Abstract
Background and objective
Reference values of physical activity to interpret longitudinal changes are not available in patients with idiopathic pulmonary fibrosis (IPF). This study aimed to define the minimal clinical important difference (MCID) of longitudinal changes in physical activity in patients with IPF.
Methods
Using accelerometry, physical activity (steps per day) was measured and compared at baseline and 6-months follow-up in patients with IPF. We calculated MCID of daily step count using multiple anchor-based and distribution-based methods. Forced vital capacity and 6-minute walk distance were applied as anchors in anchor-based methods. Effect size and standard error of measurement were used to calculate MCID in distribution-based methods.
Results
One-hundred and five patients were enrolled in the study (mean age: 68.5 ± 7.5 years). Step count significantly decreased from baseline to 6-months follow-up (−461 ± 2402,
Conclusion
Daily step count significantly declined over 6-months in patients with IPF. MCID calculated by multiple anchor-based and distribution-based methods was 570 to 1358 steps/day. These findings contribute to interpretation of the longitudinal changes of physical activity that will assist its use as a clinical and research outcome in patients with IPF.
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is a progressive disease with a median survival of only 2-3 years, 1 and its clinical course among individual patients is highly variable. 2 IPF is manifested by deterioration in pulmonary function, 3 exercise capacity, 4 physical activity 5 and health-related quality of life (HRQOL). 6 To date, longitudinal studies have shown that decline of these clinical measurements are associated with higher mortality rates in patients with IPF.3,7–9 Therefore, evaluation of longitudinal changes in these clinical outcomes is important for refinement of clinical decision making.
Minimal clinically important difference (MCID) is widely used to interpret longitudinal changes of outcomes clinically and in research trials. It is defined as the smallest difference in a measure that is interpreted to be either beneficial or harmful from the patient’s and/or clinician’s perspective. 10 The MCID helps determine whether a particular intervention or overall management has been effective. 10 Several studies have reported the MCID of pulmonary function, 11 HRQOL, 12 functional exercise capacity13–16 and extent of lung fibrosis 17 in patients with IPF. Although the MCID of physical activity has been demonstrated to be 600–1100 steps/day 18 and 350–1100 steps/day 19 in chronic obstructive pulmonary disease (COPD) patients, this has not yet been determined in IPF patients. Thus, it is difficult for clinicians and researchers to judge whether a patient’s longitudinal change in physical activity is clinically relevant in determining the decline, improvement, and management of their condition.
Common approaches of MCID are anchor-based and distribution-based methods. Anchor-based methods establish the MCID of the variable of interest to be “anchored” to the minimal change of a well-defined clinical variable. In contrast, distribution-based methods use statistical descriptors, including the effect size and standard error, to estimate the MCID. Because distribution-based methods do not integrate comparison to a well-defined clinical variable, anchor-based methods are usually recommended as the primary means to establish estimates of MCID. 20
Accelerometry, the most commonly used motion sensor, can quantify physical activity more accurately than questionnaires. 21 Physical activity questionnaires can be susceptible to recall bias in low-intensity activity for a longer survey period,22,23 as shown by patients with COPD who substantially overestimated their physical activity.21,24,25 Therefore, accelerometry is a more valid measure of physical activity than physical activity questionnaires, especially for outcomes in longitudinal studies. Moreover, step count is deemed to be the most sensitive outcome to detect longitudinal changes 26 and is readily understood by clinicians and patients. A previous investigation 27 reported MCID of physical activity in interstitial lung disease (ILD) patients, however, the study aim was to validate the MCID of an activity questionnaire against accelerometry for moderate to vigorous physical activity and 65% of the sample had an ILD diagnosis besides IPF. Further, there was a lack of consistency of the MCID derived from the anchor-based compared to the distribution-based approaches. Taken together, the estimated MCID in this study has limited applicability to the IPF population over a broader range of exertion that includes lighter physical activity. 27
This study aimed to investigate longitudinal change in physical activity, specifically daily step count measured by accelerometer, and to define the MCID of physical activity using anchor-based and distribution-based methods in patients with IPF.
Methods
Study design
This study was a retrospective chart review of patients with IPF that had physical activity assessed at baseline and a 6-months follow-up from May 2007 to March 2017 at Tosei General Hospital (Aichi, Japan).
