Abstract
Objectives
Physical activity is reduced in patients with interstitial lung disease (ILD) and physical inactivity is related to poor health outcomes. We investigated the effect of a telecoaching intervention to improve physical activity in patients with ILD.
Methods
Eighty patients with ILD were randomized into the intervention or control group. Patients in the intervention group received a 12-week telecoaching program including a step counter, a patient-tailored smartphone application, and coaching calls. Patients in the control group received usual care. Physical activity (primary outcome), physical fitness and quality of life were measured at baseline and 12 weeks later with an accelerometer, 6-min walking test and quadriceps muscle force and the King’s Brief Interstitial Lung Disease questionnaire (K-BILD).
Results
Participation in telecoaching did not improve physical activity: between-group differences for step count: 386 ± 590 steps/day, p = .52; sedentary time: 4 ± 18 min/day, p = .81; movement intensity: 0.04 ± 0.05 m/s2, p = .45). Between-group differences for the 6-min walking test, quadriceps muscle force and K-BILD were 14 ± 10 m, p = .16; 2 ± 3% predicted, p = .61; 0.8 ± 1.7 points, p = .62 respectively.
Conclusions
Twelve weeks of telecoaching did not improve physical activity, physical fitness or quality of life in patients with ILD. Future physical or behavioural interventions are needed for these patients to improve physical activity.
Introduction
Physical activity (PA) is important to maintain health in the healthy as well as chronic diseased populations. 1 Patients with interstitial lung diseases (ILD) take on average 4000 to 9000 steps per day, depending on their pathology.2–5 This corresponds to 30–80% of the activity level of healthy individuals. When followed up for 3 years, patients with idiopathic pulmonary fibrosis (IPF) showed a 50% reduction in PA 6 and low PA was associated with less quality of life, more symptoms of fatigue 7 and probably excess mortality risk in patients with ILD. 8
While pulmonary rehabilitation results in clinical benefits, 9 these translate poorly into increased PA in patients with respiratory diseases such as ILD.10–14 Interventions to improve PA are currently lacking in ILD.
Behavioural coaching aims to support individuals to increase motivation and promote positive changes in behaviour, in this context PA improvement. The COM-B conceptual model considers Capability, Opportunity, and Motivation as key components influencing behaviour. These components are used in comprehensive telecoaching for example by including an intake interview, semiautomated algorithm-based coaching and, if needed, supportive coaching calls. Such programs are successful in Chronic Obstructive Pulmonary Disease (COPD) in enhancing PA. 15 The principles and theoretical framework are disease-agnostic and have also been shown to be effective in patients with type 2 Diabetes Mellitus, cardiovascular diseases and overweight and obese adults.16–18 Since PA levels and PA patterns of patients with ILD and COPD are similar, 5 this type of intervention with limited in-person contact with a healthcare provider may be feasible for patients living remote from the centre. We speculated it may also be successful in enhancing PA in patients with ILD. However, as these patients do have a different disease progression trajectory, such a hypothesis needs to be tested. An important benefit of telecoaching in ILD, a disease with a low prevalence where specialized care is offered in reference centres that are geographically more remote from the patient’s residence, is the remote approach, which does not require much face-to-face contact. 19
To the best of our knowledge, (tele)coaching interventions to improve PA have not been investigated yet in patients with ILD. We hypothesize that a remote telecoaching intervention with the emphasis on self-management of PA with encouragement could increase PA in patients with ILD compared to usual care.
Methods
Study population and design
The study consisted of twin pilot trials conducted in Leuven, Belgium and Olomouc, Czech Republic of which data were pooled. Both were set up as a parallel-group randomized controlled trial registered at Clinicaltrials.gov (NCT04138173, NCT05147038). The studies were approved by both local ethical committees (UZ/KU Leuven (s62505) and Ethics Committee of the Faculty of Physical Culture of Palacký University Olomouc (80/2020)). Reporting procedures were in accordance with guidelines for reporting RCTs. 20 All patients provided written informed consent. Recruitment took place at University Hospitals Leuven, Belgium, and University Hospital of Olomouc, Czech Republic, between January 2020 and August 2022, using a consecutive sampling strategy: patients who attended a consultation with the chest physician and met the study’s eligibility criteria were informed about the study. Patients with a diagnosis of ILD according to the ATS/ERS statement, 21 with a stable medical condition (i.e. no infection or change in medication 4 weeks before inclusion), ≥18 years old, a diffusing capacity for carbon monoxide (DLCO) ≥ 30% predicted and able to work with a smartphone application were included. Patients were excluded if they were on the waiting list for lung transplantation, if they had a life expectancy of less than 12 weeks or any extrapulmonary conditions interfering with PA. None of the patients followed pulmonary rehabilitation during the intervention period.
