Abstract
Objective
Recently, a type 2 inflammation (T2) high endotype in bronchiectasis was identified. The fraction of exhaled nitric oxide (FeNO) and blood eosinophil count (BEC) are recognized as representative biomarkers of T2 inflammation. Herein, we investigate the clinical characteristics of T2-high endotype in non-cystic fibrosis bronchiectasis patients classified by FeNO and BEC.
Methods
This retrospective study included 164 bronchiectasis patients treated in the First Affiliated Hospital of Sun Yat-sen University from December 2017 to July 2022. Clinical characteristics were analyzed after classifying patients into four groups according to T2 inflammation biomarkers (FeNO ≥25 ppb; BEC ≥200/µL).
Results
Among the 164 bronchiectasis patients, 35.3% (58/164) presented with high FeNO, 30.5% (50/164) presented with high BEC, and 10.4% (17/164) had high FeNO and BEC. Patients with high FeNO and low BEC presented with better lung function, fewer affected lobes, and lower dyspnea prevalence compared with the three other groups. Moreover, decreased FeNO, instead of decreased BEC, is revealed to be an independent predictor for disease severity and airflow obstruction in bronchiectasis.
Conclusions
Simultaneous evaluation of FeNO and BEC proposed different endotypes of bronchiectasis established that patients with low BEC and high FeNO had better lung function, fewer affected lobes, lower dyspnea prevalence, and less disease severity. This result will contribute to a more comprehensive assessment of the disease severity and lead to more precise treatment of T2 inflammation in bronchiectasis patients.
Keywords
Introduction
Bronchiectasis, the permanent and irreversible enlargement of the airways is characterized by chronic infection or inflammation, is the third most frequent chronic respiratory disease. 1 Bronchiectasis is some geographical variation in incidence, etiology and clinical features. 2 Most bronchiectasis is idiopathic, except that in Asia it is mainly secondary to infection, especially tuberculosis infection. 3 Except for the cases caused by cystic fibrosis, no medication for bronchiectasis has been approved in any current clinical guideline globally. 4 Hence, a necessary and urgent need exists to identify an approach to evaluate different endotypes of bronchiectasis for a treatable feature.
Although airway neutrophilic inflammation has been historically recognized to be involved in the pathophysiology of bronchiectasis, recent studies reported a type 2 inflammation (T2) high endotype in bronchiectasis.5–7 Up to one-third of bronchiectasis patients show a predominant eosinophilic airway inflammation with increased blood eosinophil count (BEC) or fractional exhaled nitric oxide (FeNO), which represent systemic and airway eosinophilic inflammation, respectively.6,8 Increased FeNO or BEC appeared to identify a group of bronchiectasis patients who present with specific clinical characteristics and can be effectively treated with inhaled corticosteroids (ICS).7,9,10 To the best of our knowledge, this is the first study to combine both type 2 inflammatory biomarkers to evaluate bronchiectasis.
We conducted a retrospective and observational study by focusing on T2 inflammation and the clinical features of non-cystic fibrosis bronchiectasis patients without asthma to explore the potential association between two type 2 inflammatory biomarkers and disease severity. This study presented a better understanding of different phenotypes in patients with bronchiectasis, leading to more individualized and effective treatment.
Methods
Data source and study population
Patients with stable bronchiectasis aged 18 years or older admitted to the First Affiliated Hospital of Sun Yat-sen University from December 2017 to July 2022 were retrospectively enrolled in this study. We excluded patients with asthma, allergic bronchopulmonary aspergillosis, or any evidence of exacerbation. This research was reviewed by the Institutional Research Ethics Committee of our hospital and ethics approval was obtained ([2023]080).
Using the cut-off values of 25 ppb (FeNO) and 200 cells/µL (BEC), we divided the bronchiectasis patients into four groups: low FeNO/low BEC; high FeNO/low BEC; low FeNO/high BEC and high FeNO/high BEC.
The FACED score 11 and Bronchiectasis Severity Index (BSI) 12 were used to predict prognosis in bronchiectasis. The FACED score consists of the FEV1 (% predicted) (F), age (A), chronic colonization by Pseudomonas aeruginosa (PA) (C), radiological extension (E) and dyspnea (D). The BSI consists of age, clinical body mass index (BMI), FEV1 (% predicted), radiological extension, hospital admission prior to study, exacerbation history, MRC dyspnea scale, chronic colonization by PA, and chronic colonization by other microorganisms. According to the FACED score, the bronchiectasis cases were classified into three groups (mild, 0–2 points; moderate, 3–4 points; and severe, 5–7 points).