Subjects
Inclusion criteria were: (1) diagnosis of IPF made by a multidisciplinary team. 1 Patients diagnosed with IPF between 2007 and 2011 had their diagnosis reconfirmed using the 2011 criteria in September 2016; (2) had undergone evaluation of pulmonary function, arterial blood gases, St George’s Respiratory Questionnaire (SGRQ), 28 modified Medical Research Council (mMRC) dyspnea scale, 29 6-minute walk test (6MWT) and (3) had their physical activity measured at baseline and 6-months follow-up with a minimum accelerometer wear time of 8 h per day for 7 days. 30 Exclusion criteria were: (1) significant comorbidities that limited physical activity (i.e., cardiovascular, musculoskeletal or neurological impairment); (2) undergone any intervention to promote physical activity (i.e., pulmonary rehabilitation, counseling or motivational sessions) during study period or within 6-month prior to study enrollment; (3) initiation of long-term oxygen therapy during the study period.
Ethics approval
This study was approved by the ethics committee of Tosei General Hospital (IRB number: 742) and was conducted in accordance with principles of the Declaration of Helsinki. Written informed consent was waived because this study involved a retrospective chart review of patients’ records. Opt-out methods on the Web site were applied to obtain patient consent.
Data collection
Clinical data were derived from medical records.
Pulmonary function and arterial blood gases
Pulmonary function was measured by spirometry (CEHSTAC-55 V, Chest, Tokyo, Japan) and single-breath diffusion capacity for carbon monoxide (DLco) according to recent guidelines.31,32 Arterial blood gases were sampled at rest breathing room air in the sitting or supine position.
Health-related quality of life, dyspnea and functional exercise capacity
HRQOL was assessed using the SGRQ. 28 Functional limitation due to dyspnea was evaluated using the mMRC dyspnea scale. 29 The 6MWT was performed according to published recomendations, 33 and distance walked (6-min walk distance [6MWD]) was used as a measure of functional exercise capacity.
Physical activity
Physical activity was measured over seven consecutive days using a uniaxial accelerometer (Lifecorder GS, Suzuken, Nagoya, Japan), which has been validated for chronic respiratory disease. 34 Patients were asked to wear the activity monitor on their right waist belts for 24 h/day except for bathing and sleeping, and to maintain usual levels of physical activity. The average value of step count (steps per day) over the seven-day period was used as the measure of physical activity. A minimum of eight hours of wear time (during waking time) on each of seven days was required for data to be used in the analysis. Details of physical activity measurement according to reporting reccomendations35,36 are shown in supplementary table 2.
Calculation of minimal clinical important difference
The MCID of daily step count was established using anchor-based and distribution-based approaches.
Two distribution-based approaches were applied to estimate MCID: (1) 0.3 times the effect size (calculated as the change in step count divided the standard deviation [SD] of baseline step count), as previously described20,40 and (2) 1-standard error of measurement (SEM)40,41 of step count using following equation:
Statistical analysis
Continuous data were expressed as mean and SD. Categorical data were presented as frequencies and percentages. Paired
To determine MCID for anchor-based methods, a correlation coefficient >0.3 between external anchors and outcomes of interest is required.20,40 Therefore, the sample size was calculated to detect a correlation coefficient >0.3 between change in step count and FVC, SGRQ and 6MWD with 0.8 statistical power at the significance level set at 0.05,
42
and it required minimum sample of 89 participants. All analyses were performed using SPSS version 25.0 (IBM, Chicago, USA). Statistical significance was considered at
Results
Baseline characteristics of the 105 patients with IPF.
