Abstract
This review addresses outstanding questions regarding initial pharmacological management of chronic obstructive pulmonary disease (COPD). Optimizing initial treatment improves clinical outcomes in symptomatic patients, including those with low exacerbation risk. Long-acting muscarinic antagonist/long-acting β2-agonist (LAMA/LABA) dual therapy improves lung function versus LAMA or LABA monotherapy, although other treatment benefits have been less consistently observed. The benefits of dual bronchodilation in symptomatic patients with COPD at low exacerbation risk, and its duration of efficacy and cost effectiveness in this population, are not yet fully established. Questions remain on the impact of baseline symptom severity, prior treatment, degree of reversibility to bronchodilators, and smoking status on responses to dual bronchodilator treatment. Using evidence from EMAX (NCT03034915), a 6-month trial comparing the LAMA/LABA combination umeclidinium/vilanterol with umeclidinium and salmeterol monotherapy in symptomatic patients with COPD at low exacerbation risk who were inhaled corticosteroid-naïve, we describe how these findings can be applied in primary care.
Keywords
Introduction
Patients with poorly controlled chronic obstructive pulmonary disease (COPD) symptoms, may experience poor clinical outcomes including worse health-related quality of life (HRQoL) and greater risk of hospitalization and death than patients with well-controlled symptoms.1,2 Many patients with COPD in primary care settings are symptomatic, with activity-related breathlessness (when ascending one flight of stairs) and/or reliance on daily rescue short-acting β2-agonist (SABA).3,4 Determining the best treatment strategy for these patients is important to achieve optimal clinical outcomes.
Long-acting bronchodilators form the cornerstone of inhaled maintenance therapy in symptomatic patients at low risk of exacerbations. Long-acting muscarinic antagonist/long-acting β2-agonist (LAMA/LABA) dual bronchodilator therapy has demonstrated lung function improvements over LAMA and LABA monotherapy in COPD. However, symptoms and health status benefits have been less consistent,5,6 raising several questions about the extent of the symptomatic benefits of LAMA/LABA dual therapy over monotherapy and the factors that may influence responses to treatment.
Currently, treatment guidelines vary in their recommendations for treatment of patients with symptomatic COPD at low exacerbation risk. Previously, the Global Initiative for Chronic Obstructive Lung Disease (GOLD) strategy report recommended starting with LAMA or LABA monotherapy for most patients; LAMA/LABA dual therapy was suggested as an initial treatment option for some patients with severe breathlessness or severe symptoms, but a stepwise approach is generally recommended. 7 Notably, international and local guidelines now align in recommendations regarding dual LAMA/LABA therapy.8–10 The findings from EMAX have been used as evidence to support recommendations in the recent update from GOLD for patients with symptomatic COPD with or without exacerbations to initiate dual therapy with LAMA/LABA, preferably using a single inhaler. 10
This update aligns with guidance from the American Thoracic Society 8 and some national guidelines including the UK National Institute for Health and Care Excellence, 9 which recommend a LAMA/LABA combination as initial therapy for many patients.
Key messages from the EMAX study.

COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; SABA, short-acting β2-agonist; UMEC, umeclidinium; VI, vilanterol.