FeNO measurement
FeNO measurements were made using the NIOX MINO analyzer (Aerocrine AB, Sweden) according to the recommendations of the American Thoracic Society. 13
PFT
PFT was performed using body plethysmography (Medisoft) and flow–volume curves were obtained in accordance with the 2014 recommendations of the Chinese National Guidelines of Pulmonary Function Test . 14
Statistical analysis
Statistical analyses and figures were obtained using the GraphPad Prism eight software. The results are shown as mean ± standard deviation for normal distributed variables, median (interquartile range) for quantitative variables or for non-normal distributed variables, or as the percentage of patients (%). The comparison of differences between groups was conducted with the Student’s t-test for normally distributed variables or the Mann–Whitney U test for non-normally distributed variables. A univariate logistic regression model was utilized to evaluate the independent effects of FeNO and BEC on the clinical outcomes of bronchiectasis. p < .05 was defined as statistically significant.
Results
Baseline characteristics of bronchiectasis patients
Baseline characteristics of 164 bronchiectasis patients in stable stage.

Note: parametric data are expressed as mean (SD); nonparametric data are expressed as median (25%–75%).
Abbreviations: M: male; F: female; BMI: body mass index; FeNO: fractional exhaled nitric oxide; ppb: parts per billion; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; PA: pseudomonas aeruginosa; BSI: bronchiectasis severity index; COPD: chronic obstructive pulmonary disease.
Clinical features of the groups classified by FeNO and BEC
Demographics and clinical features of bronchiectasis patients classified by type 2 biomarker.

Note: parametric data are expressed as mean (SD); nonparametric data are expressed as median (25%–75%).
Abbreviations: M: male; F: female; BMI: body mass index; FeNO: fractional exhaled nitric oxide; ppb: parts per billion; BSI: bronchiectasis severity index; PA: pseudomonas aeruginosa; COPD: chronic obstructive pulmonary disease.
Statistical analyses and significance: *, p < .05; ****, &&&&, ^^^^, ####, p < .0001 between the patient groups.
The PFT data of bronchiectasis patients classified by type 2 biomarker.
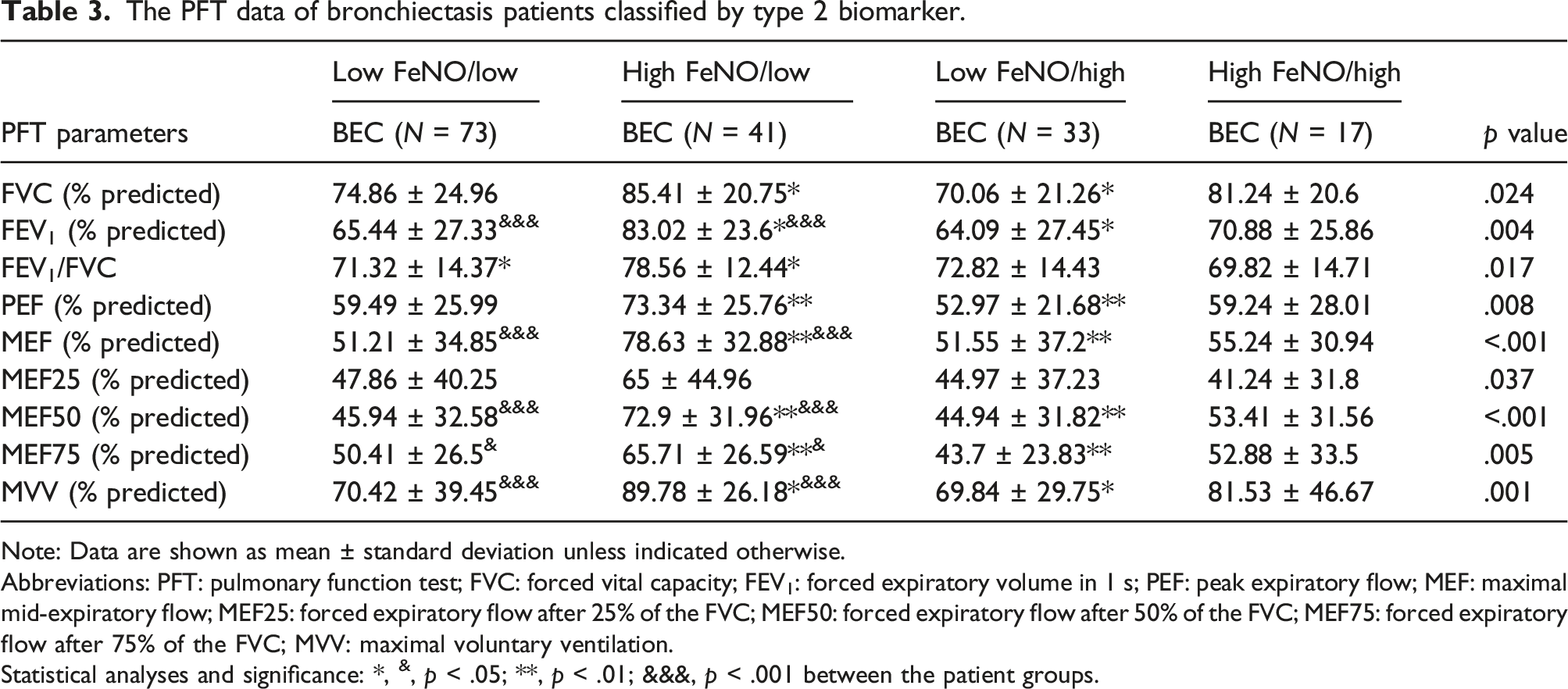
Note: Data are shown as mean ± standard deviation unless indicated otherwise.
Abbreviations: PFT: pulmonary function test; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s; PEF: peak expiratory flow; MEF: maximal mid-expiratory flow; MEF25: forced expiratory flow after 25% of the FVC; MEF50: forced expiratory flow after 50% of the FVC; MEF75: forced expiratory flow after 75% of the FVC; MVV: maximal voluntary ventilation.
Statistical analyses and significance: *, &, p < .05; **, p < .01; &&&, p < .001 between the patient groups.