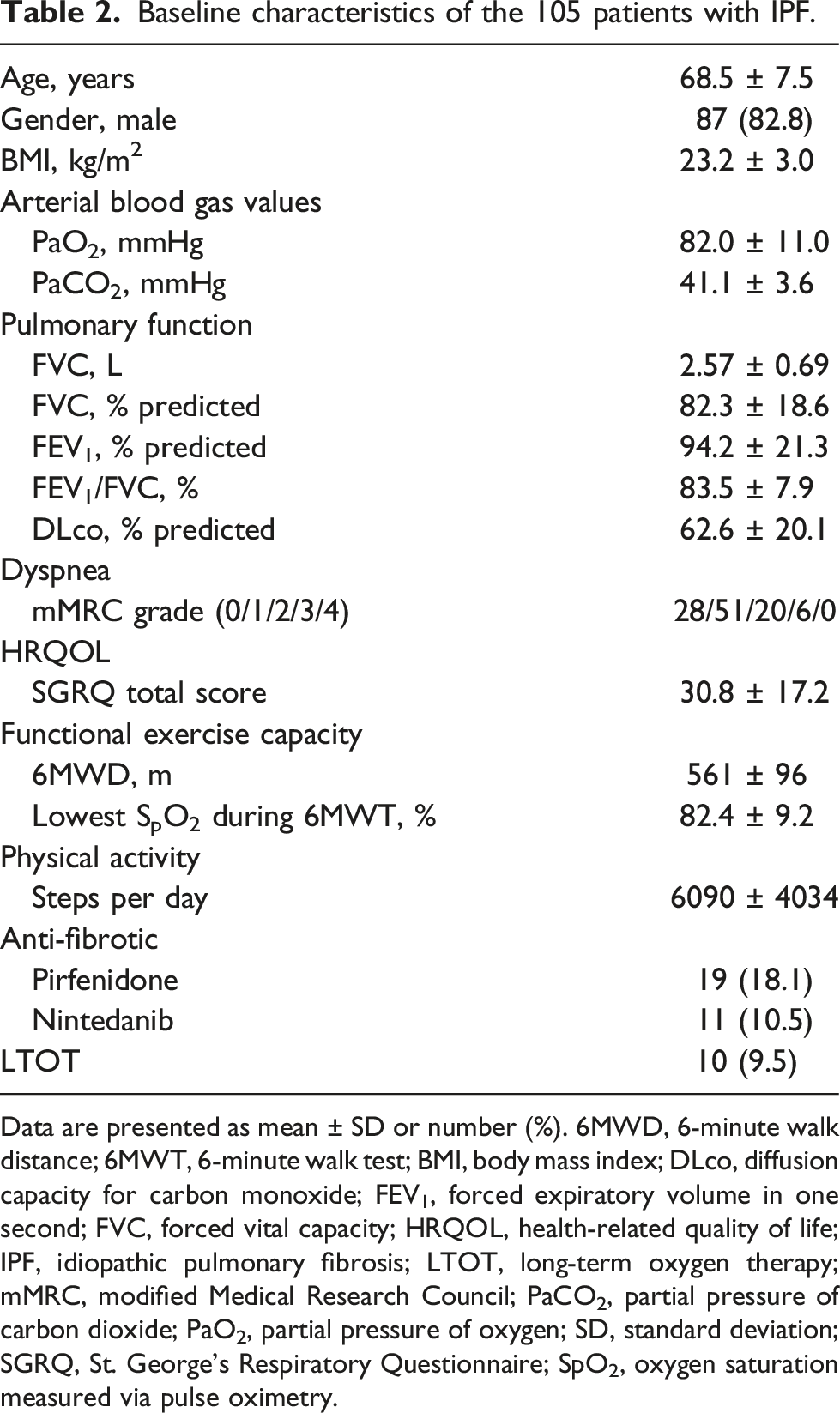
Data are presented as mean ± SD or number (%). 6MWD, 6-minute walk distance; 6MWT, 6-minute walk test; BMI, body mass index; DLco, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; HRQOL, health-related quality of life; IPF, idiopathic pulmonary fibrosis; LTOT, long-term oxygen therapy; mMRC, modified Medical Research Council; PaCO2, partial pressure of carbon dioxide; PaO2, partial pressure of oxygen; SD, standard deviation; SGRQ, St. George’s Respiratory Questionnaire; SpO2, oxygen saturation measured via pulse oximetry.
Longitudinal changes and correlation of potential anchors with physical activity.

Data are presented as mean ± SD. 6MWD, 6-minute walk distance; SD, standard deviation; FVC, forced vital capacity; SGRQ, St. George’s Respiratory Questionnaire.
a
b
cAbsolute change (6-months – baseline).
dRelative change ([6-months – baseline]/baseline * 100).

Scatter plots of change in step count and potential anchors (a), FVC; (b), SGRQ; (c), 6MWD. 6MWD, 6-minute walk distance; FVC, forced vital capacity; SGRQ, St. George’s Respiratory Questionnaire.