Which patients and treatments were included in the EMAX study?
EMAX included patients ≥40 years of age (study mean: 64.6 years) with persistent airflow limitation (pre- and post-salbutamol forced expiratory volume in 1 s [FEV1]/forced vital capacity [FVC] ratio of <0.7 and a post-salbutamol FEV1 of ≥30–≤80% predicted). Patients had symptomatic disease (COPD Assessment Test [CAT] score ≥10) with infrequent exacerbations (≤1 moderate exacerbation and no severe exacerbations in the past year; GOLD 2019 group B), and no current asthma diagnosis. 11
To avoid confounding effects from concurrent inhaled corticosteroid (ICS) use, EMAX excluded patients with ICS-containing therapy use within 6 weeks of the study run-in period, and ICS use was not permitted during the study treatment period. Patients were randomized 1:1:1 to receive dual bronchodilator therapy (once-daily UMEC/VI 62.5/25 µg, ELLIPTA inhaler) or monotherapy (once-daily UMEC 62.5 µg, ELLIPTA; or twice-daily SAL 50 µg, DISKUS inhaler) double-blind for 24 weeks (Figure 1). Patient characteristics have previously been published
11
and are summarized in Table 2. EMAX study design. CAT, COPD Assessment Test; CID, clinically important deterioration; COPD, chronic obstructive pulmonary disease; E-RS, Evaluating Respiratory Symptoms–COPD; FEV1, forced expiratory volume in 1 s; SAC-TDI, self-administered computerized-Transition Dyspnea Index; SAL, salmeterol; SGRQ, St George’s Respiratory Questionnaire; UMEC, umeclidinium; VI, vilanterol. aLABA and LAMA were the only permitted maintenance treatments. Maintenance-naïve patients used no COPD maintenance treatment from 30 days prior to screening until their first dose of study treatment. bLABA and LAMA are not mutually exclusive categories. cNumber of patients with exacerbations requiring oral or systemic corticosteroids and/or antibiotics (moderate) in 12 months prior to screening (patients with >1 moderate exacerbation or with a severe exacerbation [requiring hospitalization] were excluded). BDI, Baseline Dyspnea Index; CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; E-RS, Evaluating Respiratory Symptoms–COPD; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; LABA, long-acting β2-agonist; LABD, long-acting bronchodilator; LAMA, long-acting muscarinic antagonist; SD, standard deviation; SGRQ, St George’s Respiratory Questionnaire.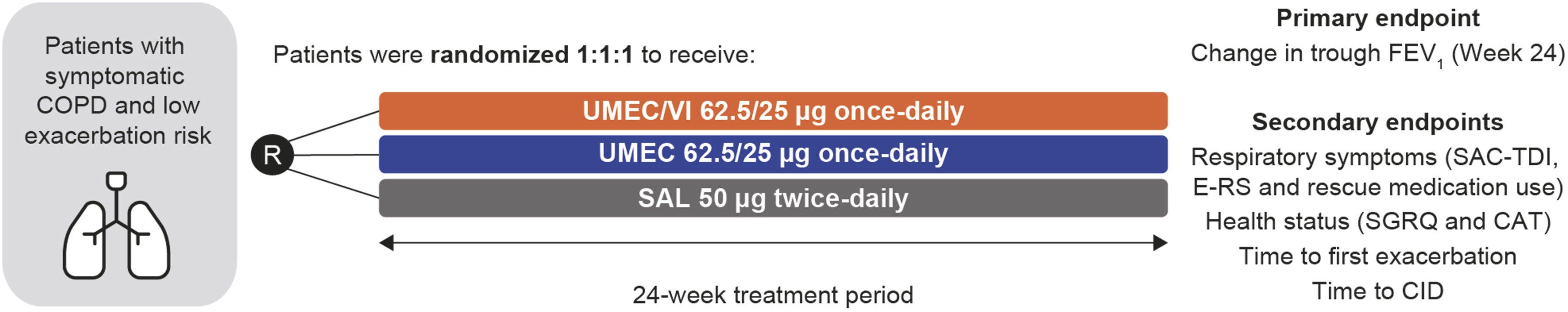
Does treatment with dual bronchodilator therapy provide better outcomes than monotherapy?
UMEC/VI dual therapy significantly improved trough FEV1 at Week 24 (primary endpoint) compared with UMEC and SAL monotherapy (Figure 2), with benefits apparent by 4 weeks and maintained for the duration of the study. Improvements in trough FVC and trough inspiratory capacity were also consistently greater with UMEC/VI versus monotherapy at all time points, indicating that patients treated with UMEC/VI were experiencing less lung hyperinflation than those treated with either monotherapy (Figure 2). Lung function improvements in dual versus monotherapy in symptomatic patients with low exacerbation risk. The primary analysis (including lung function, breathlessness, and rescue medication use) was a mixed model repeated measures analysis based on a two-sided hypothesis testing approach.
11
This article is available under a CC-BY-NC 4.0 license (http://creativecommons.org/licenses/by-nc/4.0/). CI, confidence interval; FEV1, forced expiratory volume in 1 s; SAL, salmeterol; UMEC, umeclidinium; VI, vilanterol.
Patient-reported outcomes assessed in the EMAX trial.
