Abstract
Objective
The Asthma Quality of Life Questionnaire (AQLQ) and COPD assessment test (CAT) are used to assess the health status of asthma and chronic obstructive pulmonary disease (COPD), respectively. However, whether these questionnaires are appropriate in patients with asthma-COPD overlap (ACO) has not been reported. This study aimed to evaluate the performance of the AQLQ and CAT in subjects with ACO.
Methods
Subjects were enrolled from two previously described observational studies in Beijing, China. ACO was defined by a consensus definition from a roundtable discussion. All subjects completed the AQLQ, CAT, St George’s Respiratory Questionnaire (SGRQ), pulmonary function tests, and the Asthma Control Questionnaire (ACQ)-5. Cross-sectional construct validity was evaluated by correlating the AQLQ and CAT with SGRQ score and other measures of asthma and COPD severity.
Results
147 subjects with ACO were recruited. There were floor effects on non-respiratory components of the CAT, and ceiling effects on emotion domains of the AQLQ. Both questionnaires were significantly correlated with ACQ-5 score but were not correlated with FEV1% predicted or FVC% predicted. The AQLQ and CAT were strongly correlated with SGRQ score (r = −0.657 and r = 0.623, respectively). Multivariable linear regression analysis showed that the AQLQ (standardized β-coefficient = −0.449, p < .001) had a stronger association with SGRQ score compared with CAT (standardized β-coefficient = 0.211, p = .023).
Discussion
The AQLQ and CAT were both valid for assessing the health-related quality of life in subjects with ACO, but the AQLQ performed better than CAT.
Keywords
Introduction
Asthma-chronic obstructive pulmonary disease (COPD) overlap (ACO) is characterized by persistent airflow limitation with several features usually associated with asthma and several features usually associated with COPD. The prevalence of ACO varies markedly with its definition, ranging from 0.9% to 11.1% in the general population, 11.1% to 61.0% in patients with asthma, and 4.2% to 66.0% in patients with COPD. 1 Patients with ACO have a greater burden of symptoms, frequent acute exacerbations, worse quality of life, a more rapid decline in lung function, higher mortality, and greater use of healthcare resources compared with patients with asthma or COPD alone.2–6 Consequently, more attention should be paid to ACO patients in clinical practice.
An efficient assessment of health status is of great importance to observe the effectiveness of the treatment, monitor disease progression, and rationalize the allocation of healthcare resources. The St George’s Respiratory Questionnaire (SGRQ) is widely used to assess health status in asthma patients and COPD patients 7 ; however, the routine use of SGRQ in clinical practice is limited owing to the lengthy and complex scoring algorithms of the instrument.
The Asthma Quality of Life Questionnaire (AQLQ) was developed to assess the health-related quality of life (HRQL) in patients with asthma and contains four domains: activity limitation, symptoms, emotional function, and environmental stimuli. 8 The COPD assessment test (CAT) is used to assess the disease-specific health status of COPD patients, 9 including respiratory components and non-respiratory components.10,11 These two questionnaires are widely used for asthma patients and COPD patients, respectively; however, for patients with features of both asthma and COPD (ACO), it is not known which questionnaire is more suitable to use. This study aimed to compare the performance of the AQLQ and CAT in patients with ACO. We assessed the construct validity by correlating the two questionnaires with SGRQ score and other measurements of asthma and COPD severity, with a further comparison of the associations of the AQLQ and CAT with SGRQ score.
