as means ± standard deviations or numbers (percentages) of presented cases. Bold entries in the table indicate significant differences (p ≤ .05). ABPA, allergic bronchopulmonary aspergillosis; BAL, bronchoalveolar lavage; BEC, blood eosinophil counts; COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; LABAs, long-acting beta agonists; LAMAs, long-acting muscarinic antagonists; NTM, nontuberculous mycobacteria.
Laboratory, lung function, and radiological data of patients with bronchiectasis by eosinophil count.

Data are presented as means ± standard deviations or numbers (percentages) of presented cases. Bold entries in the table indicate significant differences (p ≤ .05). BEC, blood eosinophil counts; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; TLC, total lung capacity; RV, residual volume; DLCO, diffusing lung capacity for carbon monoxide; NTM, nontuberculous mycobacteria.
Exacerbation rate
On Cox proportion analysis of the relationship between BEC and the time to first hospitalization for bronchiectasis exacerbation, compared to the normal-BEC group, the hazard ratio was 1.71 for patients with eosinopenia (95% CI 1.11–2.64, p = .01) and 1.23 for patients with eosinophilia (95% CI 0.85–1.80, p = .2)
Figure 1 illustrates the cumulative incidence of hospitalization for bronchiectasis exacerbation among the three BEC groups. The incidence was highest in the eosinopenia group (p = .04). Cumulative incidence of hospitalization for bronchiectasis exacerbation among the three BEC groups in the first year of follow up.
The relationship between BEC category and visits for bronchiectasis exacerbations was evaluated across different settings: total visits (hospital emergency room and community clinic), emergency room visits only, and community clinic visits only.
Patients with eosinopenia had a significantly higher total rate of healthcare visits per patient in the first year of follow-up compared to the normal BEC group (0.58 ± 1.26 vs 0.3 ± 0.86, p = .01), and a non-significantly higher rate compared to the eosinophilia group (0.58 ± 1.26 vs 0.32 ± 0.84, p = .1).
Patients with eosinopenia also had a significantly higher mean rate of emergency room visits due to bronchiectasis exacerbations compared to the normal BEC group (0.34 ± 0.99 vs 0.14 ± 0.54, p = .02), and a non-significantly higher rate compared to the eosinophilia group (0.34 ± 0.99 vs 0.12 ± 0.48, p = .7). Additionally, patients with eosinopenia had a non-significantly higher mean rate of community clinic visits due to bronchiectasis exacerbations compared to both the normal BEC group (0.24 ± 0.71 vs 0.16 ± 0.60, p = .2) and the eosinophilia group (0.24 ± 0.71 vs 0.21 ± 0.60, p = .9).
Survival analysis
On univariate analysis, the following variables were significantly associated with decreased survival: BEC <100 cells/µl (HR 1.57, 95% CI 1.03–2.39, p = .006), lower WBC (HR 1.03, 95% CI 1.01–1.04, p < .001), lower neutrophil count (HR 1.12, 95% CI 1.05–1.19, p < .001), and lower monocyte count (HR 3.78, 95% CI 2.03–7.05, p < .001).
The following variables were significantly associated with increased survival: higher albumin level (HR 0.31, 95% CI 0.22–0.44, p < .001), higher hemoglobin level (HR 0.76, 95% CI 0.69–0.85, p < .001), higher FVC (in liters; HR 0.37, 95% CI 0.27–0.49, p < .001), higher FEV1 (in liters’ HR 0.27, 95% CI 0.18–0.39, p < .001), and higher TLC (HR 0.96, 95% CI 0.95–0.98, p < .001).