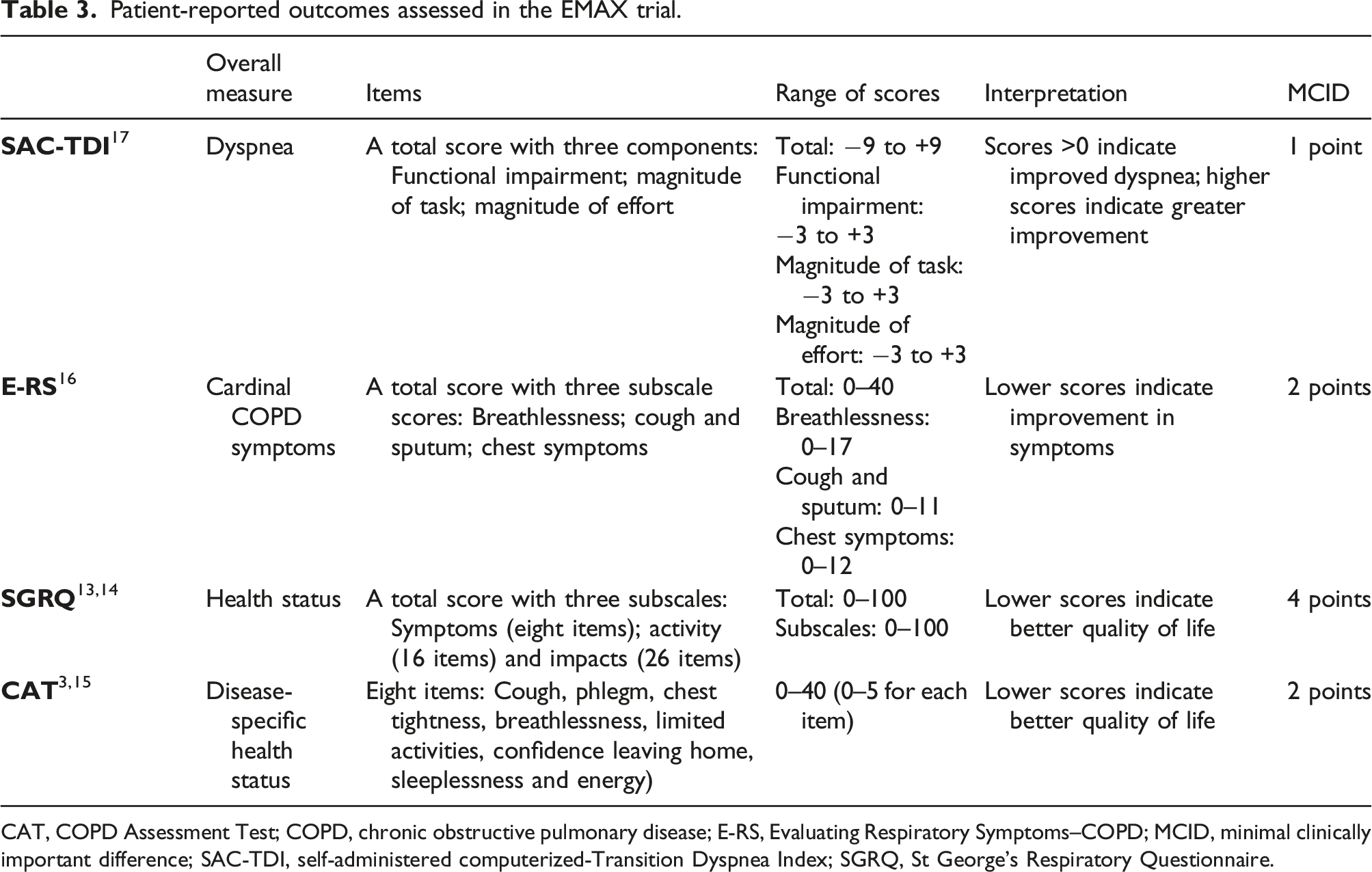
CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; E-RS, Evaluating Respiratory Symptoms–COPD; MCID, minimal clinically important difference; SAC-TDI, self-administered computerized-Transition Dyspnea Index; SGRQ, St George’s Respiratory Questionnaire.
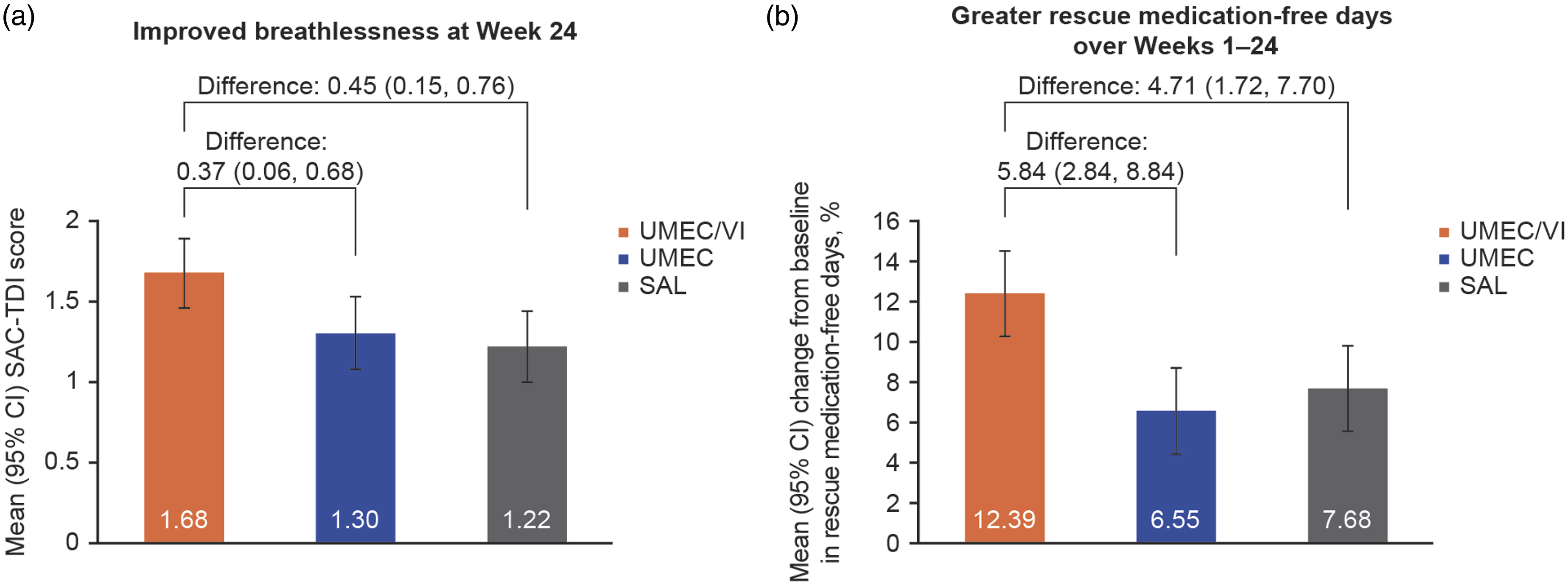
Breathlessness (a) and rescue medication-free days (b) in dual versus monotherapy in symptomatic patients with low exacerbation risk. Data shown are (a) mean (95% CI) SAC-TDI score and (b) mean (95% CI) percentage change from baseline in rescue medication-free days. The primary analysis (including lung function, breathlessness, and rescue medication use) was a mixed model repeated measures analysis based on a two-sided hypothesis testing approach. 11 This article is available under a CC-BY-NC 4.0 license (http://creativecommons.org/licenses/by-nc/4.0/). CFB, change from baseline; CI, confidence interval; SAC-TDI, self-administered computerized-Transition Dyspnea Index; SAL, salmeterol; UMEC, umeclidinium; VI, vilanterol.
Summary of the results in the EMAX trial for all patients. 11
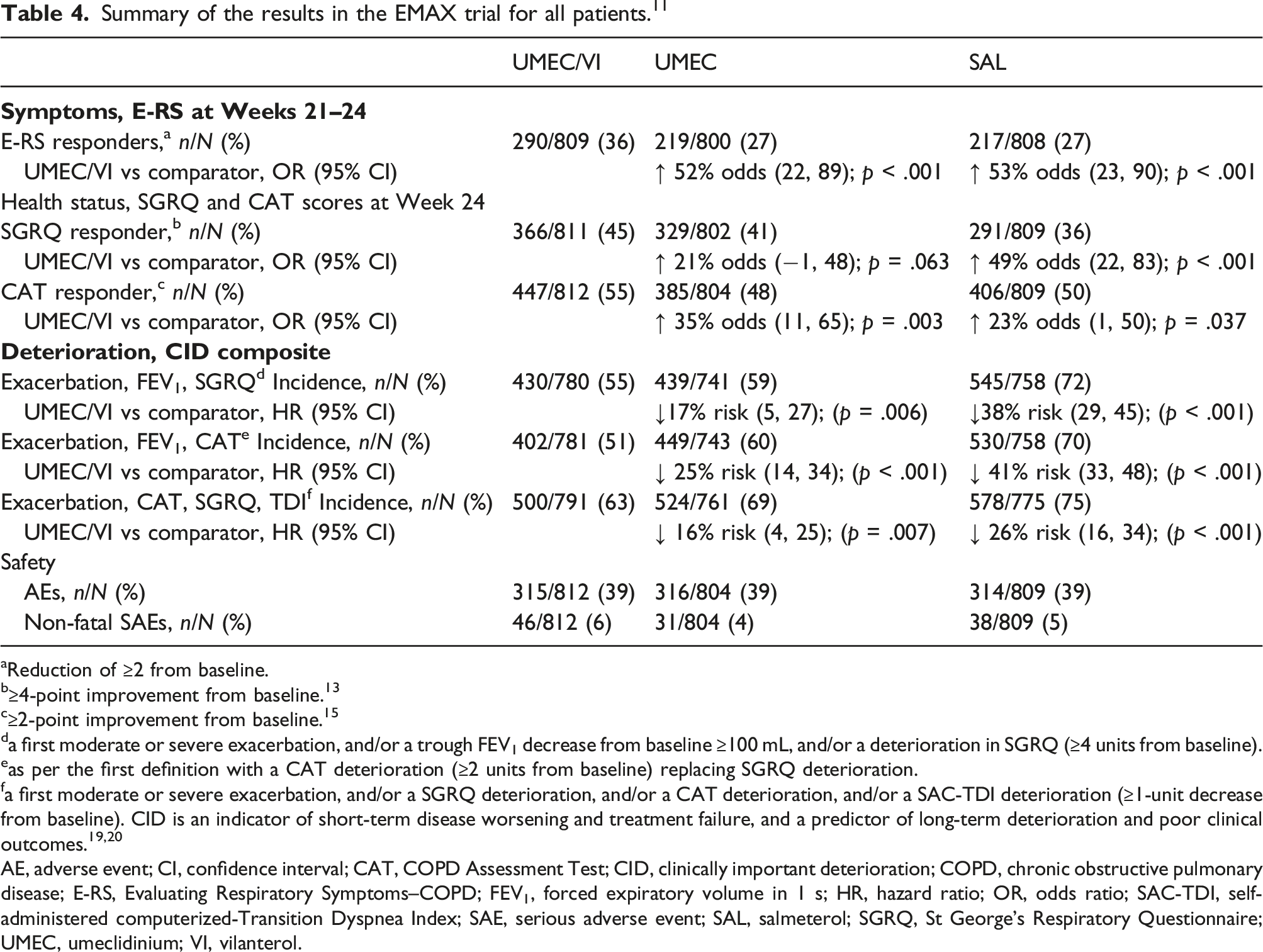
aReduction of ≥2 from baseline.
b≥4-point improvement from baseline. 13
c≥2-point improvement from baseline. 15
da first moderate or severe exacerbation, and/or a trough FEV1 decrease from baseline ≥100 mL, and/or a deterioration in SGRQ (≥4 units from baseline).