A randomization visit took place at baseline, followed by a final visit after 12 weeks. After baseline assessments, patients were randomized into the intervention (IG) or control group (CG) (1:1 allocation). Stratification was performed per centre for (1) diagnosis (IPF or not) and (2) baseline six-minute walking distance (6MWD)< or ≥450 m. The rationale was that (1) IPF is known to have a faster disease progression and (2) patients with COPD showed a better response to PA coaching with a preserved exercise tolerance (i.e. 6MWD ≥450 m). 22 Randomization was performed using computer-generated block randomization with blocks of 4 and 6 (Sealed envelope, London, UK, https://www.sealedenvelope.com) and sequentially numbered opaque sealed envelopes, prepared by an independent investigator. Due to the nature of the study, neither the investigator nor the patient could be blinded to group allocation. At the follow-up visit, all patients were asked about the occurrence of adverse events during the 12-week period.
Intervention
Patients of both groups were informed about the importance of PA and PA recommendations provided by the World Health Organization for patients with chronic conditions in a one-to-one discussion of 5 min. The usual medical care was continued in both groups and adjusted as needed by their chest physician blinded to the study group allocation.
Patients in the IG received 12 weeks of semi-automated telecoaching in addition to usual care. This telecoaching intervention was an adapted version of an intervention previously used successfully in patients with COPD 22 and included the following components (see detailed description in supplemental material): (1) a motivational face-to-face interview (30–45 min) with the patient discussing PA at baseline; (2) a step counter (Fitbit Inspire, Fitbit, Inc., San Francisco, USA) connected with a patient-tailored smartphone application (mPAC). The app provided an individualized and weekly changing step goal based on a proprietary algorithm, based on the patient’s weekly performance; and (3) biweekly supportive coaching calls with the patient throughout the study when insufficient PA progress was noted. When technical issues or changes in medication were reported patients were contacted as well. The coaches had a background of MSc in physiotherapy and had experience in pulmonary rehabilitation, in patients with ILD. On the side of the investigator, a back-end dashboard was developed to monitor the patients effectively with a ‘flagging system’ allowing for biweekly calls in case patients were not engaging in the agreed number of step counts, or in case of technical problems. The intervention is supported by the COM-B model of Michie and colleagues, as described in the study of Loeckx et al.23,24 Capability (i.e. the physical and mental capacity to engage with the behaviour) was addressed by including patients with a diffusion capacity of at least 30% of the predicted value, to ensure that PA was still possible. The six-minute walking distance indicated individual physical capability. Opportunities were addressed during the interview at baseline, where barriers and facilitators for PA in the physical and social environment were explored and an individual action plan was set up. Additionally, motivation for PA and the underlying reasons for engaging in PA were assessed. This was accompanied by the education session, but also by supporting calls and automated feedback in the application throughout the intervention to enhance and sustain motivation levels for PA.
Consistency in the interventions between the two sites was achieved by a careful translation of the application, standardization of operational procedures and regular collaborative discussions between investigators throughout the study.