Evaluation of disease severity in the different bronchiectasis patients
Disease severity related factors among the four subgroups.

Note: parametric data are expressed as mean (SD).
Abbreviations: BMI: body mass index; FEV1: forced expiratory volume in 1 s; BMI: body mass index.
Statistical analyses and significance: &, p < .01; ***, patient groups, p < .001, between the patient groups.
Independent effects of FeNO and BEC on disease severity score and pulmonary function
To investigate the clinical significance of systemic and airway eosinophilic inflammation, we compared FACED and PET scores among groups divided by BEC and FeNO. The high FeNO/low BEC group had the lowest FACED but the highest FEV1 score; by contrast, the low FeNO/high BEC group had the highest FACED and lowest FEV1 score (Figure 1). Furthermore, the independent effect of increased FeNO levels or BEC was studied by univariate logistic regression analysis. Increased FeNO levels instead of BEC were linked to the milder severity of bronchiectasis (FACED ≥3) and less airflow obstruction (FEV1 [% predicted] <50%), with ORs of 0.2609 (95% CI = 0.13–0.53, p = .0002) and 0.1557 (95% CI = 0.06–0.42, p < .0001), respectively (Table 5). The FACED score and PFT score among the four subgroups. Adjusted odds ratios (95% CIs) for disease severity and pulmonary function in univariate logistic regression. Note: Results in boldface indicates p < .05. Abbreviations: BEC: blood eosinophil count; FeNO: fractional exhaled nitric oxide; ppb: parts per billion; FEV1: forced expiratory volume in 1 s.

Discussion
We performed a retrospective study of 164 bronchiectasis patients without asthma. Idiopathic, post-infectious and COPD constitute major bronchiectasis aetiologies in our study. The high FeNO/low BEC group presented with better lung function, fewer affected lobes, lower dyspnea prevalence, and lower disease severity compared with the three other groups. Furthermore, increased FeNO but not BEC, was an independent predictor for milder severity of bronchiectasis and less airflow obstruction. As far as we know, this is the first study to combine those two biomarkers of type 2 inflammation to evaluate the clinical features of bronchiectasis patients.
High FeNO and high BEC are likely recognized as indicators of airway and systemic type 2 inflammation, respectively. In our study, 35.3% of the bronchiectasis patients presented with high FeNO, 30.5% with high BEC, and 10.4% with both high FeNO and high BEC. These outcomes indicate that persistent local and systemic T2 inflammation rather than neutrophilic airway inflammation occur in patients with specific clinical features. In turn, that observation suggests that more attention should be directed toward clinical practice.