eas per the first definition with a CAT deterioration (≥2 units from baseline) replacing SGRQ deterioration.
fa first moderate or severe exacerbation, and/or a SGRQ deterioration, and/or a CAT deterioration, and/or a SAC-TDI deterioration (≥1-unit decrease from baseline). CID is an indicator of short-term disease worsening and treatment failure, and a predictor of long-term deterioration and poor clinical outcomes.19,20
AE, adverse event; CI, confidence interval; CAT, COPD Assessment Test; CID, clinically important deterioration; COPD, chronic obstructive pulmonary disease; E-RS, Evaluating Respiratory Symptoms–COPD; FEV1, forced expiratory volume in 1 s; HR, hazard ratio; OR, odds ratio; SAC-TDI, self-administered computerized-Transition Dyspnea Index; SAE, serious adverse event; SAL, salmeterol; SGRQ, St George’s Respiratory Questionnaire; UMEC, umeclidinium; VI, vilanterol.
While UMEC/VI provided greater improvements in COPD-related symptoms versus the two monotherapies, between-treatment differences in HRQoL during the trial were generally not statistically significant versus UMEC. 11 One possible explanation for these findings is the fact that St George’s Respiratory Questionnaire (SGRQ) and CAT assess some domains that are less likely to benefit from superior bronchodilation, such as fatigue. This is in contrast with the improvements