Physical activity
Physical activity was measured using a triaxial accelerometer Dynaport Movemonitor (DAM) (McRoberts BV, The Hague, the Netherlands), a validated device in patients with COPD. 25 Following the recommendations from an International Task Force on PA in COPD, a patient population with a comparable PA pattern, 5 patients were asked to wear the DAM for seven consecutive days during waking hours at both timepoints. 26 Measurements of at least four weekdays with a minimum of 8 h of wearing time were considered valid and were included in the statistical analysis. 26 The primary outcome was the change in mean daily step count 12 weeks after randomization in the IG compared to the CG, for which the investigator was blinded. The between-group differences after 12 weeks in other PA outcomes were retrieved as secondary outcomes. A PA responder was considered as an increase of 1000 steps from baseline measurement, based on the minimally important difference for COPD. 27
Other assessments
At baseline, patients performed a complete pulmonary function test according to the ATS/ERS guidelines.28–30 Exercise tolerance was measured at both timepoints, as the best out of two 6-min walk tests following ATS/ERS recommendations, 31 as well as isometric Quadriceps force (QF), measured as the best out of four maximum voluntary isometric contractions of the Quadriceps muscle in a fixed position of 90° hip and 60° knee flexion (Biodex System II, Biodex Corporation, New York, USA). Data mentioned above were expressed as a percentage of predicted normal values.31–33 At both visits, health-related quality of life was assessed by a disease-specific questionnaire and a general questionnaire, respectively the King’s Brief Interstitial Lung Disease (K-BILD) and the 36-Item Short Form Survey Instrument (SF-36). At both visits, fatigue was assessed using the Multidimensional Assessment of Fatigue (MAF). For all patients, the importance, motivation and self-efficacy in improving PA from the patient’s perspective were assessed with the investigator during the baseline visit prior to the randomization. These questions were scored on a 10-point Likert Scale ranging from not important (1) to very important (10). Information and duration of coaching calls were collected (see supplemental material). The patient’s experience with the delivered intervention was investigated at the follow-up visit with a study-tailored questionnaire in Belgian patients (see supplemental material).
Statistics
We present the pooled analysis of the studies in Belgium and the Czech Republic. The data in both centres separately are presented in the supplemental material. Since we analysed the twin pilot studies together, no a priori sample size calculation was performed. With the achieved patient number and the standard deviation of daily steps in both groups, our sample size would have 7% power to detect a between-group difference of 386 steps per day (detailed calculation in supplemental material). Normality of baseline characteristics was assessed with the Shapiro-Wilk test. The characteristics of the intervention and control group were compared using an unpaired t-test or a Chi2 test. Mixed model analyses were performed including centre and randomization group as independent class variables. The interaction effect of the randomization group and visit was derived as main outcome. Daylight time, a proxy for seasonality, was considered as a confounder in the PA models, 26 as well as stratification variables and centre. The percentage of responders in both groups was compared with a Chi2 test. As an exploratory pre-planned analysis, the intervention effect was compared in subgroup analyses for diagnosis (IPF or other restrictive diseases) and baseline exercise tolerance (6MWD> or <450 m), using similar mixed models. A sensitivity analysis, including all PA data (valid and invalid), was performed using similar mixed models. Adherence to the use of the step counter was calculated as a percentage of days with at least 70 steps, a cut off previously used in our research group since wearing time is not available for Fitbit devices. 34 The patients’ experience with the intervention was reported descriptively. Statistical significance was set at p < .05. Data are presented as mean ± SD in the manuscript unless indicated otherwise. All statistical analyses were performed using SAS statistical package (V.9.4, SAS Institute Inc., Cary, N.C., USA).
Results
Patient population
Eighty patients with ILD were randomized to participate in the IG (n = 37) or to the CG (n = 43) (Figure 1). After 12 weeks, data from 36 patients in the IG and 41 patients in the CG were included in the analyses. Three patients were lost during follow-up due to refusal to continue participation, urgent lung transplant surgery or death. Study flowchart.
Baseline characteristics.