Figure 2 shows the Kaplan-Meier survival analysis of the three BEC categories. Patients with BEC <100 had the highest mortality rate (p = .007). Kaplan-Meier survival analysis of the three BEC categories.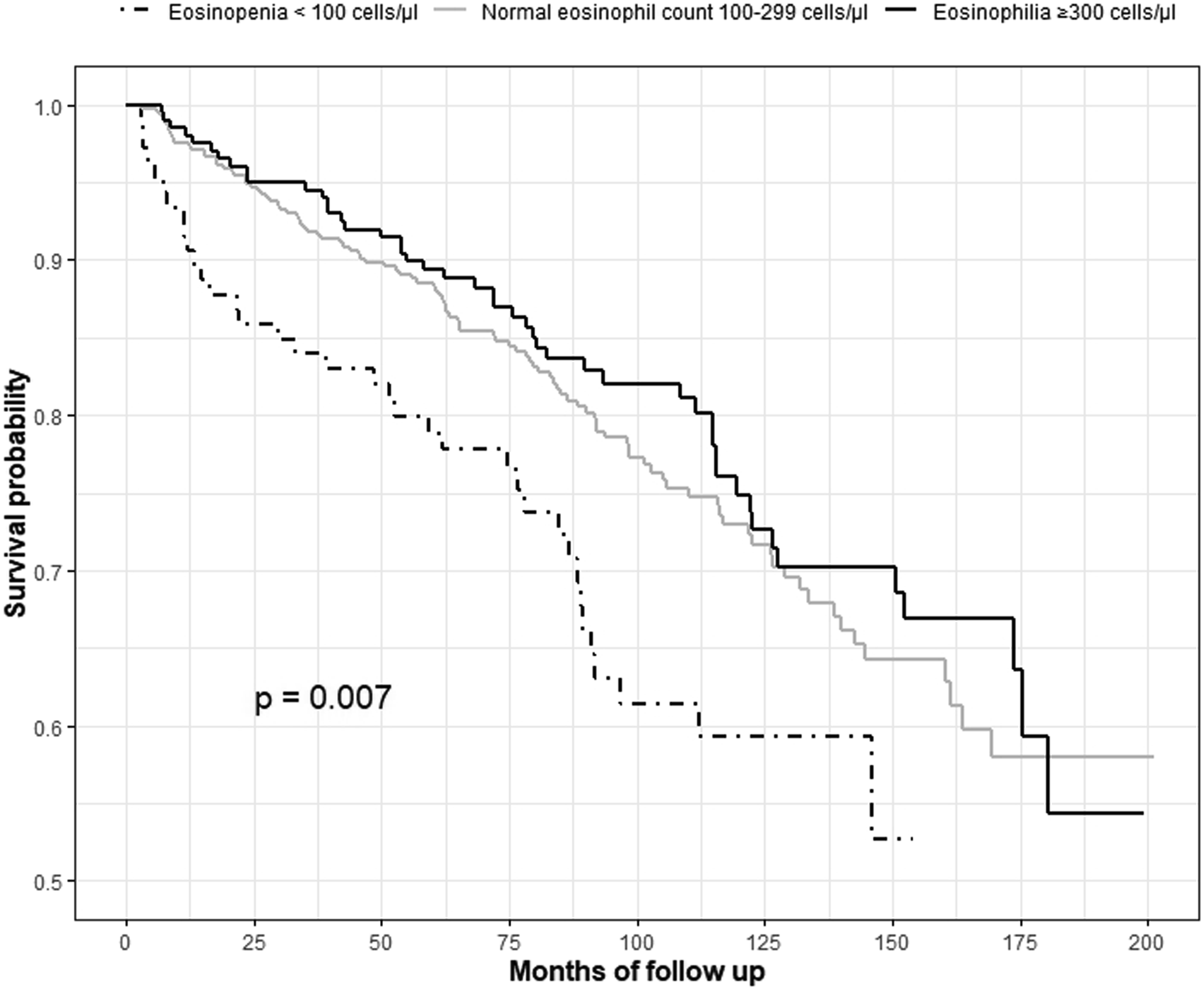
Multivariate analysis
On re-evaluation of BEC level, gender, age, albumin, FEV1 (liters) and FVC (liters) by multivariate logistic regression, BEC (HR 0.23, 95% CI 0.05–0.96, p = .04) and male gender (HR 1.93, 95% CI 1.11–3.34, p = .01) were the variables most significantly associated with decreased and increased survival, respectively.
BAL culture analysis
Data of positive BAL cultures of patients with bronchiectasis.

Data are presented as numbers (percentages) of presented cases. BAL, bronchoalveolar lavage; BEC, blood eosinophil counts; S. aureus, Staphylococcus aureus; S. pneumoniae, Streptococcus pneumoniae.
Discussion
This study aimed to explore the impact of eosinopenia on clinical outcomes in patients with bronchiectasis. Our findings reveal that eosinopenia is associated with a significantly higher risk of hospitalization due to exacerbations and increased mortality. Specifically, patients with eosinopenia had a higher hazard ratio for time to first hospitalization compared to those with a normal eosinophil count, indicating a more severe disease phenotype. Additionally, the mortality rate was notably higher in patients with eosinopenia, with a hazard ratio of 2.15 for increased mortality. These results suggest that eosinopenia may serve as a novel biomarker for adverse outcomes in bronchiectasis, highlighting its potential role in identifying patients at greater risk of severe disease progression and poor prognosis.