demonstrated by measures that solely focus on COPD-related symptoms such as the E-RS questionnaire (that assesses breathlessness, cough and sputum, and chest symptoms) or the TDI (that evaluates dyspnea) (Table 4). 11
Despite the low exacerbation risk of the EMAX population, the risk of a first moderate or severe exacerbation was significantly reduced with UMEC/VI versus SAL, in line with the previously-reported benefits of LAMA/LABA over LABA but not over LAMA monotherapy in high exacerbation risk populations. 6 The risk of short-term COPD deterioration over 24 weeks was also significantly decreased (17%–41%) with UMEC/VI versus UMEC or SAL monotherapy across three different definitions of clinically important deterioration (Table 4). These data suggest that benefits of early treatment with dual therapy are maintained for at least 6 months in symptomatic patients with COPD and low risk of exacerbations.
All treatments were well tolerated, with a similar incidence of adverse events (AEs) and serious AEs between groups, and a similar rate of completion of the study (UMEC/VI: 88%; UMEC: 81%; SAL: 84%). 11 UMEC/VI provided better outcomes with no increase in AEs versus monotherapy, with ≤5% of patients experiencing treatment-related AEs.
Predicted incremental cost-related benefits of dual versus monotherapy in symptomatic patients with low exacerbation risk over a 10-year horizon.