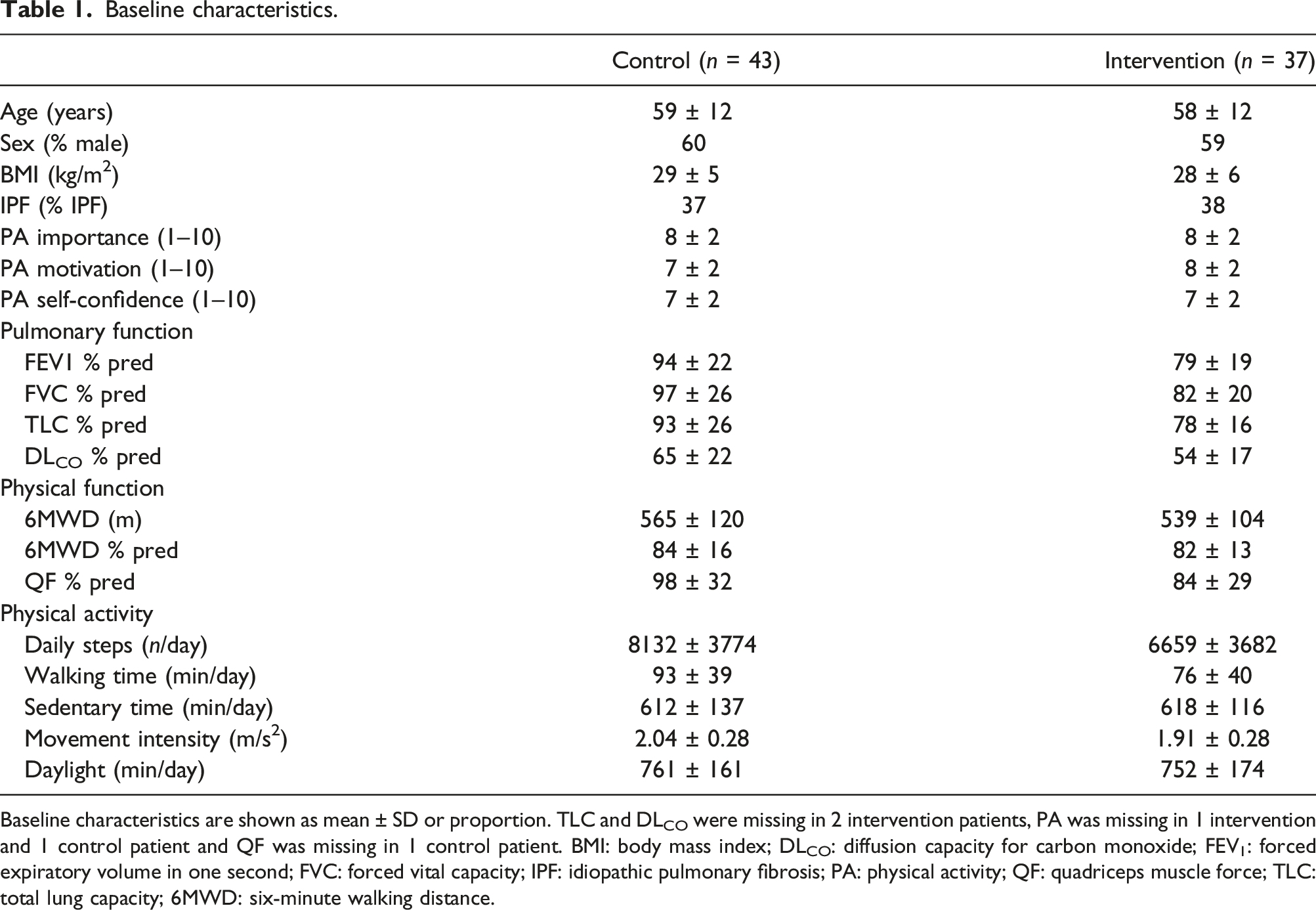
Baseline characteristics are shown as mean ± SD or proportion. TLC and DLCO were missing in 2 intervention patients, PA was missing in 1 intervention and 1 control patient and QF was missing in 1 control patient. BMI: body mass index; DLCO: diffusion capacity for carbon monoxide; FEV1: forced expiratory volume in one second; FVC: forced vital capacity; IPF: idiopathic pulmonary fibrosis; PA: physical activity; QF: quadriceps muscle force; TLC: total lung capacity; 6MWD: six-minute walking distance.
At baseline and after 12 weeks, a total of 78 and 76 valid PA measurements were analysed. Some measurements could not be included in the analyses due to incorrect positioning of the accelerometer (n = 2) and technical problems (n = 1). The accelerometer was worn 4.9 ± 0.3 week days at baseline (n = 78) with a mean wearing time of 908 ± 94 min per day, which was similar at the follow-up visit. Also, change in daylight was similar in both groups (IG: Δ−59 ± 285 and CG: Δ−91 ± 264 min/day).