Methods
Subjects
This analysis was performed using two previously described multicenter observational studies conducted in Beijing, China.12,13 Patients with ACO based on a consensus definition from a roundtable discussion 14 were included in this study. Major inclusion criteria: (1) post-bronchodilator (BD) forced expiratory volume in one second/forced vital capacity (FEV1/FVC) < 0.70 in individuals >40 years old; (2) at least 10 pack-years of tobacco smoking OR equivalent indoor or outdoor air pollution exposure; (3) documented history of asthma before age 40 years OR bronchodilator responsiveness (BDR) > 400 mL in FEV1. Minor inclusion criteria: (1) documented history of atopy or allergic rhinitis; (2) BDR of FEV1 ≥ 200 mL and 12% from baseline values on two or more visits; (3) peripheral blood eosinophil count ≥300 cells/μL. Patients who met all three major criteria and at least one minor criterion were included in this study. Exclusion criteria: serious cognitive impairment or cognitive delay that interfered with a patient’s ability to understand the questionnaire and cooperate with the study.
The protocol for this study was approved by the Ethics Committee of Beijing Chao-Yang Hospital (No. 2010-KE-65 and No. 2016-KE-183). Signed written informed consent was obtained from each patient prior to participation.
Questionnaires
St George’s respiratory questionnaire (SGRQ)
The SGRQ is used as a reference standard of quality of life in this study. It is a standardized self-reported scale to measure the impaired health and quality of life in patients with airway disease. 7 This respiratory-specific questionnaire has three domains (symptom, activity, and impact) and a total score. Scores are transformed into a point scale ranging from 0 to 100, with higher scores indicating a worse quality of life. 15
COPD assessment test (CAT)
The CAT has eight items, including respiratory components (cough, phlegm, chest tightness, and breathlessness going up a hill/stairs) and non-respiratory components (activity limitation at home, confidence leaving home, sleep, and energy). Each item is formatted as a six-point scale (0–5), making the tool easy to use for assessing the health status in COPD. 9 The total score ranges from 0 to 40, with 0 points indicating excellent health status and 40 points indicating the worst health status.
Asthma quality of life questionnaire (AQLQ)
The standardized version of the AQLQ, the AQLQ(S), is applicable for all adult patients with asthma to assess HRQL. It has 32 items in four domains: activity limitation (11 items), symptoms (12 items), emotional function (5 items), and environmental stimuli (4 items). The score ranges from 1 to 7, where one indicates maximal impairment and seven indicates no impairment. 8 The AQLQ(S) mandarin Chinese version, which has been validated in Chinese asthmatic patients, 16 was employed in this study.
Other measurements
Asthma control was assessed by the Asthma Control Questionnaire (ACQ)-5. The ACQ-5 has five items, scoring from 0 to 6, with higher scores indicating worse asthma control. 17 Ever-smokers were defined as current or former smokers with at least 10 pack-years of tobacco exposure. Spirometry and body plethysmography were performed, and a test measuring the diffusing capacity for carbon monoxide (DLCO, MasterScreen Body; Erich Jaeger, Friedberg, Germany) was administered according to American Thoracic Society/European Respiratory Society guidelines. 18 Fractional exhaled nitric oxide (FeNO) was measured by a nitric oxide analyzer (NIOX; Aerocine AB, Stockholm, Sweden) at flow rates of 50 mL/s. The count and percentage of eosinophils in peripheral blood were measured using an automated system (XN9000; Sysmex Co., Japan). The total immunoglobulin E (IgE) in peripheral blood was analyzed by means of the ImmunoCAP system (Phadia 250, Thermo Fisher Scientific).
Statistical analysis
SPSS 23.0 (IBM, Armonk, NY, USA) was used for data analysis. Normal distribution data were shown as means and standard deviations (SDs). Non-normal distribution data were shown as medians and interquartile ranges (IQRs). Categorical variables were expressed as number and percentage (%). Score distributions of the questionnaires were analyzed using histograms and Kolmogorov–Smirmov tests. Cross-sectional construct validity was evaluated by Spearman rank correlation analyses between the AQLQ and CAT with the reference standard of quality of life, namely the SGRQ. Correlations between the AQLQ, CAT, and other outcomes of asthma severity and COPD severity, namely the ACQ-5 score and lung function, were also assessed. A multivariable linear regression model including potential confounders was established to identify factors associated with the total score of SGRQ. Covariates included in the multivariable regression model were selected based on known confounders and statistical significance from the univariable analyses. To make the resulting regression coefficients comparable, standardized β-coefficients were used. p-values <0.05 were considered statistically significant.