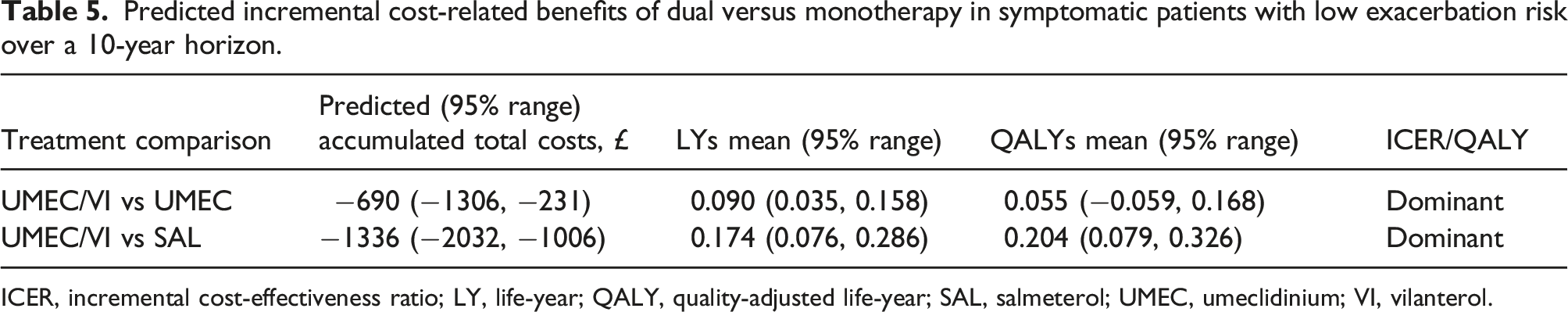
ICER, incremental cost-effectiveness ratio; LY, life-year; QALY, quality-adjusted life-year; SAL, salmeterol; UMEC, umeclidinium; VI, vilanterol.
Can monitoring of early responses help to predict sustained improvements?
It remains unclear how early responses to bronchodilator therapy relate to long-term outcomes. A post hoc analysis of EMAX showed that the majority (60%–85%) of patients who had a clinically significant symptom improvement at Weeks 1–4 retained this response at Weeks 21–24, suggesting that early symptom benefits of dual therapy translate into sustained longer-term improvements. 18 Another post hoc analysis using a novel composite endpoint of clinically important improvement at Weeks 4, 12 and 24 provided further evidence of the benefits of early treatment with dual therapy. 22
What impact do patient characteristics have on responses to bronchodilator treatment?
Secondary pre-specified and post hoc analyses of EMAX have explored how patient characteristics influence response to bronchodilator treatment and indicate that the benefits of dual bronchodilators versus monotherapy are maintained in all patient subgroups that have been evaluated.
Improvements in most study outcomes with UMEC/VI versus monotherapy were seen irrespective of the level of impairment in HRQoL at baseline, suggesting that patients are likely to benefit from dual bronchodilation across a range of symptom severities. 23
Patients with high (≥1.5 puffs/day) versus low (<1.5 puffs/day; 50%) SABA use had worse lung function, more frequent symptoms, and were less likely to use maintenance bronchodilators at baseline. 12 Nonetheless, both subgroups demonstrated significant improvements in trough FEV1 and symptoms (SAC-TDI and E-RS score) with dual versus monotherapy, although improvements in symptoms outcomes did not reach statistical significance in high SABA users. Post hoc analyses also suggested that the greatest symptomatic improvements with UMEC/VI versus monotherapy were observed in patients with <4 puffs/day of SABA, possibly because SABA use ≥4 puffs daily may diminish a patient’s ability to perceive changes in their symptoms. 12
On average, patients involved in EMAX had been carrying a diagnosis of COPD for 8 years. Despite this, 31% of the study population were not treated with a LAMA or LABA according to current recommendations7–9 at study entry, illustrating a common clinical observation that many symptomatic patients with COPD do not receive any maintenance treatment and are therefore undertreated. 21 During the study, these maintenance-naïve patients consistently demonstrated a trend for greater improvements in trough FEV1, dyspnea, rescue medication use and HRQoL than those receiving maintenance treatment with one long-acting bronchodilator at study entry. 21 This finding is consistent with previous studies comparing dual bronchodilators with monotherapy in maintenance-naïve patients.24–26
Both current and former smokers (50% each) showed improved trough FEV1, reduced rescue medication use, and improved odds of experiencing a clinically meaningful improvement in symptoms with UMEC/VI versus monotherapy. 27
Finally, improvements in trough FEV1 and reductions in rescue medication at Week 24 use were greater with UMEC/VI versus monotherapy across the full range of baseline reversibility values. 28