Physical activity
At 12 weeks, no differences in PA outcomes were observed between the IG and CG (Figure 2). In the IG, 38% of patients increased step counts by >1000 steps, compared to 23% in the CG (p = .19). Subgroup analyses stratified for diagnosis and exercise capacity revealed no differences between groups (see supplemental material). The sensitivity analysis for the main outcome rendered similar results. An overview of daily steps, measured with the step counter in the IG, is shown in Figure 3. Only small changes in steps can be observed over time. Evolution of daily physical activity measured with the dynaport movemonitor at baseline and 12 weeks, shown in mean ± SEM. Changes in physical activity within each group are presented as solid lines (intervention group) and dotted lines (control group). Between-group differences in physical activity are shown as mean ± SEM with corresponding p-value. Overview of the overall mean ± SD daily step count per week measured with fitbit inspire in the intervention group (n = 36 patients). The first week (dot) is the mean ± SD steps of 5 days baseline measurement to calculate the initial step goal. The following dots are weeks with an individualized step goal.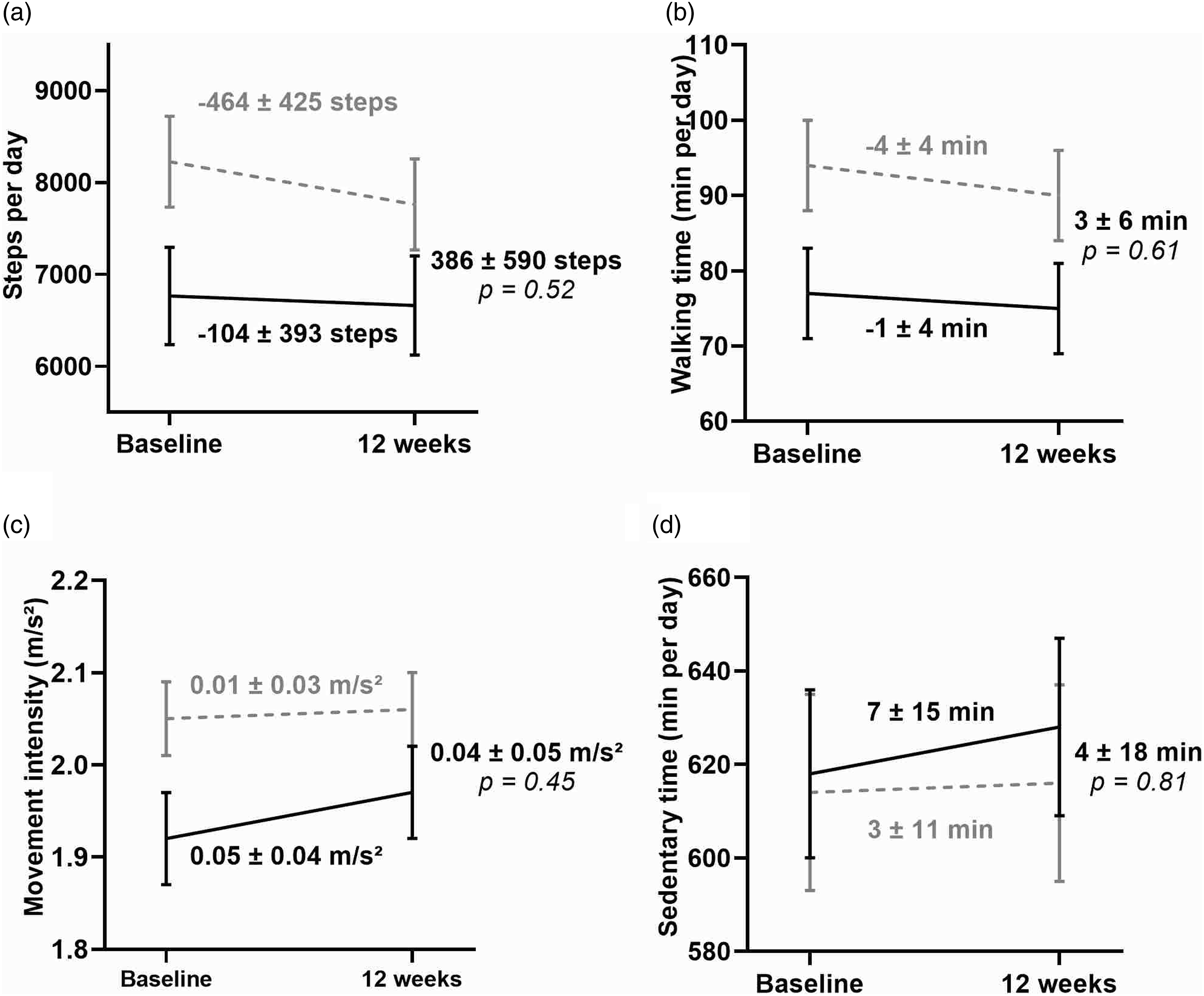

Secondary outcomes and adverse events
Secondary outcome measures at baseline, within-group differences after 12 weeks and between-group differences after 12 weeks.