Results
Patient characteristics and HRQL score distribution
Clinical characteristics of enrolled subjects with ACO (n = 147).

Data are presented as mean ± SD, median (IQR), or n (%).
ACQ-5: asthma control questionnaire-5; BD: bronchodilator; DLCO: diffusing capacity for carbon monoxide; FEV1: forced expiratory volume in one second; FEV1% predicted: forced expiratory volume in one second in percent of the predicted value; FVC: forced vital capacity; FeNO: fractional exhaled nitric oxide; IC/TLC: inspiratory capacity/total lung capacity ratio; IgE: immunoglobulin E; RV/TLC: residual volume/total lung capacity ratio.

Frequency distribution of each HRQL score. (a) SGRQ total; (b) CAT total; and (c) AQLQ total.
Distribution of AQLQ score, CAT score, and SGRQ score in ACO subjects.

AQLQ: asthma quality of life questionnaire; ACO: asthma-COPD overlap CAT: COPD assessment test; SGRQ: St George’s respiratory questionnaire; IQR: interquartile range.
aRespiratory components: cough, phlegm, chest tightness, and breathlessness going up a hill/stairs.
bNon-respiratory components: activity limitation at home, confidence leaving home, sleep, and energy.
Construct validity of the AQLQ and CAT
Correlation analyses between AQLQ, CAT, and clinical parameters of disease severity.

AQLQ: asthma quality of life questionnaire; ACQ-5: asthma control questionnaire-5; CAT: COPD assessment test; FEV1% predicted: forced expiratory volume in one second in percent of the predicted value; FVC% predicted: forced vital capacity in percent of the predicted value; SGRQ: St George’s respiratory questionnaire.
Spearman rank correlations are applied. ***p < .001.
Correlation analyses between AQLQ, CAT, and SGRQ.
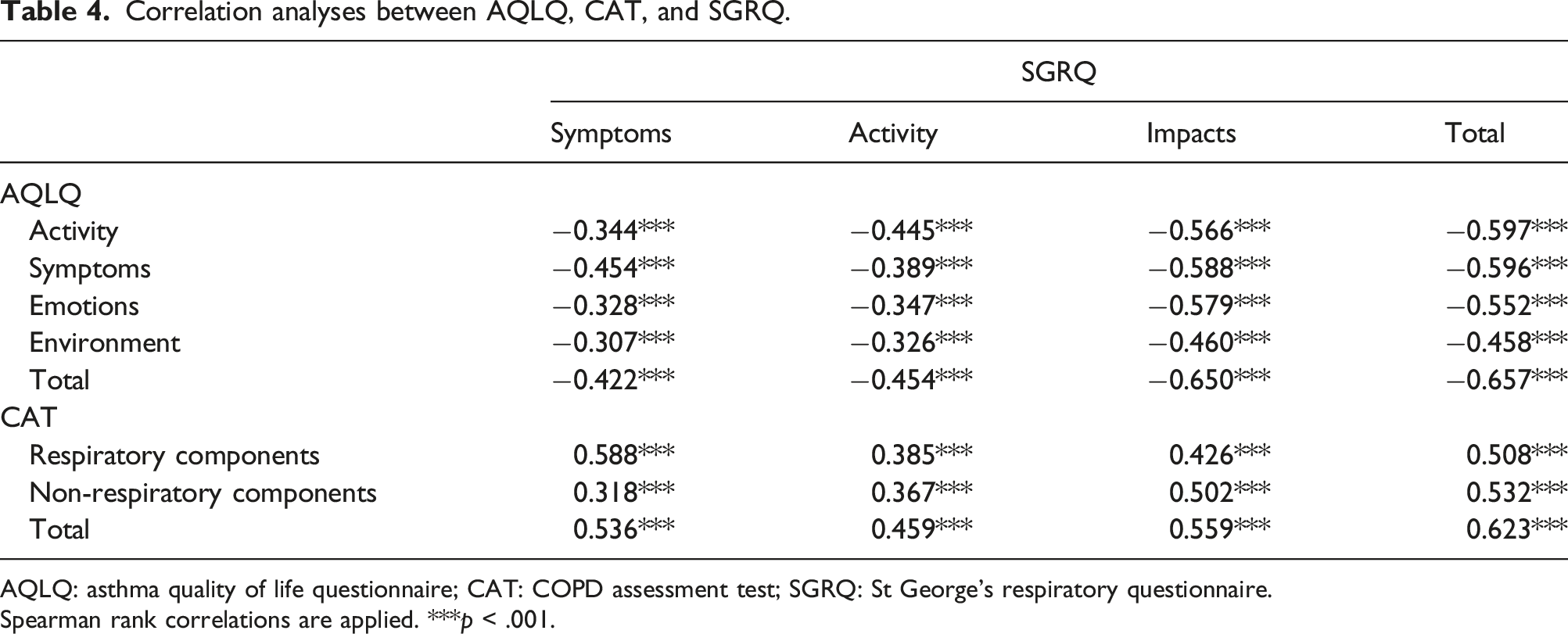
AQLQ: asthma quality of life questionnaire; CAT: COPD assessment test; SGRQ: St George’s respiratory questionnaire.
Spearman rank correlations are applied. ***p < .001.
Comparison of the associations of the AQLQ and CAT with SGRQ score
Multivariable linear regression analysis for the factors associated with SGRQ.
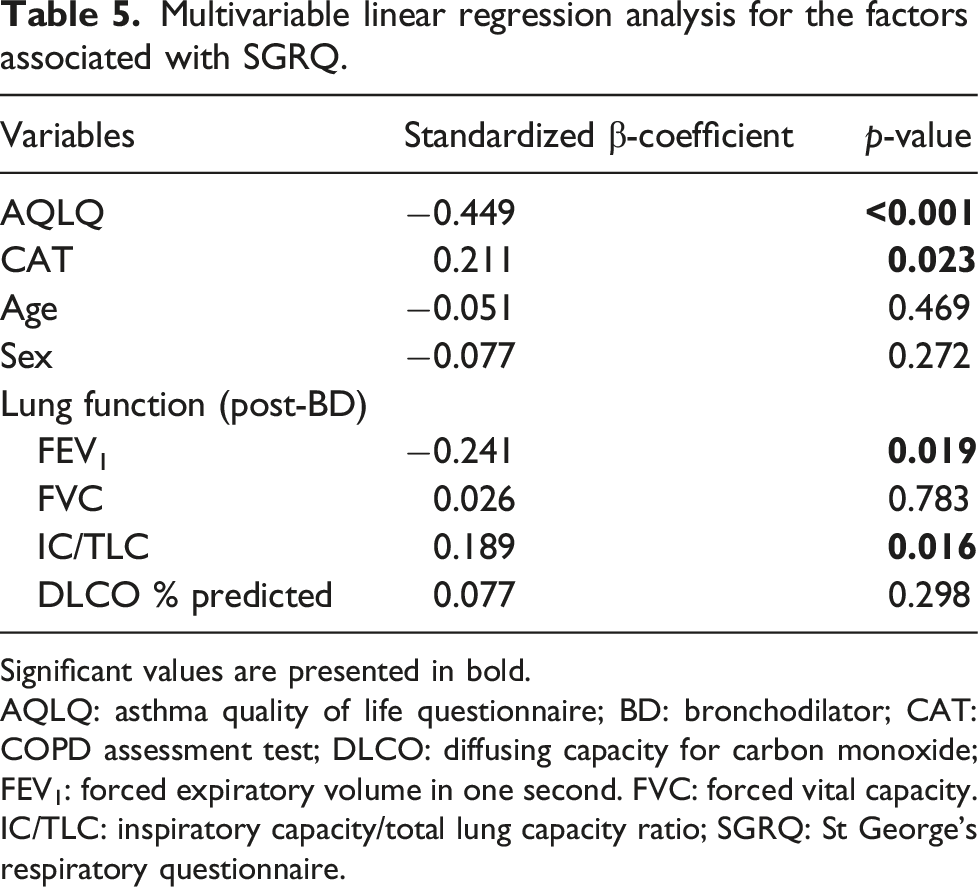
Significant values are presented in bold.