Application to clinical practice and treatment
The EMAX trial included symptomatic patients with COPD without a history of frequent exacerbations or ICS treatment, which reflect many patients seen in primary care settings. UMEC/VI dual bronchodilator therapy provided greater benefits than monotherapy across multiple endpoints and subgroups, with a safety profile similar to that of monotherapies and in a cost-effective manner. Importantly, these data are cited as rationale for the recommendations in the most recent report from GOLD, which recommends that patients with symptomatic COPD with or without exacerbations initiate treatment with a dual bronchodilator. 10
To effectively implement treatment based on these findings, it is important to identify patients who may benefit from dual bronchodilator therapy versus alternative treatments, for example, as-needed SABA, long-acting bronchodilator monotherapy or ICS-containing therapy.7,8 Post-bronchodilator FEV1 should be determined in symptomatic patients to confirm COPD diagnosis and objectively measure the degree of airflow limitation. 7 Symptom burden should also be assessed systematically, since over half of patients with COPD managed in primary care are highly symptomatic.3,4 If a validated instrument such as the CAT is unavailable, simply asking patients whether they experience breathlessness while climbing stairs or use several daily puffs of rescue medication may help identify patients who could benefit from dual bronchodilator therapy. Concurrent SABA use should also be considered when assessing treatment benefit with bronchodilator therapies, since excessive use is a marker of severe and symptomatic disease. 29 The occurrence and frequency of exacerbations should also be documented. This is reflected in the most recent report from GOLD, which suggests initial treatment based on COPD symptoms such as breathlessness and exacerbations, whereby monotherapy is recommended for patients with lower symptom burden, and dual therapy for more symptomatic patients, based on evidence from EMAX. 10
A follow-up visit 4–8 weeks after dual therapy initiation is recommended to assess patients’ response to treatment, especially since early improvements in symptoms are associated with lower risk of future deteriorations.
22
A simplified algorithm for assessment and treatment of patients with COPD in primary care is presented in Figure 4. Simplified algorithm for pharmacological management of patients with symptomatic COPD in primary care. *For patients presenting with asthma features, ICS-containing regiments should be considered; †Consideration should be given when prescribing patients with monotherapy, since dual LAMA/LABA therapy has greater efficacy across a number of outcomes, a similar safety profile, and is cost-effective compared with monotherapy. CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist mMRC, modified Medical Research Council; SABA, short-acting β2-agonist.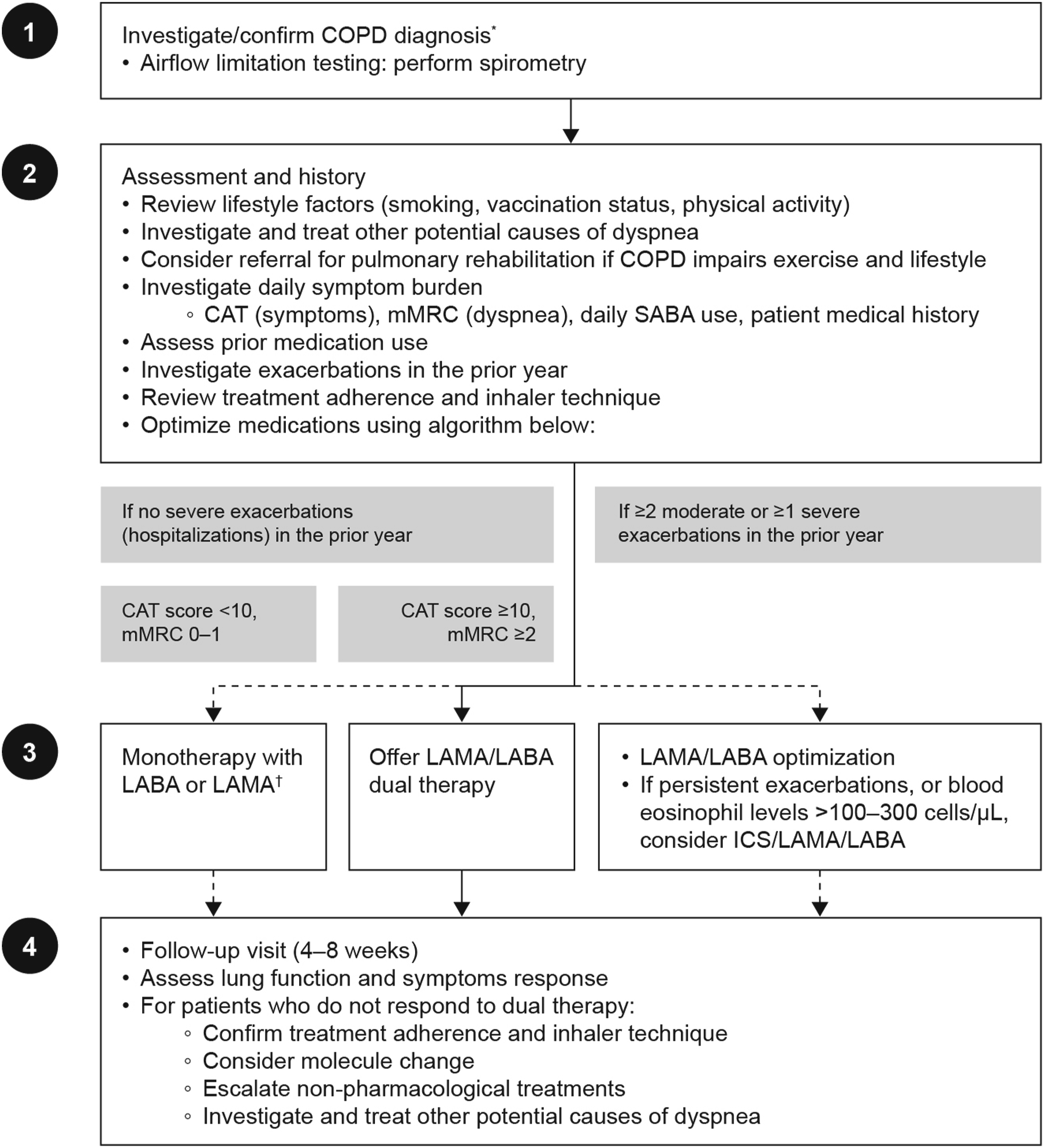
EMAX limitations and considerations
While the results of the EMAX trial have been highly informative, certain limitations resulting from the study design should be considered. The 24-week treatment period was appropriate for the trial aims but did not permit investigation of long-term exacerbation risk and disease progression. The allocation masking method, whereby all patients received treatment and placebo via a once-daily ELLIPTA inhaler and a twice-daily DISKUS inhaler, meant that patient preferences could not be evaluated. Finally, some composite endpoint analyses were conducted post hoc; however, these novel analyses provided valuable insights into treatment beyond the benefits of dual therapy over monotherapy.