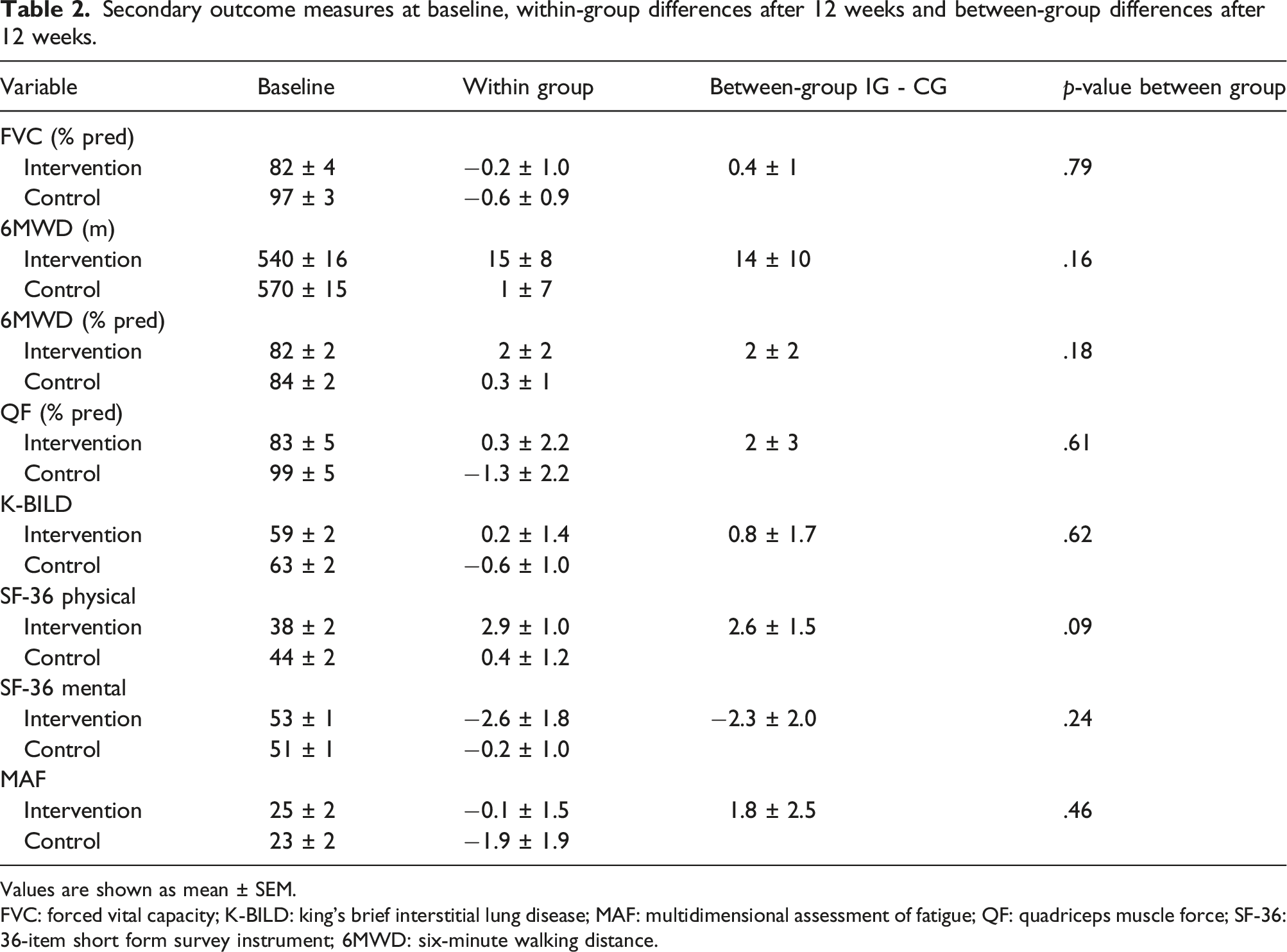
Values are shown as mean ± SEM.