In our cohort of bronchiectasis patients, 14.7% (n = 107) exhibited eosinopenia, a rate notably lower than the 27.7% reported by Shoemark et al. in a European multicohort study and the 31% observed in the Spanish Bronchiectasis Registry. The discrepancy in eosinopenia rates across these studies may be attributed to geographic and ethnic variations among the patient populations, which could influence reference laboratory values. For instance, studies have demonstrated that African populations tend to have higher eosinophil counts compared to Caucasian populations, potentially due to genetic factors and environmental exposures such as parasitic infections and allergens, which are more prevalent in certain regions. 10 Moreover, racial and ethnic differences, as seen in the eligibility for asthma biologics among pediatric populations, highlight the importance of considering such factors when interpreting laboratory results across diverse populations. 11 These differences underline the need for region- and ethnicity-specific reference intervals, especially in conditions where eosinophil counts are clinically relevant. Additionally, technical factors, such as storage conditions and the time elapsed from collection to analysis, are also thought to contribute to eosinophil count variability, although they have not been thoroughly investigated. 12 Furthermore, our study included patients on immunosuppressive agents and inhaled corticosteroids, known to reduce eosinophil levels, which might have contributed to the lower rate observed. Notably, our literature review did not reveal any studies comparing BEC in bronchiectasis patients to healthy individuals with similar baseline characteristics, highlighting a gap in current research.
We did not find statistically significant differences in neutrophil levels among the three groups, although there was a general trend of an increase in neutrophils with a decrease in BEC. These results are consistent with the study of Martinez Gracia et al., 6 suggesting that bronchiectasis is not exclusively a neutrophil-associated condition and that eosinophil levels may also have a considerable impact.
The patients with eosinopenia had a shorter time to first hospitalization due to bronchiectais exacerbations in the first year of follow-up. Accordingly, the Spanish Bronchiectasis Registry study reported that the eosinopenic group experienced both a higher number and greater severity of exacerbations, as well as a higher bronchiectasis severity index. 6 These findings are also supported by a study of phenotypic clustering of bronchiectasis wherein patients with a lower eosinophil count had more severe disease, worse lung function, and higher inflammatory parameters. 5 The European multicohort study 4 found that BEC <100 cells/µl was associated with higher bronchiectasis severity, although no clear relationship with exacerbation rates was established. Hence, we may conclude that a low BEC can be used as a biomarker of future disease behavior. This is a novel and relevant finding that warrants further attention in future studies.
In our cohort, patients with eosinopenia had a higher mortality rate, similar to the results of the European multicohort study showing that a high BEC improves survival. 4 This finding is consistent with the higher mortality associated with a relatively low BEC reported in patients with chronic obstructive pulmonary disease. 13 Evidence of the impact of eosinopenia in bronchiectasis on mortality is still scarce, and further studies are needed to corroborate this novel finding.
There are several mechanisms that might be involved in the pathophysiology linking eosinopenia with mortality. Eosinophils exert bactericidal effects via phagocytosis; they secrete eosinophil granule proteins (e.g., eosinophil cationic protein) and produce extracellular mitochondrial DNA to kill Gram-negative bacteria. 14 Some eosinophil granule proteins are also able to counteract viral infections, shaping adaptive host antiviral responses. 14 Interestingly, eosinopenia has been identified as an adverse prognostic indicator in chronic obstructive pulmonary disease (COPD), as reflected in the Dyspnoea, Eosinopenia, Consolidation, Acidaemia and atrial Fibrillation (DECAF) score, which incorporates eosinopenia as a marker of disease severity and outcome. 15 This highlights the broader implications of eosinopenia in respiratory conditions and emphasizes the need to consider its impact on disease outcomes in our discussion.
We did not find any statistically significant differences in BAL culture results among the BEC groups. However, patients with eosinopenia showed a higher culture positivity for Haemophilus sp. compared to those with normal BEC and eosinophilia. Although the difference in positivity was not statistically significant and not markedly higher than in the other groups, it warrants further investigation. The European multicohort study also observed that Haemophilus sp. was prominent in the cluster with BEC <100 cells/µl. 4 The relationship between BEC and pathogens has not been extensively studied, and further research is needed to explore this connection.
The main strengths of our study are the large number of patients as well as their thorough demographic characterization. The data represent the real-world experience that includes patients with various comorbidities using their own medications.
The limitations of this study include its retrospective, single-center design, which may limit the generalizability of the findings. Secondly, the cohort may have included patients with specific comorbidities or those on medications, such as corticosteroids, known to influence BEC. Notably, a higher proportion of patients in the low BEC group were on corticosteroids, which could have acted as a confounder, potentially contributing to both lower BEC and poorer clinical outcomes. This possibility should be considered when interpreting the association between low BEC and clinical outcomes. Thirdly, the absence of a disease severity score represents another limitation, as disease severity may have acted as an unmeasured confounder influencing the observed associations. Fourthly, the lack of a control group prevents direct comparison of BEC and comorbidities in patients with bronchiectasis. Finally, while the database captures the majority of exacerbations requiring medical attention, it is possible that some minor exacerbations managed without referral to community or private clinics were not recorded, potentially leading to underreporting
Conclusions
The patients with bronchiectasis and eosinopenia were characterized by a shorter time to hospitalization due to disease exacerbation and a higher mortality rate. Further studies are needed on the potential impact of eosinopenia on clinical outcome to enhance our understanding of the pathophysiology of bronchiectasis and guide future therapeutic approaches.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