Outstanding questions for clinical practice beyond EMAX.

COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic agonist; SABA, short-acting β2-agonist.
In conclusion, a wide range of patients with symptomatic COPD and low risk of exacerbation would benefit from initiating therapy with LAMA/LABA, rather than escalating in a stepwise approach from bronchodilator monotherapy.
Footnotes
Acknowledgments
Editorial support (in the form of writing assistance, including preparation of the draft manuscript under the direction and guidance of the authors, collating and incorporating authors’ comments for each draft, assembling tables and figures, grammatical editing and referencing) was provided by Mark Condon, Dphil, at Fishawack Indicia Ltd, UK, part of Fishawack Health, and was funded by GSK. The EMAX trial was funded by GSK (study 201749; NCT03034915).
Author contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, contributed to the writing and reviewing of the manuscript, and have given final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EMK has served on advisory boards, speaker panels or received travel reimbursement for Amphastar, AstraZeneca, Boehringer Ingelheim, Connect Biopharma, GSK, Mylan, Novartis, Pearl, Sunovion, Teva and Theravance, and has received consulting fees from Cipla and GSK. PWJ is an Emeritus Professor of Respiratory Medicine at St George’s, University of London, and a former full-time employee of GSK at the time of protocol development and contributed to study design and protocol on behalf of GSK. He is a part-time consultant at GSK and holds stocks/shares. IHB, DAL and CC are employees of GSK and hold stock and shares in GSK. LHB has received honoraria for giving a lecture or attending an advisory board for Airsonett, ALK-Abelló, AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, Meda, Novartis and Teva. FM has received research grants for participating in multicenter trials for AstraZeneca, Boehringer Ingelheim, GSK, Sanofi and Novartis, and has received unrestricted research grants and personal fees from Boehringer Ingelheim, Grifols and Novartis. IPN was an employee of GSK at the time of the EMAX trial, holds stocks and shares in GSK, and was a contingent worker on assignment at AstraZeneca. LT is a contingent worker on assignment at GSK. CFV has received grants from AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, Grifols, Mundipharma, Novartis, and the German Federal Ministry of Education and Research (BMBF) Competence Network Asthma and COPD (ASCONET), and has received personal fees from AstraZeneca, Boehringer Ingelheim, Berlin Chemie/Menarini, Chiesi, CSL Behring, GSK, Grifols, MedUpdate, Novartis and Teva. DISKUS and ELLIPTA are owned by/licensed to the GSK group of companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The EMAX trial was funded by GSK (study 201749; NCT03034915).
Data availability statement
Anonymized individual participant data and study documents can be requested for further research from www.clinicalstudydatarequest.com.