FVC: forced vital capacity; K-BILD: king’s brief interstitial lung disease; MAF: multidimensional assessment of fatigue; QF: quadriceps muscle force; SF-36: 36-item short form survey instrument; 6MWD: six-minute walking distance.
Interactions with the coach
Over 12 weeks, each patient had 6 ± 3 telephone calls with a mean contact time of 7 ± 6 min per call, resulting in a total contact time of 41 ± 34 min. Per patient, a mean of three calls was related to PA coaching, with a mean duration of 6 ± 3 min per call. Other calls were related to technical or health problems. Adherence to step counter use was high in both Belgian and Czech patients, with respectively 99.6% and 93% days of using the device.
Patients’ experience (Belgian patients)
Almost all Belgian patients (94%) in the IG found the intervention encouraging to become more active. 70 percent of the patients who followed the intervention considered themselves more active afterwards, compared to only 45% in the CG (p = .19). All (100%) patients were eager to continue using the step counter after the intervention and 63% indicated that they would like to continue using this in combination with the coaching application. The usefulness of the coaching calls was scored 8.4 out of 10 and 61% of the patients would like to keep receiving the coaching calls in combination with the step counter. The effect of the corona pandemic on spending less time outdoors (scale 1 (no impact) to 10 (very high impact)) was scored similarly between both groups, that is 5 ± 3 (IG) and 4 ± 2 (CG) (p = .3129).
Discussion
This twin trial, conducted in two countries, shows that PA in patients with ILD did not improve by providing patients a 12-week telecoaching program consisting of a step counter and smartphone application as compared to usual care. Exercise tolerance, quality of life and symptoms did not change following this program. Patients with ILD showed no intervention effect regardless the exercise tolerance at baseline or the presence of IPF.
Since no previous research on PA telecoaching using step counters, is available in patients with ILD, we can only compare to interventions in other diseases. In breast cancer survivors, integrating step counters with motivational interviewing or counselling sessions, whether delivered in person or remotely, showed significant positive effects on PA. 36 Also in overweight or obese adults, the integration of behavioural change techniques was crucial for both short and long-term success, while motivational interviewing only influenced long-term outcomes. In contrast, in individuals with high cardiovascular risk, the combination of a step counter and intensive in-person motivational interviewing did not lead to PA improvements compared to usual care. Collectively, these studies yielded mixed results and it is difficult to make suggestions for potential adaptations to the present intervention, particularly as in its current format it did improve PA in COPD. In COPD, PA improved most in those patients with a better exercise tolerance and higher PA level at baseline.15,22 Since PA characteristics of patients with COPD and ILD are comparable when matched for exercise capacity, we hypothesized that patients with ILD would have experienced a similar PA improvement. 5 Although the baseline PA level (6659 ± 3682 steps in IG and 8132 ± 3774 steps in CG) and exercise tolerance of our patients (6MWD > 500 m) showed promise in improving PA with tele-coaching, our study did not demonstrate a significant improvement in PA levels. Unfortunately, while relatively large, the present study is still underpowered and sub-analyses should be taken with caution as there is the risk of a Type 2 error. The patients with ILD in the current study started at a higher PA level than patients with COPD in the RCT of Demeyer et al. 22 Also, the lower correlation between PA and exercise tolerance in patients with ILD compared to those with COPD suggests that other factors may influence PA significantly in ILD. However, our subgroup analyses were unable to identify responders and non-responders investigating exercise tolerance and disease type.5,7 Although PA levels are similar between patient populations, the underlying pathology and attitudes towards progression are different, potentially requiring different treatment approaches. Possibly, these patients could benefit from a supervised pulmonary rehabilitation program to increase their self-efficacy regarding PA. Since this kind of program does not enhance PA, we can speculate that the addition of a coaching program using an application could support engaging in PA after a PR program. The interaction between pulmonary rehabilitation and telecoaching remains to be studied.