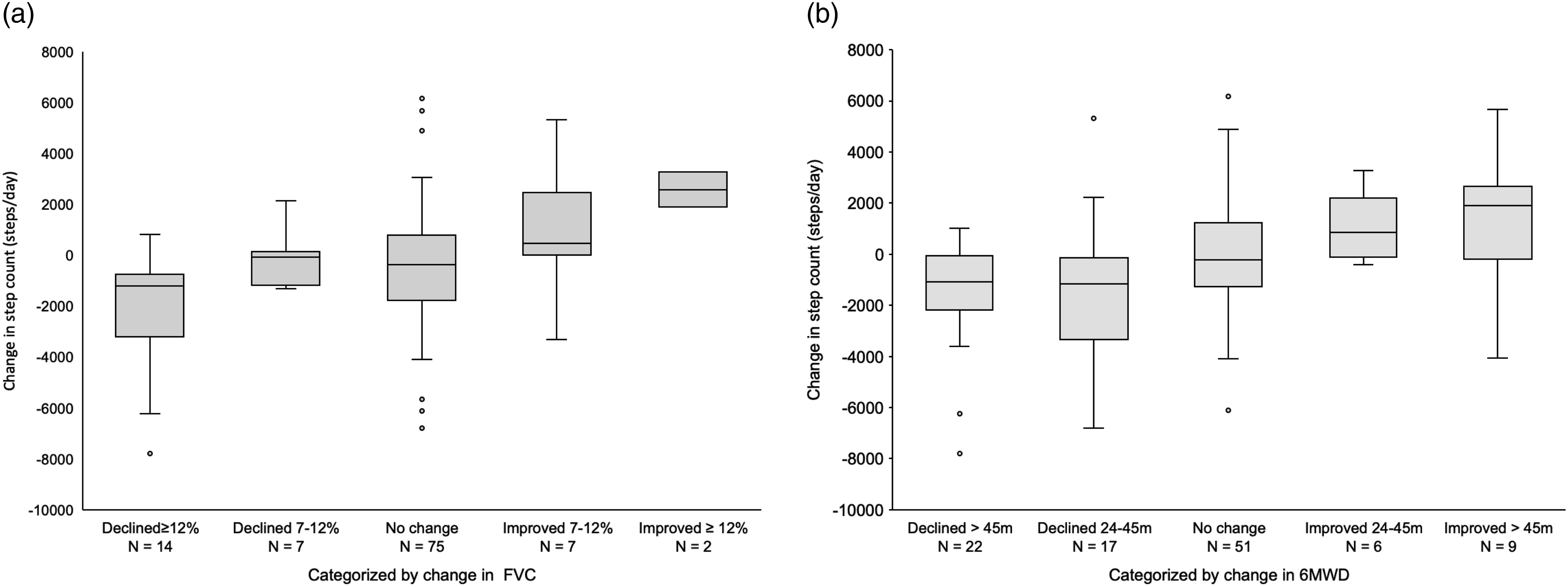
Box plots of change in step count categorized by changes in FVC (a) and 6MWD (b). 6MWD, 6-minute walk distance; FVC, forced vital capacity.
MCID estimates for step count (steps per day).

6MWD, 6-minute walk distance; CI, confidence interval; ES, effect size; FVC, forced vital capacity; MCID, minimal clinical important difference; ROC, receiver operating characteristic; SEM, standard error of measurement.
aWeighted mean of subjects whose anchor changed by the minimal (FVC = 7%–12%, 6MWD = 24–45 m).
bCut-off value to discriminate subjects whose anchor changed over the minimal or not (FVC = 10%, 6MWD = 35 m).
Discussion
We found that daily step count, an easily interpreted assessment of physical activity, significantly declined over 6-months follow-up in patients with IPF. Moreover, anchor-based and distribution-based MCID estimates of physical activity showed decreases in the range of 570 to 1358 steps/day.
We determined three anchor-based methods each using two anchors (FVC and 6MWD), and two distribution-based methods. Anchor-based methods require comparisons with clinically relevant measures and secondly, that these measures show a significant correlation of rho greater than 0.3 with the outcome of interest.20,40 We found that two of our clinically relevant measures, FVC and 6MWD, met these two criteria but SGRQ did not (Table 3). Consequently, FVC and 6MWD were further analysed as external anchors for anchor-based methods (Table 4).
Distribution-based methods, based on statistical variability, are recommended using as supportive information to anchor-based method. 20 Moreover, combination of several anchor-based methods in multiple anchors and various distribution-based methods are important. 20 Therefore, we used multiple anchor-based and distribution-based methods, and they provided a degree of reassurance on its validity. In the results, anchor-based and distribution-based MCID showed similar values, and provide further support of the robustness of MCID value determined in our study.
Two studies have investigated longitudinal declines of physical activity in patients with IPF.7,43 Bahmer et al. 43 reported that daily step count was declined 3017 steps over 3-years follow-up period in 26 patients. Badenes-Bonet et al. 7 observed 784 decline of step count during 12 months in 22 patients. These previously reported changes equate to 6-months step count declines of approximately 500 and 400 steps, respectively, similar to the decrease in our study (mean decrease of 461 steps). Compared to COPD patients, IPF patients appear to demonstrate a greater decline in physical activity as assessed by accelerometry. One report of COPD patients, revealed an annual decline of 451 steps although sit-to-stand and 6MWD remained stable. 44 Teylan et al. 19 demonstrated that after a medical event, COPD patients decreased to a MCID of between 350-1100 steps/day in patients. Our results suggest that longitudinal decline of physical activity in patients with IPF may be twice that shown in COPD 44 – 461 steps in 6-month. Moreover, the range of decline in IPF patients to a MCID of 570-1358 steps in our study appears more consistent with the range shown in COPD patients after a medical event. 19 Considering together that physical activity decline is associated with worse survival in COPD and IPF patients,7,45 judicious evaluation of physical activity may be required to optimize clinical management in IPF patients.