AQLQ: asthma quality of life questionnaire; BD: bronchodilator; CAT: COPD assessment test; DLCO: diffusing capacity for carbon monoxide; FEV1: forced expiratory volume in one second. FVC: forced vital capacity. IC/TLC: inspiratory capacity/total lung capacity ratio; SGRQ: St George’s respiratory questionnaire.
Discussion
Asthma and COPD are umbrella labels for heterogeneous conditions of chronic airway disease. 19 Distinguishing asthma and COPD is important, but in real-world settings, many patients may have features of both asthma and COPD (ACO).6,20 ACO subjects experience a greater symptom burden and a worse health status than those with asthma or COPD alone, 21 so an efficient measure of quality of life is crucial for these patients. This study is the first to compare the performance of the AQLQ and CAT in subjects with ACO. The AQLQ and CAT were valid for ACO patients owing to a strong correlation with the SGRQ score and ACQ-5 score. However, the AQLQ may be superior to CAT to assess HRQL in ACO patients as its association with SGRQ was stronger.
The distribution of the HRQL scores in this study were slightly skewed towards mild ends in the AQLQ, CAT, and SGRQ. Consequently, floor effects were shown on non-respiratory components of the CAT, and ceiling effects were shown on emotion domains of the AQLQ, but no floor or ceiling effects were shown on the SGRQ. This is probably because the ACO patients enrolled in the study were stable patients with moderate lung function decline (FEV1% predicted 60.7% ± 15.4%), and there is less impairment on activity limitation and emotional status in such patients. Our result indicated that the CAT and AQLQ were not sensitive enough to capture the health restraints for ACO patients with mild to moderate disease. Previous studies showed pronounced ceiling effects on the AQLQ in individuals with asthma from the general population, 22 which is in concordance with our findings. However, there are fewer items in the AQLQ and CAT than in SGRQ, so the former two questionnaires are superior to SGRQ in rapidly assessing quality of life in clinical practice.
In this study, the CAT and AQLQ were significantly associated with ACQ-5 score. A previous study in asthmatic subjects reported that asthma control assessed by the Global Initiative for Asthma (GINA) criteria (including occurrences of exacerbations, limitations of activities, diurnal and nocturnal symptoms, and the need for rescue/reliever treatment) was associated with quality of life assessed by the generic EuroQol 5-dimension (EQ-5D). 23 Consistent with these findings, our results showed that the association between asthma control and quality of life was still significant in patients with ACO. Another study found that asthma control assessed by asthma control test (ACT) score was closely correlated with the CAT score in patients with ACO. 24 Concordant with those results, we found a strong correlation between ACQ-5 score and CAT score in patients with ACO in this study. Our results showed that the CAT and AQLQ were well correlated with asthma control status, indicating a good construct validity in ACO patients for both questionnaires.