Strengths and limitations
An important strength of the present study is that it is the result of twin studies conducted in two countries, ensuring external validity of the results despite slight differences in the study population characteristics. Secondly, PA was measured with a valid accelerometer and data collection and processing followed recently published recommendations by an International Task Force on PA.25,26 Third, the intervention contained the most frequently used behaviour change techniques for PA improvement in patients with cardio-pulmonary diseases (education about health consequences, goal setting, self-monitoring, feedback on behaviour, and social support). 17 Lastly, the application used in the intervention of Demeyer and colleagues was slightly modified based on feedback from patients and coaches obtained in the study of Loeckx et al. 34 This led to better adherence in obtaining daily step data in both sites. Remarkably, although there was no intervention effect, Belgian patients enjoyed the intervention and self-reported to be more active. This aspect was not investigated in Czech participants. Some limitations should be considered when interpreting the present results. First, these randomized controlled pilot trials were not a priori powered. Whereas this is a limitation, the collective sample of 80 patients is, in the view of the present patient population, large. We hypothesize a larger sample would not alter the results significantly with the current intervention since the effect size of the intervention was rather small (Cohen’s d of 0.10). Second, the COVID-19 pandemic occurred at the beginning of this trial, making it difficult to complete all tests during periods of lockdowns. Also, our patients indicated that the pandemic had a major impact on their daily lives and caused them to spend less time outdoors than usual. PA levels showed to be reduced during the COVID-19 pandemic in patients, as well as in the general population. 37 As a consequence of the extended recruitment period, patients experienced different pandemic-related restrictions during the 3-month intervention. We can only speculate that this may have affected their PA behaviour.
While this semi-automated intervention does not have sufficient statistical power to conclude its effectiveness in ILD, there is a need to improve the intervention for it to serve a larger proportion of patients and to select the most appropriate patients for the intervention. Importantly, the specific barriers to increase PA in this group should be identified. Possible factors include symptoms (dyspnea, fatigue, chronic cough) or coping with disease progression. This study showed that this patient group is able to cope with remote interventions and PA monitoring, which could form the basis for a new intervention. Interventions that are more directly supervised or integrate more formal proposals for exercise may be more effective. Such interventions have recently been proposed for patients with COPD. 38
Conclusion
This randomized controlled study conducted in two countries suggests that physical activity, physical fitness, quality of life and symptoms in patients with ILD did not improve using the present wearable-driven telecoaching program, including a step counter, a smartphone application and coaching calls.
Supplemental Material
Supplemental Material - Physical activity coaching in patients with interstitial lung diseases: A randomized controlled trial
Supplemental Material for Physical activity coaching in patients with interstitial lung diseases: A randomized controlled trial by Sofie Breuls, Tamara Zlamalova, Katerina Raisova, Astrid Blondeel, Marieke Wuyts, Martin Dvoracek, Monika Zurkova, Jonas Yserbyt, Wim Janssens, Wim Wuyts, Thierry Troosters and Heleen Demeyer in Journal of Chronic Respiratory Disease.
Footnotes
Acknowledgements
The authors acknowledge the assistance of the clinical trial unit and clinical & clinical trial teams of the University Hospitals Leuven, Belgium, and University Hospital of Olomouc, Czech Republic and would like to thank all patients who participated in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Leuven cohort received an investigator-initiated and unrestricted grant from Boehringer Ingelheim #1199-0373. Astrid Blondeel was a pre-doctoral research fellow of the Flemish Research Foundation (FWO-Flanders, #1194322N) during the study period. Wim Janssens is supported by Flemish Research Foundation (FWO-Flanders). Wim Wuyts has received research grants from Boehringer Ingelheim, Roche and Galapagos. Thierry Troosters is supported by the Research Foundation-Flanders (FWO-Flanders, #G0C0720N). Heleen Demeyer is a post-doctoral research fellow of the Flemish Research Foundation (FWO Flanders, #12ZW822N). AstraZeneca Czech Republic provided an unrestricted grant for the Czech twin study.
Trial registeration
This trial was registered at ClinicalTrials.gov (https://classic.clinicaltrials.gov/ct2/show/NCT04138173 and ![]() , NCT04138173 and NCT05147038 respectively).
, NCT04138173 and NCT05147038 respectively).
Data availability statement
Data cannot be shared publicly because of patient confidentiality according to the Belgian law. New analyses are available on reasonable request from the author or UZ Leuven ethical committee (
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