Previous studies have explored the significance in bronchiectasis of either FeNO or BEC solely, but the results seem paradoxical. On the one hand, Oriano et al. 15 found that T2-high endotype bronchiectasis was associated with greater disease severity, worse dyspnea, worse respiratory function, and adverse effect on the quality of life. Cho et al. 16 confirmed that the FeNO levels tended to increase along with the disease severity scores by HRCT. On the other hand, Wang et al. 17 and Shoemark et al. 18 concluded that bronchiectasis patients with high BEC (≥100/µL) exhibited milder severity and better clinical outcomes, lung function, and nutritional status. Tsikrika et al. 19 showed that bronchiectasis subjects with high FeNO had milder disease severity and greater bronchodilator reversibility. The reasons for these contrary results may be the different inclusion criteria of patients (some studies did not exclude asthma), the varied sample sizes, the enrolment of patients from different regions, the inconsistent cutoff values. Note that eosinophil levels mainly correlate with the degree of the IL-5 pathway, and the FeNO level represents the local activity of the IL-4 and IL-13 pathways rather than the IL-5 counterpart in the bronchial mucosa in asthma cases. 20
In this study, high FeNO and low BEC jointly occurred in approximately 25% of all patients, who, in turn, presented with better condition, higher lung function, fewer affected lobes, lower dyspnea prevalence, and less disease severity compared with the three other groups. Thus, the trend of FeNO and eosinophils is inconsistent, thereby reflecting different underlying mechanisms for the two inflammatory biomarkers. Moreover, patients with bronchiectasis should be further subdivided into different groups with different treatment regimens. The high FeNO group may be a suitable target for inhaled steroids or anti-IL-4 or anti-IL-13 therapy, and the high BEC group may be more responsive to anti-IL-5 treatment. This interesting finding is in line with previous research on asthma that showed group treatment resistance with omalizumab and anti-IL-5 biologics for high FeNO/low BEC groups. 21
Furthermore, we investigated the independent effect of increased FeNO or increased BEC levels on clinical outcomes and found that FeNO, rather than BEC, is an independent risk factor for better prognosis. The lower levels of FeNO might be explained by the structural damages of lung parenchyma in most bronchiectasis patients. 22 In this study, patients with higher FeNO have fewer damaged lung lobes and thus might experience a limited extent of chronic inflammation. FeNO levels are also associated with response to inhaled steroids, so patients with high FeNO often have greater reversibility after treatment, which may explain why patients with high FeNO have better lung function and less severe disease.
BEC and FENO are commonly used to assess inflammation in asthma and COPD. BEC greater than 300/µL or FeNO ≥50 ppb indicates that eosinophilic asthma is likely to be present. COPD has been previously characterized by neutrophil airway inflammation, but eosinophilic COPD (with BEC ≥200 cells/μL or FeNO ≥25 ppb) has also been confirmed to exert an essential impact on acute exacerbation, readmission, and treatment.23,24 Therefore, for bronchiectasia, which is also characterized by neutrophil airway inflammation, the cutoff values of 25 ppb for FENO and 200/µL for BEC established a statistically significant separation between the FACED scores and lung function parameters, an outcome which indicated that adult bronchiectasis patients with FeNO greater than 25 ppb or BEC greater than 200/µL should be treated with different regimens.
This research has several limitations that should be acknowledged. First, the work is a retrospective, single-center study that involves a relatively small number of samples, and the monocentric nature of the project hinders the generalizability of our findings; that is, future investigations should consider including international data. Second, although EOS and FeNO are globally accepted as representative biomarkers of T2 inflammation, we did not consider eosinophils in sputum. To understand the relationship between clinical features and T2 inflammation in patients with bronchiectasis, further prospective and large-scale studies are therefore needed.
Conclusions
This study identified four subtypes of bronchiectasis on the basis of the BEC and FeNO levels as representative biomarkers of eosinophilic inflammation. The status of low BEC and high FeNO was associated with better lung function, fewer damaged lobes, lower dyspnea prevalence, and lower disease severity. This result suggested that the simultaneous evaluation of BEC and FENO in patients with bronchiectasis could lead to a more comprehensive assessment of disease severity and may lead to more precise treatment, including potential ICS and biologics.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (82100054, 82200029); GuangDong Basic and Applied Basic Research Foundation (2020A1515110933); Guangdong Medical Science and Technology Research Foundation (C2023044).