The relationship between lung function and quality of life in asthma and COPD has been widely reported. Previous studies showed that airflow limitation was weakly correlated with total AQLQ scores in patients with asthma and poorly related to health status in patients with COPD.25,26 In our study, neither FEV1% predicted nor FVC% predicted was correlated with the CAT score or AQLQ score in patients with ACO. These results are consistent with the observations of Kurashima et al. 24 that HRQL assessed by CAT score was not correlated with FEV1% predicted in patients with ACO. Our findings confirmed that lung function is a poor indicator of symptoms, exercise capacity, and overall quality of life in both asthma and COPD, and does not capture the heterogeneity of chronic airway disease.19,27,28
Other factors associated with HRQL in patients with ACO were also identified in this study. IC/TLC was an independent factor associated with the total SGRQ score in this study. This was not surprising since several studies reported that IC/TLC was a significant predictor of daily physical activity and mortality in patients with COPD.29–31 DLCO% predicted was associated with quality of life assessed by CAT and SGRQ score in COPD patients in a previous study, 32 but it was not an independent factor associated with HRQL in patients with ACO in this study. These results also imply that pure COPD and ACO may be two different entities for which the main physiologic impairments are located in the lung parenchyma and airways, respectively.
The multivariable regression analysis in this study showed that the AQLQ score had a stronger association with SGRQ score than the CAT score, which indicates that the AQLQ may be more suitable than the CAT for evaluating quality of life in patients with ACO. There are several speculative explanations for this result. First, the development and validation of the CAT was based on classical patients with COPD, excluding patients with asthma as a primary diagnosis. 9 The final 8-item CAT content only covers symptoms usually presented in COPD, whereas wheezing—the specific symptom for asthma—was moved out of the CAT questionnaire. Second, while the AQLQ was developed to be specific to asthmatic patients, its 32 items cover almost all aspects of the CAT. 8 In addition, in contrast to COPD which has relatively stable symptoms and fixed airflow obstruction, the variability of symptoms and airflow obstruction is more apparent in patients with ACO, 33 especially when exposed to environmental stimuli. The AQLQ has a specific domain designed for measuring impaired quality of life in relation to airway responsiveness to stimuli, 34 which facilitates a comprehensive description of health status for patients with features of asthma.
Measuring quality of life is important in clinical research and when monitoring patient progress in clinical practice. Our results indicated that some studies that only used CAT score to measure health status may not be sufficient to observe the efficacy of a certain therapy in ACO patients.35,36 Owing to the complexity and variability of symptoms in ACO patients, the results of this study recommend using the AQLQ to assess HRQL in therapeutic trials and real-world studies. Currently, more advanced treatments and biologics are being studied in asthmatic or COPD patients with target treatment traits.37,38 More studies are needed using the AQLQ to monitor health status and evaluate treatment efficacy among patients with ACO in the future.
This study does have some limitations. First, the results are restricted by the relatively small number of subjects enrolled. Second, the ACO patients in this study tended to have moderate disease severity, which may cause a small bias to our sample. In addition, the extent to which the HRQL score changes when subjects improve or deteriorate could not be analyzed in this study owing to the cross-sectional design. Further studies with a longitudinal design and a larger sample size are needed to verify our findings.
Conclusion
In summary, the AQLQ and CAT are both valid to assess the quality of life in patients with ACO. The AQLQ involves a more comprehensive measure of symptoms and disease impact and is more suitable for use in ACO patients than the CAT. We believe these findings will help to monitor quality of life more efficiently in subjects with ACO and provide HRQL measurement recommendation for future research.
Supplemental Material
Supplemental Material - Comparison of health-related quality of life measures in asthma-COPD overlap
Supplemental Material for Comparison of health-related quality of life measures in asthma-COPD overlap by Meishan Liu, Xuwen Yang, Ying Wang, Yong Lu, Lirong Liang, Hong Zhang and Kewu Huang in Journal of Chronic Respiratory Disease.
Consent to participate
Informed consent was obtained from all participants.
Footnotes
Author contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of competing interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Key R&D Program of China, Ministry of Science and Technology of China (grant number 2016YFC0901102); the Project of “Deng Feng” Talent Training, Beijing Municipal Administration of Hospitals (grant number DFL20190301); the National Natural Science Foundation of China (grant number 81870032); the Financial Budgeting Project of Beijing Institute of Respiratory Medicine (grant number YSBZ2023001); and AstraZeneca (grant number ESR-16-12485).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