Pulmonary rehabilitation and other interventions to promote physical activity may prove advantageous as reflected by an increase step counts per day following these interventions. Demeyer et al. 18 showed that distribution-based MCID of step count in a sample COPD patients undergoing pulmonary rehabilitation resulted in improvements ranging between 600-1100 steps/day. Of clinical significance, hospital admissions were reduced in patients that showed improvement more than 600 steps. 18 Accordingly, due to the potential greater downward trajectory of step count in IPF patients, interventions to mitigate decreased of physical activity might play more prominent roles in these patients.
To our knowledge, this is the first study to establish MCID of physical activity in patients with IPF. A major strength of this study is that step count is readily understood by clinicians and patients. Secondly, this study examined multiple anchor-based and distribution-based methods to define the MCID that derived similar values. A previous study reported the MCID of physical activity in patients with ILD using distribution-based methods, 27 however, the measure was moderate-to-vigorous physical activity minutes. In addition, the majority of the sample of ILD patients (65%) were not IPF, 27 which may limit the applicable of this MCID to the IPF population.
There were several limitations in this study. First, we did not consider seasonal effects on physical activity, however, data were collected across all seasons over several years. Secondly, because MCID reflected a decline of physical activity, it may not be sensitive to detect the effect of a physical activity intervention. Moreover, participants were not blinded to their step count on the accelerometer, which may have positively influenced their activity. Another consideration was that our sample consisted of patients during a relatively early stage of the IPF trajectory and there was a disproportionate dominance of males, which may limit generalizability of the MCID. Another concern may be the lack of comparability of the step counts obtained by the Lifecorder versus other pedometers but in fact a comparison the six pedometers (including Lifecorder) showed accuracy of ±1% of actual steps 46 and the Lifecorder was accurate even during slow walking speeds. 47
In conclusion, physical activity significantly declined over 6-months in patients with IPF. Additionally, MCID calculated by multiple anchor-based and distribution-based methods was 570 to 1358 steps/day. These findings provide a basis to interpret the longitudinal change of physical activity and to assist with evaluations of clinical and research outcomes in patients with IPF.
Supplemental Material
Supplemental Materials - Physical activity in idiopathic pulmonary fibrosis: Longitudinal change and minimal clinically important difference
Supplemental Materials for Physical activity in idiopathic pulmonary fibrosis: Longitudinal change and minimal clinically important difference by Kazuya Shingai, Toshiaki Matsuda, Yasuhiro Kondoh, Tomoki Kimura, Kensuke Kataoka, Toshiki Yokoyama, Yasuhiko Yamano, Tomoya Ogawa, Fumiko Watanabe, Jun Hirasawa, W. Darlene Reid, and Ryo Kozu in Chronic Respiratory Disease.
Footnotes
Acknowledgments
We thank the study participants and the staff of the Department of Rehabilitation and Respiratory Medicine, Tosei General Hospital, for their helpful. Furthermore, We acknowledge Dr Shuntaro Sato, Clinical Research Center, Nagasaki University Hospital, Nagasaki, Japan, for his valuable advice regarding the statistical analyses.
Declaration Of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yasuhiro Kondoh reports advisory board fees and personal fees from Asahi Kasei Pharma Corp., Shionogi & Co.Ltd., and Boehringer Ingelheim Co., Ltd., advisory board fees from Janssen Pharmaceutical K.K., Healios K.K., Chugai Pharmaceutical Co., Ltd., and Taiho Pharmaceutical Co., Ltd., and personal fees from AstraZeneca K.K., Eisai inc., KYORIN Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma, and Novartis Pharma K.K., outside the presentation work. Kensuke Kataoka and Yasuhiko Yamano received lecture fees from Nippon Boehringer, Ingelheim Co., Ltd., outside the submitted work. All other authors declared no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Research Grant-in-Aid for Encouragement of Scientists (20H01136) and Overseas Challenge Program for Young Researcher from Japan Society for the Promotion of Sciences.
Data Availability Statement
Data are available on reasonable request.
Supplemental material
Supplemental material for this article is available online
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

